The unit of Stefan's constant\[\sigma \]is
A)
\[W{{m}^{-2}}{{K}^{-1}}\]
done
clear
B)
\[W{{m}^{2}}{{K}^{-4}}\]
done
clear
C)
\[W{{m}^{-2}}{{K}^{-4}}\]
done
clear
D)
\[W{{m}^{-2}}{{K}^{4}}\]
done
clear
View Answer play_arrow
The displacement of a particle is given by \[y=a+bt+c{{t}^{2}}-d{{t}^{4}}.\]The initial velocity and acceleration are respectively
A)
\[b,-4d\]
done
clear
B)
\[-b,2c\]
done
clear
C)
\[b,2c\]
done
clear
D)
\[2c,-4d\]
done
clear
View Answer play_arrow
A particle [A] is dropped from a height and another particle (B) is thrown into horizontal direction with speed of 5 m/sec from the same height. The correct statement is
A)
Both particles will reach at ground simultaneously
done
clear
B)
Both particles will reach at ground with the same speed
done
clear
C)
Particle [A] will reach at ground first with respect to particle [B]
done
clear
D)
Particle [B] will reach at ground first with respect to particle [A]
done
clear
View Answer play_arrow
A vehicle of mass in is moving on a rough horizontal road with momentum P. If the coefficient of friction between the tyres and the road be\[\mu ,\] then the stopping distance is:
A)
\[\frac{P}{2\,\mu \,mg}\]
done
clear
B)
\[\frac{{{P}^{2}}}{2\,\mu \,mg}\]
done
clear
C)
\[\frac{P}{2\,\mu \,{{m}^{2}}g}\]
done
clear
D)
\[\frac{{{P}^{2}}}{2\,\mu \,{{m}^{2}}g}\]
done
clear
View Answer play_arrow
A force \[(\vec{F})=3\hat{i}+c\hat{j}+2\hat{k}\]acting on a particle causes a displacement: \[(\vec{s})=-4\hat{i}+2\hat{j}+3\hat{k}\]in its own direction. If the work done is 6 J, then the value of 'c' is
A)
0
done
clear
B)
1
done
clear
C)
6
done
clear
D)
12
done
clear
View Answer play_arrow
A constant torque of 1000 N-m turns a wheel of moment of inertia\[200\,kg-{{m}^{2}}\] about an axis through its centre. Its angular velocity after 3 seconds is
A)
1 rad/s
done
clear
B)
5 rad/s
done
clear
C)
10 rad/s
done
clear
D)
15 rad/s
done
clear
View Answer play_arrow
The moment of inertia of a uniform circular disc of radius R and mass M about an axis passing from the edge of the disc and normal to the disc is
A)
\[\frac{1}{2}M{{R}^{2}}\]
done
clear
B)
\[M{{R}^{2}}\]
done
clear
C)
\[\frac{7}{2}M{{R}^{2}}\]
done
clear
D)
\[\frac{3}{2}M{{R}^{2}}\]
done
clear
View Answer play_arrow
Potential energy of a satellite having mass m and rotating at a height of \[6.4\times {{10}^{6}}\,m\]from the earth surface is
A)
\[-0.5\,mg{{R}_{e}}\]
done
clear
B)
\[-mg{{R}_{e}}\]
done
clear
C)
\[-2\,mg{{R}_{e}}\]
done
clear
D)
\[4\,mg\,{{R}_{e}}\]
done
clear
View Answer play_arrow
Two satellites of earth, \[{{S}_{1}}\]and \[{{S}_{2}},\]are moving in the same orbit. The mass of \[{{S}_{1}}\] is four times the mass of \[{{S}_{2}}.\]Which one of the following statements is true?
A)
The time period of \[{{S}_{1}}\] is four times that of \[{{S}_{2}}\]
done
clear
B)
The potential energies of earth and satellite in the two cases are equal
done
clear
C)
\[{{S}_{1}}\]and \[{{S}_{2}}\]are moving with the same speed
done
clear
D)
The kinetic energies of the two satellites are equal
done
clear
View Answer play_arrow
Which one of the following plots represents the variation of gravitational field on a particle with distance r due to a thin spherical shell of radius R? (r is measured from the centre of the spherical shell).
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Two droplets merge with each other and forms a large droplet. In this process
A)
Energy is liberated
done
clear
B)
Energy is absorbed
done
clear
C)
Neither liberated nor absorbed
done
clear
D)
Some mass is converted into energy
done
clear
View Answer play_arrow
A certain number of spherical drops of a liquid of radius r coalesce to form a single drop of radius Rand volume V. If T is the surface tension. of the liquid, then
A)
Energy \[=4VT\left( \frac{1}{r}-\frac{1}{R} \right)\]is released
done
clear
B)
Energy \[=3VT\left( \frac{1}{r}+\frac{1}{R} \right)\] is absorbed
done
clear
C)
Energy \[=3VT\left( \frac{1}{r}-\frac{1}{R} \right)\]is released
done
clear
D)
Energy is neither released nor absorbed
done
clear
View Answer play_arrow
A black body has maximum wavelength\[{{\lambda }_{m}}\]at temperature 2000 K. Its corresponding wave- length at temperature 3000 K will be
A)
\[\frac{3}{2}{{\lambda }_{m}}\]
done
clear
B)
\[\frac{2}{3}{{\lambda }_{m}}\]
done
clear
C)
\[\frac{4}{9}{{\lambda }_{m}}\]
done
clear
D)
\[\frac{9}{4}{{\lambda }_{m}}\]
done
clear
View Answer play_arrow
A steel scale measures the length of a copper wire as 80.0 cm, when both are at \[20{}^\circ C\] (the calibration temperature for scale). What would be the scale read for the length of the wire when both are at \[40{}^\circ C\]? (Given \[{{\alpha }_{steel}}=11\times {{10}^{-6}}\]per \[{{\,}^{o}}C\] and \[{{\alpha }_{copper}}=17\times {{10}^{-6}}\]per\[{{\,}^{o}}C\])
A)
80.0096 cm
done
clear
B)
80.0272 cm
done
clear
C)
1 cm
done
clear
D)
25.2 cm
done
clear
View Answer play_arrow
The temperature of sink of Carnot engine is \[27{}^\circ \], Efficiency of engine is 25%. Then temperature of source is
A)
\[{{227}^{o}}C\]
done
clear
B)
\[{{327}^{o}}C\]
done
clear
C)
\[{{127}^{o}}C\]
done
clear
D)
\[{{27}^{o}}C\]
done
clear
View Answer play_arrow
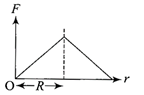
A thermodynamic system is taken through the cycle ABCD as shown in the figure. Heat rejected by the gas during the cycle is
A)
PV
done
clear
B)
2 PV
done
clear
C)
4 PV
done
clear
D)
\[{\scriptstyle{}^{1}/{}_{2}}PV\]
done
clear
View Answer play_arrow
The amount of heat energy required to raise the temperature of 1 g of Helium at NTP, from \[{{T}_{1}}K\] to \[{{T}_{2}}K\]is
A)
\[\frac{3}{8}{{N}_{a}}{{k}_{B}}({{T}_{2}}-{{T}_{1}})\]
done
clear
B)
\[\frac{3}{2}{{N}_{a}}{{k}_{B}}({{T}_{2}}-{{T}_{1}})\]
done
clear
C)
\[\frac{3}{4}{{N}_{a}}{{k}_{B}}({{T}_{2}}-{{T}_{1}})\]
done
clear
D)
\[\frac{3}{4}{{N}_{a}}{{k}_{B}}\left( \frac{{{T}_{2}}}{{{T}_{1}}} \right)\]
done
clear
View Answer play_arrow
An ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
A)
Isothermal
done
clear
B)
Adiabatic
done
clear
C)
Isobaric
done
clear
D)
Isochoric
done
clear
View Answer play_arrow
A hollow sphere is filled with water through a small hole in it. It is then hung by a long thread and made to oscillate. As the water slowly flows out of the hole at the bottom, the period of oscillation will
A)
continuously decrease
done
clear
B)
continuously increase
done
clear
C)
first decrease and then increase to original value
done
clear
D)
first increase and then decrease to original value
done
clear
View Answer play_arrow
A rectangular block of mass m and area of cross-section A floats in a liquid of density \[\rho .\] If it is given a small vertical displacement from equilibrium it undergoes oscillation with a time period T then:
A)
\[T\propto \sqrt{\rho }\]
done
clear
B)
\[T\propto \frac{1}{\sqrt{A}}\]
done
clear
C)
\[T\propto \frac{1}{\rho }\]
done
clear
D)
\[T\propto \frac{1}{\sqrt{m}}\]
done
clear
View Answer play_arrow
The equation of a wave is represented by \[y={{10}^{-4}}\sin \left[ 100t-\frac{x}{10} \right].\]The velocity of the wave will be
A)
100 m/s
done
clear
B)
250 m/s
done
clear
C)
750 m/s
done
clear
D)
1000 m/s
done
clear
View Answer play_arrow
Two sound waves with wavelengths 5.0 m and 5.5m respectively, each propagates in a gas with velocity 330 m/s. We expect the following. number of beats per second:
A)
12
done
clear
B)
0
done
clear
C)
1
done
clear
D)
6
done
clear
View Answer play_arrow
