-
question_answer1)
Which of the following elements does not form stable diatomic molecules [CBSE PMT 1989, 94]
A)
Iodine done
clear
B)
Phosphorus done
clear
C)
Nitrogen done
clear
D)
Oxygen done
clear
View Solution play_arrow
-
question_answer2)
Producer gas is a mixture of [DPMT 1982; CPMT 1978]
A)
\[CO\] and \[{{N}_{2}}\] done
clear
B)
\[C{{O}_{2}}\] and\[{{H}_{2}}\] done
clear
C)
\[CO\] and \[{{H}_{2}}\] done
clear
D)
\[C{{O}_{2}}\] and \[{{N}_{2}}\] done
clear
View Solution play_arrow
-
question_answer3)
Which one of the following combines with \[Fe(II)\] ions to form a brown complex [AIIMS 1982, 83, 87; BHU 1998; CBSE PMT 2000; Pb. PMT 2000; AFMC 1988, 92; MP PET 1997, 2000, 01]
A)
\[{{N}_{2}}O\] done
clear
B)
\[NO\] done
clear
C)
\[{{N}_{2}}{{O}_{3}}\] done
clear
D)
\[{{N}_{2}}{{O}_{5}}\] done
clear
View Solution play_arrow
-
question_answer4)
Metaphosphoric acid has the formula [CPMT 1973, 89, 93]
A)
\[{{H}_{3}}P{{O}_{4}}\] done
clear
B)
\[HP{{O}_{3}}\] done
clear
C)
\[{{H}_{2}}P{{O}_{3}}\] done
clear
D)
\[{{H}_{3}}P{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer5)
Which of the following is the most suitable drying agent for ammonia gas [MP PMT 1989; CBSE PMT 1989; DPMT 1982; CPMT 1974, 78, 91; BHU 1986, 96; 2001; IIT (Screening) 2000]
A)
Calcium oxide done
clear
B)
Anhydrous calcium chloride done
clear
C)
Phosporus pentoxide done
clear
D)
Conc. sulphuric acid done
clear
View Solution play_arrow
-
question_answer6)
Each of the following is true for white and red phosphorus except that they [CBSE PMT 1989]
A)
Are both soluble in \[C{{S}_{2}}\] done
clear
B)
Can be oxidised by heating in air done
clear
C)
Consists of same kind of atoms done
clear
D)
Can be converted into one another done
clear
View Solution play_arrow
-
question_answer7)
Which of the following is a tetrabasic acid [CPMT 1988]
A)
Orthophosphorus acid done
clear
B)
Orthophosphoric acid done
clear
C)
Metaphosphoric acid done
clear
D)
Pyrophosphoric acid done
clear
View Solution play_arrow
-
question_answer8)
Phosphine is prepared by the reaction of [MP PET/PMT 1988]
A)
\[P\] and \[{{H}_{2}}S{{O}_{4}}\] done
clear
B)
\[P\] and \[NaOH\] done
clear
C)
\[P\] and \[{{H}_{2}}S\] done
clear
D)
\[P\] and \[HN{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer9)
Which of the following is not known [MP PET/PMT 1988; NCERT 1982; CBSE PMT 1989; MP PET 1993]
A)
\[NC{{l}_{5}}\] done
clear
B)
\[N{{I}_{3}}\] done
clear
C)
\[SbC{{l}_{3}}\] done
clear
D)
\[NC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer10)
Chemical formula for the phosphorus molecule is [CPMT 1976, 80, 84, 90; BHU 1984, 86; NCERT 1977]
A)
\[P\] done
clear
B)
\[{{P}_{4}}\] done
clear
C)
\[{{P}_{2}}\] done
clear
D)
\[{{P}_{5}}\] done
clear
View Solution play_arrow
-
question_answer11)
White phosphorus \[\left( {{P}_{4}} \right)\] has [IIT 1998]
A)
Six P - P single bonds done
clear
B)
Four P - P single bonds done
clear
C)
Four lone pairs of electrons done
clear
D)
PPP angle of \[{{60}^{o}}\] done
clear
View Solution play_arrow
-
question_answer12)
Ammonium nitrate decomposes on heating into [NCERT 1974,75; CPMT 1973, 78, 88, 94; AMU 1984]
A)
Ammonia and nitric acid done
clear
B)
Nitrous oxide and water done
clear
C)
Nitrogen, hydrogen and ozone done
clear
D)
Nitric oxide, nitrogen dioxide and hydrogen done
clear
View Solution play_arrow
-
question_answer13)
In Birkeland-Eyde process, the raw material used is [CPMT 1982, 86]
A)
Air done
clear
B)
\[N{{H}_{3}}\] done
clear
C)
\[N{{O}_{2}}\] done
clear
D)
\[HN{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer14)
Among the following nitrates, Lead nitrate, Silver nitrate and Ammonium nitrate; the one that decomposes without leaving any solid residue is [NCERT 1983]
A)
Lead nitrate done
clear
B)
Ammonium nitrate done
clear
C)
Silver nitrate done
clear
D)
Sodium nitrate done
clear
View Solution play_arrow
-
question_answer15)
Of the different allotropes of phosphorus, the one which is most reactive is [CPMT 1983; NCERT 1978; CBSE PMT 1999; Kurukshetra CEE 1998]
A)
Violet phosphorus done
clear
B)
Scarlet phosphorus done
clear
C)
Red phosphorus done
clear
D)
White phosphorus done
clear
View Solution play_arrow
-
question_answer16)
Phosphine is generally prepared in the laboratory [CPMT 1983, 2003]
A)
By heating phosphorus in a current of hydrogen done
clear
B)
By heating white phosphorus with aqueous solution of caustic potash done
clear
C)
By decomposition of \[{{P}_{2}}{{H}_{4}}\] at \[{{110}^{o}}C\] done
