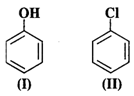
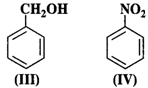
Answer:
An electrophile will always prefer
to attack centres of high electron density in the ring. Both nitro \[(-N{{O}_{2}})\]
and chloro (?Cl) groups are electron withdrawing groups. In benzyl alcohol
(III), the OH group is not directly attached to the ring and is not in a
position to activate it. Phenol (I) is the most reactive because phenolic group
activates the ring towards electrophilic attack both at the ortho and para
positions. For more details, consult section 12.16 (Chemical properties of
phenols).
You need to login to perform this action.
You will be redirected in
3 sec
