-
Find the
(a) maximum frequency, and
(b) minimum wavelength of X-rays produced by 30 kV electrons.
View Answer play_arrow
-
The work function of caesium metal is 2.14 eV. When light of frequency 6 x 1014 Hz is incident on the metal surface, photoemission
of electrons occurs. What is the
(a) maximum kinetic energy of the emitted electrons,
(b) stopping potential, and
(c) maximum speed of the emitted photoelectrons ?
View Answer play_arrow
-
The photoelectric cut-off voltage in a certain experiment is 1'5 V. What is the maximum kinetic energy of photoelectrons emitted ?
View Answer play_arrow
-
Monochromatic light of wavelength 632-8 nm is produced by a helium-neon laser. The power emitted is 9-42 mW.
(a) Find the energy and momentum of each photon in the light beam.
(b) How many photons per second, on the average, arrive at a target irradiated by this beam ? (Assume the beam to have uniform cross-section which is less than the target area), and
(c) How fast does a hydrogen atom have to travel in order to have the same momentum as that of the photon?
View Answer play_arrow
-
The energy flux of sunlight
reaching the surface of the earth is 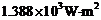 How many photons (nearly) per square metre are incident on
the Earth per second ? Assume that the photons in the sunlight have an average wavelength
of 550 nm
How many photons (nearly) per square metre are incident on
the Earth per second ? Assume that the photons in the sunlight have an average wavelength
of 550 nm
View Answer play_arrow
-
in an experiment
on photoelectric effect, the
slope of
the cut-off voltage versus frequency
of incident light
is found to be
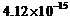
V s. Calculate the
value of Planck's constant
-
A 100 W sodium lamp radiates energy uniformly in all directions. The lamp is located at the centre of a large sphere that absorbs all the sodium light which is incident on it. The wavelength of the sodium light is 589 nm. (a) What is the energy per photon associated with the sodium light ? (b) At what rate are the photons delivered to the sphere ?
View Answer play_arrow
-
The threshold
frequency for a certain metal is
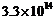
Hz. If light of
frequency 8.2 x 1014 Hz is incident on the metal, predict the cut-off
voltage for the photoelectric emission.
-
The work function for a certain metal is 4.2 eV. Will this metal give photoelectric emission for incident radiation of wavelength 330 nm ?
View Answer play_arrow
-
Light of
frequency
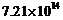
Hz is incident on a
metal surface. Electrons with a maximum speed of
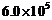
m/s are
ejected from the surface. What is the threshold frequency for photoemission of
electrons?
-
Light of wavelength 488 nm is produced by an argon laser which is used in the photoelectric effect. When light from this spectral line is incident on the cathode, the stopping (cut-off) potential of photoelectrons is 0.38 V. Find the work function of the material from which the cathode is made.
View Answer play_arrow
-
Calculate the
(a) momentum, and
(b) de Broglie wavelength of the electrons accelerated through a potential difference of 56 V.
View Answer play_arrow
-
What is the
(a) momentum, (b) speed, and
(c) de Broglie wavelength of an electron with
kinetic energy of 120 eV ?
View Answer play_arrow
-
The wavelength of light from the spectral emission line of sodium is 589 nm. Find the kinetic energy at which
(a) an electron, and
(b) a neutron, would have the same de Brogue wavelength.
View Answer play_arrow
-
What is the de
Broglie wavelength of
(a) a
bullet of mass 0.040 kg travelling at the speed of 1.0 km/s,
(b)
a ball of mass 0.060 kg moving at a speed of 1.0 m/s, and
(c) a dust
particle of mass
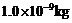
drifting with a speed
of 2.2 m/s ?
-
An electron and a photon each have a wavelength of 100 nm. Find
(a) their momenta,
(b) the energy of the photon, and
(c) the kinetic energy of electron.
View Answer play_arrow
-
(a For what
kinetic energy of a neutron will the associated de Broglie wavelength be
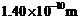
?
(b) Also
find the de Broglie wavelength of a neutron, in thermal equilibrium with matter,
having an average kinetic energy of
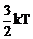
at 300 K.
-
Show that the wavelength of electromagnetic radiaion is equal to the de Broglie wavelength of its quantum (photon).
