-
question_answer1)
The volume of gases \[N{{H}_{3}},C{{O}_{2}}\] and \[{{H}_{2}}\]adsorbed by one gram of charcoal at 300 K are in order of:
A)
\[{{H}_{2}}>C{{O}_{2}}>N{{H}_{3}}\] done
clear
B)
\[N{{H}_{3}}>{{H}_{2}}>C{{O}_{2}}\] done
clear
C)
\[N{{H}_{3}}>C{{O}_{2}}>{{H}_{2}}\] done
clear
D)
\[C{{O}_{2}}>N{{H}_{3}}>{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer2)
A plot of \[log\left( \frac{x}{m} \right)\] against log P for the adsorption of a gas on a solid gives a straight line with slope equal to:
A)
\[\frac{1}{n}\] done
clear
B)
n done
clear
C)
\[log\text{ }K\] done
clear
D)
K done
clear
View Solution play_arrow
-
question_answer3)
The heat evolved in chemisorption lies in the range (in kJ/mol) of:
A)
80 to 240 done
clear
B)
20 to 40 done
clear
C)
40 to 80 done
clear
D)
20 to 100 done
clear
View Solution play_arrow
-
question_answer4)
Identify the correct statement regarding enzymes____.
A)
Enzymes are specific biological catalysts that cannot be poisoned done
clear
B)
Enzymes are normally heterogeneous catalysts that are very specific in their action done
clear
C)
Enzymes are specific biological catalysts that can normally function at very high temperatures \[(T\approx 1000K)\] done
clear
D)
Enzymes are specific biological catalysts that possess well-defined active sites done
clear
View Solution play_arrow
-
question_answer5)
Catalytic poisons act by:
A)
making the products chemically inactive. done
clear
B)
increasing the rate of the backward reaction. done
clear
C)
chemical combination with any one of the reactants. done
clear
D)
preferential adsorption on the catalyst surface. done
clear
View Solution play_arrow
-
question_answer6)
The size of particles in suspension, true solution and colloidal solution varies in the order:
A)
suspension > colloidal > true solution done
clear
B)
true solution > suspension > colloidal done
clear
C)
suspension > colloidal = true solution done
clear
D)
none of these done
clear
View Solution play_arrow
-
question_answer7)
Gold number of a lyophilic sol is such a property that:
A)
The larger its value, the greater is the peptizing power. done
clear
B)
The lower its value, the greater is the peptizing power. done
clear
C)
The lower its value, the greater is the protecting power. done
clear
D)
The larger its value, the greater is the protecting power. done
clear
View Solution play_arrow
-
question_answer8)
Among the following, correct statement is:
A)
Brownian movement is more pronounced for smaller particles than for bigger- particles. done
clear
B)
Sols of metal sulphides are lyophilic. done
clear
C)
Hardy Schuize law states that bigger the size of the ons, the greater is its coagulating power. done
clear
D)
One would expect charcoal to adsorb chlorine more than hydrogen sulphide. done
clear
View Solution play_arrow
-
question_answer9)
Under the influence of an electric field, heparticles in a sol migrate towards cathode. The coagulation of the same sol is studied using \[NaCl,N{{a}_{2}}S{{O}_{4}}\]and \[N{{a}_{3}}P{{O}_{4}}\] solutions. Their coagulating values will be in the order
A)
\[NaCl>N{{a}_{2}}S{{O}_{4}}>N{{a}_{3}}P{{O}_{4}}\] done
clear
B)
\[N{{a}_{2}}S{{O}_{4}}>N{{a}_{3}}P{{O}_{4}}>NaCl\] done
clear
C)
\[N{{a}_{3}}P{{O}_{4}}>N{{a}_{2}}S{{O}_{4}}>NaCl\] done
clear
D)
\[N{{a}_{2}}S{{O}_{4}}>NaCl>N{{a}_{3}}P{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer10)
3.6 gram of oxygen is adsorbed on 1.2 g of metal powder. What volume of oxygen adsorbed per gram of the adsorbent at 1 atm and 273 K?
