-
question_answer1)
A glass flask is filled up to a mark with 50 cc of mercury at 18ºC. If the flask and contents are heated to 38ºC, how much mercury will be above the mark? (a for glass is 9 × 10?6/ºC and coefficient of real expansion of mercury is 180 × 10?6/ºC) [EAMCET 1997]
A)
0.85 cc done
clear
B)
0.46 cc done
clear
C)
0.153 cc done
clear
D)
0.05 cc done
clear
View Solution play_arrow
-
question_answer2)
The coefficient of apparent expansion of mercury in a glass vessel is 153 × 10?6/ºC and in a steel vessel is 144 × 10?6/ºC. If a for steel is 12 × 10?6/ºC, then that of glass is [EAMCET 1997]
A)
9 × 10?6/ºC done
clear
B)
6 × 10?6/ºC done
clear
C)
36 × 10?6/ºC done
clear
D)
27 × 10?6/ºC done
clear
View Solution play_arrow
-
question_answer3)
Solids expand on heating because [CPMT 1990]
A)
Kinetic energy of the atoms increases done
clear
B)
Potential energy of the atoms increases done
clear
C)
Total energy of the atoms increases done
clear
D)
The potential energy curve is asymmetric about the equilibrium distance between neighbouring atoms done
clear
View Solution play_arrow
-
question_answer4)
An iron tyre is to be fitted on to a wooden wheel 1m in diameter. The diameter of tyre is 6 mm smaller than that of wheel. The tyre should be heated so that its temperature increases by a minimum of (the coefficient of cubical expansion of iron is 3.6 × 10?5/ºC) [CPMT 1989]
A)
167ºC done
clear
B)
334ºC done
clear
C)
500ºC done
clear
D)
1000ºC done
clear
View Solution play_arrow
-
question_answer5)
A glass flask of volume one litre at 0°C is filled, level full of mercury at this temperature. The flask and mercury are now heated to 100°C. How much mercury will spill out, if coefficient of volume expansion of mercury is \[1.82\times {{10}^{-4}}/{}^\circ C\] and linear expansion of glass is\[0.1\times {{10}^{-4}}/{}^\circ C\] respectively [MNR 1994]
A)
21.2 cc done
clear
B)
15.2 cc done
clear
C)
1.52 cc done
clear
D)
2.12 cc done
clear
View Solution play_arrow
-
question_answer6)
A steel scale measures the length of a copper wire as \[80.0\,cm,\] when both are at \[20{}^\circ C\] (the calibration temperature for scale). What would be the scale read for the length of the wire when both are at \[40{}^\circ C\]? (Given asteel \[=11\times {{10}^{-6}}\]per°C and acopper \[=17\times {{10}^{-6}}per\,{}^\circ C\]) [CPMT 2004]
A)
\[80.0096\,cm\] done
clear
B)
\[80.0272\,cm\] done
clear
C)
\[1\,cm\] done
clear
D)
\[25.2\,cm\] done
clear
View Solution play_arrow
-
question_answer7)
A bimetallic strip is formed out of two identical strips, one of copper and other of brass. The coefficients of linear expansion of the two metals are \[{{\alpha }_{C}}\] and \[{{\alpha }_{B}}\]. On heating, the temperature of the strip goes up by DT and the strip bends to form an arc of radius of curvature R. Then R is [IIT-JEE (Screening) 1999]
A)
Proportional to DT done
clear
B)
Inversely proportional to DT done
clear
C)
Proportional to \[|{{\alpha }_{B}}-{{\alpha }_{C}}|\] done
clear
D)
Inversely proportional to \[|{{\alpha }_{B}}-{{\alpha }_{C}}|\] done
clear
View Solution play_arrow
-
question_answer8)
Two metal strips that constitute a thermostat must necessarily differ in their [IIT-JEE 1992]
A)
Mass done
clear
B)
Length done
clear
C)
Resistivity done
clear
D)
Coefficient of linear expansion done
clear
View Solution play_arrow
-
question_answer9)
A metal ball immersed in alcohol weighs \[{{W}_{1}}\] at 0°C and \[{{W}_{2}}\] at 59°C. The coefficient of cubical expansion of the metal is less than that of alcohol. Assuming that the density of metal is large compared to that of alcohol, it can be shown that [CPMT 1998]
A)
\[{{W}_{1}}>{{W}_{2}}\] done
clear
B)
\[{{W}_{1}}={{W}_{2}}\] done
clear
C)
\[{{W}_{1}}<{{W}_{2}}\] done
clear
D)
\[{{W}_{2}}=({{W}_{1}}/2)\] done
clear
View Solution play_arrow
-
question_answer10)
