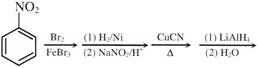
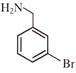
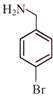
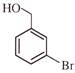
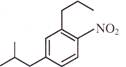
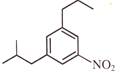
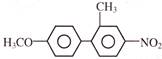
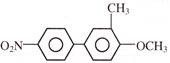
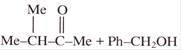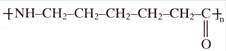Light falling on metallic surface of work function 2eV is given by \[B\text{ }=\text{ }100\text{ }\left[ sin\text{ }\left( 5\text{ }\times \text{ }{{10}^{15}} \right)\text{ }t\text{ }+\text{ }cos\text{ }\left( 8\text{ }\times \text{ }{{10}^{15}} \right)t \right]\]Then maximum kinetic energy of the photoelectrons is-
A)
1.6 Ev
done
clear
B)
2.4 eV
done
clear
C)
2.8eV
done
clear
D)
3.38eV
done
clear
View Answer play_arrow
An \[\alpha \]-particle and a deutron are moving with velocities v and 2v respectively. What will be the ratio of their de-Broglie wavelengths?
A)
1:1
done
clear
B)
\[\sqrt{2}:1\]
done
clear
C)
\[1:\text{ }\sqrt{2}\]
done
clear
D)
2:1
done
clear
View Answer play_arrow
A nucleus ruptures into two nuclear parts which have their velocity ratio equal to 2 : 1. What will be the ratio of their nuclear sizes?
A)
\[{{2}^{1/3}}\,:1\]
done
clear
B)
\[1:{{2}^{1/3}}\]
done
clear
C)
\[{{3}^{1/2}}:\text{ }1\]
done
clear
D)
\[1\text{ }:\text{ }{{3}^{1/2}}\]
done
clear
View Answer play_arrow
Two radioactive samples have decay constant 15 x and 3 x. If they have the same number of nuclei initially, the ratio of number of nuclei after a time \[\frac{1}{6x}\] is -
A)
\[\frac{1}{e}\]
done
clear
B)
\[\frac{e}{2}\]
done
clear
C)
\[\frac{1}{{{e}^{4}}}\]
done
clear
D)
\[\frac{1}{{{e}^{2}}}\]
done
clear
View Answer play_arrow
In a common emitter amplifier, using output resistance of 5000 ohm and input resistance of 2000 ohm, if the input signal voltage is 10 mV and\[P\text{ }=\text{ }50\], calculate output voltage & power gain
A)
1.25V, 6250
done
clear
B)
3V, 6250
done
clear
C)
1.5V, 3050
done
clear
D)
None of these
done
clear
View Answer play_arrow
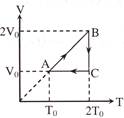
An ideal mono-atomic gas undergoes a cyclic process ABCA as shown in the figure. The ratio of heat absorbed during AB to the work done on the gas during BC is -
A)
\[\frac{5}{2\,In\,2}\]
done
clear
B)
\[\frac{5}{3}\]
done
clear
C)
\[\frac{5}{4\,In\,2}\]
done
clear
D)
\[\frac{5}{6}\]
done
clear
View Answer play_arrow
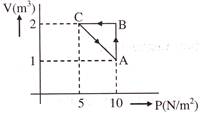
An ideal gas is taken through the cycle\[A\to B\to C\to A\], as shown in the figure. If the net heat supplied to the gas in the cycle is 5 J, the work done by the gas in the process \[C\to A\]
A)
-5 J
done
clear
B)
-10 J
done
clear
C)
-15 J
done
clear
D)
-20 J
done
clear
View Answer play_arrow
Assuming the Sun to have a spherical outer surface of radius r radiating like a black body at temperature\[t{}^\circ C\], the power received by a unit surface, (normal to the incident rays) at a distance R from the centre of the Sun is. (\[\sigma \]is Stefan constant) -
A)
\[\frac{4\pi {{r}^{2}}{{\sigma }^{4}}}{{{R}^{2}}}\]
done
clear
B)
\[\frac{{{r}^{2}}\sigma {{(t+273)}^{4}}}{4\pi {{R}^{2}}}\]
done
clear
C)
\[\frac{16{{\pi }^{2}}{{r}^{2}}\sigma t}{{{R}^{2}}}\]
done
clear
D)
\[\frac{{{r}^{2}}\sigma {{(t+273)}^{4}}}{{{R}^{2}}}\]
done
clear
View Answer play_arrow
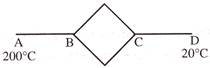
Six identical conducting rods are joined as shown in figure. Points A and D are maintained at temperatures \[200{}^\circ C\] and \[20{}^\circ \text{ }C\] respectively. The temperature of junction B will be -
A)
\[120{}^\circ C\]
done
clear
B)
\[100{}^\circ C\]
done
clear
C)
\[140{}^\circ C\]
done
clear
D)
\[\text{ }80{}^\circ C\]
done
clear
View Answer play_arrow
A closed organ pipe has length '\[\ell \]'. The air in it is vibrating in 3rd overtone with maximum amplitude 'a'. The amplitude at a distance off in from closed end of the pipe is equal to -
A)
a
done
clear
B)
\[\frac{a}{2}\]
done
clear
C)
\[\frac{a\sqrt{3}}{2}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
The speed of propagation of a wave in a medium is 300\[m{{s}^{-1}}\]. The equation of motion of point at \[x\text{ }=\text{ }0\] is given by \[y=0.04\] sin \[600\pi t\] (metre). The displacement of a point \[x=75\] cm at \[t\text{ }=\text{ }0.01\] s is-
A)
0.02 m
done
clear
B)
0.04 m
done
clear
C)
Zero
done
clear
D)
0.028 m
done
clear
View Answer play_arrow
Two identical sonometer wires have fundamental frequencies of 500 Hz, when kept under the same tension. What fractional increase in tension of one wire would cause an Occurrence of 5 beats per sec. When both wires vibrate together -
A)
1.020
done
clear
B)
0.020
done
clear
C)
1.20
done
clear
D)
0.20
done
clear
View Answer play_arrow
Two whistles A and B produce notes of frequencies 660 Hz and 596 Hz, respectively. There is a listener at the midpoint of the line joining them. Both the whistle B and the listener start moving with speed 30 m/s away from whistle A. If the speed of sound is 330 m/s, how many beats will be heard by the listener?
