question_answer 1)
Three blocks of masses\[{{m}_{1}},{{m}_{2}}\]and\[{{m}_{3}}\]are connected by massless strings as shown on a frictionless table. They are pulled with a force of 40 N. If\[{{m}_{1}}\text{=}10\text{ }kg,\text{ }{{m}_{2}}=6\]kg and\[{{m}_{3}}=4kg,\]then tension\[{{T}_{2}}\]will be:
A)
10 N
done
clear
B)
20 N
done
clear
C)
32 N
done
clear
D)
40 N
done
clear
View Answer play_arrow
question_answer 2) A rocket of mass 1000 kg is to be projected vertically upwards. The gases are exhausted vertically downwards with velocity 100 m/s with respect to the rocket. What is the minimum rate of burning of fuel, so as to just lift the rocket upwards against the gravitational attraction? (Take\[g=10\text{ }m/{{s}^{2}}\]).
A)
50 kg/s
done
clear
B)
100 kg/s
done
clear
C)
200 kg/s
done
clear
D)
400 kg/s
done
clear
View Answer play_arrow
question_answer 3) A force of 10 N acts on a body of mass 20 kg for 10 seconds. The change in its momentum is:
A)
50 kg-m/s
done
clear
B)
100 kg-m/s
done
clear
C)
300 kg-m/s
done
clear
D)
1000 kg-m/s
done
clear
View Answer play_arrow
question_answer 4) If an iron ball and a wooden ball of the same radius are released from a height \[h\] in vacuum, then time taken by both of them, to reach the ground will be:
A)
zero
done
clear
B)
unequal
done
clear
C)
roughly equal
done
clear
D)
exactly equal
done
clear
View Answer play_arrow
question_answer 5) A ball of mass 0.25 kg attached to the ends of a string of length 1.96 m is rotating in a horizontal circle. The string will break, if tension is more than 25 N. What is the maximum velocity with which the ball can be rotated?
A)
3 m/s
done
clear
B)
5 m/s
done
clear
C)
9 m/s
done
clear
D)
14 m/s
done
clear
View Answer play_arrow
question_answer 6) In uniform circular motion, the factor that remains constant is:
A)
acceleration
done
clear
B)
momentum
done
clear
C)
kinetic energy
done
clear
D)
linear velocity
done
clear
View Answer play_arrow
question_answer 7) A projectile is fired at\[30{}^\circ \]with momentum p. Neglecting friction, tine change in kinetic energy, when it returns back to the ground, will be:
A)
zero
done
clear
B)
30%
done
clear
C)
60%
done
clear
D)
100%
done
clear
View Answer play_arrow
question_answer 8) If a car at rest accelerates uniformly and attains a speed of 72 km/hr in 10 s, then it covers a distance of:
A)
50m
done
clear
B)
100m
done
clear
C)
200 m
done
clear
D)
400 m
done
clear
View Answer play_arrow
question_answer 9) Suppose refractive index\[\mu \]is given as \[\mu =A+B/{{\lambda }^{2}},\]where A and B are constants and X is wavelength then the dimensions of B are same as that of:
A)
wavelength
done
clear
B)
pressure
done
clear
C)
area
done
clear
D)
volume
done
clear
View Answer play_arrow
question_answer 10) We wish to see inside an atom. Assuming the atom to have a diameter of 100 pm, this means that one must be able to resolve a width of say 10 pm. If an electron microscope is used, the minimum electron energy required is about:
A)
15 keV
done
clear
B)
1.5 keV
done
clear
C)
150 keV
done
clear
D)
1.5 MeV
done
clear
View Answer play_arrow
question_answer 11) A particle is moving under the influence of a force which is fixed in magnitude and acting at an angle\[\theta \]in the direction of motion. The path described by the particle is:
A)
a circle
done
clear
B)
an ellipse
done
clear
C)
a parabola
done
clear
D)
a straight line
done
clear
View Answer play_arrow
question_answer 12) 300 J of work is done in sliding a 2 kg block up an inclined plane of height 10m. The work done against friction is (Take\[g=10m/{{s}^{2}}\])
A)
zero
done
clear
B)
100 J
done
clear
C)
200 J
done
clear
D)
300 J
done
clear
View Answer play_arrow
question_answer 13) The torque of force\[\overrightarrow{F}=(2\hat{i}-3\hat{j}+4\hat{k})\]newton acting at a point \[\overrightarrow{r}=(3\hat{i}+2\hat{j}+3\hat{k})\]metre about origin is:
A)
\[6\overrightarrow{i}-6\overrightarrow{j}+12\overrightarrow{k}N-m\]
done
clear
B)
\[-6\overrightarrow{i}+6\overrightarrow{j}-12\overrightarrow{k}N-m\]
done
clear
C)
\[17\overrightarrow{i}-6\overrightarrow{j}-13\overrightarrow{k}N-m\]
done
clear
D)
\[-17\overrightarrow{i}+6\overrightarrow{j}+13\overrightarrow{k}N-m\]
done
clear
View Answer play_arrow
question_answer 14) A machine which is 75% efficient, uses 12 J of energy in lifting 1 kg mass through a certain distance. The mass is then allowed to fall through the same distance. The velocity at the end of its fall is:
A)
\[\sqrt{12}m/s\]
done
clear
B)
\[\sqrt{18}m/s\]
done
clear
C)
\[\sqrt{24}m/s\]
done
clear
D)
\[\sqrt{32}m/s\]
done
clear
View Answer play_arrow
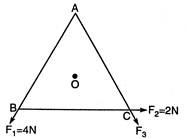
question_answer 15)
In an equilateral triangle ABC,\[{{F}_{1}},{{F}_{2}}\]and\[{{F}_{3}}\]are three forces acting along the sides AB, BC and AC as shown in the given figure. What should be the magnitude of\[{{F}_{3}}\]so that the total torque about O is zero?
A)
2 N
done
clear
B)
4 N
done
clear
C)
6 N
done
clear
D)
8 N
done
clear
View Answer play_arrow
question_answer 16) The rotational kinetic energy of a body is\[{{K}_{rot}}\] and its moment of inertia is\[I\]. The angular momentum of body is:
A)
\[I{{K}_{rot}}\]
done
clear
B)
\[2\sqrt{I{{K}_{rot}}}\]
done
clear
C)
\[\sqrt{2I{{K}_{rot}}}\]
done
clear
D)
\[2I{{K}_{rot}}\]
done
clear
View Answer play_arrow
question_answer 17) A small steel sphere of mass m is tied to a string of length r and is whirled in a horizontal circle with a uniform angular velocity\[2\omega \]. The string is suddenly pulled, so that radius of the circle is halved. The new angular velocity will be:
A)
\[2\omega \]
done
clear
B)
\[4\omega \]
done
clear
C)
\[6\omega \]
done
clear
D)
\[8\omega \]
done
clear
View Answer play_arrow
question_answer 18) A uniform disc of mass M and radius R is mounted on an axle supported in friction less bearings. A light cord is wrapped around the rim of the disc and a steady downward pull T is exerted on the cord. The angular acceleration of the disc is:
A)
\[\frac{MR}{2T}\]
done
clear
B)
\[\frac{2T}{MR}\]
done
clear
C)
\[\frac{T}{MR}\]
done
clear
D)
\[\frac{MR}{T}\]
done
clear
View Answer play_arrow
question_answer 19) If the rotational speed of earth is increased then weight of a body at the equator:
A)
increases
done
clear
B)
decreases
done
clear
C)
becomes double
done
clear
D)
does not change
done
clear
View Answer play_arrow
question_answer 20) The earth revolves around the sun in one year. If distance between them becomes double, the new time period of revolution will be:
A)
\[4\sqrt{2}\]years
done
clear
B)
\[2\sqrt{2}\] years
done
clear
C)
4 years
done
clear
D)
8 years
done
clear
View Answer play_arrow
question_answer 21) The orbital velocity of an artificial satellite in a circular orbit just above the earths surface is\[v\] The orbital velocity of a satellite orbiting at an altitude of half of the radius, is:
A)
\[\frac{3}{2}{{\upsilon }_{o}}\]
done
clear
B)
\[\frac{2}{3}{{\upsilon }_{o}}\]
done
clear
C)
\[\sqrt{\frac{2}{3}}{{\upsilon }_{o}}\]
done
clear
D)
\[\sqrt{\frac{3}{2}}{{\upsilon }_{o}}\]
done
clear
View Answer play_arrow
question_answer 22) The amplitude of an oscillating simple pendulum is 10 cm and its time period is 4 s. Its speed after 1 s when it passes through its equilibrium position is:
A)
zero
done
clear
B)
2.0 m/s
done
clear
C)
0.3 m/s
done
clear
D)
0.4 m/s
done
clear
View Answer play_arrow
question_answer 23) A simple second pendulum is mounted in a rocket. Its time period will decrease when the rocket is:
A)
moving up with uniform velocity
done
clear
B)
moving up with uniform acceleration
done
clear
C)
moving down with uniform acceleration
done
clear
D)
moving around the earth in geostationary orbit
done
clear
View Answer play_arrow
question_answer 24) A child swinging on a swing in a sitting position, stands up, then the time period of the swing will:
A)
increase
done
clear
B)
decrease
done
clear
C)
remain the same
done
clear
D)
increase if the child is long and decrease if the child is short
done
clear
View Answer play_arrow
question_answer 25) The phenomena arising due to the superposition of waves is/are:
A)
beats
done
clear
B)
stationary waves
done
clear
C)
Lissajous figures
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 26) A vehicle with horn of frequency\[\upsilon \]is moving with a velocity of 30 m/s in a direction perpendicular to the line joining the observer and the vehicle. The observer perceives the sound to have a frequency\[(v+{{v}_{1}})\]. If the sound velocity in air is 300 m/s, then.