View Answer play_arrow
-
What is the de Broglie wavelength of a nitrogen molecule in air at 300 K ? Assume that the molecule is moving with the root mean square speed of molecules at this temperature.
(Atomic mass of nitrogen = 14-0076 u)
View Answer play_arrow
-
(a) Estimate the
speed with which electrons emitted from a heated cathode of an evacuated tube
impinge on the anode maintained at a potential difference of 500 V with respect
to the cathode. Ignore the small initial speeds of the electrons. The 'specific
charge? of the electron i.e., its el m is given to be
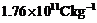
(b) Use
the same formula you employ in (a) to obtain electron speed for an anode
potential of 10 MV. Do you see what is wrong ? In what way is the formula to be
modified ?
-
(a) A
monoenergetic electron beam with electron speed of
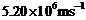
is subject
to a magnetic field of
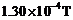
normal to the beam
velocity. What is the radius of the circle traced by the beam, given elm for
electron equals
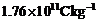
.
(b) Is the
formula you employ in (a) valid for calculating radius of the path of a 20 MeV electron
beam ? If not, in what way is it modified ?
-
An electron gun
with its anode at a potential of 100 V Ores out electrons in a spherical bulb
containing hydrogen gas at low pressure
(
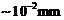
of Hg). A magnetic field of 2-83 x
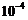
T curves the path of
the electrons in a circular orbit of radius 12*0 cm. (The path can be viewed
because the gas ions in the path focus the beam by attracting electrons, and emitting
light by electron capture; this method is known as the 'fine beam tube?
method.) Determine elm from the data.
-
(a) An X-ray tube produces a continuous spectrum of radiation with its short wavelength end at 0.45 Å. What is the maximum energy of a photon in the radiation ?
(b) From your answer to (a), guess what order of accelerating voltage (for electrons) is required in such a tube.
View Answer play_arrow
-
In an accelerator
experiment on high-energy collisions of electrons with positrons, a certain event
is interpreted as annihilation of an electron-positron pair of total energy
10.2 BeV into two y-rays of equal energy. What is the wavelength associated
with each
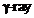
? (1 BeV = 109
eV)
-
Estimating the
following two numbers should be interesting. The first number will tell you why
radio engineers do not need to worry much about photons ! The second number
tells you why our eye can never 'count photons', even in barely detectable
light.
(a) The
number of photons emitted per second by a Medium wave transmitter of 10 kW power,
emitting radio waves of wavelength 500 m.
(b)
The number of photons entering the pupil
of our eye per
second corresponding to the minimum intensity of white light that we humans can
perceive
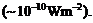
Take the area of the
pupil to be about 0.4 cm2, and the average frequency of white light
to be about 6 x 1014 Hz.
-
Ultraviolet light
of wavelength 2271 Å from a
100 W
mercury source irradiates a photo-cell
made of
molybdenum metal. If the stopping potential is 1.3 V, estimate the work
function of the metal. How would the photo-cell respond to a high intensity

red light of wavelength 6328 Å produced by a He-Ne laser ?
-
Monochromatic
radiation of wavelength 640.2 nm (
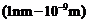
) from a neon lamp
irradiates photosensitive material made of caesium or tungsten. The stopping
voltage is measured to be 0.54 V. The source is replaced by an iron source and
its 427.2 nm line irradiates the same photo-cell. Predict the new stopping voltage.
-
A mercury lamp is
a convenient source for studying frequency dependence of photoelectric
emission, since it gives a number of spectral lines ranging from the UV to the red
end of the visible spectrum. In our experiment with rubidium photo-cell, the following
lines from a mercury source were used :
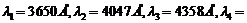
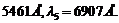
The
stopping voltages, respectively, were measured to be:
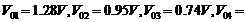
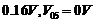
(a) Determine
the value of Planck's constant h.
(b)
Estimate the threshold frequency and work function for the material.
-
The work function for the following metals is
given :
Na : 2-75 eV ; K : 2-30 eV ; Mo : 4-17 eV ; Ni : 5-15 eV. Which of these metals will not give photoelectric emission for a radiation of wavelength 3300 Å from a He- Cd laser placed 1 m away from the photocell ?
What happens if the laser is brought nearer and placed 50 cm away ?