A)
2
done
clear
B)
4
done
clear
C)
6
done
clear
D)
8
done
clear
View Answer play_arrow
A body is displaced from \[\left( 0,\text{ }0 \right)\text{ }to\text{ }\left( 1\text{ }m,\text{ }1\text{ }m \right)\]along the path \[x\text{ }=\text{ }y\] by a force\[\overrightarrow{F}=\left( {{x}^{2}}\widehat{j}+\text{ }y \right)N\]. The work done by this force will be -
A)
\[\frac{4}{3}J\]
done
clear
B)
\[\frac{5}{6}J\]
done
clear
C)
\[\frac{3}{2}J\]
done
clear
D)
\[\frac{7}{5}J\]
done
clear
View Answer play_arrow
A person with a mass of M kg stands in contact against the wall of the cylindrical drum of radius r rotating with an angular velocity co. The coefficient of friction between the wall and the clothing is\[\mu \]. The minimum rotational speed of the cylinder which enables the person to remain stuck to the wall when the floor is suddenly removed is -
A)
\[{{\omega }_{\min }}=\sqrt{\frac{g}{\mu r}}\]
done
clear
B)
\[{{\omega }_{\min }}=\sqrt{\frac{\mu r}{g}}\]
done
clear
C)
\[{{\omega }_{\min }}=\sqrt{\frac{2g}{\mu r}}\]
done
clear
D)
\[{{\omega }_{\min }}=\sqrt{\frac{gr}{\mu }}\]
done
clear
View Answer play_arrow
A ball of mass m approaches a wall of mass \[M\text{ }\left( >>m \right)\] with speed 4 m/s along the normal to the wall. The speed of wall is 1 m/s towards the ball. The speed of the ball after an elastic collision with the wall is -
A)
5 m/s away from the wall
done
clear
B)
9 m/s away from the wall
done
clear
C)
3 m/s away from the wall
done
clear
D)
6 m/s away from the wall
done
clear
View Answer play_arrow
A body of radius R and mass m is rolling horizontally without slipping with speed v. It then rolls up a hill to a maximum height\[h=\frac{3{{v}^{2}}}{4g}\]. The body might be a -
A)
solid sphere
done
clear
B)
hollow sphere
done
clear
C)
disc
done
clear
D)
ring
done
clear
View Answer play_arrow
A body is projected up with a velocity equal to 3/4th of the escape velocity from the surface of the earth. The height it reaches from the centre of the earth is: (Radius of the earth = R):
A)
\[\frac{10R}{9}\]
done
clear
B)
\[\frac{9\,R}{7}\]
done
clear
C)
\[\frac{9\,R}{8}\]
done
clear
D)
\[\frac{10\,R}{3}\]
done
clear
View Answer play_arrow
A particle executes SHM of amplitude 5 cm and period 3s. The velocity of the particle at a distance 4 cm from the mean position-
A)
8 cm/s
done
clear
B)
12 cm/s
done
clear
C)
4 cm/s
done
clear
D)
6 m/s
done
clear
View Answer play_arrow
A soap bubble of radius r is blown upto form a bubble of radius 2r under isothermal conditions. If T is the surface tension of soap solution, the energy spent in the blowing is -
A)
\[3\pi T{{r}^{2}}\,joule\]
done
clear
B)
\[6\pi T{{r}^{2}}\,joule\]
done
clear
C)
\[12\pi T{{r}^{2}}\,joule\]
done
clear
D)
\[24\pi T{{r}^{2}}\,joule\]
done
clear
View Answer play_arrow
One end of an elastic string is attached to a block of mass m and the other end is held keeping the string relaxed and vertical. Relaxed length of the string is \[{{\ell }_{0}}\] and there is a mark P at height 0.8 \[{{\ell }_{0}}\] from the lower end. Now the upper end is slowly raised. When the block leaves the ground, the mark P reaches the point where upper end of the relaxed string was. Expression for the work done by the force applied by the hand is -
A)
\[\frac{mg{{\ell }_{0}}}{4}\]
done
clear
B)
\[\frac{mg{{\ell }_{0}}}{8}\]
done
clear
C)
\[\frac{mg{{\ell }_{0}}}{16}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A cube of iron whose sides are of length L, is put into mercury. The weight of iron cube is W. The density of iron is pp that of mercury is\[{{\rho }_{M}}\]. The depth to which the cube sinks is given by the expression -
A)
\[W{{L}^{2}}{{\rho }_{I}}\]
done
clear
B)
\[W{{L}^{2}}{{\rho }_{M}}\]
done
clear
C)
\[\frac{W}{{{L}^{2}}{{\rho }_{I}}}\]
done
clear
D)
\[\frac{W}{{{L}^{2}}{{\rho }_{M}}g}\]
done
clear
View Answer play_arrow
A block starts moving up a fixed inclined plane of inclination \[60{}^\circ \] with a velocity of 20 m/s and stops after 2 sec. The approximate value of coefficient of friction is - \[\left( g=10\text{ }m/{{s}^{2}} \right)\]
A)
3
done
clear
B)
3.3
done
clear
C)
0.27
done
clear
D)
0.33
done
clear
View Answer play_arrow
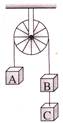
Three equal weights A, B and C of mass 2 kg each are hanging on a string passing over a fixed frictionless pulley as shown in the figure The tension in the string connecting weights B and C is-
A)
Zero
done
clear
B)
13 N
done
clear
C)
3.3 N
done
clear
D)
19.6 N
done
clear
View Answer play_arrow
At the height 80 m, an aeroplane is moving with 150 m/s. A bomb is dropped from it so as to hit a target. At what distance from the target should the bomb be dropped - (given \[g=10\text{ }m/{{s}^{2}}\])
A)
605.3 m
done
clear
B)
600 m
done
clear
C)
80 m
done
clear
D)
230 m
done
clear
View Answer play_arrow
A ball is dropped from the 9th storey of a multi-storeyed building reaches the ground in 3 second. In the first second of its free fall, it passes through n storeys, where n is equal to - (Take\[g=10\text{ }m\text{ }{{s}^{-}}^{2}\])
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
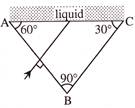
Light incident normally on face AB of a prism as shown in figure. A liquid of refractive index \[\mu \] is placed on face AC of the prism. The prism is made of glass of refractive index 3/2. The limits of p, for which total internal reflection takes place on face AC is -
A)
\[\mu >\frac{\sqrt{3}}{2}\]
done
clear
B)
\[\mu <\frac{3\sqrt{3}}{4}\]
done
clear
C)
\[\mu >\sqrt{3}\]
done
clear
D)
\[\mu <\frac{\sqrt{3}}{2}\]
done
clear
View Answer play_arrow
The focal length of a lens depends on -
A)
the radii of curvature of its surfaces
done
clear
B)
the refractive index of its material
done
clear
C)
the refractive index of the medium surrounding the lens
done
clear
D)
all the above factors
done
clear
View Answer play_arrow
An equi-convex lens has power P. It is cut into two symmetrical halves by a plane containing the principal axis and then two pieces are joined as shown in Fig. The power of the combination will be -
A)
P
done
clear
B)
2P
done
clear
C)
P/2
done
clear
D)
zero
done
clear
View Answer play_arrow
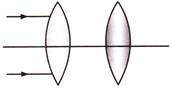
Parallel beam of light is incident on the system of two convex lenses of focal length \[{{f}_{1}}=20\] cm and \[{{t}_{2}}=10\] cm. What should be the distance between the two lenses so that rays after refraction from both the lenses pass undeviated-
A)
60 cm
done
clear
B)
30 cm
done
clear
C)
90 cm
done
clear
D)
40 cm
done
clear
View Answer play_arrow
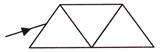
Three right angled isosceles prisms A, B and C of the same material are placed in contact with each other as shown in figure. When a ray of light passes through a single prism, if suffers deviation p. What will be the angle of deviation for same ray if it passes through the combination?