A)
\[{{v}_{1}}=0\]
done
clear
B)
\[{{v}_{1}}=0.1\,v\]
done
clear
C)
\[{{v}_{1}}=-0.1\,v\]
done
clear
D)
\[{{v}_{1}}=10\,v\]
done
clear
View Answer play_arrow
question_answer 27) ball falling in a lake of depth 200 m shows 0.1% decrease in its volume at the bottom. What is the Bulk modulus of the ball material? (Take density of water\[=1000kg/{{m}^{3}}\])
A)
\[19.6\times {{10}^{8}}N/{{m}^{2}}\]
done
clear
B)
\[19.6\times {{10}^{-10}}N/{{m}^{2}}\]
done
clear
C)
\[19.6\times {{10}^{10}}N/{{m}^{2}}\]
done
clear
D)
\[19.6\times {{10}^{-8}}N/{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 28) At what temperature, hydrogen molecules will escape from the earths surface? (Take mass of hydrogen molecule\[=0.34\times {{10}^{-26}}kg,\] Boltzmann constant\[=1.38\times {{10}^{-23}}J/K,\] Radius of earth\[=6.4\times {{10}^{6}}m\]and acceleration due to gravity\[=9.8\text{ }m/{{s}^{2}}\])
A)
10 K
done
clear
B)
\[{{10}^{2}}K\]
done
clear
C)
\[{{10}^{3}}K\]
done
clear
D)
\[{{10}^{4}}K\]
done
clear
View Answer play_arrow
question_answer 29) Real gases obey ideal gas laws more closely at:
A)
low pressure and low temperature
done
clear
B)
low pressure and high temperature
done
clear
C)
high pressure and low temperature
done
clear
D)
high pressure and high temperature
done
clear
View Answer play_arrow
question_answer 30) Carnot engine cannot give 100% efficiency, because we cannot:
A)
eleminate friction
done
clear
B)
find ideal sources
done
clear
C)
prevent radiation
done
clear
D)
reach absolute zero temperature
done
clear
View Answer play_arrow
question_answer 31) Two cylinders fitted with pistons contain equal amounts of an ideal diatomic gas at 300 K. The piston of A is free to move, while that of B is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of gas in A is 300 K, then rise in temperature of gas in B is:
A)
30 K
done
clear
B)
18 K
done
clear
C)
50 K
done
clear
D)
42 K
done
clear
View Answer play_arrow
question_answer 32) A thin square plate with each side equal to 10 cm, is heated by a blacksmith. The rate of radiated energy by the heated plate is 1134 watts. The temperature of hot square plate is (Stefans constant \[\sigma =5.67\times {{10}^{-8}}W-{{m}^{2}}{{K}^{-4}};\]Emissivity of plate =1)
A)
1000 K
done
clear
B)
1189 K
done
clear
C)
2000 K
done
clear
D)
2378 K
done
clear
View Answer play_arrow
question_answer 33) A beaker of hot water cools from\[75{}^\circ C\] to\[70{}^\circ C\] in \[{{t}_{1}}\] minutes, from \[70{}^\circ C\] to \[65{}^\circ C\] in \[{{t}_{2}}\] minutes and from\[65{}^\circ C\]to\[60{}^\circ C\]in\[{{t}_{3}}\] minutes. Then:
A)
\[{{t}_{1}}={{t}_{2}}={{t}_{3}}\]
done
clear
B)
\[{{t}_{1}}<{{t}_{2}}<{{t}_{3}}\]
done
clear
C)
\[{{t}_{1}}>{{t}_{2}}>{{t}_{3}}\]
done
clear
D)
\[{{t}_{1}}<{{t}_{2}}<{{t}_{3}}\]
done
clear
View Answer play_arrow
question_answer 34) If the ratio of specific heats of a gas at constant pressure to that at constant volume is y, the change in internal energy of a gas, when the volume changes from V to 2V at constant pressure P, is:
A)
\[PV\]
done
clear
B)
\[\frac{R}{\gamma -1}\]
done
clear
C)
\[\frac{PV}{\gamma -1}\]
done
clear
D)
\[\frac{\gamma PV}{\gamma -1}\]
done
clear
View Answer play_arrow
question_answer 35) When a charged oil drop moves upwards in an electric field of strength E, the electric force acting on the drop is:
A)
equal to the gravitational force
done
clear
B)
less than the gravitational force
done
clear
C)
greater than the gravitational force
done
clear
D)
greater than the mass of the oil drop
done
clear
View Answer play_arrow
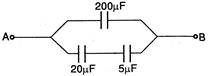
question_answer 36)
What is the resultant capacitance between points A and B?
A)
\[51\,\mu F\]
done
clear
B)
\[102\,\mu F\]
done
clear
C)
\[204\,\mu F\]
done
clear
D)
\[408\,\mu F\]
done
clear
View Answer play_arrow
question_answer 37) If a uniform wire of resistance R is uniformly stretched to n times the original length, then new resistance of the wire becomes:
A)
\[nR\]
done
clear
B)
\[{{n}^{2}}R\]
done
clear
C)
\[\frac{R}{n}\]
done
clear
D)
\[\frac{R}{{{n}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 38) A\[2{}^\circ C\]rise in temperature is observed in a conductor by passing a current. When the current is tripled, the rise in temperature will be:
A)
\[9{}^\circ C\]
done
clear
B)
\[18{}^\circ C\]
done
clear
C)
\[27{}^\circ C\]
done
clear
D)
\[36{}^\circ C\]
done
clear
View Answer play_arrow
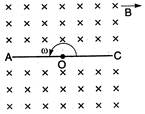
question_answer 39)
A conducting rod AC of length 4 / is rotated about appoint 0 in a uniform magnetic field B directed into the paper. \[AO=l\]and\[OC=3l\]then:
A)
\[{{V}_{A}}-{{V}_{O}}=\frac{B\omega {{l}^{2}}}{2}\]
done
clear
B)
\[{{V}_{O}}-{{V}_{C}}=\frac{3}{2}B\omega {{l}^{2}}\]
done
clear
C)
\[{{V}_{A}}-{{V}_{C}}=4B\omega {{l}^{2}}\]
done
clear
D)
\[{{V}_{C}}-{{V}_{O}}=\frac{9}{2}B\omega {{l}^{2}}\]
done
clear
View Answer play_arrow
question_answer 40) If two wires are carrying currents in opposite directions, then they will:
A)
repel each other
done
clear
B)
attract each other
done
clear
C)
become inclined to each other
done
clear
D)
neither repel nor attract each other
done
clear
View Answer play_arrow
question_answer 41) For protecting a sensitive equipment from external magnetic field, it should be:
A)
placed inside an iron can
done
clear
B)
placed inside an aluminium can
done
clear
C)
surrounded with fine copper sheet
done
clear
D)
wrapped with insulation around it when passing current through it
done
clear
View Answer play_arrow
question_answer 42) If reduction factor of a tangent galvanometer is 900 mA, then the current through it to produce a deflection of\[45{}^\circ \]is:
A)
600 mA
done
clear
B)
700 mA
done
clear
C)
750 mA
done
clear
D)
900 mA
done
clear
View Answer play_arrow
question_answer 43) The Lenzs law gives:
A)
direction of induced current
done
clear
B)
magnitude of induced emf
done
clear
C)
magnitude of induced current
done
clear
D)
magnitude and direction of induced current
done
clear
View Answer play_arrow
question_answer 44) A circuit consists of a resistor/an inductor and a capacitor connected in series. If the resistance of a resistor is\[25\,\Omega \]and the value of inductive reactance is equal to capacitive reactance, then the net impedance of the circuit will be:
A)
\[5\,\Omega \]
done
clear
B)
\[25\,\Omega \]
done
clear
C)
\[125\,\Omega \]
done
clear
D)
\[625\,\Omega \]
done
clear
View Answer play_arrow
question_answer 45) An alternating current is given by\[I={{I}_{1}}\cos \omega t+{{I}_{2}}\sin \omega t\]. The root mean square current is given by:
A)
\[\frac{({{I}_{1}}+{{I}_{2}})}{\sqrt{2}}\]
done
clear
B)
\[\frac{{{({{I}_{1}}+{{I}_{2}})}^{2}}}{2}\]
done
clear
C)
\[\sqrt{\frac{I_{1}^{2}+I_{2}^{2}}{2}}\]
done
clear
D)
\[\frac{\sqrt{I_{1}^{2}+I_{2}^{2}}}{2}\]
done
clear
View Answer play_arrow
question_answer 46) The waves which cannot travel in vacuum are:
A)
X-rays
done
clear
B)
radio-waves
done
clear
C)
infrasonic waves
done
clear
D)
ultra-violet rays
done
clear
View Answer play_arrow
question_answer 47) Which of the following is/are not electromagnetic waves?