Sound waves travel at 350 m/s through a warm air and at 3500 m/s through brass. The wave- length of a 700 Hz acoustic wave as it enters brass from warm air
A)
increases by a factor 20
done
clear
B)
increases by a factor 10
done
clear
C)
decreases by a factor 20
done
clear
D)
decreases by a factor 10
done
clear
View Answer play_arrow
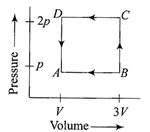
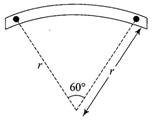
A source of sound S emitting waves of frequency 100 Hz and an observer O are located at some distance from each other. The source is moving with a speed of\[19.4\,m{{s}^{-1}}\]at an angle of \[60{}^\circ \] with the source observer line as shown in the figure. The observer is at rest. The apparent frequency observed by the observer (velocity of sound in air\[330\,m{{s}^{-1}}\]is
A)
97 Hz
done
clear
B)
100 Hz
done
clear
C)
103 Hz
done
clear
D)
106 Hz
done
clear
View Answer play_arrow
A charge q is located at the centre of a cube. The electric flux through any face is
A)
\[\frac{4\pi q}{6(4\pi {{\varepsilon }_{0}})}\]
done
clear
B)
\[\frac{4\pi q}{6(4\pi {{\varepsilon }_{0}})}\]
done
clear
C)
\[\frac{q}{6(4\pi {{\varepsilon }_{0}})}\]
done
clear
D)
\[\frac{2\pi q}{6(4\pi {{\varepsilon }_{0}})}\]
done
clear
View Answer play_arrow
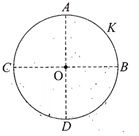
A thin conducting ring of radius R is given a charge +Q. The electric field at the centre O of the ring due the to the charge on the part AKB of the ring is E. The electric field at the centre due to the charge on the part ACDB of the ring is
A)
3E along KO
done
clear
B)
E along OK
done
clear
C)
E along KO
done
clear
D)
3E along OK
done
clear
View Answer play_arrow
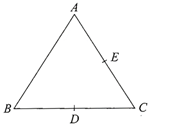
Three charges, each + q, are placed at the comers of an isosceles triangle ABC of sides BC and AC, 2a, D and E are the mid-points of BC and CA. The work done in talking a charge Q from D to E is
A)
\[\frac{eqQ}{8\pi \,{{\varepsilon }_{0}}a}\]
done
clear
B)
\[\frac{qQ}{4\pi {{\varepsilon }_{0}}a}\]
done
clear
C)
zero
done
clear
D)
\[\frac{3qQ}{4\pi {{\varepsilon }_{0}}a}\]
done
clear
View Answer play_arrow
The electric field in a certain region is acting radially outward and is given by \[E=Ar.\]A charge contained in a sphere of radius 'a' centred at the origin of the field will be given by
A)
\[4\pi {{\varepsilon }_{0}}A{{a}^{2}}\]
done
clear
B)
\[4{{\varepsilon }_{0}}{{a}^{2}}\]
done
clear
C)
\[4\pi {{\varepsilon }_{0}}A{{a}^{3}}\]
done
clear
D)
\[{{\varepsilon }_{0}}A{{a}^{2}}\]
done
clear
View Answer play_arrow
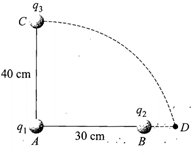
Two charges \[{{q}_{1}}\]and \[{{q}_{2}}\]are placed 30 cm apart, as shown in the figure. A third charge \[{{q}_{3}}\]is moved along the arc of a circle of radius 40 cm from C to D. The change in the potential energy of the system is \[\frac{{{q}_{3}}}{4\pi {{\varepsilon }_{0}}}k,\] where k is
A)
\[8{{q}_{2}}\]
done
clear
B)
\[8{{q}_{1}}\]
done
clear
C)
\[6{{q}_{2}}\]
done
clear
D)
\[6{{q}_{1}}\]
done
clear
View Answer play_arrow
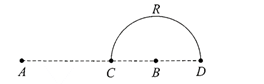
Charges +q and -q are placed at points A and B respectively which are a distance 2Z, apart, C is the midpoint between A and B. The work done in moving a charge +Q along the semicircle CRD is
A)
\[\frac{qQ}{4\pi {{\varepsilon }_{0}}L}\]
done
clear
B)
\[\frac{qQ}{2\pi {{\varepsilon }_{0}}L}\]
done
clear
C)
\[\frac{qQ}{6\pi {{\varepsilon }_{0}}L}\]
done
clear
D)
\[\frac{qQ}{6\pi {{\varepsilon }_{0}}L}\]
done
clear
View Answer play_arrow
A series combination of\[{{n}_{1}}\]capacitors, each of value \[{{C}_{1}},\]is charged by a source of potential difference 4 V. When another parallel combination of\[{{n}_{2}}\]capacitors, each of value\[{{C}_{2}}\] is charged by a source of potential difference V, it has the same (total) energy stored in it, as the first combination has. The value of \[{{C}_{2}},\]in terms of \[{{C}_{1}},\]is then
A)
\[\frac{2{{C}_{1}}}{{{n}_{1}}\,{{n}_{2}}}\]
done
clear
B)
\[16\frac{{{n}_{2}}}{{{n}_{1}}}{{C}_{1}}\]
done
clear
C)
\[2\frac{{{n}_{2}}}{{{n}_{1}}}{{C}_{1}}\]
done
clear
D)
\[\frac{16\,{{C}_{1}}}{{{n}_{1}}\,{{n}_{2}}}\]
done
clear
View Answer play_arrow
Two thin dielectric slabs of dielectric constants \[{{K}_{1}}\]and \[{{K}_{2}}(K<{{K}_{2}})\]axe inserted between plates of aparallel plate capacitor, as shown in the figure. The variation of electric field E between the plates with distance d as measured from plate P is correctly shown by
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
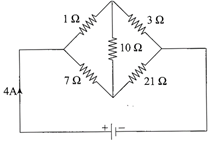
In the circuit shown in the figure, the current drawn from the battery is 4A. If\[10\,\Omega .\]resistor is replaced by \[20\,\Omega \]resistor, then current drawn from the circuit will be
A)
1 A
done
clear
B)
2 A
done
clear
C)
3 A
done
clear
D)
0 A
done
clear
View Answer play_arrow
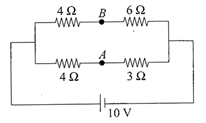
The potential difference between points A and B is
A)
\[\frac{20}{7}V\]
done
clear
B)
\[\frac{40}{7}V\]
done
clear
C)
\[\frac{10}{7}V\]
done
clear
D)
0
done
clear
View Answer play_arrow
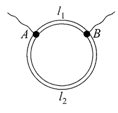
A ring is made of a wire having a resistance \[{{R}_{0}}=12\,\Omega .\]Find the points A and B, as shown in the figure, at which a current carrying conductor should be connected so that the resistance R of the sub-circuit between these points is equal to \[\frac{8}{3}\Omega .\]
A)
\[\frac{{{l}_{1}}}{{{l}_{2}}}=\frac{1}{2}\]
done
clear
B)
\[\frac{{{l}_{1}}}{{{l}_{2}}}=\frac{5}{8}\]
done
clear
C)
\[\frac{{{l}_{1}}}{{{l}_{2}}}=\frac{1}{3}\]
done
clear
D)
\[\frac{{{l}_{1}}}{{{l}_{2}}}=\frac{3}{8}\]
done
clear
View Answer play_arrow
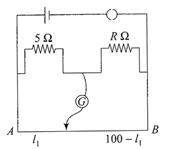
The resistances in the two arms of the meter bridge are\[5\Omega \]and \[R\Omega ,\]respectively. When the resistance R is shunted with an equal resistance, the new balance point is at \[1.6{{l}_{1}}.\] The resistance R is
A)
\[10\,\Omega \]
done
clear
B)
\[15\,\Omega \]
done
clear
C)
\[20\,\Omega \]
done
clear
D)
\[25\,\Omega \]
done
clear
View Answer play_arrow
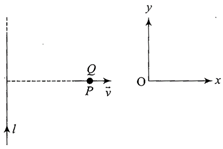
A very long straight wire carries a current I. At the instant when a charge + Q at point P has velocity \[\vec{v},\]as shown, the force on the charge is
A)
opposite to ox
done
clear
B)
along ox
done
clear
C)
opposite to oy
done
clear
D)
along oy
done
clear
View Answer play_arrow
A bar magnet of length \[l\] and magnetic dipole moment M is bent in the form of an arc as shown in the figure. The new magnetic dipole moment will be
A)
M
done
clear
B)
\[\frac{3}{\pi }M\]
done
clear
C)
\[\frac{2}{\pi }M\]
done
clear
D)
\[\frac{M}{2}\]
done
clear
View Answer play_arrow
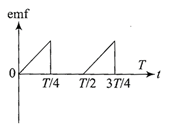
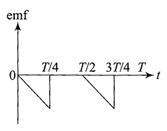
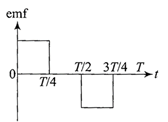
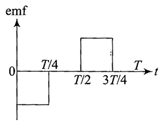
The current \[i\]in a coil varies with time as shown in the figure. The variation of induced emf with time would be
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
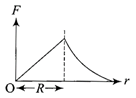
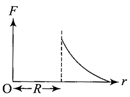
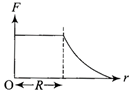
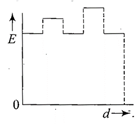
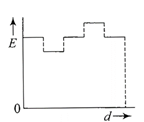
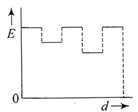
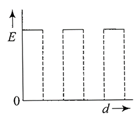
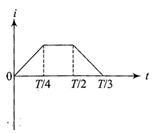
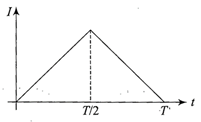
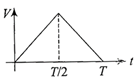
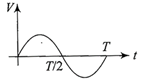
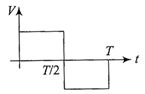
The current (I) in the inductance is varying with time according to the plot shown in figure.