clear
D)
By heating red phosphorus with an aqueous solution of caustic soda done
clear
View Solution play_arrow
-
question_answer17)
Which of the following elements is most metallic [CPMT 1983; MP PMT 1993]
A)
Phosphorus done
clear
B)
Arsenic done
clear
C)
Antimony done
clear
D)
Bismuth done
clear
View Solution play_arrow
-
question_answer18)
The basicity of orthophosphoric acid is [CPMT 1984, 91]
A)
2 done
clear
B)
3 done
clear
C)
4 done
clear
D)
5 done
clear
View Solution play_arrow
-
question_answer19)
\[HN{{O}_{2}}\] acts as [AFMC 1992]
A)
Oxidising agent done
clear
B)
Reducing agent done
clear
C)
Both a and b done
clear
D)
Its solution is stable done
clear
View Solution play_arrow
-
question_answer20)
Nitrogen dioxide cannot be obtained by heating [CPMT 1989; IIT 1985; CPMT 1993]
A)
\[KN{{O}_{3}}\] done
clear
B)
\[Pb{{(N{{O}_{3}})}_{2}}\] done
clear
C)
\[Cu{{(N{{O}_{3}})}_{2}}\] done
clear
D)
\[AgN{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer21)
When heated \[N{{H}_{3}}\] is passed over \[CuO\] gas evolved is [BCECE 2005]
A)
\[{{N}_{2}}\] done
clear
B)
\[{{N}_{2}}O\] done
clear
C)
\[HN{{O}_{3}}\] done
clear
D)
\[N{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer22)
Non-combustible hydride is [CPMT 1979]
A)
\[N{{H}_{3}}\] done
clear
B)
\[P{{H}_{3}}\] done
clear
C)
\[As{{H}_{3}}\] done
clear
D)
\[Sb{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer23)
On heating a mixture of \[N{{H}_{4}}Cl\]and\[KN{{O}_{2}}\] we get [CPMT 1972, 79; NCERT 1977]
A)
\[N{{H}_{4}}N{{O}_{3}}\] done
clear
B)
\[{{N}_{2}}\] done
clear
C)
\[{{N}_{2}}O\] done
clear
D)
\[NO\] done
clear
View Solution play_arrow
-
question_answer24)
Which of the following oxide of nitrogen is the anhydride of \[HN{{O}_{3}}\] [CPMT 1979,80,89,97; MP PET/PMT 1988; KCET 1991; CBSE PMT 1989, 91,99; EAMCET 1991; NCERT 1975; MP PET 1989; MP PMT 1994]
A)
\[NO\] done
clear
B)
\[{{N}_{2}}{{O}_{3}}\] done
clear
C)
\[{{N}_{3}}{{O}_{4}}\] done
clear
D)
\[{{N}_{2}}{{O}_{5}}\] done
clear
View Solution play_arrow
-
question_answer25)
Phosphorus is manufactured by heating in a electric furnace a mixture of NCERT 1977; CPMT 1974, 78, 81, 86]
A)
Bone ash and coke done
clear
B)
Bone ash and silica done
clear
C)
Bone ash, silica and coke done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer26)
A certain element forms a solid oxide which when dissolved in water forms an acidic solution, the element is [CPMT 1972, 78]
A)
Argon done
clear
B)
Potassium done
clear
C)
Phosphorus done
clear
D)
Sulphur done
clear
View Solution play_arrow
-
question_answer27)
Dissociation of \[{{H}_{3}}P{{O}_{4}}\] occurs in following stages [CPMT 1976]
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer28)
Nitrogen forms how many oxides
A)
3 done
clear
B)
4 done
clear
C)
5 done
clear
D)
6 done
clear
View Solution play_arrow
-
question_answer29)
The P-P-P bond angle in white phosphorus is [MP PET 1991]
A)
\[{{120}^{o}}\] done
clear
B)
\[{{109}^{o}}2{8}'\] done
clear
C)
\[{{90}^{o}}\] done
clear
D)
\[{{60}^{o}}\] done
clear
View Solution play_arrow
-
question_answer30)
Ammonium dichromate on heating gives [BHU 1973, 78; CBSE PMT 1993; MP PMT 1993]
A)
Chromium oxide and ammonia done
clear
B)
Chromic acid and nitrogen done
clear
C)
Chromium oxide and nitrogen done
clear
D)
Chromic acid and ammonia done
clear
View Solution play_arrow
-
question_answer31)
When concentrated nitric acid is heated, it decomposes to give [BHU 1973; CPMT 1996]
A)
\[{{O}_{2}}\] and \[{{N}_{2}}\] done
clear
B)
\[NO\] done
clear
C)
\[{{N}_{2}}{{O}_{5}}\] done
clear
D)
\[N{{O}_{2}}\] and \[{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer32)
The element which catches fire in air at \[{{30}^{o}}C\] and is stored under water is [BHU 1973; MP PET 1989,99]
A)
Calcium done
clear
B)
Sodium done
clear
C)
Phosphorus done
clear
D)
Zinc done
clear
View Solution play_arrow
-
question_answer33)
A solution of ammonia in water contains [CBSE PMT 1991]
A)
\[{{H}^{+}}\] done
clear
B)
\[O{{H}^{-}}\] done
clear
C)
Only \[NH_{4}^{+}\] done
clear
D)
\[O{{H}^{-}},\] \[N{{H}_{4}}N{{O}_{2}}\] and \[N{{H}_{4}}OH\] molecules done
clear
View Solution play_arrow
-
question_answer34)
Among the members of V A group (N, P, As, Sb and Bi), which of the following properties shows an increase as we go down from nitrogen to bismuth [CPMT 1982]
A)
Stability of +3 oxidation state done
clear
B)
Reducing character of hydrides done
clear
C)
Electronegativity done