A)
\[0.19L{{g}^{-1}}\] done
clear
B)
\[1L{{g}^{-1}}\] done
clear
C)
\[2.1\text{ }L\text{ }{{g}^{-1}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer11)
For the graph below, select correct order of temperature?
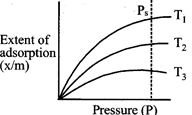
A)
\[{{T}_{1}}>{{T}_{2}}>{{T}_{3}}\] done
clear
B)
\[{{T}_{2}}>{{T}_{3}}>{{T}_{1}}\] done
clear
C)
\[{{T}_{3}}>{{T}_{2}}>{{T}_{1}}\] done
clear
D)
\[{{T}_{1}}={{T}_{2}}={{T}_{3}}\] done
clear
View Solution play_arrow
-
question_answer12)
100 mL of 0.6 M acetic acid is shaken with 2 g activated carbon. The final concentration of the solution after adsorption is 0.5 M. What is the amount of acetic acid adsorbed per gram of carbon?
A)
0.6 g done
clear
B)
0.3 g done
clear
C)
1.2 g done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer13)
The efficiency of an enzyme in catalysing a reaction is due to its capacity
A)
to form a strong enzyme-substrate complex done
clear
B)
to decrease the bond energies of substrate molecule done
clear
C)
to change the shape of the substrate molecule done
clear
D)
to lower the activation energy of the reaction done
clear
View Solution play_arrow
-
question_answer14)
A chemical reaction is catalyzed by a catalyst X. Hence X
A)
reduces enthalpy of the reaction done
clear
B)
decreases rate constant of the reaction done
clear
C)
increases activation energy of the reaction done
clear
D)
does not affect equilibrium constant of the reaction done
clear
View Solution play_arrow
-
question_answer15)
The gold numbers of protective colloids A, B, C and D are 0.04, 0.004, 10 and 40 respectively The protective powers of A, B, C and D are in the order:
A)
\[A>B>C>D\] done
clear
B)
\[B>A>C>D\] done
clear
C)
\[D>C>A>B\] done
clear
D)
\[D>C>B>A\] done
clear
View Solution play_arrow
-
question_answer16)
The density of gold is\[19\text{ }g/c{{m}^{3}}\]. If \[1.9\times {{10}^{-4}}g\] of gold is dispersed in one litre of water to give a sol having spherical gold particles of radius 10 nm, then the number of gold particles per \[m{{m}^{3}}\] of the sol will be:
A)
\[1.9\times {{10}^{12}}\] done
clear
B)
\[6.3\times {{10}^{14}}\] done
clear
C)
\[6.3\times {{10}^{10}}\] done
clear
D)
\[2.4\times {{10}^{6}}\] done
clear
View Solution play_arrow
-
question_answer17)
A thixotropic gel is characterised by
A)
reversible transformation of gel into sol on heating done
clear
B)
irreversible transformation of gel into sol by stirring done
clear
C)
reversible transformation of gel into sol by stirring done
clear
D)
swelling on placing in the dispersion medium done
clear
View Solution play_arrow
-
question_answer18)
In an experiment, addition of 4.0 ml of 0.005 M \[BaC{{l}_{2}}\] to 16.0 ml of arsenius sulphide sol just causes the complete coagulation in 2 hrs. The flocculating value of the effective ion is
A)
\[C{{l}^{-}},1.0\] done
clear
B)
\[C{{l}^{-}},2.0\] done
clear
C)
\[B{{a}^{2+}},1.0\] done
clear
D)
\[B{{a}^{2+}},0.5\] done
clear
View Solution play_arrow
-
question_answer19)
Choose the correct statement
A)
Latex is an example of solid-in-liquid sol done
clear
B)
Latex is a negative sol done
clear
C)
Latex is a positive sol done
clear
D)
None of the above is correct done
clear
View Solution play_arrow
-
question_answer20)
Arrange the following electrolytes in the increasing order of coagulating power for ferric hydroxide sol
| \[N{{a}_{3}}P{{O}_{4}}\] | \[KCl\] | \[{{K}_{2}}S{{O}_{4}}\] | \[NaCI\] |
| I | II | III | IV |
A)
I<II<III<IV done
clear
B)
II=IV<III<I done
clear
C)
II=IV<I<III done
clear
D)
II=III<IV=I done
clear
View Solution play_arrow
-
question_answer21)
Although, nitrogen does not adsorb on a surface at room temperature, it adsorbs on the surface at 83 K. Which one of the following statements is correct?
A)
At 83 K, there is formation of monolayer done
clear
B)
At 83 K, nitrogen is adsorbed as atoms done
clear
C)
At 83 K, nitrogen molecules are held by chemical bonds done
clear
D)
At 83 K, there is formation of multimolecular layers done
clear
View Solution play_arrow
-
question_answer22)
The following statements relate to the adsorption of gases on a solid surface. Identify the incorrect statement among them:
A)
Enthalpy of adsorption is negative done
clear
B)
Energy appears as heat done
clear
C)
On adsorption, the residual forces on the surface are increased done
clear
D)
Entropy of adsorption in negative done
clear
View Solution play_arrow
-
question_answer23)
If x is the mass of the gas adsorbed on mass m of the adsorbent at pressure p, Freundlich adsorption isotherm gives a straight line on plotting
A)
x/m vs p done
clear
B)
x/m vs 1/p done
clear
C)
\[\log \,x\text{/}m\,\,vs\,\,\log \,p\] done
clear
D)
\[log\,x/m\,\,vs\,p\] done
clear
View Solution play_arrow
-
question_answer24)
The correct statement for both the processes of physisorption and chemisorption is
A)
both are endothermic done
clear
B)
chemisorption is endothermic but physisorption is exothermic done
clear
C)
both are exothermic done
clear
D)
physisorption is endothermic but chemisorption is exothermic. done
clear
View Solution play_arrow
-
question_answer25)
Which one of the sols acts as protective colloid?
A)
\[A{{s}_{2}}{{S}_{3}}\] done
clear
B)
Gelatin done
clear
C)
Au done
clear
D)
\[Fe{{\left( OH \right)}_{3}}\] done
clear
View Solution play_arrow
-
question_answer26)
Given below are a few electrolytes, indicate which one among them will bring about the coagulation of a gold sol quickest and in the least of concentration?
A)
\[NaCl\] done
clear
B)
\[MgS{{O}_{4}}\] done
clear
C)
\[A{{l}_{2}}{{\left( S{{O}_{4}} \right)}_{3}}\] done
clear
D)
\[{{K}_{4}}[Fe{{(CN)}_{6}}]\] done
clear
View Solution play_arrow
-
question_answer27)
Which of the following has minimum flocculation value for positively charged sol?
A)
\[C{{l}^{-}}\] done
clear
B)
\[SO_{4}^{2-}\] done
clear
C)
\[PO_{4}^{3-}\] done
clear
D)
\[{{[Fe{{(CN)}_{6}}]}^{4-}}\] done
clear
View Solution play_arrow
-
question_answer28)
Butter is a colloid formed when
A)
fat is dispersed in water done
clear
B)
fat globules are dispersed in water done
clear
C)
water is dispersed in fat done
clear
D)
None of the these done
clear
View Solution play_arrow
-
question_answer29)
Cloud or fog is a colloidal system in which the dispersed phase and the dispersion medium are
A)
Gas, Liquid done
clear
B)
Liquid, gas done
clear
C)
Liquid, liquid done
clear
D)
Solid, Liquid done
clear
View Solution play_arrow
-
question_answer30)
Which of the following constitutes irreversible colloidal system in water as dispersion medium?
A)
Clay done
clear
B)
Platinum done
clear
C)
\[Fe{{\left( OH \right)}_{3}}\] done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer31)
At the high pressure, Langmuir adsorption isotherm takes the form
A)
\[\frac{x}{m}=\frac{ap}{1+bp}\] done
clear
B)
\[\frac{x}{m}=\frac{a}{b}\] done
clear
C)
\[\frac{x}{m}=ap\] done
clear
D)
\[\frac{m}{x}=\frac{b}{a}+\frac{1}{ap}\] done
clear
View Solution play_arrow
-
question_answer32)
Methylene blue, from its aqueous solution, is adsorbed on activated charcoal at\[25{}^\circ C\]. For this process, which of the following statement is correct?
A)
The adsorption requires activation at \[25{}^\circ C\] done
clear
B)
The adsorption is accompanied by a decrease in enthalpy done
clear
C)
The adsorption increases with increase of temperature done
clear
D)
The adsorption is irreversible done
clear
View Solution play_arrow
-
question_answer33)
In an experiment, 200 mL of 0.5 M oxalic acid is shaken with 10 g of activated charcoal and filtered. The concentration of the filtrate is reduced to 0.4 M The amount of adsorption\[\left( \frac{x}{m} \right)\] is
A)
0.9 done
clear
B)
1.8 done
clear
C)
0.18 done
clear
D)
0.09 done
clear
View Solution play_arrow
-
question_answer34)
Equal volume each of two sols of\[AgI\], one obtained by adding \[AgN{{O}_{3}}\] to slight excess of KI and another obtained by adding KI to slight excess of \[AgN{{O}_{3}}\] are mixed together. Then:
A)
the two sols will stabilize each other done
clear
B)
the sol particles will acquire more electric charge done
clear
C)
the sols will coagulate each other mutually done
clear
D)
a true solution will be obtained done
clear
View Solution play_arrow
-
question_answer35)
Under the influence of an electric field, the particles in a sol migrate towards cathode. The coagulation of the same sol is studied using \[NaCl,\]\[N{{a}_{2}}S{{O}_{4}}\] and \[N{{a}_{3}}P{{O}_{4}}\]solutions. Their coagulating values will be in maximum for:
A)
\[NaCl\] done
clear
B)
\[N{{a}_{2}}S{{O}_{4}}\] done
clear
C)
\[N{{a}_{3}}P{{O}_{4}}\] done
clear
D)
same for all done
clear
View Solution play_arrow
-
question_answer36)
According to Langmuir adsorption isotherm, the amount of gas adsorbed at very high pressures
A)
reaches a constant limiting value done
clear
B)
goes on increasing with pressure done
clear
C)
goes on decreasing with pressure done
clear
D)
increases first and then decreases with pressure done
clear
View Solution play_arrow
-
question_answer37)
Under ambient conditions, which among the following surfactants will form micelles in aqueous solution at lowest molar concentration?
A)
\[C{{H}_{3}}-{{(C{{H}_{2}})}_{8}}-CO\overset{-}{\mathop{O}}\,\,N{{a}^{+}}\] done
clear
B)
\[C{{H}_{3}}{{(C{{H}_{2}})}_{11}}\overset{+}{\mathop{N}}\,{{(C{{H}_{3}})}_{3}}\overset{-}{\mathop{B}}\,r\] done
clear
C)
\[C{{H}_{3}}-{{(C{{H}_{2}})}_{13}}-OSO_{3}^{-}N{{a}^{+}}\] done
clear
D)
\[C{{H}_{3}}{{(C{{H}_{2}})}_{15}}\overset{+}{\mathop{N}}\,{{(C{{H}_{3}})}_{3}}\overset{-}{\mathop{B}}\,r\] done
clear
View Solution play_arrow
-
question_answer38)
The migration of dispersion medium under the influence of an electric potential is called:
A)
Cataphoresis done
clear
B)
Electro-osmosis done
clear
C)
Electrophoresis done
clear
D)
Sedimentation done
clear
View Solution play_arrow
-
question_answer39)
Flocculation value of \[BaC{{l}_{2}}\] is much less than that of \[KCl\] for sol A and flocculation value of \[N{{a}_{2}}S{{O}_{4}}\] is much less than that of \[NaBr\] for sol B. The correct statement among the following is:
A)
Both the sols A and B are negatively charged. done
clear
B)
Sol A is positively charged and Sol B is negatively charged. done
clear
C)
Both the sols A and B are positively charged. done
clear
D)
Sol A is negatively charged and sol B is positively charged. done
clear
View Solution play_arrow
-
question_answer40)
Which of the following statements regarding difference between adsorption and absorption is incorrect?
A)
Adsorption is a surface whereas absorption is a bulk phenomena. done
clear
B)
Water vapours are absorbed by anhydrous \[CaC{{l}_{2}}\] but adsorbed by silica gel. done
clear
C)
Adsorption and absorption take place individually. They can not occur simultaneously. done
clear
D)
All of the above statements are correct. done
clear
View Solution play_arrow
-
question_answer41)
In Langmuir's model of adsorption of a gas on a solid surface
A)
the mass of gas striking a given area of surface is proportional to the pressure of the gas done
clear
B)
the mass of gas striking a given area of surface is independent of the pressure of the gas done
clear
C)
the rate of dissociation of adsorbed molecules from the surface does not depend on the surface covered done
clear
D)
the adsorption at a single site on the surface may involve multiple molecules at the same time. done
clear
View Solution play_arrow
-
question_answer42)
Which one of the following statements is not correct in respect of lyophilic sols?
A)
There is a considerable interaction between the dispersed phase and dispersion medium done
clear
B)
These are quite stable and are not easily coagulated done
clear
C)
They carry charge done
clear
D)
The particles are hydrated done
clear
View Solution play_arrow
-
question_answer43)
Lyophilic sols are more stable than lyophobic sols because:
A)
the colloidal particles have positive charge done
clear
B)
the colloidal particles have negative charge done
clear
C)
the colloidal particles are solvated done
clear
D)
there is strong electrostatic repulsion between the colloidal particles done
clear
View Solution play_arrow
-
question_answer44)
Select incorrect statement:
A)
Micelles are associated colloids done
clear
B)
The electrical charge on a colloid particle is indicated by electrophoresis done
clear
C)
Formation of micelles takes place above Kraft temperature done
clear
D)
Formation of micelles takes place below CMC done
clear
View Solution play_arrow
-
question_answer45)
Point out the false statement
A)
Brownian movement and Tyndall effect are shown by colloidal systems. done
clear
B)
Gold number is a measure of the protective power of a lyophillic colloid done
clear
C)
The colloidal solution of a liquid in liquid is called gel done
clear
D)
Hardy - Schuize rule is related with coagulation. done
clear
View Solution play_arrow
-
question_answer46)
Which gas will be adsorbed on a solid to greater extent?
A)
Having non - polar molecule done
clear
B)
Having highest critical temperature done
clear
C)
Having lowest critical temperature done
clear
D)
Having lowest critical pressure done
clear
View Solution play_arrow
-
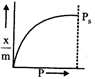
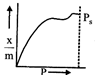
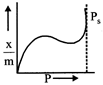
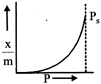
question_answer47)
Which of the following adsorption isotherms represents the absorption of a gas by a solid involving multilayers of formation? (\[{{P}_{s}}\]= saturation pressure)
A)
B)
C)
D)
View Solution play_arrow
-
question_answer48)
A detergent \[({{C}_{12}}{{H}_{25}}SO_{4}^{-}N{{a}^{+}})\] solution becomes a colloidal sol at a concentration of\[{{10}^{-3}}M\]. Onan average \[{{10}^{13}}\] colloidal particles are present in \[1\text{ }m{{m}^{3}}\]. What is the average number of ions which are contained by one colloidal particle (micelle)? [Given: \[{{N}_{A}}=6\times {{10}^{23}}mo{{l}^{-1}}\]]
A)
\[6\times {{10}^{7}}\] done
clear
B)
10 done
clear
C)
60 done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer49)
Which of the following is most powerful to coagulate the negative colloid?
A)
\[ZnS{{O}_{4}}\] done
clear
B)
\[N{{a}_{3}}P{{O}_{4}}\] done
clear
C)
\[AlC{{l}_{3}}\] done
clear
D)
\[{{K}_{4}}[Fe{{(CN)}_{6}}]\] done
clear
View Solution play_arrow
-
question_answer50)
Coagulation value of the electrolytes \[AlC{{l}_{3}}\] and \[NaCl\] for \[A{{s}_{2}}{{S}_{3}}\] sol are 0.093 and 52 respectively. How many times \[AlC{{l}_{3}}\] has greater coagulating power than\[NaCl\]?
A)
930 done
clear
B)
520 done
clear
C)
560 done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer51)
The protecting power of lyophilic colloidal sol is expressed in terms of:
A)
coagulation value done
clear
B)
gold number done
clear
C)
critical miscelle concentration done
clear
D)
oxidation number done
clear
View Solution play_arrow
-
question_answer52)
Which of the following statements is correct for the spontaneous adsorption of a gas?
A)
\[\Delta S\] is negative and, therefore, \[\Delta H\] should behighly positive done
clear
B)
\[\Delta S\] is negative and therefore, \[\Delta H\] should be highly negative done
clear
C)
\[\Delta S\] is positive and, therefore, \[\Delta H\] should be negative done
clear
D)
\[\Delta S\] is positive and therefore, \[\Delta H\] should also be highly positive done
clear
View Solution play_arrow
-
question_answer53)
Which property of colloids is not dependent on the charge on colloidal particles?
A)
Coagulation done
clear
B)
Electrophoresis done
clear
C)
Electro-osmosis done
clear
D)
Tyndall effect done
clear
View Solution play_arrow
-
question_answer54)
Smoke is an example of:
A)
Solid dispersed in solid done
clear
B)
Gas dispersed in liquid done
clear
C)
Solid dispersed in gas done
clear
D)
Gas dispersed in solid done
clear
View Solution play_arrow
-
question_answer55)
On addition of 1 mL of 10% \[NaCl\] solution to 10 mL gold sol in the presence of 0.025 g of starch, the coagulation is just prevented. Starch has gold number
A)
2.5 done
clear
B)
25 done
clear
C)
0.25 done
clear
D)
0.025 done
clear
View Solution play_arrow
-
question_answer56)
Fog is a colloidal solution of
A)
solid particles dispersed in gas done
clear
B)
solid particles dispersed in a liquid done
clear
C)
liquid particles dispersed in gas done
clear
D)
gaseous particles dispersed in a liquid done
clear
View Solution play_arrow
-
question_answer57)
Select incorrect statement:
A)
Soap and detergent lower the interfacial surface tension between oil and water done
clear
B)
Basic principle of peptization is reverse of coagulation done
clear
C)
Soap and detergent used as emulsifiers done
clear
D)
Lyophilic sols need stabilizing agent done
clear
View Solution play_arrow
-
question_answer58)
Which of the following is most effective in causing the coagulation of ferric hydroxide sol?
A)
KCl done
clear
B)
\[KN{{O}_{3}}\] done
clear
C)
\[{{K}_{2}}S{{O}_{4}}\] done
clear
D)
\[{{K}_{3}}[Fe{{(CN)}_{6}}]\] done
clear
View Solution play_arrow
-
question_answer59)
Aluminium hydroxide forms a positively charged sol. Which of the following ionic substances should be most effective in coagulating the sol?
A)
\[NaCl\] done
clear
B)
\[CaC{{l}_{2}}\] done
clear
C)
\[F{{e}_{2}}{{(S{{O}_{4}})}_{3}}\] done
clear
D)
\[{{K}_{3}}P{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer60)
Colloidal gold is prepared by
A)
Mechanical dispersion done
clear
B)
Peptisation done
clear
C)
Bredig's Arc method done
clear
D)
Hydrolisis done
clear
View Solution play_arrow
-
question_answer61)
Colloid of which one of the following can be prepared by electrical dispersion method as well as reduction method?
A)
Sulphur done
clear
B)
Ferric hydroxide done
clear
C)
Arsenious sulphide done
clear
D)
Gold done
clear
View Solution play_arrow
-
question_answer62)
The cause of Brownian movement is
A)
Heat changes in liquid state done
clear
B)
Convectional currents done
clear
C)
The impact of molecules of the dispersion medium on the colloidal particles. done
clear
D)
Attractive forces between the colloidal particles and molecules of dispersion medium. done
clear
View Solution play_arrow
-
question_answer63)
During adsorption
A)
\[T\Delta S\] is positive done
clear
B)
\[\Delta H-T\Delta S\] is negative done
clear
C)
\[\Delta H\] is positive done
clear
D)
\[T\Delta S\] and \[\Delta G\] become zero done
clear
View Solution play_arrow
-
question_answer64)
Which of the following gas molecules have maximum value of enthalpy of physisorption?
A)
\[{{C}_{2}}{{H}_{6}}\] done
clear
B)
Ne done
clear
C)
\[{{H}_{2}}O\] done
clear
D)
\[{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer65)
Adsorption is accompanied by the evolution of heat. So according to Le-Chatelier principle the amount of substance adsorbed should
A)
increase with decrease in temperature done
clear
B)
increase with increase in temperature done
clear
C)
decrease with decrease in temperature done
clear
D)
decrease with increase in temperature done
clear
View Solution play_arrow
-
question_answer66)
In Freundlich adsorption isotherm, the value of 1/n is:
A)
between 0 and 1 in all cases done
clear
B)
between 2 and 4 in all cases done
clear
C)
1 in case of physical adsorption done
clear
D)
1 in case of chemisorption done
clear
View Solution play_arrow
-
question_answer67)
The adsorption of a gas on a solid surface varies with pressure of the gas in which of the following manner.
A)
Fast \[\to \] slow \[\to \] independent of the pressure done
clear
B)
Slow \[\to \] fast \[\to \] independent of the pressure done
clear
C)
Independent of the pressure \[\to \] fast \[\to \] slow done
clear
D)
Independent of the pressure \[\to \] slow \[\to \] fast done
clear
View Solution play_arrow
-
question_answer68)
Associated colloids:
A)
raise both the surface tension and viscosity of water done
clear
B)
lower both the surface tension and viscosity of water done
clear
C)
lower the surface tension and raise the viscosity of water done
clear
D)
have greater concentration at the surface layer than the bulk of the solution done
clear
View Solution play_arrow
-
question_answer69)
Hydrolysis of urea is an example of
A)
homogeneous catalysis done
clear
B)
heterogeneous catalysis done
clear
C)
biochemical catalysis done
clear
D)
zeolite catalysis done
clear
View Solution play_arrow
-
question_answer70)
When \[KCl{{O}_{3}}\] is heated, it decomposes into \[KCl+{{O}_{2}}\]. If some \[Mn{{O}_{2}}\] is added, the reaction goes much faster because
A)
\[Mn{{O}_{2}}\] decomposes to give \[{{O}_{2}}\] done
clear
B)
\[Mn{{O}_{2}}\] provides heat by reacting done
clear
C)
better contact is provided by \[Mn{{O}_{2}}\] done
clear
D)
\[Mn{{O}_{2}}\] acts as a catalyst done
clear
View Solution play_arrow

 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