A)
\[\beta -\]rays
done
clear
B)
X-rays
done
clear
C)
Cosmic rays
done
clear
D)
Both [a] and [c]
done
clear
View Answer play_arrow
question_answer 48) The electromagnetic radiations are caused by:
A)
a stationary charge
done
clear
B)
accelerated charge
done
clear
C)
uniformly moving charge
done
clear
D)
all of the above
done
clear
View Answer play_arrow
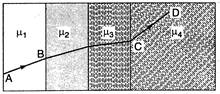
question_answer 49)
A ray of light passes through four transparent media with refractive indices \[{{\mu }_{1}},{{\mu }_{2}},{{\mu }_{3}}\]and \[{{\mu }_{4}}\]as shown in the figure. The surfaces of all media are parallel. If the emergent ray CD is parallel to the incident ray AB, we must have:
A)
\[{{\mu }_{1}}={{\mu }_{2}}\]
done
clear
B)
\[{{\mu }_{2}}={{\mu }_{3}}\]
done
clear
C)
\[{{\mu }_{3}}={{\mu }_{4}}\]
done
clear
D)
\[{{\mu }_{1}}={{\mu }_{4}}\]
done
clear
View Answer play_arrow
question_answer 50) A satisfactory photographic print is obtained at a distance of 2 m from a 60 cd lamp when the exposure time is 10 s. The time of exposure required for the same quality print at a distance 4 m from a 120 cd lamp is:
A)
5 s
done
clear
B)
10 s
done
clear
C)
20 s
done
clear
D)
25 s
done
clear
View Answer play_arrow
question_answer 51) In Youngs double-slit experiment, the angular width of fringe formed on a distant screen is\[0.1{}^\circ \]. If wavelength of light used is\[6000\overset{o}{\mathop{\text{A}}}\,,\] then spacing between the slits is:
A)
\[3.44\times {{10}^{-4}}m\]
done
clear
B)
\[4.33\times {{10}^{-4}}m\]
done
clear
C)
\[5.44\times {{10}^{-4}}m\]
done
clear
D)
\[6.33\times {{10}^{-4}}m\]
done
clear
View Answer play_arrow
question_answer 52) In Youngs double-slit experiment, distance between slits is 0.28 mm, distance between slits and screen is 1.4 m and distance between central bright fringe and third bright fringe is 0.9 cm. What is the wavelength of light used?
A)
\[5000\overset{o}{\mathop{\text{A}}}\,\]
done
clear
B)
\[6000\overset{o}{\mathop{\text{A}}}\,\]
done
clear
C)
\[7000\overset{o}{\mathop{\text{A}}}\,\]
done
clear
D)
\[9000\overset{o}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 53) Polarised light canbe produced by:
A)
scattering
done
clear
B)
dispersion
done
clear
C)
diffraction
done
clear
D)
interference
done
clear
View Answer play_arrow
question_answer 54) In a discharge tube at 0.2 mm of Hg pressure, there is a formation of:
A)
Crookes dark space
done
clear
B)
Faradays dark space
done
clear
C)
both spaces partly
done
clear
D)
Crookes dark space with glow near the electrodes
done
clear
View Answer play_arrow
question_answer 55) According to Bohrs atomic theory:
A)
electrons do not move
done
clear
B)
nucleus is negatively charged
done
clear
C)
electrons radiate energy in stationary orbits
done
clear
D)
electrons emit or absorb energy during transition state
done
clear
View Answer play_arrow
question_answer 56) In the nth orbit of hydrogen atom, ratio of its radius and de-Broglie wavelength associated with it, is:
A)
\[\frac{1}{2\pi n}\]
done
clear
B)
\[\frac{1}{2\pi {{n}^{2}}}\]
done
clear
C)
\[\frac{n}{2\pi }\]
done
clear
D)
\[\frac{{{n}^{2}}}{2\pi }\]
done
clear
View Answer play_arrow
question_answer 57) If\[_{7}{{N}^{14}}\]nucleus is bombarded with an\[\alpha -\]particle and a proton is emitted the product nucleus is:
A)
\[_{8}{{O}^{17}}\]
done
clear
B)
\[_{8}{{F}^{19}}\]
done
clear
C)
\[_{9}{{O}^{17}}\]
done
clear
D)
\[_{7}{{F}^{19}}\]
done
clear
View Answer play_arrow
question_answer 58) The relationship between decay constant\[(\lambda )\]and half-life\[{{(T)}_{1/2}}\]of a radioactive substance is:
A)
\[\lambda =\frac{{{\log }_{10}}2}{{{(T)}_{1/2}}}\]
done
clear
B)
\[\lambda =\frac{{{\log }_{2}}e}{{{(T)}_{1/2}}{{\log }_{10}}e}\]
done
clear
C)
\[\lambda =\frac{{{\log }_{2}}10}{{{(T)}_{1/2}}{{\log }_{2}}e}\]
done
clear
D)
\[\lambda =\frac{{{\log }_{10}}2}{{{(T)}_{1/2}}{{\log }_{10}}e}\]
done
clear
View Answer play_arrow
question_answer 59) At absolute zero temperature, semiconductor behaves as a/an:
A)
dielectric
done
clear
B)
conductor
done
clear
C)
insulator
done
clear
D)
super conductor
done
clear
View Answer play_arrow
question_answer 60) In an intrinsic semiconductor:
A)
\[{{n}_{e}}>{{n}_{h}}\]
done
clear
B)
\[{{n}_{h}}>{{n}_{e}}\]
done
clear
C)
\[{{n}_{e}}={{n}_{h}}\]
done
clear
D)
\[{{n}_{e}}={{n}_{h}}=0\]
done
clear
View Answer play_arrow
question_answer 61) The radius of second Bohrs orbit of hydrogen atom is:
A)
0.053 nm
done
clear
B)
0.106 nm
done
clear
C)
0.2116 nm
done
clear
D)
0.4256 nm
done
clear
View Answer play_arrow
question_answer 62) Kinetic energy of one mole of an ideal gas at 300 K is:
A)
3.48 kJ
done
clear
B)
3.74 kJ
done
clear
C)
37.4kJ
done
clear
D)
34.8kJ
done
clear
View Answer play_arrow
question_answer 63) Which of the following is the correct electronic configuration of Cr?
A)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{1}}3{{d}^{5}}\]
done
clear
B)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{2}}3{{d}^{4}}\]
done
clear
C)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{2}}3{{d}^{6}}\]
done
clear
D)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{1}}3{{d}^{8}}\]
done
clear
View Answer play_arrow
question_answer 64) If the value of\[{{C}_{p}}\]for nitrogen gas is\[j{{K}^{-1}}mo{{l}^{-1}},\]then the value of\[\Delta H\]on heating 28 g of nitrogen gas from\[0{}^\circ C\]to\[100{}^\circ C\]at constant pressure will be:
A)
1200 J
done
clear
B)
1300 J
done
clear
C)
1400 J
done
clear
D)
1500 J
done
clear
View Answer play_arrow
question_answer 65) The least count of an instrument is 0.01 cm: Taking all precautions, the most possible error in the measurement can be:
A)
0.005 cm
done
clear
B)
0.01 cm
done
clear
C)
0.0001 cm
done
clear
D)
0.1 cm
done
clear
View Answer play_arrow
question_answer 66) The phenomenon of radioactivity was discovered by:
A)
Henry Becquerrel
done
clear
B)
Madam Curie
done
clear
C)
P. Curie
done
clear
D)
Rutherford.
done
clear
View Answer play_arrow
question_answer 67) Hydrogen peroxide is prepared in the laboratory by:
A)
passing\[C{{O}_{2}}\]into\[Ba{{O}_{2}}\]
done
clear
B)
adding\[Mn{{O}_{2}}\]to dil.\[{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
adding\[N{{a}_{2}}{{O}_{2}}\]to cold water
done
clear
D)
adding\[Pb{{O}_{2}}\] into\[KMn{{O}_{4}}\].