Which one of the following is the correct variation of voltage with time in the coil?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following is positively charged?
A)
\[\alpha -\] particle
done
clear
B)
\[\beta -\]particle
done
clear
C)
\[\gamma -\]rays
done
clear
D)
X-rays
done
clear
View Answer play_arrow
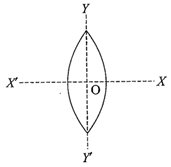
An equi-convex lens is cut into two halves along (i) XOX' and (ii) YOY' as shown in the figure. Let\[f,f',f''\] be the focal lengths of the complete lens, of each half in case (i) and of each half in case (ii), respectively
Choose the correct statement from the following
A)
\[f'=2f,f''=f\]
done
clear
B)
\[f'=f,f''=f\]
done
clear
C)
\[f'=2f,f''=2f\]
done
clear
D)
\[f'=f,f''=2f\]
done
clear
View Answer play_arrow
In the Young's double-slit experiment, the intensity of light at a point on the screen where the path difference is A is\[\lambda \](\[\lambda \]being the wave- length of light used). The intensity at a point where the path difference is \[\frac{\lambda }{4}\] will be
A)
K
done
clear
B)
\[\frac{K}{4}\]
done
clear
C)
\[\frac{K}{2}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
A photoelectric cell is illuminated by a point source of light 1 m away. When the source is shifted to 2 m then
A)
Number of electrons emitted is half the initial number
done
clear
B)
Each emitted electron carries half the initial energy
done
clear
C)
Number of electrons emitted is a quarter of the initial number
done
clear
D)
Each emitted electron carries one quarter of the initial energy
done
clear
View Answer play_arrow
Two particles of masses \[{{m}_{1}},{{m}_{2}}\]move with initial velocities\[{{u}_{1}}\]and\[{{u}_{2}}\]. On collision, one of the particles get excited to higher level, after absorbing energy e. If final velocities of particles be \[{{u}_{1}}\]and \[{{u}_{2}}\]then we must have
A)
\[{{m}_{1}}u_{1}^{2}+{{m}_{2}}u_{2}^{2}-\varepsilon ={{m}_{1}}\upsilon _{1}^{2}+{{m}_{2}}\upsilon _{2}^{2}\]
done
clear
B)
\[\frac{1}{2}{{m}_{1}}u_{1}^{2}+\frac{1}{2}{{m}_{2}}u_{2}^{2}=\frac{1}{2}{{m}_{1}}u_{1}^{2}\]\[+\frac{1}{2}{{m}_{2}}u_{2}^{2}-\varepsilon \]
done
clear
C)
\[\frac{1}{2}{{m}_{1}}u_{1}^{2}+\frac{1}{2}{{m}_{2}}u_{2}^{2}-\varepsilon =\frac{1}{2}{{m}_{1}}u_{1}^{2}\]\[+\frac{1}{2}{{m}_{2}}u_{2}^{2}\]
done
clear
D)
\[\frac{1}{2}m_{1}^{2}u_{1}^{2}+\frac{1}{2}m_{2}^{2}u_{2}^{2}+\varepsilon =\frac{1}{2}m_{1}^{2}u_{1}^{2}\]\[+\frac{1}{2}m_{2}^{2}u_{2}^{2}\]
done
clear
View Answer play_arrow
Specific volume of cylindrical virus particle is \[6.02\times {{10}^{-2}}\,cc/gm\]whose radius and length \[7\overset{\text{ }\!\!\grave{\ }\!\!\text{ o}}{\mathop{\text{A}}}\,\] and \[\text{10}\overset{\text{ }\!\!\grave{\ }\!\!\text{ o}}{\mathop{\text{A}}}\,\] respectively. If \[{{N}_{A}}=6.02\times {{10}^{23}},\]find molecular weight of virus:
A)
\[3.08\times {{10}^{3}}\,kg/mol\]
done
clear
B)
\[15.4\,kg/mol\]
done
clear
C)
\[1.54\times {{10}^{4}}\,kg/mol\]
done
clear
D)
\[3.08\times {{10}^{4}}\,kg/mol\]
done
clear
View Answer play_arrow
\[[Cr{{({{H}_{2}}O)}_{6}}]C{{l}_{3}}\](at no. of Cr = 24) has a magnetic moment of 3.83 B.M. The correct distribution of 3d electrons in the chromium of the complex
A)
\[3d_{xy}^{1},3d_{yz}^{1},3_{xz}^{1}\]
done
clear
B)
\[3_{xy}^{1},3d_{yz}^{1},3d_{{{z}^{2}}}^{1}\]
done
clear
C)
\[(3d_{{{x}^{2}}-{{y}^{2}}}^{1}),3d_{{{z}^{2}}}^{1},3d_{xz}^{1}\]
done
clear
D)
\[3d_{xy}^{1},(3d_{{{x}^{2}}-{{y}^{2}}}^{1}),3d_{yz}^{1}\]
done
clear
View Answer play_arrow
The formation of the oxide ion,\[{{O}^{2-}}(g),\]from oxygen atom requires first an exothermic and then an endothermic step as shown below:
\[O(g)+{{e}^{-}}\xrightarrow{{}}{{O}^{-}}(g);{{\Delta }_{f}}{{H}^{O-}}=-141\,kJ\,mo{{l}^{-}}\] \[{{O}^{-}}(g)+{{e}^{-}}\xrightarrow{{}}{{O}^{2-}}(g);{{\Delta }_{f}}{{H}^{O-}}=+780\,kJ\,mo{{l}^{-}}\] Thus the process of formation of \[{{O}^{2-}}\]in gas phase is neon. It is due to the fact that
A)
Oxygen is more electronegative
done
clear
B)
Addition of electron in oxygen results in larger size of the ion
done
clear
C)
Electron repulsion outweighs the stability gained by achieving noble gas configuration
done
clear
D)
\[{{O}^{-}}\]ion has comparatively smaller size than oxygen atom
done
clear
View Answer play_arrow
Among the following the pair in which the two species are not isostructural is
A)
\[Si{{F}_{4}}\]and \[S{{F}_{4}}\]
done
clear
B)
\[I{{O}^{-}}_{3}\]and\[Xe{{O}_{3}}\]
done
clear
C)
\[B{{H}^{+}}_{4}\]and\[N{{H}^{+}}_{4}\]
done
clear
D)
\[P{{F}^{-}}_{6}\]and \[S{{F}_{6}}\]
done
clear
View Answer play_arrow
A bubble of air is underwater at temperature \[{{15}^{o}}C\] and the pressure 1.5 bar. If the bubble rises to the surface where the temperature is \[{{25}^{o}}C\] and the pressure is 1.0 bar, what will happen to the volume of the bubble?
A)
Volume will become smaller by a factor of 0.70
done
clear
B)
Volume will become greater by a factor of 1.1
done
clear
C)
Volume will become greater by a factor of 1.6
done
clear
D)
Volume will become greater by a factor of 2.5
done
clear
View Answer play_arrow
For which one of the following equations is \[\Delta H_{reaction}^{O}\]equal to\[\Delta H_{f}^{o}\]the product?