A)
less than P
done
clear
B)
more than p
done
clear
C)
P
done
clear
D)
None of these
done
clear
View Answer play_arrow
Two monochromatic waves each of intensity I have a constant phase difference of\[\phi \]. If these waves superpose, then the intensity of the resultant wave is -
A)
4I
done
clear
B)
\[4I\text{ }cos\text{ }\phi \]
done
clear
C)
\[4I\text{ }co{{s}^{2}}\,\phi \]
done
clear
D)
\[4I\text{ }co{{s}^{2}}\,(\phi \,2)\]
done
clear
View Answer play_arrow
A student has measured the length of a wire equal to 0.04580 m. This value of length has the number of significant figures equal to -
A)
Five
done
clear
B)
Four
done
clear
C)
Six
done
clear
D)
None of these
done
clear
View Answer play_arrow
Some charge is being given to a conductor, then its potential is -
A)
maximum at surface
done
clear
B)
maximum at centre
done
clear
C)
same throughout the conductor
done
clear
D)
maximum somewhere between surface and centre
done
clear
View Answer play_arrow
64 small drops of mercury, each of radius r and charge q, coalesce to form a big drop. The ratio of the surface density of charge of each small drop with that of the big drop is -
A)
64 : 1
done
clear
B)
1 : 64
done
clear
C)
1 : 4
done
clear
D)
4 : 1
done
clear
View Answer play_arrow
Two batteries, one of e.m.f. 18 volts and internal resistance 2\[\Omega \], and the other of e.m.f. 12 volts and internal resistance 1\[\Omega \], are connected as shown in the figure. The voltmeter V will record a reading of -
A)
30 volt
done
clear
B)
18 volts
done
clear
C)
15 volt
done
clear
D)
14 volts
done
clear
View Answer play_arrow
If the potential difference across the internal resistance \[{{r}_{1}}\] is equal to the e.m.f. E of the battery, then -
A)
\[R\text{ }=\text{ }{{r}_{1}}+{{r}_{2}}\]
done
clear
B)
\[R\text{ }=\text{ }{{r}_{1}}/{{r}_{2}}\]
done
clear
C)
\[R\text{ }=\text{ }{{r}_{1}}-{{r}_{2}}~\]
done
clear
D)
\[R\text{ }=\text{ }{{r}_{2}}/{{r}_{1}}\]
done
clear
View Answer play_arrow
A parallel plate capacitor carries a charge q. The distance between the plates is doubled by application of a force. The work done by the force is -
A)
zero
done
clear
B)
\[{{q}^{2}}/C\]
done
clear
C)
\[{{q}^{2}}/2C\]
done
clear
D)
\[{{q}^{2}}/4C\]
done
clear
View Answer play_arrow
A 10 uF capacitor is charged to 500 V and then its plates are joined together through a resistance of 10 ohm. The heat produced in the resistance is -
A)
500 J
done
clear
B)
250 J
done
clear
C)
125 J
done
clear
D)
1.25 J
done
clear
View Answer play_arrow
Two similar coils of radius R and number of turns N are lying concentrically with their planes at right angles to each other. The currents flowing in them are I and 21 respectively. The resultant magnetic induction at the centre will be
A)
\[\frac{{{\mu }_{0}}NI}{R}\]
done
clear
B)
\[\frac{{{\mu }_{0}}NI}{2R}\]
done
clear
C)
\[\frac{\sqrt{3}{{\mu }_{0}}NI}{2R}\]
done
clear
D)
\[\frac{\sqrt{5}\,{{\mu }_{0}}NI}{2R}\]
done
clear
View Answer play_arrow
Two circular coils made of similar wires but of radius 20 cm and 40 cm are connected in parallel. The ratio of magnetic fields at their centre is -
A)
4 : 1
done
clear
B)
1 : 4
done
clear
C)
2 : 1
done
clear
D)
1 : 2
done
clear
View Answer play_arrow
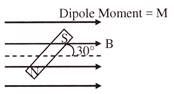
The potential energy of magnet is:
A)
\[U=-\frac{MB}{2}\]
done
clear
B)
\[U=-\sqrt{3}\,\,\,\frac{MB}{2}\]
done
clear
C)
\[U\,\,=\,\,\frac{MB}{2}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
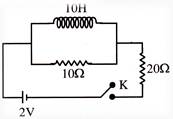
Two resistors of \[10\,\Omega \] and \[20\,\Omega \] and an ideal inductor of 10H are connected to a 2 V battery as shown. The key K is inserted at time\[t\text{ }=\text{ }0\]. The initial (\[t\text{ }=\text{ }0\] ) and final \[(t\text{ }\to \text{ }\infty )\] currents through battery are -
A)
\[\frac{1}{15}A,\,\frac{1}{10}A\]
done
clear
B)
\[\frac{1}{10}A,\,\frac{1}{15}A\]
done
clear
C)
\[\frac{2}{15}A,\,\frac{1}{10}A\]
done
clear
D)
\[\frac{1}{15}A,\,\frac{2}{25}A\]
done
clear
View Answer play_arrow
The following truth table corresponds to the logic gate -
A B X 0 0 0 0 1 1 1 0 1 1 1 1
A)
NAND
done
clear
B)
AND
done
clear
C)
XOR
done
clear
D)
OR
done
clear
View Answer play_arrow
A silicon specimen is made into a p-type semiconductor by doping on an average one indium atom per \[5\times {{10}^{7}}\] silicon atoms. If the number density of atoms in the silicon specimen is \[5\times {{10}^{26}}\] atoms/metre3 then the number of acceptor atoms in silicon per cubic centimeter will be -
A)
\[2.5\times {{10}^{30}}\]
done
clear
B)
\[1.0\times {{10}^{13}}\]
done
clear
C)
\[1.0\times {{10}^{15}}\]
done
clear
D)
\[2.5\times {{10}^{36}}\]
done
clear
View Answer play_arrow
The simplest formula of the compound containing \[50\,%\] X (atomic mass 10 amu) and \[50\,%\] Y (atomic mass 20 amu) is:
A)
\[X{{Y}_{2}}\]
done
clear
B)
\[{{X}_{2}}Y\]
done
clear
C)
\[{{X}_{2}}{{Y}_{3}}\]
done
clear
D)
\[X{{Y}_{3}}\]
done
clear
View Answer play_arrow
If the principal quantum number is 3, the azimuthal quantum number can have values -
A)
1, 2, 3
done
clear
B)
3, 2, 1, 0-1, -2, -3
done
clear
C)
0, 1, 2
done
clear
D)
\[\frac{1}{2},\,\,-\frac{1}{2}\]
done
clear
View Answer play_arrow
Oxidation state of nitrogen is incorrectly given in -
A)
\[{{N}_{2}}{{H}_{4}},\,\left( -2 \right)\]
done
clear
B)
\[AIN,\left( -\,3 \right)\]
done
clear
C)
\[{{\left( {{N}_{2}}{{H}_{5}} \right)}_{2}}S{{O}_{4}},\,\left( +2 \right)\]
done
clear
D)
\[KN{{O}_{2}},\left( +\,3 \right)\]
done
clear
View Answer play_arrow
One mole of \[{{N}_{2}}{{O}_{4}}(g)\] at 300 K is kept in a closed container at 1 atm. It is heated to 600 K when \[20\,%\] by mass of \[{{N}_{2}}{{O}_{4}}(g)\] decomposes to\[N{{O}_{2}}\left( g \right)\]. The resultant pressure is -
A)
1.2 atm
done
clear
B)
2.0 atm
done
clear
C)
2.4 atm
done
clear
D)
1.0 atom
done
clear
View Answer play_arrow
The property of colloid is
A)
Scattering of light
done
clear
B)
settling under gravity
done
clear
C)
Dialysis
done
clear
D)
Emulsion
done
clear
View Answer play_arrow
Which gives \[{{N}_{2}}\] on heating -
A)
\[N{{H}_{4}}N{{O}_{3}}\]
done
clear
B)
\[N{{H}_{4}}N{{O}_{2}}\]
done
clear
C)
\[Na{{N}_{3}}\]
done
clear
D)
[b], [c] both
done
clear
View Answer play_arrow
Which among the following species does not have bond angles of \[90{}^\circ \] -
A)
\[\left[ Ni{{\left( CO \right)}_{4}} \right]\]
done
clear
B)
\[\left[ Fe{{\left( CO \right)}_{5}} \right]\]
done
clear
C)
\[S{{F}_{6}}\]
done
clear
D)
\[Xe{{F}_{4}}\]
done
clear
View Answer play_arrow
Which among the following possess only ionic bond -
A)
\[{{B}_{4}}C\]
done
clear
B)
WC
done
clear
C)
\[A{{l}_{4}}{{C}_{3}}\]
done
clear
D)
\[\text{ }M{{g}_{2}}{{C}_{3}}\]
done
clear
View Answer play_arrow
Which of the following statement is correct for\[CsB{{r}_{3}}\]:
A)
It is a covalent compound
done
clear
B)
It contains \[C{{s}^{3+}}\] and \[B{{r}^{-}}\] ions
done
clear
C)
It contains \[C{{s}^{+}}\] and \[B{{r}^{-}}_{3}\] ions
done
clear
D)
It contains\[C{{s}^{+}}\], \[B{{r}^{-}}\] and lattice \[B{{r}_{2}}\] molecule
done
clear
View Answer play_arrow
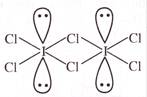
What is wrong about \[{{I}_{2}}C{{l}_{6}}\] molecule -
A)
It is a planar molecule
done
clear
B)
It is dimmer of \[IC{{l}_{3}}\]
done
clear
C)
Iodine is \[s{{p}^{3}}{{d}^{2}}\] hybridised
done
clear
D)
It is a polar molecule
done
clear
View Answer play_arrow
Pick out the incorrect order of given properties-
A)
Electronegativity -\[P<S<N<O\]
done
clear
B)
Stability - \[-\,{{O}_{2}}<{{O}_{2}}^{-}<{{O}^{2-}}_{2}<\,{{O}^{\,2+}}_{2}\]
done
clear
C)
Dipole moment - \[\,P{{H}_{3}}<As{{H}_{3}}<Sb{{H}_{3}}<N{{H}_{3}}\]
done
clear
D)
Vander wall radius - \[Ne<F<O<N\]
done
clear
View Answer play_arrow
Hybridisation at the central atom of the following molecules are respectively. \[C{{O}_{3}}^{-2},\text{ }B{{F}_{4}}^{-},\text{ }{{I}_{3}}^{-},\,\,CI{{F}_{5}}\]
A)
\[s{{p}^{2}},\,s{{p}^{3}},\,s{{p}^{3}}{{d}^{2}},\,s{{p}^{3}}d\]
done
clear
B)
\[s{{p}^{2}},\,s{{p}^{2}}s{{p}^{3}}d,\,s{{p}^{3}}{{d}^{2}}\]
done
clear
C)
\[s{{p}^{2}},\,s{{p}^{3}},\,s{{p}^{3}}d,\,\,s{{p}^{3}}{{d}^{2}}\]
done
clear
D)
\[s{{p}^{2}},s{{p}^{3}},s{{p}^{3}}{{d}^{3}},s{{p}^{3}}d\]
done
clear
View Answer play_arrow
Which of the following statement is incorrect -
A)
The stability constant of \[{{\left[ Cu{{\left( N{{H}_{3}} \right)}_{4}} \right]}^{2+}}\] is greater than \[{{\left[ Cu{{(en)}_{2}} \right]}^{2+}}\]
done
clear
B)
\[\left[ Ag{{\left( N{{H}_{3}} \right)}_{2}} \right]\,\,\,\text{ }\left[ Ag{{\left( CN \right)}_{2}} \right]\] has only one coordination isomer
done
clear
C)
It \[{{\Delta }_{0}}\,<\,P.E.\] then 4th electron enters in eg. Set
done
clear
D)
Cis-platin is a heteroleptic complex
done
clear
View Answer play_arrow
Coordination number and oxidation number of Cr in \[{{K}_{3}}\left[ Cr{{({{C}_{2}}{{O}_{4}})}_{3}} \right]\] are respectively?