done
clear
View Answer play_arrow
question_answer 68) Vinegar is:
A)
\[HCHO\]
done
clear
B)
\[HCOOH\]
done
clear
C)
\[C{{H}_{3}}CHO\]
done
clear
D)
\[C{{H}_{3}}COOH\]
done
clear
View Answer play_arrow
question_answer 69) Which of the following product is formed in the reaction \[C{{H}_{3}}MgBr\xrightarrow[(ii)\,{{H}_{2}}O]{(i)\,C{{O}_{2}}}?\]
A)
Acetic acid
done
clear
B)
Methanoic acid
done
clear
C)
Methanol
done
clear
D)
Ethanal
done
clear
View Answer play_arrow
question_answer 70) Ethyl chloride is converted into diethyl ether by:
A)
Perkins reaction
done
clear
B)
Grignard reagent
done
clear
C)
Wurtz reaction
done
clear
D)
Williamsons synthesis
done
clear
View Answer play_arrow
question_answer 71) lodoform test is not answered by:
A)
\[C{{H}_{3}}OH\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
C)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{CH}}\,C{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 72) The equivalent weight of a solid element is found to be 9. If the specific heat of this element is\[1.05\,J{{g}^{-1}}{{K}^{-1}},\]then its atomic weight is:
A)
17
done
clear
B)
21
done
clear
C)
25
done
clear
D)
27
done
clear
View Answer play_arrow
question_answer 73) The maximum energy is possessed by an electron, when it is present:
A)
in nucleus
done
clear
B)
in ground energy state
done
clear
C)
in first excited state
done
clear
D)
at infinite distance from the nucleus
done
clear
View Answer play_arrow
question_answer 74) A gas has a vapour density 11.2. The volume occupied by 1 gm of gas at NTP is:
A)
1 litre
done
clear
B)
11.2 litre
done
clear
C)
22.4 litre
done
clear
D)
unpredictable
done
clear
View Answer play_arrow
question_answer 75) Particle nature of electron was experimently demonstrated by:
A)
Max Bon
done
clear
B)
J.J. Thomson
done
clear
C)
de Broglie
done
clear
D)
Schrondinger
done
clear
View Answer play_arrow
question_answer 76) Which of the following is a magic number?
A)
10
done
clear
B)
16
done
clear
C)
55
done
clear
D)
50
done
clear
View Answer play_arrow
question_answer 77) The Ostwald dilution law is expressed as:
A)
\[{{K}_{a}}=\frac{c{{\alpha }^{2}}}{(1-\alpha )}\]
done
clear
B)
\[{{K}_{a}}=\frac{c\alpha }{(1-\alpha )}\]
done
clear
C)
\[{{K}_{a}}=\frac{1-\alpha }{c{{\alpha }^{2}}}\]
done
clear
D)
\[{{K}_{a}}=\frac{c(1-\alpha )}{{{\alpha }^{2}}}\]
done
clear
View Answer play_arrow
question_answer 78) The pH value of an acid is 5 and its concentration is 1 M. What is the value of\[{{K}_{a}}\]for the acid?
A)
\[{{10}^{-7}}\]
done
clear
B)
\[{{10}^{-5}}\]
done
clear
C)
\[{{10}^{-10}}\]
done
clear
D)
\[{{10}^{-8}}\]
done
clear
View Answer play_arrow
question_answer 79) To calculate the amount of work done in joules during reversible isothermal expansion of an ideal gas, the volume must be expressed in:
A)
\[{{m}^{3}}\] only
done
clear
B)
\[d{{m}^{3}}\] only
done
clear
C)
\[c{{m}^{3}}\] only
done
clear
D)
any of these
done
clear
View Answer play_arrow
question_answer 80) In the reaction, \[{{H}_{2}}(g)+{{I}_{2}}(g)\] \[2HI(g).\]Which of the following is correct?
A)
\[{{K}_{p}}={{K}_{c}}\]
done
clear
B)
\[{{K}_{p}}>{{K}_{c}}\]
done
clear
C)
\[{{K}_{p}}<{{K}_{c}}\]
done
clear
D)
\[{{K}_{p}}\ge {{K}_{c}}\]
done
clear
View Answer play_arrow
question_answer 81) The mass of an object changes from 0.2 g to 0.025 g in 1500 years. The half life of the object is:
A)
500 yrs
done
clear
B)
600 yrs
done
clear
C)
800 yrs
done
clear
D)
950 yrs
done
clear
View Answer play_arrow
question_answer 82) Which of the following is the correct nuclear reaction, when\[_{90}^{233}Th\]undergoes one\[\beta -\] emission?
A)
\[_{90}^{233}Th\xrightarrow[{}]{{}}\,_{90}^{233}Pa{{+}_{-2}}{{e}^{0}}\]
done
clear
B)
\[_{90}^{233}Th\xrightarrow[{}]{{}}\,_{91}^{233}Pa{{+}_{-1}}{{e}^{0}}\]
done
clear
C)
\[_{90}^{233}Th\xrightarrow[{}]{{}}\,_{90}^{292}Pa{{+}_{0}}{{e}^{4}}\]
done
clear
D)
\[_{90}^{233}Th\xrightarrow[{}]{{}}\,_{91}^{229}Pa{{+}_{-1}}{{e}^{4}}\]
done
clear
View Answer play_arrow
question_answer 83) Which of the following is the chief ore of copper?
A)
\[C{{u}_{2}}S\]
done
clear
B)
\[C{{u}_{2}}O\]
done
clear
C)
\[CuFe{{S}_{2}}\]
done
clear
D)
\[CuC{{O}_{3}}.Cu{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 84) Which of the following is an epsom salt?
A)
\[2CaS{{O}_{4}}.{{H}_{2}}O\]
done
clear
B)
\[MgS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
C)
\[MgS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
D)
\[BaS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 85) Bromine can be liberated from\[KBr\]solution by the action of:
A)
\[KI\]
done
clear
B)
\[NaCl\]
done
clear
C)
\[C{{l}_{2}}\]
done
clear
D)
12 solution
done
clear
View Answer play_arrow
question_answer 86) When lead nitrate is heated, it produces:
A)
\[N{{O}_{2}}\]
done
clear
B)
\[NO\]
done
clear
C)
\[{{N}_{2}}{{O}_{5}}\]
done
clear
D)
\[{{N}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 87) Which of the following lanthanide is commonly used?
A)
Lanthanum
done
clear
B)
Nobelium
done
clear
C)
Thorium
done
clear
D)
Cerium
done
clear
View Answer play_arrow
question_answer 88) The IUPAC name of \[{{(CH{{ }_{3}})}_{3}}CCH=C{{H}_{2}}\]is:
A)
3, 3-dimethyl-1-butene
done
clear
B)
2, 2-dimethyl 3-butene
done
clear
C)
1, 1, 1-trimethyl-2-propene
done
clear
D)
3, 3, 3-trimethyl 2-propene
done
clear
View Answer play_arrow
question_answer 89) The order of reactivities of methyl halide in the formation of Grignard reagent is:
A)
\[C{{H}_{3}}Cl>C{{H}_{3}}Br>C{{H}_{3}}I\]
done
clear
B)
\[C{{H}_{3}}I>C{{H}_{3}}Br>C{{H}_{3}}Cl\]
done
clear
C)
\[C{{H}_{3}}Br>C{{H}_{3}}I>C{{H}_{3}}Cl\]
done
clear
D)
\[C{{H}_{3}}Br>C{{H}_{3}}Cl>C{{H}_{3}}I\]
done
clear
View Answer play_arrow
question_answer 90) Which of the following waste materials can be used for producing bio-gas?
A)
Thermocoal
done
clear
B)
Glass bottle
done
clear
C)
Tin cans
done
clear
D)
Paper scrap
done
clear
View Answer play_arrow
question_answer 91) Which of the following is called Marsh gas?
A)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
D)
\[C{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 92) The hardness of water is estimated by:
A)
EDTA method
done
clear
B)
Titrimetric method
done
clear
C)
Conductivity method
done
clear
D)
Distillation method
done
clear
View Answer play_arrow
question_answer 93) The addition of 1% alcohol to chloroform acts as:
A)
auto-catalyst
done
clear
B)
bio-catalyst
done
clear
C)
positive catalyst
done
clear
D)
negative catalyst
done
clear
View Answer play_arrow
question_answer 94) Which one of the following order of acid strength is correct?