View Answer play_arrow
-
Light of intensity

falls
on a sodium photo-cell of surface area 2 cm2. Assuming that the top
5 layers of sodium absorb the incident energy, estimate time required for photoelectric
emission in the wave-picture of radiation. The work function for the metal is given
to be about 2eV. What is the implication of your answer?
-
Crystal
diffraction experiments can be performed using X-rays, or electrons accelerated
through appropriate voltage.
Which probe has
greater energy? (For quantitative comparison, take the wavelength of the probe
equal to 1 Å, which is of the order of inter-atomic spacing in the
lattice)
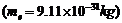
.
-
(a) Obtain
the de Broglie wavelength of a neutron of kinetic energy 150 e. As you have
seen in Exercise 44, an electron beam of this energy is suitable for crystal diffraction
experiments. Would a neutron beam of the same energy be equally suitable ?
Explain,
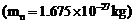
.
(b) Obtain the de
Broglie wavelength associated with thermal neutrons at room temperature (27°C).
Hence explain why a fast neutron beam needs to be thermalised with the
environment before it can be used for neutron diffraction experiments.
-
An electron microscope uses electrons accelerated by a voltage of 50 kV. Determine
the de Broglie wavelength associated with the electrons. If other factors (such as numerical aperture, etc.) are taken to be roughly the same, how does the resolving power of an electron microscope compare with that of an optical microscope which uses yellow light ?
View Answer play_arrow
-
The wavelength of a probe is roughly a measure of the size
of a structure that it can probe in some detail. The quark structure of protons
and neutrons appears at the minute length -scale of 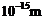 or less. This structure was first probed in early 1970's
using high energy electron beams produced by a linear accelerator at Stanford,
USA. Guess what might have been the order of energy of these electron beams.
(Rest mass energy of electron = 0-511 Me V).
or less. This structure was first probed in early 1970's
using high energy electron beams produced by a linear accelerator at Stanford,
USA. Guess what might have been the order of energy of these electron beams.
(Rest mass energy of electron = 0-511 Me V).
View Answer play_arrow
-
Find the typical de Broglie wavelength associated with a He atom in helium gas at room temperature (27°C) and 1 atm pressure ; and compare it with the mean separation between two atoms under these conditions.
View Answer play_arrow
-
Compute the
typical de Broglie wavelength of an electron in a metal at 27°C and compare it with
the mean separation between two electrons in a metal which is given to be about
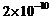
m.
-
Answer the
following questions :
(a) Quarks
inside protons and neutrons are thought to carry fractional charges [
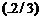
e;(-1/3)e].
Why do they not show up in *Millikan's oil-drop experiment ?
(b)
What is so special about the combination el m ? Why do we not simply talk of e
and m separately ?
(c) Why
should gases be insulators at ordinary pressures and start conducting at very
low pressures ?
(d)
Every metal has a definite work function.
Why do
all photoelectrons not come out with the same energy if incident radiation is
monochromatic ? Why is there an energy distribution of photoelectrons ?
(e) The
energy and momentum of an electron are related to the frequency and wavelength of
the associated matter wave by the relations :
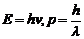
But while the value
of
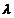
is physically significant,
the value of v (and therefore, the value of the phase speed
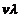
)
has no physical significance. Why?
-
question_answer38)
A particle is
dropped from a height H. The de Broglie wavelength of the particle as a function
of height is proportional to
(a)H
(b) H1/2
(c)H0
(d) H-1/2
View Answer play_arrow
-
question_answer39)
The wavelength of a
photon needed to remove a proton from a nucleus which is bound to the nucleus
with 1 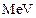 energy is nearly
(o)
1.2nm (b) 1.2 x 10-3 nm
(c)
1.2 x 10-6 nm (d) 1.2 x 101 nm
energy is nearly
(o)
1.2nm (b) 1.2 x 10-3 nm
(c)
1.2 x 10-6 nm (d) 1.2 x 101 nm
View Answer play_arrow
-
question_answer40)
Consider a beam of
electrons (each electron with energy E0) incident on a metal surface
kept in an evacuated chamber. Then
(a) no electrons will be emitted as only photons can emit
electrons
(b)
electrons can be emitted but all with an
energy,
E0
(c)
electrons can be emitted with any energy, with a maximum of E0 - 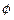 (
( 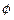 is
the work function)
(d)
electrons can be emitted with any energy, with a maximum of E0
is
the work function)
(d)
electrons can be emitted with any energy, with a maximum of E0
View Answer play_arrow
-
question_answer41)
Consider Fig. 7.14
in the Fundamental Physics for Class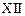 .