A)
4 and +2
done
clear
B)
6 and +3
done
clear
C)
3 and +3
done
clear
D)
3 and 0
done
clear
View Answer play_arrow
Pick out the wrong statement about wilkinson?s catalyst -
A)
It is \[\pi \] bonded organometallic compound
done
clear
B)
It is \[\left[ RhCl{{\left( PP{{h}_{3}} \right)}_{3}} \right]\]
done
clear
C)
Central metal ion is \[ds{{p}^{2}}\] hybridised
done
clear
D)
It is low spin complex
done
clear
View Answer play_arrow
The momentum of a particle associated with de-Broglie?s wavelength of \[6\text{ }\overset{{}^\circ }{\mathop{A}}\,\] is-
A)
\[~1.1\,\,\times \,\,{{10}^{-24}}\,kg\,m\,{{s}^{\,-\,1}}\]
done
clear
B)
(b)\[~1.1\,\,\times \,\,{{10}^{34}}\,kg\,m\,{{s}^{\,-\,1}}\]
done
clear
C)
(c)\[~39.6\,\,\times \,\,{{10}^{-34}}\,kg\,m\,{{s}^{\,-\,1}}\]
done
clear
D)
\[~39.6\,\,\times \,\,{{10}^{-24}}\,kg\,m\,{{s}^{\,-\,1}}\]
done
clear
View Answer play_arrow
CsBr crystallizes in a body centred cubic lattice. The unit cell length is 436.6 pm. Given that the atomic mass of \[Cs=\text{ }133\] and of \[Br\text{ }=\text{ }80\] amu and Avogadro?s number being\[6.02\times {{10}^{23}}mo{{l}^{-1}}\], the density of CsBr is:
A)
8.50 g/\[c{{m}^{3}}\]
done
clear
B)
4.25 g/\[c{{m}^{3}}\]
done
clear
C)
42.5 g/\[c{{m}^{3}}\]
done
clear
D)
0.425 g/\[c{{m}^{3}}\]
done
clear
View Answer play_arrow
Match the following:
I II [A] Calcination (1) \[2C{{u}_{2}}S\,+\,3{{O}_{2}}\,\,\to \]\[\,\,2C{{u}_{2}}O\,+\,2S{{O}_{2}}\] [B] Roasting (2) \[F{{e}_{2}}{{O}_{3}}.n{{H}_{2}}O\,\,\to \] \[F{{e}_{2}}{{O}_{3}}+n{{H}_{2}}O\,\,\] [C] Flux (3) \[C{{r}_{2}}{{O}_{3}}+2Al\,\,\to \,\,\]\[A{{l}_{2}}{{O}_{3}}+2Cr\] [D] Thermite (4) \[Si{{O}_{2}}+\,FeO\,\to \,FeSi{{O}_{3}}\]
A)
A - 1 B - 2 C - 3 D - 4
done
clear
B)
A - 2 B - 1 C - 4 D - 3
done
clear
C)
A - 4 B - 1 C - 2 D - 3
done
clear
D)
A - 3 B - 1 C - 2 D - 4
done
clear
View Answer play_arrow
Which of the following rules could explain the presence of three unpaired electrons in N-atom-
A)
Hund's rule
done
clear
B)
Aufbau's principle
done
clear
C)
Heisenberg's uncertainty principle
done
clear
D)
Pauli's exclusion principle
done
clear
View Answer play_arrow
The molal elevation constant of water = 0.52 K\[molalit{{y}^{-1}}\]. The boiling point of 1.0 molal aqueous \[KCl\] solution (assuming complete dissociation of\[KCl\]), should be -
A)
\[100.52{}^\circ C\]
done
clear
B)
\[101.04{}^\circ C\]
done
clear
C)
\[99.48{}^\circ C\]
done
clear
D)
\[98.96{}^\circ C\]
done
clear
View Answer play_arrow
2 mole of ideal gas at \[27{}^\circ C\] temp. is expanded reversibly from 2 lit to 20 lit. Find entropy change in cal -
A)
92.1
done
clear
B)
0
done
clear
C)
4
done
clear
D)
9.2
done
clear
View Answer play_arrow
Which of the following statement is incorrect -
A)
Activation energy for the forward reaction is always equal to activation energy for the reverse reaction
done
clear
B)
An increase in temperature increases the reaction rate
done
clear
C)
The larger the initial reactant concentration for a second order reaction the shorter its half life
done
clear
D)
When \[\Delta t\] is infinitesimally small, the average rate equals the instantaneous rate
done
clear
View Answer play_arrow
Three test tubes contain aqueous solutions as under
I. KCN II. \[Fe{{\left( CN \right)}_{2}}\] III: Mixture of KCN, \[Fe{{\left( CN \right)}_{2}}\] in molar ratio 4:1 pick up the correct statement
A)
Both I and III will give test for \[C{{N}^{-}}\]
done
clear
B)
Both II and III will give test for \[F{{e}^{+2}}\]
done
clear
C)
Solution III will neither give test for GST nor \[F{{e}^{+2}}\] ions
done
clear
D)
Electrical conductivity of all solution will be same
done
clear
View Answer play_arrow
What will be the \[{{H}^{+}}\] ion concentration when 4 gm \[NaOH\] dissolved in 1000 ml. of water -
A)
\[{{10}^{-1}}\]
done
clear
B)
\[{{10}^{-13}}\]
done
clear
C)
\[{{10}^{-4}}\]
done
clear
D)
\[{{10}^{-10}}\]
done
clear
View Answer play_arrow
The compressibility factor of a gas is less than unity at STP, therefore -
A)
\[{{V}_{m}}>22.4\text{ }lit\]
done
clear
B)
\[{{V}_{m}}<22.4\text{ }lit\]
done
clear
C)
\[{{V}_{m}}=\,\,22.4\,\,lit\]
done
clear
D)
\[{{V}_{m}}=\,\,44.8\,\,lit\]
done
clear
View Answer play_arrow
For the cell given below \[\left( {{E}_{cell}}-E{{{}^\circ }_{cell}} \right)\] is\[-0.15\text{ }V\]. \[Zn/Z{{n}^{+2}}\parallel \,C{{u}^{+2}}/Cu\] then -
A)
\[\frac{\left[ Z{{n}^{+2}} \right]}{\left[ C{{u}^{+2}} \right]}\,\,=\,\,{{10}^{2}}\]
done
clear
B)
\[\frac{\left[ Z{{n}^{+2}} \right]}{\left[ C{{u}^{+2}} \right]}\,\,=\,\,{{10}^{-\,2}}\]
done
clear
C)
\[\frac{\left[ Z{{n}^{+2}} \right]}{\left[ C{{u}^{+2}} \right]}\,\,=\,\,{{10}^{-\,4}}\]
done
clear
D)
\[\frac{\left[ Z{{n}^{+2}} \right]}{\left[ C{{u}^{+2}} \right]}\,\,=\,\,{{10}^{5}}\]
done
clear
View Answer play_arrow
The unit cell cube length for LiCI (just like NaCI structure) is \[5.14\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]. Assuming anion- anion contact, the ionic radius for chloride ion is:
A)
\[1.815\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[2.8\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[3.8\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[4.815\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
On passing 3 amp. of electricity for 50 minutes, 1.8 g of metal deposits. The equivalent mass of metal is -
A)
10.5
done
clear
B)
25.8
done
clear
C)
19.3
done
clear
D)
30.7
done
clear
View Answer play_arrow
Cane sugar \[+\text{ }{{H}_{2}}O\,\,\to \] Aldo sugar + keto sugar what is the final product of reduction of above keto sugar by HI (in Presence of red Phosphorous)
A)
Fructosazone
done
clear
B)
Penta Acetyl Galactose
done
clear
C)
Sorbitol and Penta acetyl fructose
done
clear
D)
Sixth Homologhe of Paraffin series
done
clear
View Answer play_arrow
For the reaction \[{{X}_{2}}{{O}_{4(l)}}\,\,\to \,\,2X{{O}_{2(g)}}\,\,\Delta E\,\,=\,\,2.1\] \[Kcal,\,\,\Delta s\,=\,20\,cal/K\] at 300 K. Hence \[\Delta \]G is -