A)
\[RCOOH>HOH>ROH>HC\equiv CH\]
done
clear
B)
\[RCOOH>HOH>CH\equiv CH>ROH\]
done
clear
C)
\[RCOOH>HC\equiv CH>HOH>ROH\]
done
clear
D)
\[RCOOH>ROH>HOH>CH\equiv CH\]
done
clear
View Answer play_arrow
question_answer 95) In order to convert aniline into chlorobenzene the reagent used is:
A)
\[NaN{{O}_{2}}/HCl,\text{ }CuCl\]
done
clear
B)
\[C{{l}_{2}}/CC{{l}_{4}}\]
done
clear
C)
\[C{{l}_{2}}/AlC{{l}_{3}}\]
done
clear
D)
\[CuC{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 96) A drug that is antipyretic as well as analgesic is:
A)
chloroquine
done
clear
B)
penicillin
done
clear
C)
paracetamol
done
clear
D)
chloropromazine hydrochloride
done
clear
View Answer play_arrow
question_answer 97) The basic component of the smog is:
A)
PAN
done
clear
B)
PBN
done
clear
C)
\[N{{O}_{2}}\]
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 98) DNA multiplication is called:
A)
translation
done
clear
B)
transduction
done
clear
C)
transcription
done
clear
D)
replication
done
clear
View Answer play_arrow
question_answer 99) Hexoses and pentoses are:
A)
disaccharides
done
clear
B)
Monosaccharides
done
clear
C)
Polysaccharides
done
clear
D)
Oligosaccharides
done
clear
View Answer play_arrow
question_answer 100) An ester used as medicine is:
A)
ethyl acetate
done
clear
B)
methyl salicylate
done
clear
C)
ethyl benzoate
done
clear
D)
methyl benzoate
done
clear
View Answer play_arrow
question_answer 101) What is formed when nitrobenzene is reduced using zinc and alkali?
A)
Phenol
done
clear
B)
Hydroazobenzene
done
clear
C)
Aniline
done
clear
D)
Nitrosobenzene
done
clear
View Answer play_arrow
question_answer 102) Which of the following test is not used for testing of proteins?
A)
Millons test
done
clear
B)
Molishs test
done
clear
C)
Biuret test
done
clear
D)
Ninhydrin test
done
clear
View Answer play_arrow
question_answer 103) Which of the following is not a synthetic fibre?
A)
Rubber
done
clear
B)
Nylon-6
done
clear
C)
Nylon-6, 6
done
clear
D)
Nylon-6, 10
done
clear
View Answer play_arrow
question_answer 104) A liquid decomposes at its normal boiling point. It can be purified by:
A)
sublimation
done
clear
B)
steam distillation
done
clear
C)
vacuum distillation
done
clear
D)
fractional distillation
done
clear
View Answer play_arrow
question_answer 105) Which of the following is obtained when auric chloride reacts with sodium chloride?
A)
\[Na[AuCl]\]
done
clear
B)
\[Na[AuC{{l}_{2}}]\]
done
clear
C)
\[Na[AuC{{l}_{3}}]\]
done
clear
D)
\[Na[AuC{{l}_{4}}]\]
done
clear
View Answer play_arrow
question_answer 106) The limiting radius ratio for tetrahedral shape is:
A)
0 to 0.155
done
clear
B)
0.225 to 0.414
done
clear
C)
0.155 to 0.225
done
clear
D)
0.414 to 0.732
done
clear
View Answer play_arrow
question_answer 107) When a capillary tube of diameter 0.8 mm is dipped in a liquid having density\[800\text{ }kg{{m}^{-3}},\]then the height of liquid in the capillary tube rises to 4 cm. The surface tension of liquid is \[(g=9.8m/{{s}^{2}}):\]
A)
\[4.3\times {{10}^{-2}}N{{m}^{-1}}\]
done
clear
B)
\[5.6\times {{10}^{-2}}N{{m}^{-1}}\]
done
clear
C)
\[6.3\times {{10}^{-2}}N{{m}^{-1}}\]
done
clear
D)
\[7.3\times {{10}^{-2}}N{{m}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 108) The\[BC{{l}_{3}}\]is a planar molecule whereas\[NC{{l}_{3}}\]is pyramidal, because:
A)
nitrogen atom is smaller than boron atoms
done
clear
B)
\[B-Cl\]bond is more polar than\[N-Cl\]bond.
done
clear
C)
\[N-Cl\]bond is more covalent than \[B-Cl\] bond
done
clear
D)
\[BC{{l}_{3}}\]has no lone pair but\[NC{{l}_{3}}\]has a lone pair of electrons
done
clear
View Answer play_arrow
question_answer 109) Railway wagon axles are made by heating iron rods embedded in charcoal powder. This process is known as:
A)
tempering
done
clear
B)
anneling
done
clear
C)
sherardising
done
clear
D)
case hardening
done
clear
View Answer play_arrow
question_answer 110) The reaction \[Ag_{(aq)}^{2+}+A{{g}_{(s)}}2Ag_{(aq)}^{+}\]is an example of:
A)
reduction
done
clear
B)
oxidation
done
clear
C)
disproportionation
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 111) Number of\[\sigma \]and\[\pi \]bonds in acetylene are:
A)
3 and 2
done
clear
B)
2 and 2
done
clear
C)
2 and 3
done
clear
D)
4 and 3
done
clear
View Answer play_arrow
question_answer 112) The percentage s-character of the hybrid orbitals in methane, ethene and ethyne are respectively:
A)
25, 50, 75
done
clear
B)
25, 33, 50
done
clear
C)
10, 20, 40
done
clear
D)
50, 75, 100
done
clear
View Answer play_arrow
question_answer 113) Which of the following statements is true?
A)
\[\Delta G\]is always less than\[\Delta H\]
done
clear
B)
\[\Delta G\]is always more than\[\Delta H\]
done
clear
C)
\[\Delta G\]is always proportional to\[\Delta H\]
done
clear
D)
\[\Delta G\]may be lesser, greater or equal to\[\Delta H\]
done
clear
View Answer play_arrow
question_answer 114) Which of the following is not a characteristic of chemisorption?
A)
Adsorption is specific
done
clear
B)
Adsorption is irreversible
done
clear
C)
\[\Delta H\]is of the order of 400 kJ
done
clear
D)
Adsorption may be multimolecular layer
done
clear
View Answer play_arrow
question_answer 115) Which of the following process is not used for the purification of solid impurities?
A)
Distillation
done
clear
B)
Sublimation
done
clear
C)
Crystallisation
done
clear
D)
Vaporisation
done
clear
View Answer play_arrow
question_answer 116) Which of the following organization is engaged in the promotion and construction of bio-gas plants in India?
A)
KBIC
done
clear
B)
GVD
done
clear
C)
KVIC
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 117) Which of the following is not true about the urea?
A)
It can be stored easily
done
clear
B)
It should be applied at sowing time
done
clear
C)
It cannot be used for all types of crops and soils
done
clear
D)
The cost of production of urea is cheap.
done
clear
View Answer play_arrow
question_answer 118) Which of the following reagent can distinguish between 1-Butyne and 2-Butyne?
A)
Aqueous\[NaOH\]
done
clear
B)
Bromine water
done
clear
C)
Fehlings solution
done
clear
D)
Ammonical\[AgN{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 119) At 530 K, glycerol reacts with oxalic acid to produce:
A)
allyl alcohol
done
clear
B)
formic acid
done
clear
C)
glyceraldehyde
done
clear
D)
formaldehyde
done
clear
View Answer play_arrow
question_answer 120) Which of the following compound does not undergo schotten Baumann reaction?
A)
Phenol
done
clear
B)
Primary amine
done
clear
C)
Secondary amine
done
clear
D)
Tertiary amine
done
clear
View Answer play_arrow
question_answer 121) The main function of lysosome is:
A)
sexual reproduction
done
clear
B)
extracellular digestion
done
clear
C)
intracellular digestion
done
clear
D)
both b and c
done
clear
View Answer play_arrow
question_answer 122) Which of the following maintains continuity between the water and lipid phases inside and outside the cells?
A)
Cell wall
done
clear
B)
Lecithin
done
clear
C)
Cell vacuole
done
clear
D)
Cell membrane of woody plants
done
clear
View Answer play_arrow
question_answer 123) The method of classification, called cladistics, is based on:
A)
natural system
done
clear
B)
mutation theory
done
clear
C)
artificial system
done
clear
D)
evolutionary history
done
clear
View Answer play_arrow
question_answer 124) In many bacteria, the cell membrane becomes invaginated and folded to form:
A)
pili
done
clear
B)
cristae
done
clear
C)
fimbriae
done
clear
D)
mesosomes
done
clear
View Answer play_arrow
question_answer 125) Lomasomes are found in:
A)
algal cell
done
clear
B)
fungal cell
done
clear
C)
bacterial cell
done
clear
D)
cyanobacterial cell
done
clear
View Answer play_arrow
question_answer 126) Zygote of Spirogyra produces four haploid nuclei, in which:
A)
one is functional
done
clear
B)
two are functional
done
clear
C)
three are functional
done
clear
D)
all are functional
done
clear
View Answer play_arrow
question_answer 127) Which of the following part of Fimaria, sporophyte is involved in the dispersal of spores?
