-
question_answer1)
The nature of binding for a crystal with alternate and evenly spaced positive and negative ions is [CBSE PMT 2000]
A)
Covalent done
clear
B)
Metallic done
clear
C)
Dipolar done
clear
D)
Ionic done
clear
View Solution play_arrow
-
question_answer2)
For a crystal system, a = b = c, a = b = g ¹ 90o, the system is [BHU 2000]
A)
Tetragonal system done
clear
B)
Cubic system done
clear
C)
Orthorhombic system done
clear
D)
Rhombohedral system done
clear
View Solution play_arrow
-
question_answer3)
Biaxial crystal among the following is [Pb. CET 1998]
A)
Calcite done
clear
B)
Quartz done
clear
C)
Selenite done
clear
D)
Tourmaline done
clear
View Solution play_arrow
-
question_answer4)
The temperature coefficient of resistance of a conductor is [AFMC 1998]
A)
Positive always done
clear
B)
Negative always done
clear
C)
Zero done
clear
D)
Infinite done
clear
View Solution play_arrow
-
question_answer5)
Potassium has a bcc structure with nearest neighbour distance 4.525 Å. Its molecular weight is 39. Its density in kg/m3 is [DCE 1997]
A)
900 done
clear
B)
494 done
clear
C)
602 done
clear
D)
802 done
clear
View Solution play_arrow
-
question_answer6)
The expected energy of the electrons at absolute zero is called [RPET 1996]
A)
Fermi energy done
clear
B)
Emission energy done
clear
C)
Work function done
clear
D)
Potential energy done
clear
View Solution play_arrow
-
question_answer7)
In a triclinic crystal system [EAMCET (Med.) 1995]
A)
\[a\ne b\ne c\], \[\alpha \ne \beta \ne \gamma \] done
clear
B)
\[a=b=c\], \[\alpha \ne \beta \ne \gamma \] done
clear
C)
\[a\ne b\ne c\], \[\alpha \ne \beta =\gamma \] done
clear
D)
\[a=b\ne c\], \[\alpha =\beta =\gamma \] done
clear
View Solution play_arrow
-
question_answer8)
Metallic solids are always opaque because [AFMC 1994]
A)
Solids effect the incident light done
clear
B)
Incident light is readily absorbed by the free electron in a metal done
clear
C)
Incident light is scattered by solid molecules done
clear
D)
Energy band traps the incident light done
clear
View Solution play_arrow
-
question_answer9)
In which of the following ionic bond is present [EAMCET (Med.) 1994]
A)
NaCl done
clear
B)
Ar done
clear
C)
Si done
clear
D)
Ge done
clear
View Solution play_arrow
-
question_answer10)
Which of the following materials is non crystalline [CBSE PMT 1993]
A)
Copper done
clear
B)
Sodium chloride done
clear
C)
Wood done
clear
D)
Diamond done
clear
View Solution play_arrow
-
question_answer11)
The coordination number of \[Cu\] is [AMU 1992]
A)
1 done
clear
B)
6 done
clear
C)
8 done
clear
D)
12 done
clear
View Solution play_arrow
-
question_answer12)
Which one of the following is the weakest kind of bonding in solids [CBSE PMT 1992; KCET 1992]
A)
Ionic done
clear
B)
Metallic done
clear
C)
Vander Waals done
clear
D)
Covalent done
clear
View Solution play_arrow
-
question_answer13)
In a crystal, the atoms are located at the position of [AMU 1985]
A)
Maximum potential energy done
clear
B)
Minimum potential energy done
clear
C)
Zero potential energy done
clear
D)
Infinite potential energy done
clear
View Solution play_arrow
-
question_answer14)
Crystal structure of \[NaCl\] is [NCERT 1982]
A)
Fcc done
clear
B)
Bcc done
clear
C)
Both of the above done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer15)
What is the coordination number of sodium ions in the case of sodium chloride structure [CBSE PMT 1988]
A)
6 done
clear
B)
8 done