clear
D)
Acidic nature of the pentoxide done
clear
View Solution play_arrow
-
question_answer35)
The important method for the fixation of nitrogen is [CPMT 1985, 94]
A)
Haber done
clear
B)
Solvay done
clear
C)
Deacon done
clear
D)
Fischer method done
clear
View Solution play_arrow
-
question_answer36)
Which of the following is oxidised in air [AFMC 1987; KCET 1991]
A)
White phosphorus done
clear
B)
\[C{{H}_{4}}\] done
clear
C)
\[{{H}_{2}}O\] done
clear
D)
\[NaCl\] done
clear
View Solution play_arrow
-
question_answer37)
A pure sample of nitrogen is prepared by heating
A)
Calcium cyanamide done
clear
B)
Barium azide done
clear
C)
Ammonium hydroxide done
clear
D)
Ammonium nitrite done
clear
View Solution play_arrow
-
question_answer38)
Nitrous oxide
A)
Is a mixed oxide done
clear
B)
Is an acidic oxide done
clear
C)
Is highly soluble in hot water done
clear
D)
Supports the combustion of sulphur done
clear
View Solution play_arrow
-
question_answer39)
Which of the following represents laughing gas [CPMT 1986, 89; Manipal MEE 1995;MP PMT 1990; MP PET 1995; RPMT 1999; AFMC 2002]
A)
\[NO\] done
clear
B)
\[{{N}_{2}}O\] done
clear
C)
\[N{{O}_{2}}\] done
clear
D)
\[{{N}_{2}}{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer40)
\[N{{O}_{2}}\] is a mixed oxide is proved by the first that with \[NaOH\], it forms
A)
Nitrites salt done
clear
B)
Nitrates salt done
clear
C)
Mixture of nitrate and nitrite done
clear
D)
Ammonia done
clear
View Solution play_arrow
-
question_answer41)
Which of the following metal produces nitrous oxide with dil. \[Zn\]
A)
\[Sn\] done
clear
B)
\[Zn\] done
clear
C)
\[Cu\] done
clear
D)
\[Ag\] done
clear
View Solution play_arrow
-
question_answer42)
Which of the following acid exist in polymeric form
A)
\[HP{{O}_{3}}\] done
clear
B)
\[{{H}_{4}}{{P}_{2}}{{O}_{7}}\] done
clear
C)
\[{{H}_{3}}P{{O}_{4}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer43)
Superphosphate of lime is [AMU 1985]
A)
A mixture of normal calcium phosphate and gypsum done
clear
B)
A mixture of primary calcium phosphate and gypsum done
clear
C)
Normal calcium phosphate done
clear
D)
Soluble calcium phosphate done
clear
View Solution play_arrow
-
question_answer44)
If phosphoric acid is allowed to react with sufficient quantity of \[NaOH\], the product obtained is [DPMT 1983; MP PMT 1983]
A)
\[NaHP{{O}_{3}}\] done
clear
B)
\[N{{a}_{2}}HP{{O}_{4}}\] done
clear
C)
\[Na{{H}_{2}}P{{O}_{4}}\] done
clear
D)
\[N{{a}_{3}}P{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer45)
White phosphorus contains [CPMT 1978; KCET (Med.) 2000; MP PET 1990]
A)
\[{{P}_{5}}\] molecules done
clear
B)
\[{{P}_{4}}\] molecules done
clear
C)
\[{{P}_{6}}\] molecules done
clear
D)
\[{{P}_{2}}\] molecules done
clear
View Solution play_arrow
-
question_answer46)
In the catalytic oxidation of ammonia an oxide is formed which is used in the preparation of \[HN{{O}_{3}}\]. This oxide is [CPMT 1984; KCET 1990; AIIMS 1996]
A)
\[{{N}_{2}}{{O}_{5}}\] done
clear
B)
\[{{N}_{2}}{{O}_{4}}\] done
clear
C)
\[N{{O}_{2}}\] done
clear
D)
\[NO\] done
clear
View Solution play_arrow
-
question_answer47)
Nitric acid oxidises phosphorus to [CPMT 1984; JIPMER 2002]
A)
\[{{H}_{2}}{{P}_{2}}{{O}_{7}}\] done
clear
B)
\[{{H}_{3}}P{{O}_{3}}\] done
clear
C)
\[AgN{{O}_{3}}\] done
clear
D)
\[NO\] done
clear
View Solution play_arrow
-
question_answer48)
Which one of the following statements is true for \[HN{{O}_{2}}\] [CPMT 1980, 84]
A)
It is very stable in aqueous solution done
clear
B)
It cannot act both as an oxidant and as a reductant done
clear
C)
It cannot act as an oxidising agent done
clear
D)
It cannot act as reducing agent done
clear
View Solution play_arrow
-
question_answer49)
Which oxide is alkaline [MP PET 1990]
A)
\[{{P}_{2}}{{O}_{3}}\] done
clear
B)
\[B{{i}_{2}}{{O}_{3}}\] done
clear
C)
\[A{{s}_{2}}{{O}_{3}}\] done
clear
D)
\[{{B}_{2}}{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer50)
Which acid is formed by \[{{P}_{2}}{{O}_{3}}\] [MP PET 1991]
A)
\[{{H}_{3}}P{{O}_{4}}\] done
clear
B)
\[{{H}_{3}}P{{O}_{3}}\] done
clear
C)
\[HP{{O}_{3}}\] done
clear
D)
\[{{H}_{4}}{{P}_{2}}{{O}_{7}}\] done
clear
View Solution play_arrow
-
question_answer51)
Which nitrogen trihalides is least basic [IIT 1987; Kurukshetra CEE 1998; CPMT 1999]
A)
\[N{{F}_{3}}\] done
clear
B)
\[NC{{l}_{3}}\] done
clear
C)
\[NB{{r}_{3}}\] done
clear
D)
\[N{{I}_{3}}\] done
clear
View Solution play_arrow
-
question_answer52)
