A) \[\xleftarrow[\Pr ocess]{Wac\ker }C{{H}_{2}}=C{{H}_{2}}\] and three \[{{H}_{3}}C-C\equiv C-C{{H}_{3}}\]
B) \[{{H}_{3}}C-C{{H}_{2}}-C\equiv CH\], one \[{{H}_{2}}C=CH-C\equiv CH\] and two \[HC\equiv C-C{{H}_{2}}-C\equiv CH\]
C) \[NaOH\] and three \[{{104}^{o}}40\]
D) \[{{103}^{o}}\] and two \[{{107}^{o}}\]
Correct Answer: B
Solution :
Sulphuric anhydride is \[{{H}_{3}}C-C\equiv C-C{{H}_{3}}\] and its structure is Xs follows: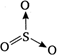 \[\Rightarrow \]\[3\sigma \], \[1p\pi -p\pi \], \[2p\pi -p\pi \] bonds are present.
\[\Rightarrow \]\[3\sigma \], \[1p\pi -p\pi \], \[2p\pi -p\pi \] bonds are present.
You need to login to perform this action.
You will be redirected in
3 sec