A)
2.7 Kcal
done
clear
B)
-2.7 Kcal
done
clear
C)
9.3 Kcal
done
clear
D)
-9.3 Kcal
done
clear
View Answer play_arrow
The relative reactivity towards reaction with MeMgBr of the following is:
A)
(i)>(ii)>(iu)
done
clear
B)
(ii)>(iii)>(i)
done
clear
C)
(i)>(iii)>(u)
done
clear
D)
(iii)>(i)>(ii)
done
clear
View Answer play_arrow
How many stereo isomers are possible from- \[C{{H}_{3}}-CH\left( Br \right)-CH\left( Br \right)-CH=CH-C{{H}_{3}}\]
A)
4
done
clear
B)
6
done
clear
C)
8
done
clear
D)
10
done
clear
View Answer play_arrow
How are the following compound related?
A)
Identical
done
clear
B)
Enantiomers
done
clear
C)
Structural isomer
done
clear
D)
Distereomers
done
clear
View Answer play_arrow
For the given compound which of the following statement(s) are correct?
A)
It has 8 stereoisomers
done
clear
B)
It has 3 stereo centres
done
clear
C)
The compound does not show tautomerism
done
clear
D)
All the above
done
clear
View Answer play_arrow
The major product (Z) can be:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
o-xylene on ozonolysis may give:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Arrange stability of the given carbocations in decreasing order:
A)
(iii)>(ii)>(i)>(iv)
done
clear
B)
(i) > (ii) > (iii) > (iv)
done
clear
C)
(iii) > (i) > (ii) > (iv)
done
clear
D)
(u)>(iii)>(i)>(iv)
done
clear
View Answer play_arrow
Identify correct reactivity order for \[{{S}_{N}}1\] reaction:
A)
(i)>(ii)>(iii)
done
clear
B)
(ii)>(i)>(iii)
done
clear
C)
(i)>(iii)>(ii)
done
clear
D)
(iii)>(i)>(ii)
done
clear
View Answer play_arrow
In the given reaction
A and B are:
A)
\[M{{e}_{3}}C-C{{H}_{2}}OH+Ph-COONa\]
done
clear
B)
\[M{{e}_{3}}C-COONa+Ph-C{{H}_{2}}OH\]
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
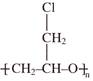
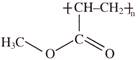
Identify structure of Nylon-6:
A)
\[~+C{{H}_{2}}-C{{H}_{2}}-O-C{{H}_{2}}-C{{H}_{2}}-O{{+}_{n}}\]
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
What could be the product for the following reaction?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Predict the major product of the following reaction:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Predict the end product of the following reaction sequence:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The major product [X] of the given reaction is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
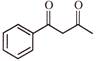
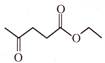
What is the name of the following compound?
A)
Ethyl-4-oxopentanoate
done
clear
B)
Ethyl ester methyl ketone
done
clear
C)
Ethyl-2-ketonepentanoate
done
clear
D)
Ethyl-4-ketone pentyl ester
done
clear
View Answer play_arrow
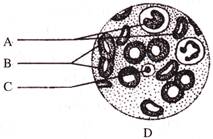
Identify the type of connective tissue and their components given in the figure.
A)
A-White blood cells, B-Red blood cells, C-Platelets, D-Blood
done
clear
B)
A-Haversian system, B-Central canal, C-Lacuna, D-Bone
done
clear
C)
A-Macrophages, B-Collagenous fibre, C-Elastic fibre, D-Loose connective tissue
done
clear
D)
A-Haversian system, B-Red blood cells, C-While blood cells, D-Blood
done
clear
View Answer play_arrow
Benign tumours are enclosed in
A)
nervous tissue
done
clear
B)
epithelial tissue
done
clear
C)
muscular tissue
done
clear
D)
connective tissue
done
clear
View Answer play_arrow
Choose the odd pair out in the following column.
A)
Areolar connective tissue - Collagen
done
clear
B)
Epithelium - Keratin
done
clear
C)
Neuron - Melanin
done
clear
D)
Muscle fibre - Actin
done
clear
View Answer play_arrow
When breast feeding is replaced by less nutritive food low in proteins and calories, the infants below the age of one year are likely to suffer from -
A)
Marasmus
done
clear
B)
Rickets
done
clear
C)
Kwashiorkor
done
clear
D)
Pellagra
done
clear
View Answer play_arrow
Which of these terms is used for two different parts applied with a slight difference in is spelling, not in pronunciation?
A)
Pelvi's
done
clear
B)
Ileum
done
clear
C)
Cloaca
done
clear
D)
Intestine
done
clear
View Answer play_arrow
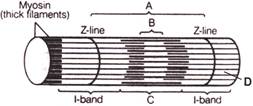
Identify the marked structures in the given figure.
A)
A-Sarcomere, B-H-zone, C-A-band, D-Actin
done
clear
B)
A-H-zone, B-A-band, C-Actin, D-Sacromere
done
clear
C)
A-Actin, B-H-zone, C-A-band, D-Sarcomere
done
clear
D)
A-Sarcomere, B-A-band, C-H-zone, D-Actin
done
clear
View Answer play_arrow
Which one of the following is the correct difference between rod cells and cone cells of our retina?
A)
Feature Rod cells Cone cells Visual acuity High Low
done
clear
B)
Feature Rod cells Cone cells Visual pigment Iogopsin Contained Rhodopsin
done
clear
C)
Feature Rod cells Cone cells Overall function Vision in poor light Colour vision & detailed vision in bright light
done
clear
D)
Feature Rod cells Cone cells Distribution More concentrated in centre Evenly distributed all over retina of retina
done
clear
View Answer play_arrow
In a man, abducens nerve is injured. Which one of the following functions will be affected?
A)
Movement of the eye ball
done
clear
B)
Swallowing
done
clear
C)
Movement of the tongue
done
clear
D)
Movement of the neck
done
clear
View Answer play_arrow
\[N{{a}^{+}}-{{K}^{\text{+}}}\] pump is found in membranes of many cells, like nerve cells. It works against electro- chemical gradient and involve in integral protein ATPase. For each molecule of ATP used-
A)
Three ions of \[N{{a}^{+}}\] are pumped out and two \[{{K}^{+}}\] are taken in
done
clear
B)
Three ions of \[N{{a}^{+}}\] are taken in and two \[{{K}^{+}}\]are pumped out
done
clear
C)
Two ions of \[N{{a}^{+}}\] are thrown out and three \[{{K}^{+}}\] are absorbed
done
clear
D)
Three ions of \[{{K}^{+}}\] are absorbed and three \[N{{a}^{+}}\] are pumped out
done
clear
View Answer play_arrow
Which of the following is known as ?cochlear duct??