Dehydrated phosphorus trichloride in water gives [MP PET 1990]
A)
\[HP{{O}_{3}}\] done
clear
B)
\[{{H}_{3}}P{{O}_{4}}\] done
clear
C)
\[{{H}_{3}}P{{O}_{2}}\] done
clear
D)
\[{{H}_{3}}P{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer53)
Which is used in the manufacture of safe matchsticks [DPMT 1982, CPMT 1974, 75]
A)
White phosphorus done
clear
B)
Sulphur done
clear
C)
Red phosphorus done
clear
D)
Selenium done
clear
View Solution play_arrow
-
question_answer54)
Which oxide of nitrogen is coloured gas [IIT 1987; Kurukshetra CEE 1998]
A)
\[{{N}_{2}}O\] done
clear
B)
\[NO\] done
clear
C)
\[{{N}_{2}}{{O}_{5}}\] done
clear
D)
\[N{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer55)
Which oxide do not act as a reducing agent [MP PET 1990]
A)
\[NO\] done
clear
B)
\[N{{O}_{2}}\] done
clear
C)
\[{{N}_{2}}O\] done
clear
D)
\[{{N}_{2}}{{O}_{5}}\] done
clear
View Solution play_arrow
-
question_answer56)
In \[N{{H}_{4}}N{{O}_{2}},\] the oxidation number of nitrogen will be [MP PET 1990]
A)
+ 3 done
clear
B)
+ 5 done
clear
C)
- 3 and + 3 done
clear
D)
+ 3 and + 5 done
clear
View Solution play_arrow
-
question_answer57)
In which compound, the oxidation state of phosphorus is+4 [MP PET 1991]
A)
\[{{P}_{4}}{{O}_{11}}\] done
clear
B)
\[{{P}_{4}}{{O}_{8}}\] done
clear
C)
\[PC{{l}_{5}}\] done
clear
D)
\[NC{{l}_{5}}\] done
clear
View Solution play_arrow
-
question_answer58)
In which compound, the oxidation state of nitrogen is -1 [MP PMT 1989]
A)
\[NO\] done
clear
B)
\[{{N}_{2}}O\] done
clear
C)
\[N{{H}_{2}}OH\] done
clear
D)
\[{{N}_{2}}{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer59)
Which of the following oxide is least acidic [MP PMT 1990; CBSE PMT 1996]
A)
\[{{P}_{4}}{{O}_{6}}\] done
clear
B)
\[{{P}_{4}}{{O}_{10}}\] done
clear
C)
\[A{{s}_{4}}{{O}_{6}}\] done
clear
D)
\[A{{s}_{4}}{{O}_{19}}\] done
clear
View Solution play_arrow
-
question_answer60)
The basic character of hydrides of the V-group elements decreases in the order [CBSE PMT 1996]
A)
\[Sb{{H}_{3}}>P{{H}_{3}}>As{{H}_{3}}>N{{H}_{3}}\] done
clear
B)
\[N{{H}_{3}}>Sb{{H}_{3}}>P{{H}_{3}}>As{{H}_{3}}\] done
clear
C)
\[N{{H}_{3}}>P{{H}_{3}}>As{{H}_{3}}>Sb{{H}_{3}}\] done
clear
D)
\[Sb{{H}_{3}}>As{{H}_{3}}>P{{H}_{3}}>N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer61)
Which is least stable [MP PET 1989]
A)
\[Bi{{H}_{3}}\] done
clear
B)
\[Sb{{H}_{3}}\] done
clear
C)
\[As{{H}_{3}}\] done
clear
D)
\[P{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer62)
Which of the following is not hydrolysed [DPMT 2005]
A)
\[AsC{{l}_{3}}\] done
clear
B)
\[P{{F}_{3}}\] done
clear
C)
\[SbC{{l}_{3}}\] done
clear
D)
\[N{{F}_{3}}\] done
clear
View Solution play_arrow
-
question_answer63)
Electrolysis temperature is maximum for [MP PET 1990]
A)
\[As{{H}_{3}}\] done
clear
B)
\[N{{H}_{3}}\] done
clear
C)
\[P{{H}_{3}}\] done
clear
D)
\[Sb{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer64)
Which of the following is kept in water? [BCECE 2005]
A)
White phosphorous done
clear
B)
Sodium done
clear
C)
Potassium done
clear
D)
Calcium done
clear
View Solution play_arrow
-
question_answer65)
Which of the following substances is used as a fertilizer
A)
\[{{K}_{2}}S{{O}_{4}}.A{{l}_{2}}{{(S{{O}_{4}})}_{3}}.24{{H}_{2}}O\] done
clear
B)
\[Ca{{({{H}_{3}}P{{O}_{4}})}_{2}}{{H}_{2}}O+CaS{{O}_{4}}\] done
clear
C)
\[NaAl{{O}_{2}}\] done
clear
D)
\[Ca{{C}_{2}}\] done
clear
View Solution play_arrow
-
question_answer66)
On adding water to \[BiC{{l}_{3}}\] solution in \[HCl\], the compound produced is
A)
\[B{{i}_{2}}{{O}_{3}}\] done
clear
B)
\[Bi{{(OH)}_{3}}\] done
clear
C)
\[BiOCl\] done
clear
D)
\[BiOC{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer67)
V-A group precipitate was dissolved in \[HN{{O}_{3}}\] and treated with excess of \[N{{H}_{4}}OH\]. It gives a white ppt. because of
A)
\[Cu{{(OH)}_{2}}\] done
clear
B)
\[Cd{{(OH)}_{2}}\] done
clear
C)
\[Bi{{(OH)}_{3}}\] done
clear
D)
\[Hg{{(OH)}_{2}}\] done
clear
View Solution play_arrow
-
question_answer68)
N, P, As, Sb, Bi elements belong to [DPMT 1982]
A)
VA group done
clear
B)
IVA group done
clear
C)
VIIA group done
clear
D)
VB group done
clear
View Solution play_arrow
-
question_answer69)
Which one of the following elements occur free in nature [CPMT 1988]
A)
Nitrogen done
clear
B)
Phosphorus done
clear
C)
Arsenic done
clear
D)
Antimony done
clear
View Solution play_arrow
-
question_answer70)
Which of the following elements of group VA does not show allotropy [CPMT 1980]
A)
\[N\] done
clear
B)
