Answer:
Due
to -I and -R-effect of the - ![]() group,
the electron density in the O?H bond decreases relative to O?H group in phenol.
group,
the electron density in the O?H bond decreases relative to O?H group in phenol.
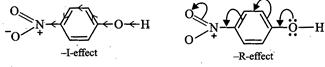
![]() As a result, p-nitrophenol is much more acidic than phenol.
As a result, p-nitrophenol is much more acidic than phenol.
You need to login to perform this action.
You will be redirected in
3 sec