A)
Scala media
done
clear
B)
Scala vestibuli
done
clear
C)
Scala tympani
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which one is related to urine concentration in mammals?
A)
Testosterone hormone
done
clear
B)
Antidiuretic hormone
done
clear
C)
Oxytocin hormone
done
clear
D)
All of the above
done
clear
View Answer play_arrow
In column-I (the endocrine structure) and column-II (the corresponding hormones). Match the columns and identify the correct option from those given.
Column I
Column II
[A] Hypothalamus
I. Cortisol
[B] Anterior pituitary
II. Oestrogen
[C] Testis
III. FSH and LH
[D] Ovary
IV. Androgens
V. Gonadrotropin releasing hormone
A)
A - V B - III C - IV D - II
done
clear
B)
A - V B - III C - I D - IV
done
clear
C)
A - I B - II C - IV D - III
done
clear
D)
A - III B - V C - IV D - II
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II I. Herbarium 1. Preserved plant and animal specimen and also artistic and educational material II. Botanical Garden 2. Living wild animal III. Zoological Park 3. Preserved plant specimen on sheet IV. Museum 4. Diversity of living plant
Codes
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Most abundant bacteria are
A)
Chemosynthetic bacteria
done
clear
B)
Heterotrophic bacteria
done
clear
C)
Heterotrophic decomposers
done
clear
D)
Archaebacteria
done
clear
View Answer play_arrow
Which of the following statements about Euglena is true?
A)
Euglenoids bear flagella
done
clear
B)
Euglena when placed in continuous darkness, losses their photosynthetic activity and die
done
clear
C)
The pigments of Euglena are quite different from those of green plants
done
clear
D)
Euglena is a marine protest
done
clear
View Answer play_arrow
Identify the edible and delicate Ascomycetes members.
A)
Agaricus and Puccinia
done
clear
B)
morels and truffles
done
clear
C)
puffball and Agaricus
done
clear
D)
puffball and mushrooms
done
clear
View Answer play_arrow
Which of the following statements are false about viruses?
I. Viruses are facultative parasites. II. Viruses can multiply only when they are inside the living cells. III. Viruses cannot pass bacterial proof filters. IV. Viruses do not contains proteins DNA and RNA.
Code:
A)
I, II and III
done
clear
B)
II, III and IV
done
clear
C)
I, III and IV
done
clear
D)
I, II, III and IV
done
clear
View Answer play_arrow
The members of Chlorophyceae usually have a rigid cell wall made up of-
A)
cellulose (outer layer) and algin (inner layer)
done
clear
B)
pectose (inner layer) and peptidoglycan (outer layer)
done
clear
C)
cellulose (inner layer) and pectose (outer layer)
done
clear
D)
chitin (inner layer) and pectose (outer layer)
done
clear
View Answer play_arrow
In bryophytes antheridium produces ...A... and female sex organ archegonium produces ...B... Here A and B refer to -
A)
A-uniflagellate antherozoids; B-two egg
done
clear
B)
A-biflagellate antherozoids; B-one egg
done
clear
C)
A-non-motile antherozoids; B-one egg
done
clear
D)
A-non-motile antherozoids; B-two egg
done
clear
View Answer play_arrow
In pteridophytes spore germinate to give rise to
A)
thalloid gametophytes called prothallus
done
clear
B)
thalloid sporophytes called prothallus
done
clear
C)
thalloid sporocarp
done
clear
D)
thalloid, photosynthesis sporophyte
done
clear
View Answer play_arrow
In gymnosperms the dominate phase is ...A... They are heterosporous, produce ...B... and...C.... Here, A, B and C refers to -
A)
A-sporophyte, B-haploid microspores, C-haploid megaspores
done
clear
B)
A-gametophyte, B-haploid microspores, C-diploid megaspores
done
clear
C)
A-sporophyte, B-diploid microspores, C-diploid megaspores
done
clear
D)
A-gametophyte, B-diploid microspores, C-haploid megaspores
done
clear
View Answer play_arrow
How many organisms in the list given below are autotrophs? Lactobacillus, Nostoc, Chara, Nitrosomonas, Nitrobacter, Streptomyces, Trypanosoma, Porphyra, Wolffia -
A)
Four
done
clear
B)
Five
done
clear
C)
Six
done
clear
D)
Three
done
clear
View Answer play_arrow
Two male gametes in angiosperms are produced by -
A)
Meiosis of microspore mother cell
done
clear
B)
Meiosis of megaspore mother cell
done
clear
C)
Mitosis of generative cell
done
clear
D)
Mitosis of vegetative cell
done
clear
View Answer play_arrow
During the germination of seeds of cereals the stored food hydrolyzing enzymes an synthesized in-
A)
Cotyledon
done
clear
B)
Endosperm
done
clear
C)
Aleurone layer
done
clear
D)
Plumule
done
clear
View Answer play_arrow
Egg apparatus of an embryo sac consists of -
A)
Egg cell only
done
clear
B)
Egg cell + 3 antipodal cells
done
clear
C)
One egg cell + 2 synergids
done
clear
D)
One egg cell + 2 synergids + 2 antipodal cells
done
clear
View Answer play_arrow
The given kind of pyramid cannot be possible for-
A)
Pyramid ofbiomass in forest ecosystem
done
clear
B)
Pyramid of number in grassland ecosystem
done
clear
C)
Pyramid of number in lake ecosystem
done
clear
D)
Pyramid of biomass in lake ecosystem
done
clear
View Answer play_arrow
Birds and mammal species does not show -
A)
Migration
done
clear
B)
Produce small sized, large number of offesprings
done
clear
C)
Regulation by physical mean
done
clear
D)
Breed more than once in life time
done
clear
View Answer play_arrow
Choose the number of correct statements regarding secondary succession -
[A] Rate of succession is slow [B] Pioneer species depends on soil conditions and water availability [C] Climax reach fastly [D] Occurs on bare rock leads to mesopytic conditions [E] Climax community are forest
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
1
done
clear
View Answer play_arrow
How many of the following are the characteristic features of stable ecosystem -
[A] Less seasonal variations [B] Photosynthetis/Respiration = 1 [C] Few number of keystone species [D] Resistant to occasional of disturbances [E] Annual net community productivity almost stable [F] High degree of exotic species
A)
3
done
clear
B)
4
done
clear
C)
5
done
clear
D)
2
done
clear
View Answer play_arrow
Select the number of correct statements regarding stomatal apparatus -
[A] Dorsiventral leaf has greater number of stomata on lower surface [B] Isobilateral leaf has less number of stomata over upper surface [C] Cellulosic microfibrils radially arranged on outer wall of guard cells [D] Roating hydrophytes posses hypostomatic leaves [E] Stomata are sunken in xerophytes
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
Which of the following is associated with principal mechanism of asexual reproduction and vegetative propagation?
A)
Mitosis
done
clear
B)
Meiosis
done
clear
C)
Both [a] and [b]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Respectively in \[{{C}_{3}}\] and \[{{C}_{4}}\] plants weather photorespiration would be present at low \[C{{O}_{2}}\] concentration -
A)
High, High
done
clear
B)
Low, High
done
clear
C)
High, Negligible
done
clear
D)
Neglible, High
done
clear
View Answer play_arrow
Choose the steps of glycolysis where substrate level phosphorylation occurs -
[A]\[\,Glucose\,\,\to \,\,Glucose\text{ }6P\] [B] \[1,\,\,3\,\,Bi\,\,PGA\,\,\to \,\,3\,PGA\] [C] \[1,\text{ }3\text{ }Bi\text{ }PGAL\,\,\to \,\,1,\text{ }3,\text{ }Bi\text{ }PGA\] [D] \[PEP\text{ }\to \text{ }Pyruvate\] [E] \[Fructose\text{ }6\text{ }P\,\,\to \,\,Fructose\text{ }1,\text{ }6\text{ }Bi\text{ }P\]
A)
A, E
done
clear
B)
A, D
done
clear
C)
B, D
done
clear
D)
A, B, D, E
done
clear
View Answer play_arrow
A plant is kept in 24 hr. darkness and a flash of light is provided in middle of it's dark period. The plant show flowering. This plant should be a
A)
Long day plant
done
clear
B)
Short day plant
done
clear
C)
Day neutral plant
done
clear
D)
\[{{C}_{4}}\] plant
done
clear
View Answer play_arrow
Which of the following statements is not true?