\[Bi\] done
clear
C)
\[P\] done
clear
D)
As done
clear
View Solution play_arrow
-
question_answer71)
Which does not form complex [CPMT 1986]
A)
\[N\] done
clear
B)
\[P\] done
clear
C)
\[As\] done
clear
D)
\[Bi\] done
clear
View Solution play_arrow
-
question_answer72)
The strongest base is [IIT 1989; CPMT 1997; MP PET 2001,03]
A)
\[N{{H}_{3}}\] done
clear
B)
\[P{{H}_{3}}\] done
clear
C)
\[As{{H}_{3}}\] done
clear
D)
\[Sb{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer73)
The most stable hydride is [EAMCET 1988]
A)
\[N{{H}_{3}}\] done
clear
B)
\[P{{H}_{3}}\] done
clear
C)
\[As{{H}_{3}}\] done
clear
D)
\[Sb{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer74)
Which has the lowest boiling point [CBSE PMT 1989]
A)
\[N{{H}_{3}}\] done
clear
B)
\[P{{H}_{3}}\] done
clear
C)
\[As{{H}_{3}}\] done
clear
D)
\[Sb{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer75)
Which is the most explosive [BHU 1984; Roorkee 1989; AIIMS 1996; MP PMT 1985, 2001]
A)
\[NC{{l}_{3}}\] done
clear
B)
\[PC{{l}_{3}}\] done
clear
C)
\[AsC{{l}_{3}}\] done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer76)
Of the following, the most acidic is [EAMCET 1980]
A)
\[A{{s}_{2}}{{O}_{3}}\] done
clear
B)
\[{{P}_{2}}{{O}_{3}}\] done
clear
C)
\[S{{b}_{2}}{{O}_{3}}\] done
clear
D)
\[B{{i}_{2}}{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer77)
Of the following, non-existent compound is [NCERT 1975, 79]
A)
\[P{{H}_{4}}I\] done
clear
B)
\[A{{s}_{2}}{{O}_{3}}\] done
clear
C)
\[SbC{{l}_{2}}\] done
clear
D)
\[A{{s}_{2}}{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer78)
Pure \[{{N}_{2}}\] gas is obtained from [CBSE PMT 1991]
A)
\[N{{H}_{3}}+NaN{{O}_{2}}\] done
clear
B)
\[N{{H}_{4}}Cl+NaN{{O}_{2}}\] done
clear
C)
\[{{N}_{2}}O+Cu\] done
clear
D)
\[{{(N{{H}_{4}})}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
View Solution play_arrow
-
question_answer79)
Pure nitrogen can be prepared from [KCET 1991; AFMC 1993; AMU 1985]
A)
\[N{{H}_{4}}OH\] done
clear
B)
\[C{{a}_{3}}{{N}_{2}}\] done
clear
C)
\[N{{H}_{4}}N{{O}_{2}}\] done
clear
D)
\[Ba{{(N{{O}_{3}})}_{2}}\] done
clear
View Solution play_arrow
-
question_answer80)
Nitrogen combines with metals to form [CPMT 1981, 93]
A)
Nitrites done
clear
B)
Nitrates done
clear
C)
Nitrosyl chloride done
clear
D)
Nitrides done
clear
View Solution play_arrow
-
question_answer81)
Nitrogen is relatively inactive element because [CBSE PMT 1992]
A)
Its atom has a stable electronic configuration done
clear
B)
It has low atomic radius done
clear
C)
Its electronegativity is fairly high done
clear
D)
Dissociation energy of its molecule is fairly high done
clear
View Solution play_arrow
-
question_answer82)
The cyanide ion, \[C{{N}^{-}}\] and \[{{N}_{2}}\] are isoelectronic. But in contrast to \[C{{N}^{-}},\] \[{{N}_{2}}\] is chemically inert because of [IIT 1992]
A)
Low bond energy done
clear
B)
Absence of bond polarity done
clear
C)
Unsymmetrical electron distribution done
clear
D)
Presence of more number of electrons in bonding orbitals done
clear
View Solution play_arrow
-
question_answer83)
Which statement is not correct for nitrogen [AIIMS 1991]
A)
It has a small size done
clear
B)
It does not readily react with \[{{O}_{2}}\] done
clear
C)
It is a typical non-metal done
clear
D)
d-orbitals are available for bonding done
clear
View Solution play_arrow
-
question_answer84)
The element which is essential in nitrogen fixation is [NCERT 1981]
A)
Zinc done
clear
B)
Copper done
clear
C)
Molybdenum done
clear
D)
Boron done
clear
View Solution play_arrow
-
question_answer85)
Laughing gas is prepared by heating [EAMCET 1980]
A)
\[N{{H}_{4}}Cl\] done
clear
B)
\[{{(N{{H}_{4}})}_{2}}S{{O}_{4}}\] done
clear
C)
\[N{{H}_{4}}Cl+NaN{{O}_{3}}\] done
clear
D)
\[N{{H}_{4}}N{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer86)
Nitrogen (I) oxide is produced by [IIT 1989]
A)
Thermal decomposition of ammonium nitrate done
clear
B)
Disproportionation of \[{{N}_{2}}{{O}_{4}}\] done
clear
C)
Thermal decomposition of ammonium nitrite done
clear
D)
Interaction of hydroxyl amine and nitrous acid done
clear
View Solution play_arrow
-
question_answer87)
Which of the following is not correct for \[{{N}_{2}}O\] [CPMT 1984]
A)
It is called laughing gas done
clear
B)
It is nitrous oxide done
clear
C)
It is not a linear molecule done
clear
D)
It is least reactive in all oxides of nitrogen done
clear
View Solution play_arrow
-
question_answer88)