A)
\[{{N}_{2}}(g)+{{O}_{3}}(g)\to {{N}_{2}}{{O}_{3}}(g)\]
done
clear
B)
\[C{{H}_{4}}(g)+2C{{l}_{2}}(g)\to C{{H}_{2}}C{{l}_{2}}(l)+2HCl(g)\]
done
clear
C)
\[Xe(g)+2{{i}_{2}}(g)\to Xe{{F}_{4}}(g)\]
done
clear
D)
\[2CO(g)+{{O}_{2}}(g)\to 2C{{O}_{2}}(g)\]
done
clear
View Answer play_arrow
If the value of equilibrium constant for a particular reaction is\[1.6\times {{10}^{12}},\]then at equilibrium the system will contain
A)
mostly products
done
clear
B)
similar amounts of reactants and products
done
clear
C)
all reactants
done
clear
D)
mostly reactants
done
clear
View Answer play_arrow
Solubility of\[M{{X}_{2}}\]type electrolytes is \[0.5\times {{10}^{-4}}\,mol/L,\]then find out\[{{K}_{sp}}\]of electrolytes.
A)
\[5\times {{10}^{-12}}\]
done
clear
B)
\[25\times {{10}^{-10}}\]
done
clear
C)
\[1\times {{10}^{-13}}\]
done
clear
D)
\[5\times {{10}^{-13}}\]
done
clear
View Answer play_arrow
A mixture of potassium chlorate, oxalic acid and sulphuric acid is heated. During the reaction which element undergoes maximum change in the oxidation number?
A)
Cl
done
clear
B)
C
done
clear
C)
S
done
clear
D)
H
done
clear
View Answer play_arrow
When \[{{H}_{2}}{{O}_{2}}\]is oxidised, the product is
A)
\[O{{H}^{-}}\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[{{O}^{2-}}\]
done
clear
D)
\[HO_{2}^{-}\]
done
clear
View Answer play_arrow
Property of the alkaline earth metals that increases with their atomic number
A)
solubility of their hydroxide in water
done
clear
B)
solubility of their sulphates in water
done
clear
C)
ionisation energy
done
clear
D)
electronegativity
done
clear
View Answer play_arrow
The stability of+1 oxidation state among Al, Ga, In and Ti increases in the sequence
A)
Ti < In < Ga < Al
done
clear
B)
In < Ti < Ga < Al
done
clear
C)
Ga < In < Al < Ti
done
clear
D)
Al < Ga < In < Ti
done
clear
View Answer play_arrow
An organic compound containing C, H and N have the percentage 40, 13.33 and 46.67 respectively. Its empirical formula may be:
A)
\[{{C}_{2}}{{H}_{7}}N\]
done
clear
B)
\[{{C}_{2}}{{H}_{7}}{{N}_{2}}\]
done
clear
C)
\[C{{H}_{4}}N\]
done
clear
D)
\[C{{H}_{5}}N\]
done
clear
View Answer play_arrow
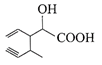
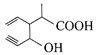
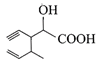
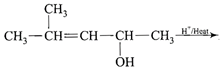
Structure of the compound whose IUPAC name is 3-ethyl-2-hydroxy-4-methylhex-3-en-5-ynoi acid is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Geometrical isomers differ in
A)
position of functional groups
done
clear
B)
position of atoms
done
clear
C)
spatial arrangement of atoms
done
clear
D)
length of carbon chain
done
clear
View Answer play_arrow
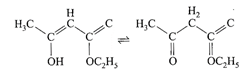
The enolic form of ethyl acetoacetate as shown below has
A)
9 sigma bonds and 2 pi-bonds
done
clear
B)
9 sigma bonds and 1 pi-bond
done
clear
C)
18 sigma bonds and 2 pi-bonds
done
clear
D)
16 sigma bonds and 1 pi-bond
done
clear
View Answer play_arrow
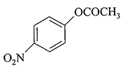
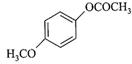
In the following reactions,
[a] [b]
A)
done
clear
B)
and
done
clear
C)
and
done
clear
D)
and
done
clear
View Answer play_arrow
The oxidation of benzene by\[{{V}_{2}}{{O}_{5}}\] in the presence of air produces
A)
benzoic acid
done
clear
B)
benzaldehyde
done
clear
C)
benzoic anhydride
done
clear
D)
maleic anhydride
done
clear
View Answer play_arrow
Which one of the following statement is not true?
A)
pH of drinking water should be between 5.5 and 9.5.
done
clear
B)
Concentration of DO below 6 ppm is good for the growth of fish.
done
clear
C)
Clean water would have a BOD value of less than 5 ppm.
done
clear
D)
Oxides of sulphur, nitrogen and carbon are the most widespread air pollutant.
done
clear
View Answer play_arrow
If species \[_{a}^{b}X\]first emits a position, then two a and two\[\beta \]and in last one a and finally converted to species \[_{c}^{d}Y,\]so the correct relation is
A)
\[c=a-5,d=b-12\]
done
clear
B)
\[c=a-6,d=b-8\]
done
clear
C)
\[c=a-4,d=b-12\]
done
clear
D)
\[c=a-5,d=b-8\]
done
clear
View Answer play_arrow
With which one of the following element silicon should be doped so as to give p-type semiconductor?
A)
As
done
clear
B)
Se
done
clear
C)
B
done
clear
D)
Ge
done
clear
View Answer play_arrow
The vacant space in bcc lattice unit cell is:
A)
23%
done
clear
B)
32%
done
clear
C)
26%
done
clear
D)
48%
done
clear
View Answer play_arrow
During osmosis, flow of water through a semi- permeable membrane is
A)
from both sides of semipermeable membrane with unequal flow rates
done
clear
B)
from solution having lower concentration only
done
clear
C)
from solution having higher concentration only
done
clear
D)
from both sides of semipermeable membrane with equal flow rates
done
clear
View Answer play_arrow
The boiling point of \[0.2\,mol\,k{{g}^{-1}}\] solution of X in water is greater than equimolal solution of Y in water. Which one of the following statements is true in the case?
A)
Molecular mass of X is less than the molecular mass of Y.
done
clear
B)
Y is undergoing dissociation in water while X undergoes no change.
done
clear
C)
X is undergoing dissociation in water.
done
clear
D)
Molecular mass of X is-greater than the molecular mass of Y.
done
clear
View Answer play_arrow
The emf of a Daniel cell at 298 K is \[{{E}_{1}}\]\[\underset{(0.01\,M)}{\mathop{Zn|ZnS{{O}_{4}}|}}\,\underset{(1.0\,M)}{\mathop{|CuS{{O}_{4}}|Cu}}\,\] when the concentration of\[ZnS{{O}_{4}}\]is 1.0 M and that of \[CuS{{O}_{4}}\] is 0.01 M, the emf changed to \[{{E}_{2}}.\]What is the relationship between \[{{E}_{1}}\]and\[{{E}_{2}}\]
A)
\[{{E}_{2}}=0\ne {{E}_{1}}\]
done
clear
B)
\[{{E}_{1}}>{{E}_{2}}\]
done
clear
C)
\[{{E}_{1}}<{{E}_{2}}\]
done
clear
D)
\[{{E}_{1}}={{E}_{2}}\]
done
clear
View Answer play_arrow
Kohlrausch's law states that at:
A)
Infinite dilution, each ion makes definite contribution to equivalent conductance of an electrolyte depending on the nature of the other ion of the electrolyte
done
clear
B)
Infinite dilution, each ion makes definite contribution to conductance of an electrolyte whatever be the nature of the other ion of the electrolyte
done
clear
C)
Infinite dilution, each ion makes definite contribution to equivalent conductance of an electrolyte, whatever be the nature of the other ion of the electrolyte
done
clear
D)
Finite dilution, each ion makes definite contribution to equivalent conductance of an electrolyte, whatever be the nature of the other ion electrolyte
done
clear
View Answer play_arrow
When \[\,0.1\,mol\,MnO_{4}^{2-}\]is oxidised the quantity of electricity required to completely oxidise\[MnO_{4}^{2-}\]to\[MnO_{4}^{-}\]is
A)
96500C
done
clear
B)
\[2\times 96500\,C\]
done
clear
C)
9650 C
done
clear
D)
96.50 C
done
clear
View Answer play_arrow
\[2A\to B+C.\]It would be a zero-order reaction when
A)
the rate of reaction is proportional to square of cone. of A
done
clear
B)
the rate of reaction remains same at any cone. of A
done
clear
C)
the rate remains unchanged at any cone. of B and C
done
clear
D)
the rate of reaction doubles if cone. of B is increased to double.
done
clear
View Answer play_arrow
In the reaction.\[Br{{O}^{-}}_{3}(aq)+5B{{r}^{-}}(aq)+6{{H}^{+}}\to 3B{{r}_{2}}(l)+3{{H}_{2}}O(l)\] The rate of appearance of bromine \[(B{{r}_{2}})\]Is related to rate of disappearance .of bromide ions as following:
A)
\[\frac{d(B{{r}_{2}})}{dt}=\frac{3}{5}\frac{d(B{{r}^{-}})}{dt}\]
done
clear
B)
\[\frac{d(B{{r}_{2}})}{dt}=-\frac{3}{5}\frac{d(B{{r}^{-}})}{dt}\]
done
clear
C)
\[\frac{d(B{{r}_{2}})}{dt}=-\frac{5}{3}\frac{d(B{{r}^{-}})}{dt}\]
done
clear
D)
\[\frac{d(B{{r}_{2}})}{dt}=\frac{5}{3}\frac{d(B{{r}^{-}})}{dt}\]
done
clear
View Answer play_arrow
The Langmuir adsorption isotherm is deduced using the assumption:
A)
The adsorption sites are equivalent in their ability to adsorb the particles
done
clear
B)
The heat of adsorption varies with coverage
done
clear
C)
The adsorbed molecules interact with each other
done
clear
D)
The adsorption takes place in multilayers
done
clear
View Answer play_arrow
"Metals are usually not found as nitrates in their ores." Out of the following two (I and II) reasons which is/are true for the above observation?