The coefficient of volumetric expansion of mercury is 18 × 10?5/ºC. A thermometer bulb has a volume 10?6 m3 and cross section of stem is 0.004 cm2. Assuming that bulb is filled with mercury at 0ºC then the length of the mercury column at 100ºC is [Pb. PMT 1998, DPMT 1997, 2001]
A)
18.8 mm done
clear
B)
9.2 mm done
clear
C)
7.4 cm done
clear
D)
4.5 cm done
clear
View Solution play_arrow
-
question_answer11)
A piece of metal weight 46 gm in air, when it is immersed in the liquid of specific gravity 1.24 at 27ºC it weighs 30 gm. When the temperature of liquid is raised to 42ºC the metal piece weight 30.5 gm, specific gravity of the liquid at 42ºC is 1.20, then the linear expansion of the metal will be [BHU 1995]
A)
3.316 × 10?5/ºC done
clear
B)
2.316 × 10?5/ºC done
clear
C)
4.316 × 10?5/ºC done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer12)
It is known that wax contracts on solidification. If molten wax is taken in a large vessel and it is allowed to cool slowly, then [CBSE PMT 1994]
A)
It will start solidifying from the top downward done
clear
B)
It will start solidifying from the bottom upward done
clear
C)
It will start solidifying from the middle, upward and downward at equal rates done
clear
D)
The whole mass will solidify simultaneously done
clear
View Solution play_arrow
-
question_answer13)
A substance of mass m kg requires a power input of P watts to remain in the molten state at its melting point. When the power is turned off, the sample completely solidifies in time t sec. What is the latent heat of fusion of the substance [IIT JEE 1992]
A)
\[\frac{Pm}{t}\] done
clear
B)
\[\frac{Pt}{m}\] done
clear
C)
\[\frac{m}{Pt}\] done
clear
D)
\[\frac{t}{Pm}\] done
clear
View Solution play_arrow
-
question_answer14)
Steam at 100ºC is passed into 1.1 kg of water contained in a calorimeter of water equivalent 0.02 kg at 15ºC till the temperature of the calorimeter and its contents rises to 80ºC. The mass of the steam condensed in kg is [IIT 1995]
A)
0.130 done
clear
B)
0.065 done
clear
C)
0.260 done
clear
D)
0.135 done
clear
View Solution play_arrow
-
question_answer15)
2 kg of ice at ? 20°C is mixed with 5 kg of water at 20°C in an insulating vessel having a negligible heat capacity. Calculate the final mass of water remaining in the container. It is given that the specific heats of water and ice are 1 kcal/kg per °C and 0.5 kcal/kg/°C while the latent heat of fusion of ice is 80 kcal/kg [IIT-JEE (Screening) 2003]
A)
7 kg done
clear
B)
6 kg done
clear
C)
4 kg done
clear
D)
2 kg done
clear
View Solution play_arrow
-
question_answer16)
Water of volume 2 litre in a container is heated with a coil of \[1\,kW\] at \[27\,{}^\circ C\]. The lid of the container is open and energy dissipates at rate of \[160\,J/s.\] In how much time temperature will rise from \[27\,{}^\circ C\] to \[77\,{}^\circ C\] [Given specific heat of water is \[4.2\,kJ/kg\]] [IIT-JEE (Screening) 2004]
A)
8 min 20 s done
clear
B)
6 min 2 s done
clear
C)
7 min done
clear
D)
14 min done
clear
View Solution play_arrow
-
question_answer17)
A lead bullet at 27°C just melts when stopped by an obstacle. Assuming that 25% of heat is absorbed by the obstacle, then the velocity of the bullet at the time of striking (M.P. of lead = 327°C, specific heat of lead = 0.03 cal/gm°C, latent heat of fusion of lead = 6 cal/gm and J = 4.2 joule/cal) [IIT 1981]
A)
410 m/sec done
clear
B)
1230 m/sec done
clear
C)
307.5 m/sec done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer18)
If two balls of same metal weighing 5 gm and 10 gm strike with a target with the same velocity. The heat energy so developed is used for raising their temperature alone, then the temperature will be higher
A)
For bigger ball done
clear
B)
For smaller ball done
clear
C)
Equal for both the balls done
clear
D)
None is correct from the above three done
clear
View Solution play_arrow
-
question_answer19)
The temperature of equal masses of three different liquids A, B and C are 12ºC, 19ºC and 28ºC respectively. The temperature when A and B are mixed is 16ºC and when B and C are mixed is 23ºC. The temperature when A and C are mixed is [Kerala PET 2005]