A)
Both mitochondria and chloroplasts provide energy to cells in the same way
done
clear
B)
Both mitochondria and chloroplasts have more than one membrane
done
clear
C)
Only chloroplasts contain the pigment chlorophyll
done
clear
D)
Both animal and plant cells contain mitochondria
done
clear
View Answer play_arrow
Girdling experiment cannot be performed in sugarcane because:
A)
It cannot withstand injury
done
clear
B)
Vascular bundles in it are scattered
done
clear
C)
Phloem in it situated internal to xylem
done
clear
D)
Sugarcane plants are too delicate
done
clear
View Answer play_arrow
Select the incorrect match -
A)
Bakanae disease - Gibberellin
done
clear
B)
Adenine derivative - Cytokinin
done
clear
C)
Oxidation of carotene - IBA
done
clear
D)
Methionine - Ethylene
done
clear
View Answer play_arrow
Select incorrect statement regarding \[{{N}_{2}}\] fixing microbes -
A)
Some of the \[{{N}_{2}}\] fixers are free living while some are symbiotic
done
clear
B)
All \[{{N}_{2}}\] fixing prokaryotes posses leghaemoglobin as \[{{O}_{2}}\] scavenger
done
clear
C)
Symbiotic \[{{N}_{2}}\] fixers performs anaerobic respiration in symbiotic state
done
clear
D)
Cyanobacteria can fix \[{{N}_{2}}\] in both symbiotic and free living state
done
clear
View Answer play_arrow
Which of the following is a polyunsaturated fatty acid?
A)
Palmitic acid
done
clear
B)
Arachidonic acid
done
clear
C)
Stereic acid
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Which of the following is true?
[A] The all molecules in the acid insoluble fraction are polymer substance [B] Collgen is a intracellular ground substance [C] Protein is a homopolymer of a amino acid [D] The polypeptide chain formed during translation is primary configuration of protein.
A)
B, C & D
done
clear
B)
A, B & D
done
clear
C)
B & D
done
clear
D)
Only D
done
clear
View Answer play_arrow
Thymine increase the stability of DNA because-
A)
It reduce activity of DNA
done
clear
B)
It prevent degradation of DNA
done
clear
C)
It reduce rate of mutation in DNA
done
clear
D)
[a] & [c] both
done
clear
View Answer play_arrow
Few statement regarding cell wall is given below. How many of these are correct?
[A] Cellulosic cell wall is present in plant and few fungi [B] Cell wall is present in all plants and few animals [C] Cell wall is common among bacteria, plant and fungi [D] Cell wall is dead, flexible and permeable
A)
Four
done
clear
B)
One
done
clear
C)
Two
done
clear
D)
Three
done
clear
View Answer play_arrow
RER is frequently involved in -
A)
Protein synthesis
done
clear
B)
Carbohydrate synthesis
done
clear
C)
Lipid synthesis
done
clear
D)
[a] & [c] both
done
clear
View Answer play_arrow
Oxidative enzyme in plant cell are present in -
A)
Mitochondria and peroxysome
done
clear
B)
Only mitochondria
done
clear
C)
Mitochondria and chloroplast
done
clear
D)
Mitochondria, chloroplast and peroxisome
done
clear
View Answer play_arrow
Chromosomal structure which are attached to each other by centromere during meiosis-I is known as -
A)
Homologous chromosome
done
clear
B)
Sister chromatid
done
clear
C)
Daughter chromosome
done
clear
D)
[b] & [c] both
done
clear
View Answer play_arrow
Golgi complex and ER disappear during..... A.....and reappear during..... B..... . What is A and B respectively?
A)
Prophase and cytokinesis
done
clear
B)
Prophase and Telophase
done
clear
C)
Metaphase and Telophase
done
clear
D)
Metaphase and cytokinesis
done
clear
View Answer play_arrow
Which of the following property of phospholipids is responsible for lipid bilayer formation?
A)
Zwittorio
done
clear
B)
Amphoteric nature
done
clear
C)
Amphipathic nature
done
clear
D)
All of above
done
clear
View Answer play_arrow
If the temperature is increased above \[35\text{ }{}^\circ C\]
A)
Rate of decline of respiration will be earlier than decline of photosynthesis
done
clear
B)
Rate of decline of photosynthesis will be earlier than decline of respiration
done
clear
C)
Both decline simultaneously
done
clear
D)
Both do not show fixed pattern
done
clear
View Answer play_arrow
Quiescent centre is found in plants at the -
A)
Root tip
done
clear
B)
Cambium
done
clear
C)
Shoot tip
done
clear
D)
Tip
done
clear
View Answer play_arrow
Cambium is a type of -
A)
Apical meristem
done
clear
B)
Intercalary meristem
done
clear
C)
Lateral meristem
done
clear
D)
Permanent or mature meristem
done
clear
View Answer play_arrow
The first few leaves after the cotyledons in the seeding of Acacia are pinnately compound but the petioles of the upper compound leaves are flattened and they bear few pinnae. The adult plant has only parallel veined leaf like flattened petioles but without any pinnae. These observation lead us to the conclusion that:
A)
The leaves in the adult plant are reduced to phyllode while the seeding leaves are unreduced
done
clear
B)
The green parallel veined structures in the adult plant are phylloclades
done
clear
C)
The plant exhibits heterophyJJy bearing simple leaves in the adult plant and compound leaves in the seeding
done
clear
D)
The adult plant leaves are a case of reversion where they become simple like cotyledons
done
clear
View Answer play_arrow
Endospermic seed are present in -
[A] Tulip [B] Petunia [C] Trifolium [D] Castor
A)
A & D
done
clear
B)
A, B & D
done
clear
C)
B & C
done
clear
D)
A, B, C & D
done
clear
View Answer play_arrow
Which of the following is true regarding the figure given below?
A)
Leaves are modified for protection
done
clear
B)
Phyllode is present
done
clear
C)
Phylloclade is present
done
clear
D)
[a] & [c] both
done
clear
View Answer play_arrow
During viral infection the protein formed in host cells to resist is:
A)
Interferon
done
clear
B)
Antitoxin
done
clear
C)
Antibody
done
clear
D)
Histone
done
clear
View Answer play_arrow
Saline solution is given to patients of Cholera because:
A)
\[N{{a}^{+}}\] prevents water loss from body
done
clear
B)
NaCI function as regulatory material
done
clear
C)
NaCI produces energy
done
clear
D)
NaCI is antibacterial
done
clear
View Answer play_arrow
What is true for monoclonal antibodies?
A)
These antibodies obtained from one parent and for one antigen
done
clear
B)
These obtained from different parents and for one antigen
done
clear
C)
These obtained from one parent and for many antigens
done
clear
D)
These obtained from many parents and for many antigen
done
clear
View Answer play_arrow
Salmonella is related with -
A)
Typhoid
done
clear
B)
Polio
done
clear
C)
T.B.
done
clear
D)
Tetanus
done
clear
View Answer play_arrow
Mental retardness in man occur due to -
A)
Loss of one X chromosome
done
clear
B)
Addition of one X chromosome
done
clear
C)
Slight growth in Y
done
clear
D)
Overgrowth in Y
done
clear
View Answer play_arrow
Symptoms of Lathyrism -
A)
Bone deformation
done
clear
B)
Muscular dystrophy and paralysis
done
clear
C)
Asphyxia
done
clear
D)
Cordiac arrest
done
clear
View Answer play_arrow
Segments of DNA which can move in genome-
A)
Transposons
done
clear
B)
Introns
done
clear
C)
Exons
done
clear
D)
Cistrons
done
clear
View Answer play_arrow
Common bacterium used in genetic engineering is-
A)
E. Coli
done
clear
B)
Diplococcus
done
clear
C)
Rhizobium
done
clear
D)
Spirillum
done
clear
View Answer play_arrow
An example of gene therapy is -
A)
Production of injectable Hepatitis B vaccine
done
clear
B)
Production of vaccines in food crops like potatoes which can be eaten
done
clear
C)
Production of test tube babies by artificial insemination and implantation of fertilized eggs
done
clear
D)
Introduction of gene for adenosine deaminase in persons suffering from Severe Combined Immune-Deficiency (SCID)
done
clear
View Answer play_arrow
Which of these is not correct regarding Bt cotton?
