Thomson's Atomic Model
Category : JEE Main & Advanced
J.J. Thomson gave the first idea regarding structure of atom. According to this model.
(1) An atom is a solid sphere in which entire and positive charge and it's mass is uniformly distributed and in which negative charge (i.e. electron) are embedded like seeds in watermelon.
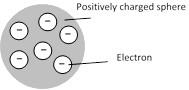
(2) This model explained successfully the phenomenon of thermionic emission, photoelectric emission and ionization.
(3) The model fail to explain the scattering of \[\alpha -\]particles and it cannot explain the origin of spectral lines observed in the spectrum of hydrogen and other atoms.
You need to login to perform this action.
You will be redirected in
3 sec