I. Metal nitrates are highly unstable. II. Metal nitrates are highly soluble in water.
A)
I is false but II is true
done
clear
B)
I is true but II is false.
done
clear
C)
I and II are true
done
clear
D)
I and II are false
done
clear
View Answer play_arrow
Strong reducing behaviour of\[{{H}_{3}}P{{O}_{2}}\]is due to:
A)
High oxidation state of phosphorus
done
clear
B)
Presence of two -OH groups and one P-H bond
done
clear
C)
Presence of one -OH group and two P-H bonds
done
clear
D)
High electron gain enthalpy of phosphorus
done
clear
View Answer play_arrow
Which one is responsible for depletion of ozone layer in the upper strata of the atmosphere?
A)
Polyhalogens
done
clear
B)
Ferrocene
done
clear
C)
Fullerenes
done
clear
D)
Freons
done
clear
View Answer play_arrow
A salt, which on heating with cone.\[{{H}_{2}}S{{O}_{4}}\]gives violet vapours, is
A)
Iodide
done
clear
B)
Nitrate
done
clear
C)
Sulphate
done
clear
D)
Bromide
done
clear
View Answer play_arrow
Which of the following gases exists more abundantly in nature than the others?
A)
Helium
done
clear
B)
Neon
done
clear
C)
Argon
done
clear
D)
Krypton
done
clear
View Answer play_arrow
Gadolinium belong to 4f series. Its atomic number is 64. Which of the following is the correct electronic configuration of gadolinium?
A)
\[[Xe]4{{f}^{7}}5{{d}^{1}}6{{s}^{2}}\]
done
clear
B)
\[[Xe]4{{f}^{6}}5{{d}^{2}}6{{s}^{2}}\]
done
clear
C)
\[[Xe]4{{f}^{8}}5{{d}^{2}}\]
done
clear
D)
\[[Xe]4{{f}^{9}}6{{s}^{1}}\]
done
clear
View Answer play_arrow
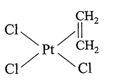
Which is considered to be an anticancer species?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
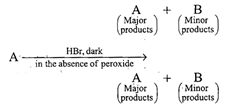
Which of the following reaction(s) can be used for the preparation of alkyl halides?
(i)\[C{{H}_{3}}C{{H}_{2}}OH+HCl\xrightarrow{anh.ZnC{{l}_{2}}}\] (ii)\[C{{H}_{3}}C{{H}_{2}}OH+HCl\xrightarrow{{}}\] (iii)\[{{(C{{H}_{3}})}_{3}}COH+HCl\xrightarrow{{}}\] (iv)\[{{(C{{H}_{3}})}_{2}}CHOH+HCl\xrightarrow{anh.ZnC{{l}_{2}}}\]
A)
(IV) only
done
clear
B)
(III) and (IV) only
done
clear
C)
(I), and (IV) only
done
clear
D)
(I) and (II) only
done
clear
View Answer play_arrow
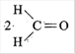
\[HO{{H}_{2}}C.C{{H}_{2}}OH\]on heating with periodic acid gives
A)
done
clear
B)
\[2C{{O}_{2}}\]
done
clear
C)
2 HCOOH
done
clear
D)
done
clear
View Answer play_arrow
Treatment of cyclopentanone with
methyl lithium gives which of the following species?
A)
Cyclopentanonyl radical
done
clear
B)
Cyclopentanonyl biradical
done
clear
C)
Cyclopentanonyl anion
done
clear
D)
Cyclopentanonyl cation
done
clear
View Answer play_arrow
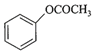
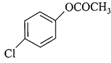
Which one of the following esters gets hydro- lysed most easily under alkaline conditions?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The hormone that helps in conversion of glucose to glycogen is:
A)
cortisone
done
clear
B)
bile salt
done
clear
C)
adrenaline
done
clear
D)
insulin
done
clear
View Answer play_arrow
Caprolactam is used for the manufacture of:
A)
Trylene
done
clear
B)
Nylon-6, 6
done
clear
C)
Bylon-6
done
clear
D)
Teflon
done
clear
View Answer play_arrow
Aspirin is an acetylation product of
A)
\[m-\]ydroxybenzoic acid
done
clear
B)
o-hydroxybenzoic acid
done
clear
C)
\[p-\]dihydroxybenzene
done
clear
D)
o-dihydroxybenzene
done
clear
View Answer play_arrow
If we use phenolphthalein as an indicator in a titration of \[N{{a}_{2}}C{{O}_{3}}\]with HCl, the usual result is
A)
No visible change will occur
done
clear
B)
The indicator reacts with the acid
done
clear
C)
The indicator reacts with the base
done
clear
D)
Sodium chloride and carbonic acid will be formed
done
clear
View Answer play_arrow
Which of the following is less general in characters as compared to genus?
A)
Species
done
clear
B)
Division
done
clear
C)
Class
done
clear
D)
Family
done
clear
View Answer play_arrow
Which fungal disease spreads by seed and flowers?
A)
Loose smut of Wheat
done
clear
B)
Corn stunt
done
clear
C)
Covered smut of Barley
done
clear
D)
Soft rot of Potato
done
clear
View Answer play_arrow
Which one of the following is living fossil?
A)
Moss
done
clear
B)
Saccharomyces
done
clear
C)
spirogyra
done
clear
D)
Cycas
done
clear
View Answer play_arrow
Sycon belongs to a group of animals, which are best described as:
A)
Unicellular or acellular
done
clear
B)
Multicellular without any tissue organization
done
clear
C)
Multicellular with a gastrovascular system
done
clear
D)
Multicellular having tissue organization, but no body cavity
done
clear
View Answer play_arrow
Which of the following is a correct pair?
A)
Cuscuta - parasite
done
clear
B)
Dischidia - insectivorous
done
clear
C)
Opuntia - predator
done
clear
D)
Capsella - hydrophyte
done
clear
View Answer play_arrow
Which one of the following contains the largest quantity of extracellular material?
A)
Striated muscle
done
clear
B)
Aerolar tissue
done
clear
C)
Stratified epithelium
done
clear
D)
Myelinated nerve fibres
done
clear
View Answer play_arrow
Earthworms have no skeleton but during bur- rowing the anterior end becomes turgid and acts as a hydraulic skeleton. It is due to:
A)
gut peristalsis
done
clear
B)
setae
done
clear
C)
coelomic fluid
done
clear
D)
blood
done
clear
View Answer play_arrow
The kind of epithelium which forms the inner walls of blood vessels is:
A)
Cuboidal epithelium
done
clear
B)
Columnar epithelium
done
clear
C)
Ciliated columnar epithelium
done
clear
D)
Squamous epithelium
done
clear
View Answer play_arrow
Choose the correctly matched pair:
A)
Inner lining of salivary ducts-Ciliated epithelium
done
clear
B)
Moist surface of buccal cavity-Glandular epithelium
done
clear
C)
Tubular parts of nephrons-Cuboidal epithelium
done
clear
D)
Inner surface of bronchioles-Squamous epithelium
done
clear
View Answer play_arrow
Which of the following characteristics is mainly responsible for diversification of insects on land?
A)
Eyes
done
clear
B)
Segmentation
done
clear
C)
Bilateral symmetry
done
clear
D)
Exoskeleton
done
clear
View Answer play_arrow
Mitotic spindle is mainly composed of which protein:
A)
Actin
done
clear
B)
Tubulin
done
clear
C)
Actomyosin
done
clear
D)
Myoglobin
done
clear
View Answer play_arrow
A student wishes to study the cell structure under a light microscope having \[10\times \]eyepiece and \[45\times \]objective. He should illuminate the object by which one of the following colours of light so as to get the best possible resolution:
A)
Red
done
clear
B)
Green
done
clear
C)
Yellow
done
clear
D)
Blue
done
clear
View Answer play_arrow
Which of the following statements regarding mitochondrial membrane is not correct?
