-
question_answer1)
Consider a beam of electrons (each electron with energy \[{{\operatorname{E}}_{0}}\]) incident on a metal surface kept in an evacuated chamber. Then,
A)
no electrons will be emitted as only photons can emit electrons. done
clear
B)
electrons can be emitted but all with an energy, \[{{\operatorname{E}}_{0}}\]. done
clear
C)
electrons can be emitted with any energy, with a maximum of \[{{\operatorname{E}}_{0}}\] - \[\phi \](\[\phi \] is the work function). done
clear
D)
electron can be omitted with energy, with a maximum of \[{{\operatorname{E}}_{0}}\]. done
clear
View Solution play_arrow
-
question_answer2)
The wavelength of a photon needed to remove a proton from a nucleus which is bound to the nucleus with 1 MeV energy is nearly
A)
1.2 nm done
clear
B)
1.2\[\times {{10}^{-3}}\]nm done
clear
C)
1.2 \[\times {{10}^{-6}}\] nm done
clear
D)
1.2 \[\times \] 10 nm done
clear
View Solution play_arrow
-
question_answer3)
The phenomenon which shows quantum nature of electromagnetic radiation is:
A)
photoelectric effect. done
clear
B)
tyndall effect. done
clear
C)
interference. done
clear
D)
reflection and refraction. done
clear
View Solution play_arrow
-
question_answer4)
Kinetic energy of electrons emitted in photoelectric effect is
A)
directly proportional to the intensity of incident light. done
clear
B)
inversely proportional to the intensity of incident line. done
clear
C)
independent of the intensity of incident light. done
clear
D)
independent of the frequency of light. done
clear
View Solution play_arrow
-
question_answer5)
Threshold wavelength of a photoelectric emission from a material is 600 nm. Which of the following illuminating source will emit photoelectrons?
A)
400 W, infrared lamp done
clear
B)
10 W, ultraviolet lamp done
clear
C)
100 W, ultraviolet lamp done
clear
D)
Both [b] & [c] done
clear
View Solution play_arrow
-
question_answer6)
Photoelectrons emitted from a metal have
A)
different speeds starting from 0 to certain maximum. done
clear
B)
same kinetic energy. done
clear
C)
same frequency. done
clear
D)
Both [b] & [c] done
clear
View Solution play_arrow
-
question_answer7)
At stopping potential, the kinetic energy of emitted photoelectron is
A)
minimum. done
clear
B)
maximum. done
clear
C)
zero. done
clear
D)
cannot de predicted done
clear
View Solution play_arrow
-
question_answer8)
Photons are
A)
electrically neutral and not deflected by electric or magnetic field. done
clear
B)
electrically neutral and deflected by magnetic field. done
clear
C)
electrically charged and not deflected by electric or magnetic field. done
clear
D)
electrically charged and not deflected by electric field. done
clear
View Solution play_arrow
-
question_answer9)
A particle is dropped from a height H. The de-Broglie wavelength of the particle as a function of height is proportional to
A)
H done
clear
B)
\[{{\operatorname{H}}^{1/2}}\] done
clear
C)
\[{{\operatorname{H}}^{o}}\] done
clear
D)
\[{{\operatorname{H}}^{-1/2}}\] done
clear
View Solution play_arrow
-
question_answer10)
A proton, a neutron, an electron and an a-particle have same energy. Then, their de-Broglie wavelengths compare as
A)
\[{{\lambda }_{p}}={{\lambda }_{n}}>{{\lambda }_{e}}>{{\lambda }_{a}}\] done
clear
B)
\[{{\lambda }_{a}}<{{\lambda }_{p}}={{\lambda }_{n}}>{{\lambda }_{e}}\] done
clear
C)
\[{{\lambda }_{e}}<{{\lambda }_{p}}={{\lambda }_{n}}>{{\lambda }_{a}}\] done
clear
D)
\[{{\lambda }_{e}}={{\lambda }_{p}}={{\lambda }_{n}}={{\lambda }_{a}}\] done
clear
View Solution play_arrow
-
question_answer11)
An electron is moving with an initial velocity \[v={{v}_{0}}\overset{}{\mathop{i}}\,\] and is in a magnetic field \[B={{B}_{0}}\overset{}{\mathop{j}}\,\]. Then, its de-Broglie wavelength
A)
remains constant. done
clear
B)
increases with time. done
clear
C)
decreases with time. done
clear
D)
increases and decreases periodically. done
clear
View Solution play_arrow
-
question_answer12)
An electron (mass m) with an initial velocity \[v\ \ v\ i\ ({{v}_{0}}>0)\] is in an electric field \[\operatorname{E}=\ -{{E}_{0}}\overset{}{\mathop{l}}\,\] (\[{{E}_{0}}\]=constant > 0). Its de-Broglie wavelength at time t is given by
A)
\[\frac{{{\lambda }_{0}}}{\left[ 1+\frac{e{{\operatorname{E}}_{0}}t}{m\ {{v}_{0}}} \right]}\] done
clear
B)
\[{{\lambda }_{0}}\left[ 1+\frac{e{{\operatorname{E}}_{0}}t}{m{{v}_{0}}} \right]\] done
clear
C)
\[{{\lambda }_{0}}\] done
clear
D)
\[{{\lambda }_{0}}t\] done
clear
View Solution play_arrow
-
question_answer13)
An electron (mass m) with an initial velocity \[v={{v}_{0}}\hat{l}\] is in an electric field \[E={{E}_{0}}\hat{j}\]. If \[{{\lambda }_{0}}=\frac{h}{m{{v}_{0}}}\] its de-Broglie wavelength at time t is given by
A)
\[{{\lambda }_{0}}\] done
clear
B)
\[{{\lambda }_{0}}\sqrt{1+\frac{{{e}^{2}}\operatorname{E}_{0}^{2}{{t}^{2}}}{{{m}^{2}}v_{0}^{2}}}\] done
clear
C)
\[\frac{{{\lambda }_{0}}}{\sqrt{1+\frac{{{e}^{2}}\operatorname{E}_{0}^{2}{{t}^{2}}}{{{m}^{2}}v_{0}^{2}}}}\] done
clear
D)
\[\frac{{{\lambda }_{0}}}{\left( 1+\frac{{{e}^{2}}\operatorname{E}_{0}^{2}{{t}^{2}}}{{{m}^{2}}v_{0}^{2}} \right)}\] done
clear
View Solution play_arrow
-
question_answer14)
The ratio of de-Broglie wavelength associated with two electrons accelerated through 25 V and 36 V is
A)
25/36 done
clear
B)
36/25 done
clear
C)
5/6 done
clear
D)
6/5 done
clear
View Solution play_arrow
-
question_answer15)
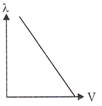
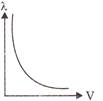
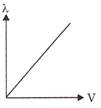
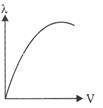
Which of the following graphs shows the variation of de-Broglie wavelength with potential through which a particle of charge q and mass m is accelerated?
A)
B)
C)
D)
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