Suppose the voltage applied to A is increased. The diffracted beam will have
the maximum at a value of
.
Suppose the voltage applied to A is increased. The diffracted beam will have
the maximum at a value of 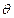 that
( a) will be larger than that earlier value
(b) will be the same as the earlier value
(c) will be less than the earlier value
(d) will depend on the target
that
( a) will be larger than that earlier value
(b) will be the same as the earlier value
(c) will be less than the earlier value
(d) will depend on the target
View Answer play_arrow
-
question_answer42)
A proton, a
neutron, an electron and an 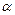 - particle
have same energy. Then their de Broglie wavelengths compare as
(a)
- particle
have same energy. Then their de Broglie wavelengths compare as
(a) 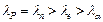 (b)
(b)
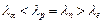 (c)
(c)
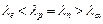 (d)
(d)
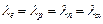
View Answer play_arrow
-
question_answer43)
An electron is
moving with an initial velocity 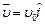 and is in
a magnetic field
and is in
a magnetic field 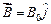 . Then it's de
Broglie wavelength
(a) remains constant
(b) increases with time
(c) decreases with time
(d) increases and decreases periodically
. Then it's de
Broglie wavelength
(a) remains constant
(b) increases with time
(c) decreases with time
(d) increases and decreases periodically
View Answer play_arrow
-
question_answer44)
An electron (mass
m) with an initial velocity 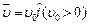 is in
electric field
is in
electric field 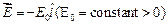 . It?s de Broglie
wavelength at time t is given by
(a)
. It?s de Broglie
wavelength at time t is given by
(a)  (b)
(b) 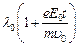 (c)
(c)
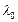 (d)
(d)
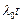
View Answer play_arrow
-
question_answer45)
An electron (mass
m) with an initial velocity 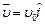 is in an
electric field
is in an
electric field 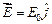 If
If 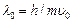 it?s de Brogue wavelength at
time is given by
(a)
it?s de Brogue wavelength at
time is given by
(a) 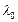 (b)
(b)
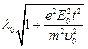 (c)
(c)
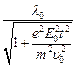 (d)
(d) 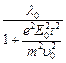
View Answer play_arrow
-
question_answer46)
Relativistic corrections
become necessary when the expression for the kinetic energy 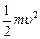 , becomes comparable with mc2,
where m is the mass of the particle. At what de Broglie wavelength will
relativistic corrections become important for an electron?
(a)
, becomes comparable with mc2,
where m is the mass of the particle. At what de Broglie wavelength will
relativistic corrections become important for an electron?
(a) 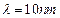 (b)
(b) 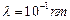 (c)
(c)
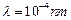 (d)
(d) 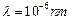
View Answer play_arrow
-
question_answer47)
Two particles A1
and A2 of masses m1, m2 (m1 > m2)
have the same de Broglie wave- length, Then
(a) their momenta are the same
(b )their energies are the same
(c) energy of A1 is less than the energy of A2
(d) energy
of A1 is more than the energy of A2
View Answer play_arrow
-
question_answer48)
The de Broglie
wavelength of a photon is twice the de Broglie wavelength of an electron. The
speed of the electron is 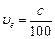 .Then
(a)
.Then
(a) 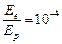 (b)
(b) 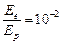 (c)
(c)
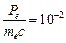 (d)
(d) 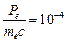
View Answer play_arrow
-
question_answer49)
Photons absorbed in
matter are converted to heat. A source emitting n photon/sec of frequency v is
used to convert 1 kg of ice at 0°C to water at 0°C. Then, the time T
taken for the conversion
(a) decreases with increasing n, with v fixed
(b) decreases with n fixed, v increasing
(c) remains constant with n and v changing such that 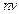 = constant
(d) increases when the product
= constant
(d) increases when the product 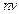 increases
increases
View Answer play_arrow
-
question_answer50)
A particle moves in a
closed orbit around the origin, due to a force which is directed towards the
origin. The de Broglie wavelength of the particle varies cyclically between two
values 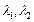 and
and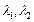 .Which
of the following statement are true?
