A) Hydrogen bonding
B) Dipole-dipole interaction
C) Covalent bonds
D) London dispersion force
Correct Answer: A
Solution :
Key Idea When H atom is directly linked with N or O, or F, inter or intermolecular H-bonding is formed. In between \[{{\,}_{Z+}}_{1}^{A}Y\xrightarrow[{}]{{}}\,_{Z-1}^{A-4}\beta +\alpha _{2}^{4}\] molecules intermolecular H-bonding exist.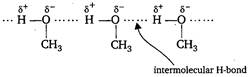 Hence, it is the intermolecular H-bonding that must be overcome in converting liquid \[_{Z+1}^{A-4}\beta \xrightarrow[{}]{{}}\,_{Z-1}^{A-4}\beta +\gamma _{0}^{0}\] to gas.
Hence, it is the intermolecular H-bonding that must be overcome in converting liquid \[_{Z+1}^{A-4}\beta \xrightarrow[{}]{{}}\,_{Z-1}^{A-4}\beta +\gamma _{0}^{0}\] to gas.
You need to login to perform this action.
You will be redirected in
3 sec