A)
Calyptra
done
clear
B)
Operculum
done
clear
C)
Peristome
done
clear
D)
Annulus
done
clear
View Answer play_arrow
question_answer 128) In Selaginella, the adaxial outgrowth, from the base of leaf, is called:
A)
ligule
done
clear
B)
velum
done
clear
C)
rhizophore
done
clear
D)
glossopodium
done
clear
View Answer play_arrow
question_answer 129) In Pinus, the third tier of embryonal cells from below is known as:
A)
rosette tier
done
clear
B)
suspensor tier
done
clear
C)
embryonal tier
done
clear
D)
free-nuclear tier
done
clear
View Answer play_arrow
question_answer 130) Most of the petrocrops belongs to family:
A)
Malvaceae
done
clear
B)
Rutaceae
done
clear
C)
Leguminosae
done
clear
D)
Euphorbiaceae
done
clear
View Answer play_arrow
question_answer 131) Amia belongs to family:
A)
Labiatae
done
clear
B)
Fabaceae
done
clear
C)
Solanaceae
done
clear
D)
Euphorbiaceae
done
clear
View Answer play_arrow
question_answer 132) Tuberous roots are found in:
A)
Beta vulgaris
done
clear
B)
Daucus carota
done
clear
C)
Ipomoea batatas
done
clear
D)
Raphanus sativus
done
clear
View Answer play_arrow
question_answer 133) Velamen is found in:
A)
Vanda
done
clear
B)
Rosa
done
clear
C)
Viscum
done
clear
D)
Santahim
done
clear
View Answer play_arrow
question_answer 134) The type of leaf in Dazicus carota is:
A)
simple
done
clear
B)
bipinnate
done
clear
C)
tripinnate
done
clear
D)
decompound
done
clear
View Answer play_arrow
question_answer 135) Aerenchyma provides:
A)
flexibility to plants
done
clear
B)
mechanical strength to plants
done
clear
C)
buoyancy to hydrophytic plants
done
clear
D)
promoting nature of photosynthesis
done
clear
View Answer play_arrow
question_answer 136) Two cross-sections of stem and root appear simple, when viewed by naked eye. But under microscope, they can be differentiated by:
A)
exarch condition of root and stem
done
clear
B)
endarch condition of stem and root
done
clear
C)
endarch condition of root and exarch condition of stem
done
clear
D)
endarch condition of stem and exarch condition of root
done
clear
View Answer play_arrow
question_answer 137) Umbel inflorescence is found in:
A)
Musa
done
clear
B)
Colocasia
done
clear
C)
Coriandrum
done
clear
D)
Helianthus
done
clear
View Answer play_arrow
question_answer 138) When gynoecium is present in the topmost position of thalamus, the flower is known as:
A)
inferior
done
clear
B)
epigynous
done
clear
C)
perigynous
done
clear
D)
hypogynous
done
clear
View Answer play_arrow
question_answer 139) Orthotropus ovule belongs to:
A)
Urtica
done
clear
B)
Polygonum
done
clear
C)
Peperomea
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 140) In a flowering plant, megaspore develops into an embryo sac, which contains:
A)
4 cell, one of which is an egg
done
clear
B)
6 cells, one of which is an egg
done
clear
C)
8 cells, one of which is an egg
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 141) The type of pollination found in Calotropis is:
A)
dicliny
done
clear
B)
herkogamy
done
clear
C)
heterostyly
done
clear
D)
dichogamy
done
clear
View Answer play_arrow
question_answer 142) Apogamy is:
A)
reproduction of virus
done
clear
B)
failure of fusion of gametes
done
clear
C)
development of bacteria
done
clear
D)
loss of function of reproduction
done
clear
View Answer play_arrow
question_answer 143) If a sporangium is derived from a single cell, it is called:
A)
leptosporangiate
done
clear
B)
eusporangiate
done
clear
C)
heterosporangiate
done
clear
D)
monosporangiate
done
clear
View Answer play_arrow
question_answer 144) Drupe contains:
A)
stony endocarp
done
clear
B)
stony mesocarp
done
clear
C)
edible epicarp
done
clear
D)
edible endocarp
done
clear
View Answer play_arrow
question_answer 145) When stomata open, the pH of guard cells:
A)
increases
done
clear
B)
decreases
done
clear
C)
remains same
done
clear
D)
both a and b
done
clear
View Answer play_arrow
question_answer 146) Water lost in guttation is:
A)
pure water
done
clear
B)
impure water
done
clear
C)
in vapour form
done
clear
D)
either a and b
done
clear
View Answer play_arrow
question_answer 147) Photochemical reactions in the chloroplasts are directly involved in:
A)
fixation of carbon dioxide
done
clear
B)
synthesis of glucose and starch
done
clear
C)
formation of phosphoglyceric acid
done
clear
D)
photolysis of water and phosphorylation of ADP to ATP
done
clear
View Answer play_arrow
question_answer 148) Which of the following elements are essential for the photolysis of water?
A)
\[Ca\]and\[Cl\]
done
clear
B)
\[Mn\] and\[Cl\]
done
clear
C)
\[Zn\]and\[I\]
done
clear
D)
\[Cu\]and \[Fe\]
done
clear
View Answer play_arrow
question_answer 149) Respiratory Quotient (R.Q.) is defined as:
A)
vol. of\[{{O}_{2}}\] /vol. of\[C{{O}_{2}}\]
done
clear
B)
vol. of \[C{{O}_{2}}\] /vol. of\[{{O}_{2}}\]
done
clear
C)
vol. of \[{{O}_{2}}\] /vol. of \[{{N}_{2}}\]
done
clear
D)
vol. of\[{{N}_{2}}\]/vol. of \[C{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 150) A sleep movement in plants is a nastic response, that occurs daily in respose to:
A)
dark
done
clear
B)
light
done
clear
C)
water
done
clear
D)
both a and b
done
clear
View Answer play_arrow
question_answer 151) Plant hormones are:
A)
growth regulators
done
clear
B)
growth promoters
done
clear
C)
growth inhibitors
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 152) The most common auxin is:
A)
GA
done
clear
B)
ABA
done
clear
C)
kinetuv
done
clear
D)
IAA
done
clear
View Answer play_arrow
question_answer 153) Gibberellin promotes cell division and elongation in:
A)
leaves
done
clear
B)
roots
done
clear
C)
shoots
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 154) The deteriorative processes in plants, that naturally terminate their functional life, are collectively called:
A)
wilting
done
clear
B)
abscission
done
clear
C)
plasmolysis
done
clear
D)
senescence
done
clear
View Answer play_arrow
question_answer 155) Mendelian recombinations are due to:
A)
linkage
done
clear
B)
mutations
done
clear
C)
dominant characters
done
clear
D)
independent assortment
done
clear
View Answer play_arrow
question_answer 156) Chromosome nui \[2n-1\]is an example of:
A)
trisomy
done
clear
B)
euploidy
done
clear
C)
polyploidy
done
clear
D)
monosomy
done
clear
View Answer play_arrow
question_answer 157) Agrobacterium tumefaciens contains a large plasmid, which induces tumour in the plants, it is termed as:
A)
\[Ti\]plasmid
done
clear
B)
\[Ri\]plasmid
done
clear
C)
recombinant plasmid
done
clear
D)
shine delgrano sequence
done
clear
View Answer play_arrow
question_answer 158) The organisms inhabiting a common environment belong to the same:
A)
species
done
clear
B)
genus
done
clear
C)
population
done
clear
D)
community
done
clear
View Answer play_arrow
question_answer 159) Minerals, metals and fossil fuels are which type of resources of energy?
A)
renewable
done
clear
B)
non-renewable
done
clear
C)
biodegradable
done
clear
D)
degradable
done
clear
View Answer play_arrow
question_answer 160) Foul smell in the water bodies of tanks, ponds etc. is due to:
A)
aerobiosis
done
clear
B)
ariaerobiosis
done
clear
C)
psammophytes
done
clear
D)
biological magnification
done
clear
View Answer play_arrow
question_answer 161) Ethology is the study of:
A)
behaviour of animals
done
clear
B)
past life of organism
done
clear
C)
disease causing pathogens
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 162) The study of molluscan is called:
A)
Nerology
done
clear
B)
Conchology
done
clear
C)
Malacology
done
clear
D)
Craniology
done
clear
View Answer play_arrow
question_answer 163) The globular proteins undergo structural changes, in response to extremes of pH or temperature, are called:
A)
renaturation
done
clear
B)
denaturation
done
clear
C)
combination
done
clear
D)
both a and b
done
clear
View Answer play_arrow
question_answer 164) Sex chromosomes are also known as:
A)
automomes
done
clear
B)
allosomes
done
clear
C)
genome
done
clear
D)
karyotype
done
clear
View Answer play_arrow
question_answer 165) The lytic enzyme present in semen is:
A)
ligase
done
clear
B)
estrogenase
done
clear
C)
androgenase
done
clear
D)
hyaluronidase
done
clear
View Answer play_arrow
question_answer 166) The fastest acting enzyme, in the biological kingdom, is:
A)
lipase
done
clear
B)
amylase
done
clear
C)
peptidase
done
clear
D)
carbonic anhydrase
done
clear
View Answer play_arrow
question_answer 167) Chymotrypsin is produced by:
A)
liver
done
clear
B)
pancreas
done
clear
C)
stomach
done
clear
D)
duodenum
done
clear
View Answer play_arrow
question_answer 168) In which of the following kingdom diatoms are placed?