(a) The particle could be moving in a circular orbit with
origin as centre
(b) The
particle could be moving in an elliptic orbit with origin as its focus
(c)
When the de Broglie wavelength is
.Which
of the following statement are true?
(a) The particle could be moving in a circular orbit with
origin as centre
(b) The
particle could be moving in an elliptic orbit with origin as its focus
(c)
When the de Broglie wavelength is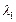 , the
particle is
nearer the origin than when its value is
, the
particle is
nearer the origin than when its value is 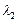 (d) When the de Broglie wavelength is
(d) When the de Broglie wavelength is 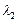 the particle is nearer the
origin than when its value is
the particle is nearer the
origin than when its value is 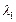
View Answer play_arrow
-
question_answer51)
A proton and an 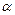 -particle are accelerated,
using the same potential difference. How are the de Broglie wavelengths Ay and
Ag related to each other?
-particle are accelerated,
using the same potential difference. How are the de Broglie wavelengths Ay and
Ag related to each other?
View Answer play_arrow
-
question_answer52)
(i) In the
explanation of photo electric effect, we assume one photon of frequency v
collides with an electron and transfers its energy. This leads to the equation
for the maximum energy 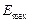 of the emitted
electron as
of the emitted
electron as 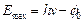 where
where 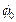 g is the work function of the
metal. If an electron absorbs 2 photons (each of frequency v ) what will be the
maximum energy for the emitted electron?
(ii)
Why is this fact (two photon absorption not taken into consideration in our
discussion of the stopping potential?
g is the work function of the
metal. If an electron absorbs 2 photons (each of frequency v ) what will be the
maximum energy for the emitted electron?
(ii)
Why is this fact (two photon absorption not taken into consideration in our
discussion of the stopping potential?
View Answer play_arrow
-
question_answer53)
There are materials
which absorb photons of shorter wavelength and emit photons of longer wavelength.
Can there be stable substances which absorb photons of larger wavelength and
emit light of shorter wavelength.
View Answer play_arrow
-
question_answer54)
Do all the
electrons that absorb a photon come out as photoelectrons?
View Answer play_arrow
-
question_answer55)
There are two
sources of light, each emitting with a power of 100 W. One emits X-rays of
wavelength 1 nm and the other visible light at 500 nm. Find the ratio of number
of photons of X-rays to the photons of visible light of the given wavelength?
View Answer play_arrow
-
question_answer56)
Consider Fig.
7(EP).2 for photoemission. How would you reconcile with momentum-conservation?
Note light (photons) have momentum in a different direction than the emitted
electrons.
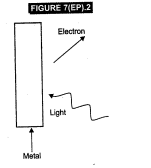
View Answer play_arrow
-
question_answer57)
Consider a metal
exposed to light of wavelength 600 nm. The maximum energy of the electron
doubles when light of wavelength 400 nm is used. Find the work function in eV.
View Answer play_arrow
-
question_answer58)
Assuming an electron
is confined to a 1 nm wide region, find the uncertainty in momentum using Heisenberg
Uncertainty principle (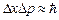 ). You can assume the
uncertainty in position
). You can assume the
uncertainty in position 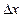 as 1 nm.
Assuming
as 1 nm.
Assuming 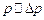 , find the energy of the
electron in electron volts.
, find the energy of the
electron in electron volts.
View Answer play_arrow
-
question_answer59)
Two monochromatic
beams A and B of equal intensity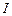 , hit a
screen. The number of photons hitting the screen by beam A is twice that by
beam B. Then what inference can you make about their frequencies?
i.e., Frequency of beam B is twice of beam A.
, hit a
screen. The number of photons hitting the screen by beam A is twice that by
beam B. Then what inference can you make about their frequencies?
i.e., Frequency of beam B is twice of beam A.
View Answer play_arrow
-
question_answer60)
Two particles A and
B of de Broglie wavelengths 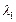 and
and 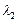 combine to form a particle
C. The process conserves momentum. Find the de Broglie wavelength of the particle
C. (The motion is one Dimensional).
combine to form a particle
C. The process conserves momentum. Find the de Broglie wavelength of the particle
C. (The motion is one Dimensional).
View Answer play_arrow
-
question_answer61)
A neutron beam of
energy E scatters from atoms on a surface with a spacing d = 0.1 nm. The first
maximum of intensity in the reflected beam occurs at 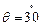 = . What is the kinetic
energy E of the beam in
= . What is the kinetic
energy E of the beam in 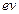 ?