A)
The outer membrane is permeable to all kinds of molecules
done
clear
B)
The enzymes of the electron transfer chain are embedded in the outer membrane
done
clear
C)
The inner membrane is highly convoluted forming a series of infoldings
done
clear
D)
The outer membrane resembles a sieve.
done
clear
View Answer play_arrow
What are those structures that appear as 'beads- on-string' in the chromosomes when viewed under electron microscope?
A)
Genes
done
clear
B)
Nucleotides
done
clear
C)
Nucleosomes
done
clear
D)
Base pairs
done
clear
View Answer play_arrow
Which one of the following organelle in the figure correctly matches with its function?
A)
Rough endoplasmic reticulum, formation of glycoproteins
done
clear
B)
Golgi apparatus, protein synthesis
done
clear
C)
Golgi apparatus, formation ofglycolipids
done
clear
D)
Rough endoplasmic reticulum, protein synthesis
done
clear
View Answer play_arrow
Match the columns and identify the correct option:
Column-I Column-II [a] Thylakoids (i) Disc-shaped sacs in Golgi apparatus [b] Cristae (i.i) Condensed structure of DNA [c] Cistemae (lii) Flat membranous sacs in stroma [d] Chromatin (IV) Infoldings in mitochondria
A)
[a] [b] [c] [d] (iii) (iv) (ii) (i)
done
clear
B)
[a] [b] [c] [d] (iv) (iii) (i) (ii)
done
clear
C)
[a] [b] [c] [d] (iii) (iv) (i) (ii)
done
clear
D)
[a] [b] [c] [d] (iii) (i) (iv) (ii)
done
clear
View Answer play_arrow
Types of RNA polymerase required in nucleus for RNA synthesis are:
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Enzymes, vitamins and hormones can be classified into a single category of biological chemicals, because all of these:
A)
Are exclusively synthesized in the body of a living organism as at present
done
clear
B)
enhance oxidative metabolism
done
clear
C)
Are conjugated proteins
done
clear
D)
Help in regulating metabolism
done
clear
View Answer play_arrow
The curve given below shows enzymatic activity with relation to three conditions (pH, temperature and substrate concentration). What do the two axis (\[x\]and y) represent?
A)
\[x-\]axis y-axis enzymatic activity Temperature
done
clear
B)
\[x-\]axis y-axis enzymatic activity pH
done
clear
C)
\[x-\]axis y-axis Temperature enzyme activity
done
clear
D)
\[x-\]axis y-axis substrate concentration enzymatic activity
done
clear
View Answer play_arrow
Macro molecule chitin is:
A)
nitrogen containing polysaccharide
done
clear
B)
phosphorus containing polysaccharide
done
clear
C)
sulphur containing polysaccharide
done
clear
D)
simple polysaccharide
done
clear
View Answer play_arrow
The chitinous exoskeleton of arthropods is formed by the polymerisation of:
A)
lipoglycans
done
clear
B)
keratin sulphate and chondroitin sulphate
done
clear
C)
D-glucosamine
done
clear
D)
N-acetyl glucosamine
done
clear
View Answer play_arrow
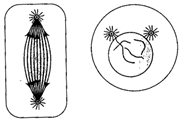
Which stages of cell division do the following figures A and B represent respectively?
A)
Metaphase - Telophase
done
clear
B)
Telophase - Metaphase
done
clear
C)
Late Anaphase - Prophase
done
clear
D)
Prophase-Anaphase
done
clear
View Answer play_arrow
Select the correct option:
I II [a] Synapsis aligns homologous chromosomes (i) AnaphaseII [b] Synthesis ofRNA and protein (ii) Zygotene [c] Action of enzyme recombinase (iii) \[{{G}_{2}}\] phase [d] Centromeres do not separate but chromatids move towards opposite poles (iv) Anaphase I (v) Pachytene
A)
[a] [b] [c] [d] (ii) (iii) (iv) (v)
done
clear
B)
[a] [b] [c] [d] (ii) (i) (iii) (iv)
done
clear
C)
[a] [b] [c] [d] (ii) (iii) (v) (iv)
done
clear
D)
[a] [b] [c] [d] (i) (ii) (v) (iv)
done
clear
View Answer play_arrow
In which of the following plant sunken stomata, are found:
A)
Nerium
done
clear
B)
Hydrilla
done
clear
C)
Mango
done
clear
D)
Guava
done
clear
View Answer play_arrow
Carbohydrates are commonly found as starch in plant storage organs. Which of the following five (a-e) properties of starch make it useful as a storage material?
[a] Easily translocated [b] Chemically non-reactive [c] Easily digested by animals [d] Osmotically inactive [e] Synthesized during photosynthesis
A)
[a], (3) and (5)
done
clear
B)
[a] and (5)
done
clear
C)
[b] and (3)
done
clear
D)
[b] and (4)
done
clear
View Answer play_arrow
Choose the correct match Bladderwort, sundew Venus flytrap:
A)
Nepanthes, Dionaea, Drosera
done
clear
B)
Nepanthes, Utricularia, Vanda
done
clear
C)
Utricularia, Drosera, Dionaea
done
clear
D)
Dionaea, Trapa, Vanda
done
clear
View Answer play_arrow
Which one of the following elements is not an essential micronutrient for plant growth?
A)
Ca
done
clear
B)
Mn
done
clear
C)
Zn
done
clear
D)
Cu
done
clear
View Answer play_arrow
Which one gives the most valid and recent explanation for stomatal movements?
A)
Guard cell photosynthesis
done
clear
B)
Transpiration
done
clear
C)
Potassium influx and efflux
done
clear
D)
Starch hydrolysis
done
clear
View Answer play_arrow
Cytochrome is:
A)
Metallo flavoprotein
done
clear
B)
Fe containing porphyrin pigment
done
clear
C)
Glycoprotein
done
clear
D)
Lipid
done
clear
View Answer play_arrow
In \[{{C}_{3}}\] plants, the first stable product of photo- synthesis during the dark reaction is:
A)
Oxaloacetic acid
done
clear
B)
3-phosphoglyceric acid
done
clear
C)
Phosphoglyceraldehyde
done
clear
D)
Malic acid
done
clear
View Answer play_arrow
The first acceptor of electrons from an excited chlorophyll molecule of photosystem II is:
A)
Quinone
done
clear
B)
Cytochrome
done
clear
C)
Iron-sulphur protein
done
clear
D)
Ferredoxin
done
clear
View Answer play_arrow
The oxygen evolved during photosynthesis comes from water molecules. Which one of the following pairs of elements is involved in this reaction?
A)
Magnesium and Chlorine
done
clear
B)
Manganese and Chlorine
done
clear
C)
Manganese and Potassium
done
clear
D)
Magnesium and Molybdenum
done
clear
View Answer play_arrow
Which one of the following concerns photo- phosphorylation?
A)
\[ADP+AMO\xrightarrow{Light\,energy}ATP\]
done
clear
B)
\[ADP+Inorganic\,P{{O}_{4}}\xrightarrow{Light\,energy}ATP\]
done
clear
C)
\[ADP+Inorganic\,P{{O}_{4}}\xrightarrow{{}}ATP\]
done
clear
D)
\[AMP+Inorganic\,P{{O}_{4}}\xrightarrow{Light\,energy}ATP\]
done
clear
View Answer play_arrow
Aerobic respiratory pathway is appropriately termed:
A)
Anabolic
done
clear
B)
Catabolic
done
clear
C)
Parabolic
done
clear
D)
Amphibolic
done
clear
View Answer play_arrow
Glycolate induces opening of stomata in:
A)
Presence of oxygen
done
clear
B)
Low \[C{{O}_{2}}\]cone.
done
clear
C)
High\[C{{O}_{2}}\]
done
clear
D)
\[C{{O}_{2}}\]absent
done
clear
View Answer play_arrow
Senescence as an active developmental cellular process in the growth and functioning of a flowering plant is indicated in:
A)
Annual plants
done
clear
B)
Floral parts
done
clear
C)
Vessels and tracheid differentiation
done
clear
D)
Leaf abscission
done
clear
View Answer play_arrow
Auxin can be bioassayed by:
A)
Lettuce hypocotyl elongation
done
clear
B)
Avena coleoptile curvature
done
clear
C)
Hydroponics
done
clear
D)
Potometer
done
clear
View Answer play_arrow
Stool of a person contains whitish grey colour due to malfunction of which type of organ:
A)
Pancreas
done
clear
B)
Spleen
done
clear
C)
Kidney
done
clear
D)
Liver
done
clear
View Answer play_arrow
In which group of three of the following five statements (a-e) contains all the three correct statements regarding beriberi:
[a] A crippling disease prevalent among the native population of sub-Saharan Africa [b] A deficiency disease caused by lack of thiamine (vitamin B1) [c] A nutritional disorder in infants and young children when the diet is persistently deficient in essential protein [d] Occurs in those countries where the staple diet is polished rice [e] The symptoms are pain from neuritis, paralysis, muscle wasting, progressive oedema, mental deterioration and finally heart failure
A)
[b], [c] and [e]
done
clear
B)
[a], [b] and [d]
done
clear
C)
[b], [d] and [e]
done
clear
D)
[a], [c] and [e]
done
clear
View Answer play_arrow
Which one of the following pairs of food com- pounds in humans reaches the stomach totally undigested?
