Matter
Category : 5th Class
Matter
Matter
Matter i s anything that occupies space and has weight. It is classified into three categories. Solid, liquid and gas.
All matter can be split into tiny units that are not visible to the naked eye. These small units are called molecules.
Salt molecule is the smallest particle of salt.
Atom
A molecule has smaller units called atoms. Atoms are building blocks of matter. There are 117 kinds of atoms known to us. Atoms of same kind form elements. Atoms of different kinds combine and form compounds.
Molecules are constantly in a state of motion and are attracted towards each other. In solids, molecules are tightly packed.
In liquids, molecules are less closely packed compared to solids. In liquid, molecules can move freely and can flow. That is why a liquid has definite volume but no definite shape. In gases, the distance between molecules is large. The molecules in a gas move faster than the molecules in a solid or a liquid. Gas has no definite shape and volume.
Combination of States of Matter
States of matters may be combined as follows:
Solid in water
Sugar and salt are soluble in water. The molecules of these substances occupy the space between the molecules of water.
Liquids in water
When two liquids mix in each other, liquids are said to be miscible. Alcohol is miscible with water whereas kerosene is immiscible with water.
Gases in water
Carbon dioxide, nitrogen, oxygen and ammonia dissolve in water. Water in oceans and rivers contains dissolved oxygen and nitrogen.
When water is boiled, bubbles are formed of gases which are dissolved in water. Aerated drinks contain carbon dioxide.
Change of State
Matter changes its state with change in temperature and pressure. If we heat ice, it changes into water that is state changes from solid to liquid.
Physical change
A physical change is a temporary change in which no new substance is formed. Physical changes can also be reversed. For example,
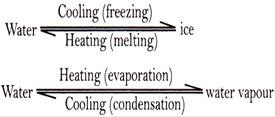
These changes involve change:
Chemical change
In a chemical change, on heating or cooling we get a new substance that cannot be changed into the old one. For example, burning of paper, rusting of iron, etc.
Chemical changes:
(i) are permanent.
(ii) cannot be reversed.
(iii) result in the formation of new substances.
You need to login to perform this action.
You will be redirected in
3 sec