A)
Plantae
done
clear
B)
Fungi
done
clear
C)
Protozoans
done
clear
D)
Protista
done
clear
View Answer play_arrow
question_answer 169) Nucleus of Monocystis is:
A)
bilobed
done
clear
B)
spherical
done
clear
C)
multilobed
done
clear
D)
bean shaped
done
clear
View Answer play_arrow
question_answer 170) Plasmodium is an:
A)
endoparasite
done
clear
B)
ectoparasite
done
clear
C)
intercellular parasite
done
clear
D)
both a and b
done
clear
View Answer play_arrow
question_answer 171) Choanocyte is an ascon type of canal system, which is lined by:
A)
porocyte
done
clear
B)
tncurrent
done
clear
C)
apopyle
done
clear
D)
spongocoel
done
clear
View Answer play_arrow
question_answer 172) Which of the following are also known as collar cells?
A)
Choanocytes
done
clear
B)
Pinocytes
done
clear
C)
Thesocytes
done
clear
D)
Cnidoblast
done
clear
View Answer play_arrow
question_answer 173) Rostellum and hooks are absent in the scolex of:
A)
Taenia seginata
done
clear
B)
Taenia solwm
done
clear
C)
Echinococcus granulosus
done
clear
D)
Fasciola hepatica
done
clear
View Answer play_arrow
question_answer 174) Coelom in Ascaris lumbricoides, is:
A)
truecoelom
done
clear
B)
pseudocoelom
done
clear
C)
schizocoelom
done
clear
D)
phycocoelom
done
clear
View Answer play_arrow
question_answer 175) Which of the following animal is sanguivorous?
A)
Nereis
done
clear
B)
Tapeworm
done
clear
C)
Earthworm
done
clear
D)
Hirudinaria
done
clear
View Answer play_arrow
question_answer 176) Sand fly spreads a particular type of disease by its:
A)
proboscis
done
clear
B)
mandible
done
clear
C)
salivary gland
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 177) Which of the following belongs to the class Amphineura?
A)
Chiton
done
clear
B)
Nautilus
done
clear
C)
Dentalium
done
clear
D)
Pinctada
done
clear
View Answer play_arrow
question_answer 178) Ambulacral system is mainly useful for:
A)
locomotion
done
clear
B)
circulation
done
clear
C)
feeding
done
clear
D)
defence
done
clear
View Answer play_arrow
question_answer 179) Swim bladder is present in:
A)
star fishes
done
clear
B)
bony fishes
done
clear
C)
carp fishes
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 180) The Indian salamander is:
A)
Siren
done
clear
B)
Toilotriton
done
clear
C)
Amblystoma
done
clear
D)
Nectzirus
done
clear
View Answer play_arrow
question_answer 181) The character of birds, without exception, is:
A)
omnivorous
done
clear
B)
flying wings
done
clear
C)
beak without teeth
done
clear
D)
lay eggs with calcareous shell
done
clear
View Answer play_arrow
question_answer 182) Blood platelets are found only in the blood of:
A)
birds
done
clear
B)
reptiles
done
clear
C)
mammals
done
clear
D)
amphibians
done
clear
View Answer play_arrow
question_answer 183) Ligament connects:
A)
bone to bone
done
clear
B)
bone to muscle
done
clear
C)
muscle to muscle
done
clear
D)
both b and c
done
clear
View Answer play_arrow
question_answer 184) Which of the following fatty acid is not synthesised in the human body?
A)
Glycerol
done
clear
B)
Cholesterol
done
clear
C)
Linoleic acid
done
clear
D)
Both a and b
done
clear
View Answer play_arrow
question_answer 185) Which of the following blood vessel in the circulatory system of frog has more deoxygenated blood?
A)
Pulmonary artery
done
clear
B)
Precaval veins
done
clear
C)
Pulmocutaneous artery
done
clear
D)
Pulmocutaneous vein
done
clear
View Answer play_arrow
question_answer 186) Tachycardia is:
A)
fast heart rate
done
clear
B)
slow heart rate
done
clear
C)
stop heart rate
done
clear
D)
normal heart rate
done
clear
View Answer play_arrow
question_answer 187) Atrial natriuretic hormone is produced by:
A)
kidney
done
clear
B)
heart
done
clear
C)
duodenum
done
clear
D)
thyroid gland
done
clear
View Answer play_arrow
question_answer 188) Cells present in the inner lining of kidneys are:
A)
podocytes
done
clear
B)
choanocytes
done
clear
C)
pinocytes
done
clear
D)
nephrocytes
done
clear
View Answer play_arrow
question_answer 189) Which of the following does not control the heart beat?
A)
Vagus
done
clear
B)
Epinephrine
done
clear
C)
Norepinephrine
done
clear
D)
Glossopharyngeal nerve
done
clear
View Answer play_arrow
question_answer 190) The egg of frog is:
A)
telolecithal
done
clear
B)
microlecithal
done
clear
C)
alecithal
done
clear
D)
centrolecithal
done
clear
View Answer play_arrow
question_answer 191) Which of the following undergoes spermigenesis?
A)
Spermatids
done
clear
B)
Spermatogoma
done
clear
C)
Primary spermatocytes
done
clear
D)
Secondary spermatocytes
done
clear
View Answer play_arrow
question_answer 192) Antifertilizin is present on:
A)
egg
done
clear
B)
tail
done
clear
C)
ovum
done
clear
D)
spermatozoa
done
clear
View Answer play_arrow
question_answer 193) Swan neck flask experiment proved:
A)
biogenesis
done
clear
B)
abiogenesis
done
clear
C)
gene therapy
done
clear
D)
both a and b
done
clear
View Answer play_arrow
question_answer 194) Cosmozoic theory was proposed by:
A)
Helmholtz
done
clear
B)
Richter
done
clear
C)
Pasteur
done
clear
D)
Arrhenius
done
clear
View Answer play_arrow
question_answer 195) The idea, not related to the Darwinian evolutionary theory is:
A)
survival of the fittest
done
clear
B)
struggle for existence
done
clear
C)
inheritance of acquired characters
done
clear
D)
origin of species by natural selection
done
clear
View Answer play_arrow
question_answer 196) Which of the following has the least number of chromosomes?
A)
Amoeba
done
clear
B)
Drosophila
done
clear
C)
Pheretima
done
clear
D)
Ascaris megalocephala
done
clear
View Answer play_arrow
question_answer 197) Exotoxins are related with:
A)
tetanus
done
clear
B)
diphtheria
done
clear
C)
cancer
done
clear
D)
both a and b
done
clear
View Answer play_arrow
question_answer 198) Argentaffin may arise in:
A)
pancreas
done
clear
B)
bile duct
done
clear
C)
intestinal tract
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 199) Which of these may cause hypothermia in humans?
A)
Smoking
done
clear
B)
LSD
done
clear
C)
Dopamine
done
clear
D)
Alcohol consumption
done
clear
View Answer play_arrow
question_answer 200) Which of the following is a dual purpose breed?
A)
Sindhi
done
clear
B)
Deoni
done
clear
C)
Jersey
done
clear
D)
Sahiwal
done
clear
View Answer play_arrow
question_answer 201) The various practices and norms for banks transactions are laid down by the Reserve Bank of India.
A)
are laid up
done
clear
B)
are lead down
done
clear
C)
are lead up
done
clear
D)
no improvement
done
clear
View Answer play_arrow
question_answer 202) She is having a lot of money:
A)
is having lot of
done
clear
B)
has lot of
done
clear
C)
has a lot of
done
clear
D)
no improvement
done
clear
View Answer play_arrow
question_answer 203) They all loved each other:
A)
one another
done
clear
B)
for each other
done
clear
C)
among each other
done
clear
D)
no improvement
done
clear
View Answer play_arrow
question_answer 204) Directions: In each of the following questions, find out which part of the sentence has an error. In each of the following questions, find out which part of the sentence has an error.
A)
He
done
clear
B)
worked
done
clear
C)
hard and
done
clear
D)
failed
done
clear
View Answer play_arrow
question_answer 205) Directions: In each of the following questions, find out which part of the sentence has an error. In each of the following questions, find out which part of the sentence has an error.
A)
The Jury
done
clear
B)
is
done
clear
C)
divided
done
clear
D)
on the issue
done
clear
View Answer play_arrow
question_answer 206) Directions: In each of the following questions, find out which part of the sentence has an error. In each of the following questions, find out which part of the sentence has an error.