A)
No such plant is heard of
done
clear
B)
It is a disease/resistant plant
done
clear
C)
It produces more yield of cotton
done
clear
D)
It has been obtained by recombination
done
clear
View Answer play_arrow
Genetic material of prokaryotic cell -
A)
Non histonic double stranded DNA
done
clear
B)
Histonic double stranded DNA
done
clear
C)
Histone & DNA both are absent
done
clear
D)
Histone without DNA
done
clear
View Answer play_arrow
In inducible operon, regulatory gene synthesize
A)
Promoter
done
clear
B)
Operator
done
clear
C)
Represser
done
clear
D)
Aporepressor
done
clear
View Answer play_arrow
Irregularity is found in drosophila during the organ differentiation for example- inplace of wing, long legs are formed. Which gene is responsible for-
A)
Double dominant gene
done
clear
B)
Homeotic gene
done
clear
C)
Complimentary gene
done
clear
D)
Plastid
done
clear
View Answer play_arrow
Dioecious organisms are -
A)
Neries
done
clear
B)
Apis
done
clear
C)
PUa
done
clear
D)
All of above
done
clear
View Answer play_arrow
Alimentary canal without anus is found in -
A)
Platyhelminthese
done
clear
B)
Nemathelminthese
done
clear
C)
Annelida
done
clear
D)
Mollusca
done
clear
View Answer play_arrow
What is the value of temperature coefficient \[\left( {{Q}_{10}} \right)\] dark reaction
A)
1
done
clear
B)
2 or 3
done
clear
C)
Both
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Parapodia are locomotory organ in -
A)
Terrestrial annelids
done
clear
B)
Parasitic annelids
done
clear
C)
Aquatic annelids
done
clear
D)
All of above
done
clear
View Answer play_arrow
A soft and spongy layer of skin forms a mantle over the-
A)
Head
done
clear
B)
Muscular foot
done
clear
C)
Visceral hump
done
clear
D)
Whole body
done
clear
View Answer play_arrow
A reduction of NADP to \[NADP{{H}_{2}}\] is associated with-
A)
EMP-pathway
done
clear
B)
HMP-shunt
done
clear
C)
Calvin cycle
done
clear
D)
Glycolysis
done
clear
View Answer play_arrow
Number of vasa effentia in each testies of male frog are -
A)
15-17
done
clear
B)
10-12
done
clear
C)
8-10
done
clear
D)
14-16
done
clear
View Answer play_arrow
Functional connection between kidney and gonads are found in -
A)
Male frog
done
clear
B)
Female frog
done
clear
C)
Both of above
done
clear
D)
None of above
done
clear
View Answer play_arrow
Ommatidia in each compound eye of cockroach are-
A)
Hexagonal and 2500 in no.
done
clear
B)
Pentagonal and 2000 in no.
done
clear
C)
Hexagonal and 2000 in no.
done
clear
D)
Pentagonal and 2500 in no.
done
clear
View Answer play_arrow
Number of ovariole or ovarian tubule in both ovaries are -
A)
8
done
clear
B)
16
done
clear
C)
32
done
clear
D)
64
done
clear
View Answer play_arrow
Calciferous gland are present in which part of elementary canal of earthworm -
A)
Buccal cavity
done
clear
B)
Pharynx
done
clear
C)
Oesophagus
done
clear
D)
Stomach
done
clear
View Answer play_arrow
Which one is false?
A)
Pleura is double covering of kidney
done
clear
B)
Scurvy is due to vilatmin C deficiency
done
clear
C)
Pancreas is both exocrine and endocrine gland
done
clear
D)
Blood from right side of heart is carried to lungs by pulmonary artery
done
clear
View Answer play_arrow
Forced deep breathing for a few minutes by a person sitting at rest may be followed by a temporary cessation of breathing. This is due to-
A)
Too much Oz in the blood
done
clear
B)
Too much COs in the blood
done
clear
C)
Very little C02 in the blood
done
clear
D)
Both too much 02 and very little \[C{{O}_{2}}\] in the blood
done
clear
View Answer play_arrow
Which of the following statements regarding brown fat is correct?
A)
It is poorly vascularized
done
clear
B)
It functions in unilocular energy storage
done
clear
C)
It is directly innervated by the parasympathetic nervous system
done
clear
D)
It produces heat through the uncoupling of the electron transport chain from oxidative phosphorylation.
done
clear
View Answer play_arrow
The period when menstrual cycle stops is -
A)
Menarche
done
clear
B)
Luteal phase
done
clear
C)
Menopause
done
clear
D)
Heat period
done
clear
View Answer play_arrow
Heart of crocodile is
A)
single chambered
done
clear
B)
two chambered
done
clear
C)
three chambered
done
clear
D)
four chambered
done
clear
View Answer play_arrow
Myocardial infarction is caused by
A)
Hardening of arteries
done
clear
B)
lumpy thickness develop in the inner walls of arteries
done
clear
C)
clot may occur in the lumen of a pulmonary artery
done
clear
D)
sudden interruption in blood flow towards a portion of heart
done
clear
View Answer play_arrow
In kidney the high capillary bed is found -
A)
in Bowman's capsule
done
clear
B)
around anterior tubules
done
clear
C)
around loop of Henie
done
clear
D)
absent
done
clear
View Answer play_arrow
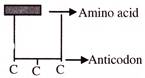
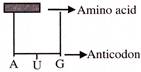
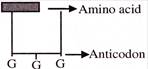
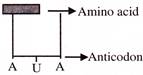
Find out the sequence of binding of the following ammo acyl -t-RNA complexes during translation to an m-RNA transcribed by a DNA segment having the base sequence. 3' ATACCCATGGGG 5' Choose the answer showing the correct order of alphabets-
A)
A, B, C, D
done
clear
B)
D, A, B, C
done
clear
C)
A, B, D, C
done
clear
D)
B, A, C, D
done
clear
View Answer play_arrow
De Vries gave his mutation theory on organic evolution while working on -
A)
Althea rosea
done
clear
B)
Drosophila melangaster
done
clear
C)
Oenothera lamarckiana
done
clear
D)
Pisum sativum
done
clear
View Answer play_arrow
When two related populations occupy geographically or spatially separate areas, they are called -
A)
Allopatric population
done
clear
B)
Quantum population
done
clear
C)
Saltational population
done
clear
D)
Parapatric population
done
clear
View Answer play_arrow
Which of the following is a correct match for connecting link between two phyla?
A)
Archeopteryx - aves and mammalian
done
clear
B)
Amphioxus - echinodermata and chordate
done
clear
C)
Ornithorhynchus - aves and reptilia
done
clear
D)
Peripatus - annelida and arthopoda
done
clear
View Answer play_arrow
Convergent evolution is illustrated by -
A)
Rat and dog
done
clear
B)
Bacterium and protozoan
done
clear
C)
Starfish and cutle fish
done
clear
D)
Dogfish and whale
done
clear
View Answer play_arrow
According to chemisomotic theory of P. Mitchell (1978), ATPs are synthesised on membrance due to the -
A)
Proton gradient
done
clear
B)
Electron gradient
done
clear
C)
Osmosis
done
clear
D)
From \[{{H}_{2}}S{{O}_{4}}\]
done
clear
View Answer play_arrow

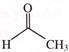
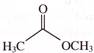
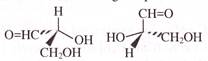
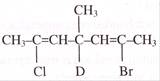
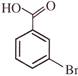
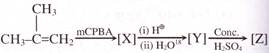 The major product (Z) can be:
The major product (Z) can be: 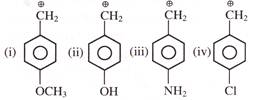
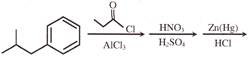
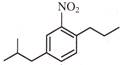
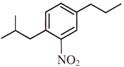
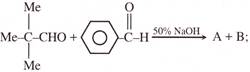 A and B are:
A and B are: