question_answer 1)
Adsorption of gases on solid surface is generally exothermic because [IIT JEE (Screening) 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 2)
Two mole of an ideal gas is expanded isothermally and reversibly from 1 litre ot 10 litre at \[300K\]. The enthalpy change (in \[kJ\]) for the process is [IIT JEE (Screening) 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 3)
Heat of neutralization of strong acid against strong base is constant and is equal to [EAMCET 1980; AFMC 1989; DPMT 1991; MP PET 1999; BHU 1999; MP PMT 1995]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 4)
The value of heat generated when 36.5 gm \[HCl\] and 40 gm of \[NaOH\] reacts during neutralization [NCERT 1984; CPMT 1993]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 5)
When a gas undergoes adiabatic expansion, it gets cooled due to [DCE 2002]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 6)
The heat content of a system is called
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 7)
To calculate the amount of work done in joules during reversible isothermal expansion of an ideal gas, the volume must be expressed in [KCET (Med.) 2001]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 8)
In an isobaric process, the ratio of heat supplied to the system \[(dQ)\] and work done by the system \[(dW)\] for diatomic gas is [AFMC 2002]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 9)
The enthalpy change for the reaction of 50.00 ml of ethylene with 50.00 ml of \[{{H}_{2}}\] at 1.5 atm pressure is \[\Delta H=-0.31\,kJ\]. The value of \[\Delta E\] will be [DPMT 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 10)
Enthalpy of solution of \[NaOH\] (solid) in water is \[-41.6\,kJ\,mo{{l}^{-1}}\]. When \[NaOH\] is dissolved in water, the temperature of water [UPSEAT 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 11)
In which of the following entropy decreases ? [CPMT 1988, 94; MP PMT 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 12)
For conversion C (graphite) ® C (diamond) the \[\Delta S\] is [MP PMT 2001; MP PET 2003]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 13)
For a reaction \[\Delta H=9.08\,kJ\,mo{{l}^{-1}}\] and \[\Delta S=35.7\,J{{K}^{-1}}mo{{l}^{-1}}\] Which of the following statements is correct for the reaction [AMU (Engg.) 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 14)
For a reaction to occur spontaneously [CBSE PMT 1995]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 15)
The total amount of energy in the universe is fixed, but [AMU (Engg.) 1999]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 16)
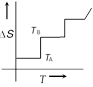
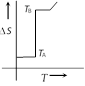
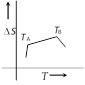
If for a given substance melting point is \[{{T}_{B}}\] and freezing point is \[{{T}_{A}}\], then correct variation shown by graph between entropy change and temperature is [DCE 2001]
A)
B)
C)
D)
View Solution play_arrow
question_answer 17)
In endothermic reaction, the value of change in enthalpy \[(\Delta H)\] is [Pb. CET 2001]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 18)
Which of the following would be expected to have the largest antropy per mole [MP PMT 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 19)
The enthalpies of formation of \[A{{l}_{2}}{{O}_{3}}\] and \[C{{r}_{2}}{{O}_{3}}\] are \[-1596\,kJ\] and \[-1134\,kJ\] respectively. \[\Delta H\] for the reaction \[2Al+C{{r}_{2}}{{O}_{3}}\to 2Cr+A{{l}_{2}}{{O}_{3}}\] is [KCET 2003]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 20)
Heat of reaction at constant volume is measured in the apparatus
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 21)
Which of the following gas has the highest heat of combustion
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 22)
The amount of heat measured for a reaction in a bomb calorimeter is [AIIMS 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 23)
For an endothermic reaction where \[\Delta H\] represents the enthalpy of the reaction in kJ/mole, the minimum value for the energy of activation will be [IIT JEE 1992; MP PET 1993]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 24)
The heat of neutralization of a strong acid by a strong base is nearly equal to [MP PET 1993; BCECE 2005]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 25)
A Beckmann thermometer is used to measure [Kurukshetra CEE 2002]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 26)
The heat required to raise the temperature of a body by 1 K is called [AIEEE 2002]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 27)
Mechanical work is specially important in system that contain [Kerala (Med.) 2002]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
E)
done
clear
View Solution play_arrow
question_answer 28)
?The quantity of heat which must be supplied to decompose a compound into its element is equal to the heat evolved during the formation of that compound from the elements.? This statement is known as
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 29)
Hess law deals with [BHU 1979]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 30)
For which one of the following reactions, \[\Delta H\] is not equal to \[\Delta E\] [IIT JEE 1995]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 31)
The heat liberated when 1.89 g of benzoic acid is burnt in a bomb calorimeter at \[{{25}^{o}}C\] increases the temperature of 18.94 kg of water by \[{{0.632}^{o}}C\].If the specific heat of water at \[{{25}^{o}}C\] is 0.998 cal/g-deg, the value of the heat combustion of benzoic acid is [CPMT 1999; BHU 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 32)
For a hypothetic reaction \[A\to B\], the activation energies for forward and backward reactions are 19 kJ/mole and 9 kJ/mole respectively. The heat of reaction is [CBSE PMT 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 33)
The enthalpy of neutralization of \[HCN\] by \[NaOH\] is \[-12.13\,kJ\,mo{{l}^{-1}}\]. The enthalpy of ionisation of \[HCN\] will be [BHU 1997]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 34)
In thermodynamics, a process is called reversible when [AIIMS 2001]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 35)
Which of the following unit represents largest amount of energy [AMU 2001]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 36)
Which of the following will have the highest bond energy [MP PMT 1990]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 37)
\[{{C}_{\text{(graphite)}}}+{{O}_{2}}(g)\,\to \,C{{O}_{2}}(g)\] \[\Delta H=-94.05\,kcal\,mo{{l}^{-1}}\] \[{{C}_{\text{(diamond)}}}+{{O}_{2}}(g)\,\to \,C{{O}_{2}}(g);\,\]\[\Delta H=-94.50\,kcal\,mo{{l}^{-1}}\] therefore [DPMT 2001]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 38)
Enthalpy of formation of two compounds x and y are ? 84 kJ and ? 156 kJ respectively. Which of the following statements is correct [CBSE PMT 2001]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 39)
For the process dry ice \[\xrightarrow{{}}\,C{{O}_{2}}(g)\] [KCET 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 40)
For melting of 3 moles of water at \[{{0}^{o}}C\] the \[\Delta {{G}^{o}}\] is [MP PMT 2001]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 41)
The equilibrium constant of a reaction at \[298\,K\] is \[5\times {{10}^{-3}}\] and at \[1000\,K\] is \[2\times {{10}^{-5}}\]. What is the sign of \[\Delta H\] for the reaction [Pb. CET 1998]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 42)
Which of the following has lowest fusion temperature [MP PET 2003]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 43)
Consider the reactions \[C(s)+2{{H}_{2}}(g)\to C{{H}_{4}}(g),\Delta H=-x\,kcal\] \[C(g)+4H(g)\to C{{H}_{4}}(g),\,\Delta H=-{{x}_{1}}\,kcal\] \[C{{H}_{4}}(g)\to C{{H}_{3}}(g)+H(g),\,\Delta H=+y\,kcal\] The bond energy of \[C-H\] bond is [JIPMER 1997]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 44)
Given the bond energies \[N\equiv N,\,\,H-H\] and \[N-H\] bonds are \[945,\,436\] and \[391\,kJ\,mol{{e}^{-1}}\] respectively, the enthalpy of the following reaction \[{{N}_{2}}(g)+3{{H}_{2}}(g)\to 2N{{H}_{3}}(g)\] is [EAMCET 1992; JIPMER 1997]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 45)
The relation between \[\Delta G\] and \[E\] for a cell is \[\Delta G=-nFE\]; the cell reaction will be spontaneous if [MP PET 1995]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 46)
Which of the following is not a correct statement? [AMU (Engg.) 2002]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 47)
If at 298 K the bond energies of \[C-H,\,C-C,\] \[C=C\] and \[H-H\] bonds are respectively 414, 347, 615 and 435 kJ \[mo{{l}^{-1}}\], the value of enthalpy change for the reaction \[{{H}_{2}}C=C{{H}_{2}}(g)+{{H}_{2}}(g)\to {{H}_{3}}C-C{{H}_{3}}(g)\] at 298 K will be [AIEEE 2003]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 48)
The equation \[\frac{1}{2}{{H}_{2}}+\frac{1}{2}C{{l}_{2}}\to HCl\] (\[\Delta {{H}_{298}}=-22060\,kcal\]) means [CMC Vellore 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
E)
done
clear
View Solution play_arrow
question_answer 49)
The \[{{H}_{2}}O(g)\] molecule dissociates as (i) \[{{H}_{2}}O(g)\to H(g)+OH(g);\,\Delta H=490\,kJ\] (ii) \[OH(g)\to H(g)+O(g);\,\Delta H=424\,kJ\] The average bond energy (in kJ) for water is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 50)
When \[50c{{m}^{3}}\] of \[0.2\,N\,{{H}_{2}}S{{O}_{4}}\] is mixed with \[50c{{m}^{3}}\] of \[1\,N\,KOH\], the heat liberated is [KCET 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 51)
Following reaction occurring in an automobile \[2{{C}_{8}}{{H}_{18}}(g)+25{{O}_{2}}(g)\to 16C{{O}_{2}}(g)+18{{H}_{2}}O(g).\] The sign of \[\Delta H,\,\Delta S\] and \[\Delta G\] would be [CBSE PMT 1994; KCET 1999]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 52)
For the reaction \[{{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\,\,\to \,\,{{H}_{2}}O(l),\,\Delta H=-285.8\,\,kJ\,\,mo{{l}^{-1}}\]\[\Delta S=-0.163\,kJ\,mo{{l}^{-1}}{{K}^{-1}}.\] What is the value of free energy change at \[{{27}^{o}}C\] for the reaction? [KCET 1999]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
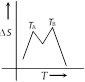 done
clear
done
clear