Which of the following oxides of nitrogen is the anhydride of nitrous acid [NCERT 1975; AIIMS 1991]
A)
\[NO\] done
clear
B)
\[{{N}_{2}}{{O}_{3}}\] done
clear
C)
\[{{N}_{2}}{{O}_{4}}\] done
clear
D)
\[{{N}_{2}}{{O}_{5}}\] done
clear
View Solution play_arrow
-
question_answer89)
Which of the following is a true acidic anhydride [NCERT 1977]
A)
\[CO\] done
clear
B)
\[NO\] done
clear
C)
\[Cl{{O}_{2}}\] done
clear
D)
\[{{N}_{2}}{{O}_{5}}\] done
clear
View Solution play_arrow
-
question_answer90)
On strongly heating \[Pb{{(N{{O}_{3}})}_{2}}\] crystals, the gas formed is [NCERT 1980; CPMT 1997]
A)
\[N{{O}_{2}}\] done
clear
B)
\[{{O}_{2}}\] done
clear
C)
\[N{{O}_{2}}+{{O}_{2}}\] done
clear
D)
\[NO\] done
clear
View Solution play_arrow
-
question_answer91)
Nitrogen dioxide is released by heating [AFMC 1992]
A)
\[Pb{{(N{{O}_{3}})}_{2}}\] done
clear
B)
\[KN{{O}_{3}}\] done
clear
C)
\[NaN{{O}_{2}}\] done
clear
D)
\[NaN{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer92)
Nitric oxide is prepared by the action of \[HN{{O}_{3}}\] on [AFMC 1990]
A)
\[Fe\] done
clear
B)
\[Cu\] done
clear
C)
\[Zn\] done
clear
D)
\[Sn\] done
clear
View Solution play_arrow
-
question_answer93)
When lightning flash is produced, which gas may form [EAMCET 1992; AFMC 1989]
A)
Nitrous oxide done
clear
B)
Nitrogen dioxide done
clear
C)
Dinitrogen pentoxide done
clear
D)
Nitric oxide done
clear
View Solution play_arrow
-
question_answer94)
Of the following, which has three electron bond in its structure [CPMT 1986]
A)
Nitrous oxide done
clear
B)
Nitric oxide done
clear
C)
Dinitrogen trioxide done
clear
D)
Nitrogen pentoxide done
clear
View Solution play_arrow
-
question_answer95)
Which of the following oxides of nitrogen is neutral [CPMT 1988]
A)
\[{{N}_{2}}{{O}_{5}}\] done
clear
B)
\[{{N}_{2}}{{O}_{3}}\] done
clear
C)
\[{{N}_{2}}{{O}_{4}}\] done
clear
D)
\[{{N}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer96)
Oxidation of \[NO\] in air produces [KCET 1992]
A)
\[{{N}_{2}}O\] done
clear
B)
\[{{N}_{2}}{{O}_{3}}\] done
clear
C)
\[N{{O}_{2}}\] done
clear
D)
\[{{N}_{2}}{{O}_{5}}\] done
clear
View Solution play_arrow
-
question_answer97)
The reddish brown coloured gas formed when nitric oxide is oxidised by air is [IIT 1979]
A)
\[{{N}_{2}}{{O}_{5}}\] done
clear
B)
\[{{N}_{2}}{{O}_{4}}\] done
clear
C)
\[N{{O}_{2}}\] done
clear
D)
\[{{N}_{2}}{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer98)
When \[AgN{{O}_{3}}\] is heated strongly, the products formed are [Roorkee 1990]
A)
\[NO\] and \[N{{O}_{2}}\] done
clear
B)
\[N{{O}_{2}}\] and \[{{O}_{2}}\] done
clear
C)
\[N{{O}_{2}}\] and \[{{N}_{2}}O\] done
clear
D)
\[NO\] and \[{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer99)
Which of the nitrates on strong heating leaves the metal as the residue [KCET 1990]
A)
\[AgN{{O}_{3}}\] done
clear
B)
\[Pb{{(N{{O}_{3}})}_{2}}\] done
clear
C)
\[Cu{{(N{{O}_{3}})}_{2}}\] done
clear
D)
\[Al{{(N{{O}_{3}})}_{3}}\] done
clear
View Solution play_arrow
-
question_answer100)
Nitrogen dioxide [KCET 1989]
A)
Dissolves in water forming nitric acid done
clear
B)
Does not dissolve in water done
clear
C)
Dissolves in water to form nitrous acid and gives off oxygen done
clear
D)
Dissolves in water to form a mixture of nitrous and nitric acids done
clear
View Solution play_arrow
-
question_answer101)
Concentrated nitric acid oxidises cane sugar to [CBSE PMT 1991]
A)
\[C{{O}_{2}}\] and \[{{H}_{2}}O\] done
clear
B)
\[CO\] and \[{{H}_{2}}O\] done
clear
C)
\[CO,\] \[C{{O}_{2}}\] and \[{{H}_{2}}O\] done
clear
D)
Oxalic acid and water done
clear
View Solution play_arrow
-
question_answer102)
A mixture of ammonia and air at about \[{{800}^{o}}C\] in the presence of Pt gauze forms [Pb. CET 1989]
A)
\[{{N}_{2}}O\] done
clear
B)
\[NO\] done
clear
C)
\[N{{H}_{2}}OH\] done
clear
D)
\[{{N}_{2}}{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer103)
Which of the following acid possesses oxidising, reducing and complex forming properties [MNR 1985]
A)
\[HN{{O}_{3}}\] done
clear
B)
\[{{H}_{2}}S{{O}_{4}}\] done
clear
C)
\[HCl\] done
clear
D)
\[HN{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer104)
Nitrogen is essential constituent of all [MP PMT 1990]
A)
Proteins done
clear
B)
Fats done
clear
C)
Proteins and fats done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer105)
Ammonia gas can be collected by the displacement of [NCERT 1989, 90]
A)
Conc. \[{{H}_{2}}S{{O}_{4}}\] done
clear
B)
Brine done
clear
C)
Water done
clear
D)
Mercury done
clear