A)
18.2ºC done
clear
B)
22ºC done
clear
C)
20.2ºC done
clear
D)
25.2ºC done
clear
View Solution play_arrow
-
question_answer20)
In an industrial process 10 kg of water per hour is to be heated from 20°C to 80°C. To do this steam at 150°C is passed from a boiler into a copper coil immersed in water. The steam condenses in the coil and is returned to the boiler as water at 90°C. how many kg of steam is required per hour. (Specific heat of steam = 1 calorie per gm°C, Latent heat of vaporisation = 540 cal/gm)
A)
1 gm done
clear
B)
1 kg done
clear
C)
10 gm done
clear
D)
10 kg done
clear
View Solution play_arrow
-
question_answer21)
In a vertical U-tube containing a liquid, the two arms are maintained at different temperatures \[{{t}_{1}}\] and \[{{t}_{2}}\]. The liquid columns in the two arms have heights \[{{l}_{1}}\] and \[{{l}_{2}}\] respectively. The coefficient of volume expansion of the liquid is equal to

A)
\[\frac{{{l}_{1}}-{{l}_{2}}}{{{l}_{2}}{{t}_{1}}-{{l}_{1}}{{t}_{2}}}\] done
clear
B)
\[\frac{{{l}_{1}}-{{l}_{2}}}{{{l}_{1}}{{t}_{1}}-{{l}_{2}}{{t}_{2}}}\] done
clear
C)
\[\frac{{{l}_{1}}+{{l}_{2}}}{{{l}_{2}}{{t}_{1}}+{{l}_{1}}{{t}_{2}}}\] done
clear
D)
\[\frac{{{l}_{1}}+{{l}_{2}}}{{{l}_{1}}{{t}_{1}}+{{l}_{2}}{{t}_{2}}}\] done
clear
View Solution play_arrow
-
question_answer22)
The coefficient of linear expansion of crystal in one direction is \[{{\alpha }_{1}}\] and that in every direction perpendicular to it is \[{{\alpha }_{2}}\]. The coefficient of cubical expansion is
A)
\[{{\alpha }_{1}}+{{\alpha }_{2}}\] done
clear
B)
\[2{{\alpha }_{1}}+{{\alpha }_{2}}\] done
clear
C)
\[{{\alpha }_{1}}+2{{\alpha }_{2}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer23)
Three rods of equal length l are joined to form an equilateral triangle PQR. O is the mid point of PQ. Distance OR remains same for small change in temperature. Coefficient of linear expansion for PR and RQ is same i.e. \[{{\alpha }_{2}}\] but that for PQ is \[{{\alpha }_{1}}\]. Then
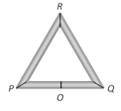
A)
\[{{\alpha }_{2}}=3{{\alpha }_{1}}\] done
clear
B)
\[{{\alpha }_{2}}=4{{\alpha }_{1}}\] done
clear
C)
\[{{\alpha }_{1}}=3{{\alpha }_{2}}\] done
clear
D)
\[{{\alpha }_{1}}=4{{\alpha }_{2}}\] done
clear
View Solution play_arrow
-
question_answer24)
A one litre glass flask contains some mercury. It is found that at different temperatures the volume of air inside the flak remains the same. What is the volume of mercury in this flask if coefficient of linear expansion of glass is 9 ´ 10?6/°C while of volume expansion of mercury is 1.8 ´ 10?4/°C
A)
50 cc done
clear
B)
100 cc done
clear
C)
150 cc done
clear
D)
200 cc done
clear
View Solution play_arrow
-
question_answer25)
10 gm of ice at ? 20°C is dropped into a calorimeter containing 10 gm of water at 10°C; the specific heat of water is twice that of ice. When equilibrium is reached, the calorimeter will contain
A)
20 gm of water done
clear
B)
20 gm of ice done
clear
C)
10 gm ice and 10 gm water done
clear
D)
5 gm ice and 15 gm water done
clear
View Solution play_arrow
-
question_answer26)
A rod of length 20 cm is made of metal. It expands by 0.075cm when its temperature is raised from 0°C to 100°C. Another rod of a different metal B having the same length expands by 0.045 cm for the same change in temperature. A third rod of the same length is composed of two parts, one of metal A and the other of metal B. This rod expands by 0.060 cm for the same change in temperature. The portion made of metal A has the length [CPMT 1991]
A)
20 cm done
clear
B)
10 cm done
clear
C)
15 cm done
clear
D)
18 cm done
clear
View Solution play_arrow
-
question_answer27)
Steam is passed into 22 gm of water at 20°C. The mass of water that will be present when the water acquires a temperature of 90°C (Latent heat of steam is 540 cal/gm) is [SCRA 1994]
A)
24.8 gm done
clear
B)
24 gm done
clear
C)
36.6 gm done
clear
D)
30 gm done
clear
View Solution play_arrow