clear
C)
4 done
clear
D)
12 done
clear
View Solution play_arrow
-
question_answer16)
The distance between the body centred atom and a corner atom in sodium \[(a=4.225{AA})\] is [CBSE PMT 1995]
A)
\[3.66\,{AA}\] done
clear
B)
\[3.17\,{AA}\] done
clear
C)
\[2.99\,{AA}\] done
clear
D)
\[2.54\,{AA}\] done
clear
View Solution play_arrow
-
question_answer17)
A solid that transmits light in visible region and has a very low melting point possesses [J & K CET 2001]
A)
Metallic bonding done
clear
B)
Ionic bonding done
clear
C)
Covalent bonding done
clear
D)
Vander Waal?s bonding done
clear
View Solution play_arrow
-
question_answer18)
Atomic radius of fcc is [J & K CET 2001]
A)
\[\frac{a}{2}\] done
clear
B)
\[\frac{a}{2\sqrt{2}}\] done
clear
C)
\[\frac{\sqrt{3}}{4}a\] done
clear
D)
\[\frac{\sqrt{3}}{2}a\] done
clear
View Solution play_arrow
-
question_answer19)
A solid reflects incident light and it?s electrical conductivity decreases with temperature. The binding in this solids
A)
Ionic done
clear
B)
Covalent done
clear
C)
Metallic done
clear
D)
Molecular done
clear
View Solution play_arrow
-
question_answer20)
The laptop PC?s modern electronic watches and calculators use the following for display
A)
Single crystal done
clear
B)
Poly crystal done
clear
C)
Liquid crystal done
clear
D)
Semiconductors done
clear
View Solution play_arrow
-
question_answer21)
The nearest distance between two atoms in case of a bcc lattice is equal to [J & K CET 2004]
A)
\[a\frac{\sqrt{2}}{3}\] done
clear
B)
\[a\frac{\sqrt{3}}{2}\] done
clear
C)
\[q\sqrt{3}\] done
clear
D)
\[\frac{a}{\sqrt{2}}\] done
clear
View Solution play_arrow
-
question_answer22)
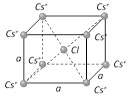
What is the net force on a Cl? placed at the centre of the bcc structure of CsCl [DCE 2003; AIIMS 2004]
A)
Zero done
clear
B)
\[k{{e}^{2}}/{{a}^{2}}\] done
clear
C)
\[k{{e}^{2}}{{a}^{2}}\] done
clear
D)
Data is incomplete done
clear
View Solution play_arrow
-
question_answer23)
Sodium has body centred packing. If the distance between two nearest atoms is \[3.7{AA}\], then its lattice parameter is [Pb. PET 2002]
A)
\[4.8\,{AA}\] done
clear
B)
\[4.3\,{AA}\] done
clear
C)
\[3.9\,{AA}\] done
clear
D)
\[23.3\,{AA}\] done
clear
View Solution play_arrow
-
question_answer24)
Which of the following is an amorphous solid [AIIMS 2005; J & K CET 2004]
A)
Glass done
clear
B)
Diamond done
clear
C)
Salt done
clear
D)
Sugar done
clear
View Solution play_arrow
-
question_answer25)
Copper has face centered cubic (fcc) lattice with interatomic spacing equal to \[2.54{AA}\]. The value of the lattice constant for this lattice is [CBSE PMT 2005]
A)
\[1.27\,{AA}\] done
clear
B)
\[5.08\,{AA}\] done
clear
C)
\[2.54\,{AA}\] done
clear
D)
\[3.59\,{AA}\] done
clear
View Solution play_arrow
-
question_answer26)
In good conductors of electricity, the type of bonding that exists is [CBSE PMT 1995]
A)
Ionic done
clear
B)
Vander Waals done
clear
C)
Covalent done
clear
D)
Metallic done
clear
View Solution play_arrow
-
question_answer27)
Bonding in a germanium crystal (semi- conductor) is [CPMT 1986; KCET 1992; EAMCET (Med.) 1995; MP PET/PMT 2004]
A)
Metallic done
clear
B)
Ionic done
clear
C)
Vander Waal's type done
clear
D)
Covalent done
clear
View Solution play_arrow
-
question_answer28)
The ionic bond is absent in [J & K CET 2005]
A)
NaCl done
clear
B)
CsCl done
clear
C)
LiF done
clear
D)
\[{{H}_{2}}O\] done
clear
View Solution play_arrow