?
View Answer play_arrow
-
question_answer62)
Consider a thin
target (10-2 m square, 10-3 m thickness) of sodium, which
produces a photocurrent of 100 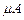 when a
light of intensity
when a
light of intensity 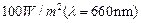 falls
on it. Find the probability that a photoelectron is produced when a photon
strikes a sodium atom. [Take density of Na = 0.97 kg/m3, Avogadro's
number = 6 x 1026 kg atom],
falls
on it. Find the probability that a photoelectron is produced when a photon
strikes a sodium atom. [Take density of Na = 0.97 kg/m3, Avogadro's
number = 6 x 1026 kg atom],
View Answer play_arrow
-
question_answer63)
Consider an
electron in front of metallic surface at a distance d (treated as an infinite
plane surface). Assume the force of attraction by the plate is given as 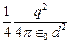 Calculate work in taking
the charge to an infinite distance from the plate. Taking d = 0.1 nm, find the work
done in electron volts. [Such a force law is not valid for
Calculate work in taking
the charge to an infinite distance from the plate. Taking d = 0.1 nm, find the work
done in electron volts. [Such a force law is not valid for 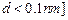 .
.
View Answer play_arrow
-
question_answer64)
A student performs
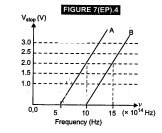
an experiment on photoelectric effect, using two materials A and B. A plot of 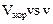 is given in Fig. 7(EP).4 (i)
Which material A or 5 has a higher work function? (ii) Given the electric
charge of an electron = 1.6 x 10-19 C, find the value of h obtained
from the experiment for both A and B. Comment on whether it is consistent with
Einstein's theory.
is given in Fig. 7(EP).4 (i)
Which material A or 5 has a higher work function? (ii) Given the electric
charge of an electron = 1.6 x 10-19 C, find the value of h obtained
from the experiment for both A and B. Comment on whether it is consistent with
Einstein's theory.
View Answer play_arrow
-
question_answer65)
A particle A with a
mass 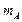 is moving with a velocity
is moving with a velocity 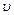 and hits a particle B (mass
and hits a particle B (mass
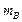 ) at rest (one dimensional
motion). Find the change in the de Broglie wavelength of the particle A. Treat
the collision as elastic.
) at rest (one dimensional
motion). Find the change in the de Broglie wavelength of the particle A. Treat
the collision as elastic.
View Answer play_arrow
-
question_answer66)
Consider a 20 W
bulb emitting light of wavelength 5000 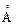 and
shinning on a metal surface kept at a distance 2 m. Assume that the metal surface
has work function of 2
and
shinning on a metal surface kept at a distance 2 m. Assume that the metal surface
has work function of 2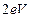 and
that each atom on the metal surface can be treated as a circular disk of radius
1.5
and
that each atom on the metal surface can be treated as a circular disk of radius
1.5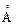 .
(i) Estimate no. of photons emitted by the bulb per
second. [Assume no other losses] (ii) Will there be photoelectric emission?
(ii'i) How much time would be required by the atomic disk to receive energy
equal to work function (
.
(i) Estimate no. of photons emitted by the bulb per
second. [Assume no other losses] (ii) Will there be photoelectric emission?
(ii'i) How much time would be required by the atomic disk to receive energy
equal to work function (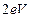 )? (iv)How
many photons would atomic disk receive within time duration calculated in (iii)
above? (v) Can you explain how photoelectric effect was observed
instantaneously? [Hint: Time calculated in part (iii) is from classical
consideration and you may further take the target of surface area say 1 cm2
and estimate what would happen ?]
)? (iv)How
many photons would atomic disk receive within time duration calculated in (iii)
above? (v) Can you explain how photoelectric effect was observed
instantaneously? [Hint: Time calculated in part (iii) is from classical
consideration and you may further take the target of surface area say 1 cm2
and estimate what would happen ?]
View Answer play_arrow