View Solution play_arrow
-
question_answer106)
The chemical used for cooling in refrigeration is [CPMT 1981, 88]
A)
\[C{{O}_{2}}\] done
clear
B)
\[N{{H}_{4}}OH\] done
clear
C)
\[N{{H}_{4}}Cl\] done
clear
D)
Liquid \[N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer107)
A hydride of nitrogen which is acidic is [NCERT 1978, 80; CPMT 1980; BHU 1986]
A)
\[N{{H}_{3}}\] done
clear
B)
\[{{N}_{2}}{{H}_{4}}\] done
clear
C)
\[{{N}_{2}}{{H}_{2}}\] done
clear
D)
\[{{N}_{3}}H\] done
clear
View Solution play_arrow
-
question_answer108)
\[PC{{l}_{5}}\] exists but \[NC{{l}_{5}}\] does not because [EAMCET 1977, 82]
A)
Nitrogen has no vacant orbitals done
clear
B)
\[NC{{l}_{5}}\] is unstable done
clear
C)
Nitrogen atom is much smaller done
clear
D)
Nitrogen is highly inert done
clear
View Solution play_arrow
-
question_answer109)
Phosphide ion has the electronic structure similar to that of [CPMT 1988]
A)
Nitride ion done
clear
B)
Fluoride ion done
clear
C)
Sodium ion done
clear
D)
Chloride ion done
clear
View Solution play_arrow
-
question_answer110)
Which of the following phosphorus is most stable [AFMC 1992]
A)
Red done
clear
B)
White done
clear
C)
Black done
clear
D)
All stable done
clear
View Solution play_arrow
-
question_answer111)
Red phosphorus can be obtained from white phosphorus by [KCET 1989]
A)
Heating it with a catalyst in an inert atmosphere done
clear
B)
Distilling it in an inert atmosphere done
clear
C)
Dissolving it in carbon disulphide and crystallizing done
clear
D)
Melting it and pouring the liquid into water done
clear
View Solution play_arrow
-
question_answer112)
Bones glow in the dark because [EAMCET 1980]
A)
They contain shining material done
clear
B)
They contain red phosphorus done
clear
C)
White phosphorus undergoes slow combustion in contact with air done
clear
D)
White phosphorus changes into red form done
clear
View Solution play_arrow
-
question_answer113)
Which of the following properties of white phosphorus are shared by red phosphorus [NCERT 1973, 74]
A)
It shows phosphorescenes in air done
clear
B)
It reacts with hot aqueous \[NaOH\] to give phosphine done
clear
C)
It dissolves in carbon disulphide done
clear
D)
It burns when heated in air done
clear
View Solution play_arrow
-
question_answer114)
Mixture used for the tips of match stick is [DPMT 1984]
A)
\[S+K\] done
clear
B)
\[S{{b}_{2}}{{S}_{3}}\] done
clear
C)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}+S+\]red P done
clear
D)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}+K+S\] done
clear
View Solution play_arrow
-
question_answer115)
In modern process phosphorus is manufactured by [CPMT 1974, 78, 81]
A)
Heating a mixture of phosphorite mineral with sand and coke in electric furnace done
clear
B)
Heating calcium phosphate with coke done
clear
C)
Heating bone ash with coke done
clear
D)
Heating the phosphate mineral with sand done
clear
View Solution play_arrow
-
question_answer116)
White phosphorus when boiled with strong solution of caustic soda produces [CPMT 1989,03]
A)
Phosphine done
clear
B)
Phosphoric acid done
clear
C)
Phosphorus acid done
clear
D)
No reaction done
clear
View Solution play_arrow
-
question_answer117)
White phosphorus reacts with caustic soda. The products are \[P{{H}_{3}}\] and \[Na{{H}_{2}}P{{O}_{2}}\]. This reaction is an example of [IIT 1980; KCET 1993]
A)
Oxidation done
clear
B)
Reduction done
clear
C)
Oxidation and reduction done
clear
D)
Neutralisation done
clear
View Solution play_arrow
-
question_answer118)
Phosphine is not obtained by the reaction [MP PMT 1989]
A)
White P is heated with \[NaOH\] done
clear
B)
Red P is heated with \[NaOH\] done
clear
C)
\[C{{a}_{3}}{{P}_{2}}\] reacts with water done
clear
D)
Phosphorus trioxide is boiled with water done
clear
View Solution play_arrow
-
question_answer119)
\[P{{H}_{4}}I+NaOH\] forms [CBSE PMT 1991]
A)
\[P{{H}_{3}}\] done
clear
B)
\[N{{H}_{3}}\] done
clear
C)
\[{{P}_{4}}{{O}_{6}}\] done
clear
D)
\[{{P}_{4}}{{O}_{10}}\] done
clear
View Solution play_arrow
-
question_answer120)
Phosphine is produced by adding water to [KCET 1991]
A)
\[Ca{{C}_{2}}\] done
clear
B)
\[HP{{O}_{3}}\] done
clear
C)
\[C{{a}_{3}}{{P}_{2}}\] done
clear
D)
\[{{P}_{4}}{{O}_{10}}\] done
clear
View Solution play_arrow
-
question_answer121)
When aluminium phosphide is treated with dil. sulphuric acid [KCET 1989]
A)
\[S{{O}_{2}}\] is liberated done
clear
B)
\[P{{H}_{3}}\] is evolved done
clear
C)
\[{{H}_{2}}S\] is evolved done
clear
