a-Scattering Experiment
Category : JEE Main & Advanced
'Geiger and Marsden (students of Rutherford) studied the scattering of \[\alpha -\]particles by gold foil on the advice of Rutherford and made the following observations.
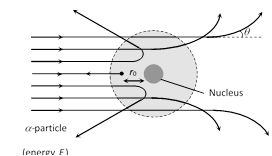
(1) Most of the \[\alpha -\]particles pass through the foil straight away undeflected.
(2) Some of them are deflected through small angles.
(3) A few \[\alpha -\]particles (1 in 1000) are deflected through the angle more than \[{{90}^{o}}\].
(4) A few \[\alpha -\]particles (very few) returned back i.e. deflected by \[{{180}^{o}}\].
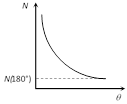
(5) Number of scattered particles : \[N\propto \frac{1}{{{\sin }^{4}}(\theta /2)}\]
(6) If t is the thickness of the foil and N is the number of \[\alpha -\]particles scattered in a particular direction (i.e. \[\theta =\]constant), it was observed that \[\frac{N}{t}=\text{constant}\]\[\Rightarrow \]\[\frac{{{N}_{1}}}{{{N}_{2}}}=\frac{{{t}_{1}}}{{{t}_{2}}}\]
(7) Distance of closest approach (Nuclear dimension) : The minimum distance from the nucleus up to which the \[\alpha -\]particle approach, is called the distance of closest approach \[({{r}_{0}})\]. At this distance the entire initial kinetic energy has been converted into potential energy so \[\frac{1}{2}m{{v}^{2}}=\frac{1}{4\pi {{\varepsilon }_{0}}}.\,\frac{(Ze)\,2e}{{{r}_{0}}}\]\[\Rightarrow \]\[{{r}_{0}}=\frac{Z{{e}^{2}}}{m{{v}^{2}}\pi {{\varepsilon }_{0}}}\]\[=\frac{4kZ{{e}^{2}}}{m{{v}^{2}}}\]
(8) Impact parameter (b) : The perpendicular distance of the velocity vector \[(\vec{\upsilon })\] of the \[\alpha -\]particle from the centre of the nucleus when it is far away from the nucleus is known as impact parameter. It is given as
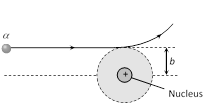
\[b=\frac{Z{{e}^{2}}\cot (\theta /2)}{4\pi {{\varepsilon }_{0}}\left( \frac{1}{2}m{{v}^{2}} \right)}\,\]\[\Rightarrow \]\[b\propto \cot (\theta /2)\]
For large b, \[\alpha \]particles will go undeviated and for small b the \[\alpha -\]particle will suffer large scattering.
You need to login to perform this action.
You will be redirected in
3 sec