A)
Starch and cellulose
done
clear
B)
Protein and starch
done
clear
C)
Starch and fat
done
clear
D)
Fat and cellulose
done
clear
View Answer play_arrow
Which of the following statements is not correct?
A)
Acini are present in the pancreas and secrete carboxypeptidase
done
clear
B)
Brunner's glands are present in the submucosa of stomach and secrete pepsinogen
done
clear
C)
Goblet cells are present in the mucosa of intestine and secrete mucus
done
clear
D)
Oxyntic cells are present in the mucosa of stomach and secrete HCl.
done
clear
View Answer play_arrow
When \[C{{O}_{2}}\]concentration in blood increases, breathing becomes
A)
There is no effect on breathing
done
clear
B)
Slow and deep
done
clear
C)
Faster and deeper
done
clear
D)
Shallower and slow
done
clear
View Answer play_arrow
The figure given below shows a small part of human lung where exchange of gases takes place. In which one of the options given below, the one part A, B, C or D is correctly identified along with its function.
A)
B: red blood cell-transport of \[C{{O}_{2}}\]mainly
done
clear
B)
C: arterial capillary - passes oxygen to tissues
done
clear
C)
A: alveolar cavity - main site of exchange of respiratory gases
done
clear
D)
D: Capillary wall - exchange of \[{{O}_{2}}\] and \[C{{O}_{2}}\]takes place here
done
clear
View Answer play_arrow
Impulse of heart beat originates from:
A)
S. A. Node
done
clear
B)
A. V. Node
done
clear
C)
Vagus Nerve
done
clear
D)
Cardiac Nerve
done
clear
View Answer play_arrow
Which type of white blood cells are concerned with the release of histamine and the natural anticoagulant heparin?
A)
Eosinophils
done
clear
B)
Monocytes
done
clear
C)
Neutrophils
done
clear
D)
Basophils
done
clear
View Answer play_arrow
Which two of the following changes (a - d) usually tend to occur in the plain dwellers when they move to high altitudes (3,500 m or more)?
[a] Increase in red blood cell size [b] Increase in red blood cell production [c] Increased breathing rate [d] Increase in thrombocyte count
Changes occurring are:
A)
[b] and [c]
done
clear
B)
[c] and [d]
done
clear
C)
[a] and [d]
done
clear
D)
[a] and [b]
done
clear
View Answer play_arrow
How do parasympathetic neural signals affect the working of the heart?
A)
Reduce both heart rate and cardiac output
done
clear
B)
Heart rate is increased without affecting the cardiac output.
done
clear
C)
Both heart rate and cardiac output increase
done
clear
D)
Heart rate decreases but cardiac output increases.
done
clear
View Answer play_arrow
A terrestrial animal must be able to:
A)
Conserve water
done
clear
B)
Actively pump salts out through the skin
done
clear
C)
Excrete large amounts of salts in urine
done
clear
D)
Excrete large amounts of water in urine
done
clear
View Answer play_arrow
A person who is on a long hunger strike and is surviving only on water will have:
A)
Less urea in his urine
done
clear
B)
More sodium in his urine
done
clear
C)
Less amino acids in his urine
done
clear
D)
More glucose in his blood
done
clear
View Answer play_arrow
Which one of the following statements is correct with respect to kidney function regulation?
A)
During summer when body loses of lot of water by evaporation, the release of ADH is suppressed
done
clear
B)
When someone drinks lot of water, ADH release is suppressed
done
clear
C)
Exposure to cold temperature stimulates ADH release
done
clear
D)
An increase in glomerular blood flow stimulates formation of Angiotensin II.
done
clear
View Answer play_arrow
Which of the following does not favour the formation of large quantities of dilute urine?
A)
Atrial-natriuretic factor
done
clear
B)
Alcohol
done
clear
C)
Caffeine
done
clear
D)
Renin
done
clear
View Answer play_arrow
What is sarcomere?
A)
Part between two H-lines
done
clear
B)
Part between two A-lines
done
clear
C)
Part between two I-bands
done
clear
D)
Part between two Z-lines
done
clear
View Answer play_arrow
Select the correct statement with respect to locomotion in humans:
A)
A decreased level of progesterone causes osteoporosis in old people.
done
clear
B)
Accumulation of uric acid crystals in joints causes their inflammation.
done
clear
C)
The vertebral column has 10 thoracic vertebrae.
done
clear
D)
The joint between adjacent vertebrae is a fibrous joint.
done
clear
View Answer play_arrow
When we migrate from dark to light, we fail 10 see for sometimes but after a time visibility becomes normal. It is an example of
A)
Accommodation
done
clear
B)
Adaptation
done
clear
C)
Mutation
done
clear
D)
Photoperiodism
done
clear
View Answer play_arrow
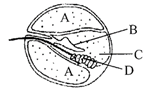
Given below is a diagrammatic cross section of a single loop of human cochlea:
Which one of the following options correctly represents the names of three different parts?
A)
D: Sensory hair cells.
A: Endolymph, B: Tectorial membrane
done
clear
B)
A: Perilymph, B: Tectorial membrane, C: Endolymph
done
clear
C)
B: Tectorial membrane, C: Perilymph, D: Secretory cells
done
clear
D)
C: Endolymph, D: Sensory hair cells, A: Serum
done
clear
View Answer play_arrow
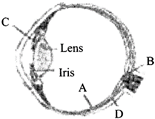
Parts A, B, C and D of the human eye are shown in the diagram. Select the option which gives correct identification along with its functions/ characteristics:
A)
A-Retina - contains photo receptors-rods and cones
done
clear
B)
B-Blind spot - has only a few rods and cones
done
clear
C)
C-Aqueous chamber - reflects the light which does not pass through the lens
done
clear
D)
D-choroid - its anterior part forms ciliary body
done
clear
View Answer play_arrow
Acromegaly is caused by:
A)
Excess of STH
done
clear
B)
Excess of thyroxin
done
clear
C)
Deficiency of thyroxin
done
clear
D)
Excess of adrenalin
done
clear
View Answer play_arrow
Which one of the following hormones is a modified amino acid?
A)
Progesterone
done
clear
B)
Prostaglandin
done
clear
C)
Estrogen
done
clear
D)
Epinephrine
done
clear
View Answer play_arrow
Match the source gland with its respective hormone as well as the function.
A)
Source gland Hormone Function Thyroid Thyroxine Regulates blood calcium level
done
clear
B)
Source gland Hormone Function Anterior pituitary Oxytocin Contraction of uterus muscles during child birth
done
clear
C)
Source gland Hormone Function Posterior pituitary Vasopressin Stimulates resorption of water in the distal tubules in the nephron
done
clear
D)
Source gland Hormone Function Corpus luteum Estrogen Supports Pregnancy
done
clear
View Answer play_arrow
Which of the following events is not associated with ovulation in human female?
A)
LH surge
done
clear
B)
Decrease in estradiol
done
clear
C)
Full development of Graafian follicle
done
clear
D)
Release of secondary oocyte
done
clear
View Answer play_arrow
Vegetative propagation in mint occurs by:
A)
Sucker
done
clear
B)
Runner
done
clear
C)
Offset
done
clear
D)
Rhizome
done
clear
View Answer play_arrow
In oogamy fertilization involves:
A)
A large non-motile female gamete and a small motile male gamete
done
clear
B)
A large non-motile female gamete and a small non-motile male gamete
done
clear
C)
A large motile female gamete and a small non-motile male gamete
done
clear
D)
A small non-motile female gamete and a large motile male gamete
done
clear
View Answer play_arrow
What would be the number of chromosomes in the cells of the aleurone layer in a plant species with 8 chromosomes in its synergids?
A)
16
done
clear
B)
24
done
clear
C)
32
done
clear
D)
8
done
clear
View Answer play_arrow
The scutellum observed in a grain of wheat or maize is comparable to which part of the seed in other monocotyledons?
A)
Cotyledon
done
clear
B)
Endosperm
done
clear
C)
Aleurone layer
done
clear
D)
Plumule
done
clear
View Answer play_arrow
Both autogamy and geitonogamy are prevented in:
A)
Castor
done
clear
B)
Maize
done
clear
C)
Papaya
done
clear
D)
Cucumber
done
clear
View Answer play_arrow
Which one of the following statements in not true?
