A) \[NaOH\]
B) \[{{104}^{o}}40\]
C) \[{{103}^{o}}\]
D) \[{{107}^{o}}\]
Correct Answer: C
Solution :
\[{{\mathbf{H}}_{\mathbf{3}}}\mathbf{P}{{\mathbf{O}}_{\mathbf{3}}}\] \[\Rightarrow \] \[H-O-\underset{O}{\overset{H}{\mathop{\underset{\downarrow }{\overset{|}{\mathop{P}}}\,}}}\,\overset{P-H\,bonds}{\mathop{-O-H\}}}\,\] \[{{\mathbf{H}}_{\mathbf{3}}}\mathbf{P}{{\mathbf{O}}_{\mathbf{2}}}\] \[\Rightarrow \]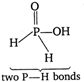
You need to login to perform this action.
You will be redirected in
3 sec