A)
I will wait here
done
clear
B)
until you
done
clear
C)
do not
done
clear
D)
come
done
clear
View Answer play_arrow
question_answer 207) Directions: In each of the following questions, find out which part of the sentence has an error. In each of the following questions, find out which part of the sentence has an error.
A)
Please open
done
clear
B)
the book
done
clear
C)
on page
done
clear
D)
five
done
clear
View Answer play_arrow
question_answer 208) Directions: In each of the following questions one word in bold is followed by four words. Choose the word opposite in meaning. Cronies:
A)
comer
done
clear
B)
friends
done
clear
C)
enemies
done
clear
D)
pals
done
clear
View Answer play_arrow
question_answer 209) Directions: In each of the following questions one word in bold is followed by four words. Choose the word opposite in meaning. Glint:
A)
gleam
done
clear
B)
happiness
done
clear
C)
excitement
done
clear
D)
despair
done
clear
View Answer play_arrow
question_answer 210) Directions: In each of the following questions one word in bold is followed by four words. Choose the word opposite in meaning. Apprehensive:
A)
question
done
clear
B)
worried
done
clear
C)
relaxed
done
clear
D)
anxious
done
clear
View Answer play_arrow
question_answer 211) In 2003, in an eco-marathon contest in Hiroshima Fancy Carol recorded the mileage of distance per litre of petrol as:
A)
396 km/litre
done
clear
B)
936 km/litre
done
clear
C)
1962 km/litre
done
clear
D)
3962 km/litre
done
clear
View Answer play_arrow
question_answer 212) Since the Britishers wanted India to produce and supply raw materials to feed English factories, they pushed a policy that encouraged the cultivation of:
A)
jute
done
clear
B)
indigo
done
clear
C)
cotton
done
clear
D)
commercial crop
done
clear
View Answer play_arrow
question_answer 213) Which of the following was a medical doctor?
A)
John Webster
done
clear
B)
Thomas Gray
done
clear
C)
Somerset Maugham
done
clear
D)
Samuel Taylor Coleridge
done
clear
View Answer play_arrow
question_answer 214) Georgetown is the capital of:
A)
Greece
done
clear
B)
Guatemala
done
clear
C)
Guinea
done
clear
D)
Guyana
done
clear
View Answer play_arrow
question_answer 215) Tea is grown on:
A)
plateaus
done
clear
B)
mountains
done
clear
C)
hill slopes
done
clear
D)
mountain valley
done
clear
View Answer play_arrow
question_answer 216) Which is the chief air pollutant?
A)
Nitrogen
done
clear
B)
Ammonia
done
clear
C)
Carbon dioxide
done
clear
D)
Sulphur dioxide
done
clear
View Answer play_arrow
question_answer 217) Normal blood pressure of man is:
A)
80/120 mm Hg
done
clear
B)
90/140 mm Hg
done
clear
C)
120/160 mm Hg
done
clear
D)
85/120 mm Hg
done
clear
View Answer play_arrow
question_answer 218) How much noise is produced when a man breathes?
A)
35 decibel
done
clear
B)
0.1 decibel
done
clear
C)
0.5 decibel
done
clear
D)
10 decibel
done
clear
View Answer play_arrow
question_answer 219) A myopic eye can be corrected by using a:
A)
convex lens
done
clear
B)
concave lens
done
clear
C)
plane mirror
done
clear
D)
cylindrical lens
done
clear
View Answer play_arrow
question_answer 220) National income accounting is the study of the income and expenditure of the entire:
A)
family
done
clear
B)
state
done
clear
C)
economy
done
clear
D)
organization
done
clear
View Answer play_arrow
question_answer 221) The solution of the equations \[-6+5y=2\]and\[-5x+6y=9\]is:
A)
\[x=1\]and \[y=2\]
done
clear
B)
\[x=2\]and\[y=3\]
done
clear
C)
\[x=3\]and \[y=4\]
done
clear
D)
\[x=4\]and \[y=5\]
done
clear
View Answer play_arrow
question_answer 222) The\[g.c.d.\]of the numbers 96, 528, 1584 and 2016 is:
A)
24
done
clear
B)
48
done
clear
C)
72
done
clear
D)
96
done
clear
View Answer play_arrow
question_answer 223) If\[{{2}^{x}}={{8}^{y+1}}\]and\[{{9}^{y}}={{3}^{x-9}},\]then the values of\[x\]and y are:
A)
\[x=18\]and \[y=9\]
done
clear
B)
\[x=21\]and \[y=6\]
done
clear
C)
\[x=23\]arid \[y=4\]
done
clear
D)
\[x=25\]and \[y=10\]
done
clear
View Answer play_arrow
question_answer 224) The 1 c.m. of the polynomials\[20{{x}^{2}}y({{x}^{2}}-{{y}^{2}})\]and\[35x{{y}^{2}}(x-y)\]is:
A)
\[24({{x}^{2}}-{{y}^{2}})\]
done
clear
B)
\[140{{x}^{2}}{{y}^{2}}({{x}^{2}}-{{y}^{2}})\]
done
clear
C)
\[35{{x}^{3}}{{y}^{3}}(x-y)\]
done
clear
D)
\[80xy({{x}^{2}}-{{y}^{2}})\]
done
clear
View Answer play_arrow
question_answer 225) If\[15({{x}^{2}}+{{y}^{2}})-34xy=0,\]then the value of\[x:y\]is:
A)
3 : 5 or 5 : 3
done
clear
B)
4 : 3 or 3 : 4
done
clear
C)
1 : 2 or 2 : 1
done
clear
D)
3:1 or 1 : 3
done
clear
View Answer play_arrow
question_answer 226) The solution of the rational expression\[\frac{2{{x}^{2}}+x-3}{{{(x-1)}^{2}}}+\frac{2{{x}^{2}}+5x+3}{({{x}^{2}}-1)}\]is:
A)
\[\frac{(2x+3)}{(x-1)}\]
done
clear
B)
\[\frac{(x+1)}{(2x+3)}\]
done
clear
C)
\[\frac{(x-1)}{(x+1)}\]
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 227) What number must be added to each term of the ratio\[4:5\]so that it may become equal to \[5:6\]?
A)
1
done
clear
B)
2
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 228) If\[\alpha \]and\[\beta \]are the roots of the quadratic equation\[2{{x}^{2}}+5x+k=0\]and\[{{\alpha }^{2}}+{{\beta }^{2}}+\alpha \beta =\frac{21}{4},\]then the value of k is:
A)
1
done
clear
B)
2
done
clear
C)
6
done
clear
D)
7
done
clear
View Answer play_arrow
question_answer 229) The time taken by a train 120 m long, running at 54 km/hr, in crossing an electric pole is:
A)
2 sec
done
clear
B)
4 sec
done
clear
C)
8 sec
done
clear
D)
10 sec
done
clear
View Answer play_arrow
question_answer 230) A vendor bought toffees at 6 for a rupee. How many for a rupee must he sell to gain 20%?
A)
3 toffees
done
clear
B)
4 toffees
done
clear
C)
5 toffees
done
clear
D)
7 toffees
done
clear
View Answer play_arrow
question_answer 231)
Statements: Some politicians are honest. All the bonest persons are respected. Conclusions I. All politicians are respected. II. Some politicians are respected.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
question_answer 232)
Statements: All scholars are thinkers. Some thinkers are wise. Conclusions I. All scholars are not wise. II. Some scholars are wise.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
question_answer 233)
Statements: All flies are insects, m All insects .can fly Conclusions I. Some flies are not insects II. All insects are flies.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
question_answer 234)
Statements: Some engineers are not competent. Some competent persons are educated. Conclusions I. All engineers are not competent. II. All competent persons are not educated.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
question_answer 235)
Statements: All scholars are genius. No woman is a scholar. Conclusions I. No woman is a genius. II. All genius are men.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
question_answer 236) Wood : Charcoal : : Coal : ?
A)
smoke
done
clear
B)
coke
done
clear
C)
ash
done
clear
D)
fire
done
clear
View Answer play_arrow
question_answer 237) What is related to health as clean is related to contamination?
A)
Pollution
done
clear
B)
Heartiness
done
clear
C)
Affliction
done
clear
D)
Therapeutics
done
clear
View Answer play_arrow
question_answer 238) If you are going towards South, then turn left, then left again, then right and right again. In which direction are you going now?
A)
East
done
clear
B)
West
done
clear
C)
North
done
clear
D)
South
done
clear
View Answer play_arrow
question_answer 239) Complete the series: 84, 103, 120, 135, 148,:
A)
137
done
clear
B)
148
done
clear
C)
159
done
clear
D)
161
done
clear
View Answer play_arrow
question_answer 240) Find ODD man out:
A)
IGLOO
done
clear
B)
HUT
done
clear
C)
FLAT
done
clear
D)
WORKSHOP
done
clear
View Answer play_arrow