A)
Honey is made by bees by digesting pollen collected from flowers
done
clear
B)
Pollen grains are rich in nutrients, and they are used in the form of tablets and syrups
done
clear
C)
Pollen grains of some plants cause severe allergies and bronchial afflictions in some people
done
clear
D)
The flowers pollinated by flies and bats secrete foul odour to attract them
done
clear
View Answer play_arrow
In angiosperms, microsporogenesis and megasporogenesis :
A)
occur in ovule
done
clear
B)
occur in anther
done
clear
C)
form gametes without furthers divisions
done
clear
D)
involve meiosis
done
clear
View Answer play_arrow
Sertoli cells are regulated by the pituitary hormone known as:
A)
FSH
done
clear
B)
GH
done
clear
C)
Prolactin
done
clear
D)
LH
done
clear
View Answer play_arrow
Which one of the following is the most likely root cause why menstruation is not taking place in regularly cycling human female?
A)
Retention of well-developed corpus luteum
done
clear
B)
Fertilization of the ovum
done
clear
C)
Maintenance of the hypertrophical endo- metrial lining
done
clear
D)
Maintenance of high concentration of sex hormones in the blood stream
done
clear
View Answer play_arrow
The permissible use of the technique amniocentesis is for:
A)
detecting sex of the unborn foetus
done
clear
B)
artificial insemination
done
clear
C)
transfer of embryo into the uterus of a surrogate mother
done
clear
D)
Detecting any genetic abnormality
done
clear
View Answer play_arrow
In a normal pregnant woman, the amount of total gonadotropin activity was assessed. The results expected was:
A)
High levels of FSH and LH in uterus to stimulate endometrial thickening
done
clear
B)
High level of circulating HCG to stimulate estrogen and progesterone synthesis
done
clear
C)
High level of circulating FSH and LH in the uterus to stimulate implantation of the embryo
done
clear
D)
High level of circulating HCG to stimulate endometrial thickening
done
clear
View Answer play_arrow
In human females, meiosis II is not completed until?
A)
birth
done
clear
B)
puberty
done
clear
C)
fertilization
done
clear
D)
uterine implantation
done
clear
View Answer play_arrow
Given below are four methods (A-D) and their modes of action (1-4) in achieving contraception. Select their correct matching from the four options that follow:
Method Mode of Action 1. The pill [a] Prevents sperms reaching cervix 2. Condom [b] Prevents implantation 3. Vasectomy [c] Prevents ovulation 4. Copper-T [d] Semen contains no sperms
A)
1 = [c], 2 = [d], 3 = [a], 4 = [b]
done
clear
B)
1 = [b], 2 = [c], 3 = [a], 4 = [d]
done
clear
C)
1 = [c], 2 = [a], 3 = [d], 4 = [b]
done
clear
D)
1 = [d], 2 = [a], 3 = [b], 4 = [c]
done
clear
View Answer play_arrow
Hysterectomy is surgical removal of:
A)
Mammary glands
done
clear
B)
Uterus
done
clear
C)
Prostate gland
done
clear
D)
Vas deference
done
clear
View Answer play_arrow
Probability of four son to a couple is:
A)
1/4
done
clear
B)
1/8
done
clear
C)
1/16
done
clear
D)
1/32
done
clear
View Answer play_arrow
A diseased man marries a normal woman. They get three daughter and five sons. All the daughters were diseased and sons were normal. The gene of this disease is:
A)
Sex-linked dominant
done
clear
B)
Sex-linked recessive
done
clear
C)
Sex-limited character
done
clear
D)
Autosomal dominant
done
clear
View Answer play_arrow
One of the parents of a cross has a mutation in its mitochondria. In that cross, that parent is taken as a male. During segregation of \[{{F}_{2}}\]progenies that mutation is found in:
A)
None of the progenies
done
clear
B)
All the progenies
done
clear
C)
50% of the progenies
done
clear
D)
1/3 of the progenies
done
clear
View Answer play_arrow
In Mendel's experiments with garden pea, round seed shape (RR) was dominant over wrinkled seeds (rr), yellow cotyledon (YY) was dominant over green cotyledon (yy). What are the expected phenotypes in the\[{{F}_{2}}\]generation of the cross\[\text{RRYY }\!\!\times\!\!\text{ rryy}\]?
A)
Only round seeds with green cotyledons
done
clear
B)
Only wrinkled seeds with yellow cotyledons
done
clear
C)
Only wrinkled seeds with green cotyledons
done
clear
D)
Round seeds with yellow cotyledons, and wrinkled seeds with yellow cotyledons
done
clear
View Answer play_arrow
The two polynucleotide chains in DNA are:
A)
Semiconservative
done
clear
B)
Parallel
done
clear
C)
Discontinuous
done
clear
D)
Antiparallel
done
clear
View Answer play_arrow
Which one of the following symbols and its representation used in human pedigree analysis is correct?
A)
mating between relatives
done
clear
B)
unaffected male
done
clear
C)
unaffected female
done
clear
D)
male affected
done
clear
View Answer play_arrow
The following ratio is generally constant for a given species:
A)
T+C/G+A
done
clear
B)
G+C/A+T
done
clear
C)
A+C/T+G
done
clear
D)
A+G/C+T
done
clear
View Answer play_arrow
One gene-one enzyme hypothesis was postulated by
A)
R. Franklin
done
clear
B)
Hershey and Chase
done
clear
C)
A. Garrod
done
clear
D)
Beadle and Tatum
done
clear
View Answer play_arrow
Whose experiments cracked the DNA and dis- covered unequivocally that a genetic code is a "triplet"?
A)
Beadle and Tatum
done
clear
B)
Nirenberg and Mathaei
done
clear
C)
Hershey and Chase
done
clear
D)
Morgan and Sturtevant
done
clear
View Answer play_arrow
The diagram shows an important concept in the genetic implication of DNA. Fill in the blanks A to C. \[DNA\xrightarrow{A}mRNA\xrightarrow{B}protein\xrightarrow[C]{\Pr oposed\,by}\]
A)
A-transcription B-replication C-James Watson
done
clear
B)
A-translation B-transcription C-Erevin Chargaff
done
clear
C)
A-transcription B-translation C-Francis Crick
done
clear
D)
A-translation B-extension C-Rosalind Franklin
done
clear
View Answer play_arrow
The introduction of t -DNA into plants involves:
A)
Allowing the plant roots to stand in water
done
clear
B)
Infection of the plant by Agrobacterium tumefaciens
done
clear
C)
Altering the pH of the soil, then heat shocking the plants
done
clear
D)
Exposing the plants to cold for a brief period
done
clear
View Answer play_arrow
Which one of the following options gives one correct example each of convergent evolution and divergent evolution?
A)
Convergent evolution Divergent evolution Bones of forelimbs of vertebrates Wings of butterfly and birds
done
clear
B)
Convergent evolution Divergent evolution Thorns of Bougainvillia and tendrils of Cucurbita Eyes of octopus and mammals
done
clear
C)
Convergent evolution Divergent evolution Eyes of octopus and mammals Bones of forelimpbs of vertebrates
done
clear
D)
Convergent evolution Divergent evolution Thorns of Bougainvillia and ten drils of Cucurbita Wings of butterflies and birds
done
clear
View Answer play_arrow
LSD is:
A)
hallucinogenic
done
clear
B)
sedative
done
clear
C)
stimulant
done
clear
D)
tranquiliser
done
clear
View Answer play_arrow
To obtain virus-free healthy plants from a diseased one by tissue culture technique, which part/parts of the diseased plant will be taken?
A)
Apical meristem only
done
clear
B)
Palisade parenchyma
done
clear
C)
Both apical and axillary meristems
done
clear
D)
Epidermis only
done
clear
View Answer play_arrow
Match the following list of microbes and their importance:
[a] [b] Saccharomyces cerevisiae Production of immuno- suppressive agents Monascus pufpiireiis Ripening of Swiss cheese Trichoderma polysporum Commerical production ofethanol Propiombacterium shannanii Production of blood cho- lesterol lowering agents
A)
[a] [b] [c] [d] (iii) (i) (iv) (ii)
done
clear
B)
[a] [b] [c] [d] (iii) (iv) (i) (ii)
done
clear
C)
[a] [b] [c] [d] (iv) (iii) (ii) (i)
done
clear
D)
[a] [b] [c] [d] (iv) (ii) (i) (iii)
done
clear
View Answer play_arrow
Golden rice is a genetically modified crop plant where the incorporated gene is meant for bio- synthesis of:
A)
Vitamin A
done
clear
B)
Vitamin B
done
clear
C)
Vitamin C
done
clear
D)
Omega 3
done
clear
View Answer play_arrow
 Which one of the following is the correct variation of voltage with time in the coil?
Which one of the following is the correct variation of voltage with time in the coil? 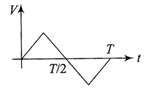
 Choose the correct statement from the following
Choose the correct statement from the following 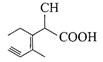
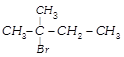
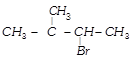
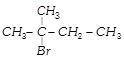





 Which one of the following options correctly represents the names of three different parts?
Which one of the following options correctly represents the names of three different parts?