D)
\[{{H}_{2}}\] is evolved done
clear
View Solution play_arrow
-
question_answer122)
With reference to protonic acids, which of the following statements is correct [CPMT 1990]
A)
\[P{{H}_{3}}\] is more basic than \[N{{H}_{3}}\] done
clear
B)
\[P{{H}_{3}}\] is less basic than \[N{{H}_{3}}\] done
clear
C)
\[P{{H}_{3}}\] is equally basic as \[N{{H}_{3}}\] done
clear
D)
\[P{{H}_{3}}\] is amphoteric while \[N{{H}_{3}}\] is basic done
clear
View Solution play_arrow
-
question_answer123)
One of the acid listed below is formed from \[{{P}_{2}}{{O}_{3}}\] and the rest are formed from \[{{P}_{2}}{{O}_{5}}\]. The acid formed from phosphorus (III) oxide is [NCERT 1975]
A)
\[HP{{O}_{3}}\] done
clear
B)
\[{{H}_{4}}{{P}_{2}}{{O}_{7}}\] done
clear
C)
\[{{H}_{3}}P{{O}_{4}}\] done
clear
D)
\[{{H}_{3}}P{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer124)
\[{{P}_{2}}{{O}_{5}}\] is heated with water to give [CBSE PMT 1991; DPMT 2000]
A)
Hypophosphorus acid done
clear
B)
Orthophosphorus acid done
clear
C)
Hypophosphoric acid done
clear
D)
Orthophosphoric acid done
clear
View Solution play_arrow
-
question_answer125)
Hypophosphorus acid is [NCERT 1977; MP PMT 1992]
A)
A tribasic acid done
clear
B)
A dibasic acid done
clear
C)
A monobasic acid done
clear
D)
Not acidic at all done
clear
View Solution play_arrow
-
question_answer126)
\[PC{{l}_{3}}\] reacts with water to form [KCET 1990; CBSE PMT 1991; CPMT 2003]
A)
\[P{{H}_{3}}\] done
clear
B)
\[{{H}_{3}}P{{O}_{3}},\]\[HCl\] done
clear
C)
\[POC{{l}_{3}}\] done
clear
D)
\[{{H}_{3}}P{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer127)
\[{{H}_{3}}P{{O}_{3}}\] is [CPMT 1977, 79, 94; NCERT 1981; MP PMT 1980]
A)
A tribasic acid done
clear
B)
A dibasic acid done
clear
C)
Neutral done
clear
D)
A monobasic acid done
clear
View Solution play_arrow
-
question_answer128)
Oxidation state of + 1 for phosphorus is found in [MP PMT 1991; MP PET 2001]
A)
\[{{H}_{3}}P{{O}_{3}}\] done
clear
B)
\[{{H}_{3}}P{{O}_{4}}\] done
clear
C)
\[{{H}_{3}}P{{O}_{2}}\] done
clear
D)
\[{{H}_{4}}{{P}_{2}}{{O}_{7}}\] done
clear
View Solution play_arrow
-
question_answer129)
Which is not an acid salt [MNR 1989]
A)
\[Na{{H}_{2}}P{{O}_{3}}\] done
clear
B)
\[Na{{H}_{2}}P{{O}_{2}}\] done
clear
C)
\[N{{a}_{3}}H{{P}_{2}}{{O}_{6}}\] done
clear
D)
\[N{{a}_{4}}{{P}_{2}}{{O}_{7}}\] done
clear
View Solution play_arrow
-
question_answer130)
By the action of hot conc. \[{{H}_{2}}S{{O}_{4}},\] phosphorus changes to [Roorkee 1992]
A)
Phosphorus acid done
clear
B)
Orthophosphoric acid done
clear
C)
Metaphosphoric acid done
clear
D)
Pyrophosphoric acid done
clear
View Solution play_arrow
-
question_answer131)
Cyanamide process is used in the formation of [BHU 1995]
A)
\[{{N}_{2}}\] done
clear
B)
\[HN{{O}_{3}}\] done
clear
C)
\[N{{H}_{3}}\] done
clear
D)
\[P{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer132)
The number of hydroxyl group in pyrophosphoric acid is [KCET 1993]
A)
3 done
clear
B)
4 done
clear
C)
5 done
clear
D)
7 done
clear
View Solution play_arrow
-
question_answer133)
There is very little difference in acid strength in the series \[{{H}_{3}}P{{O}_{4}},\] \[{{H}_{3}}P{{O}_{3}},\] and \[{{H}_{3}}P{{O}_{2}}\] because [KCET 1990]
A)
Phosphorus in these acids exists in different oxidation states done
clear
B)
The hydrogen in these acids are not all bounded to the phosphorus done
clear
C)
Phosphorus is not a highly electronegative element done
clear
D)
Phosphorus oxides are less basic done
clear
View Solution play_arrow
-
question_answer134)
\[BiC{{l}_{3}}\] on hydrolysis forms a white precipitate of [CPMT 1988]
A)
Bismuthio acid done
clear
B)
Bismuth oxychloride done
clear
C)
Bismuth pentachloride done
clear
D)
Bismuth hydroxide done
clear
View Solution play_arrow
-
question_answer135)
At high temperature nitrogen combines with calcium carbide \[(Ca{{C}_{2}})\] to give [DPMT 1981, 85; AFMC 1998; MP PET 2000]
A)
Calcium cyanide done
clear
B)
Calcium cyanamide done
clear
C)
Calcium carbonate done
clear
D)
Calcium nitride done
clear
View Solution play_arrow
-
question_answer136)
Calcium cyanamide on treatment with steam under pressure gives ammonia and [EAMCET 1984, 88; CPMT 1986]
A)
Calcium carbonate done
clear
B)
Calcium hydroxide done
clear
C)
Calcium oxide done
clear
D)
Calcium bicarbonate done
clear
View Solution play_arrow
-
question_answer137)
Which one has the highest percentage of nitrogen [KCET 1991; CBSE PMT 1993; AIIMS 1996, MP PET 2001; RPET 2003]
A)
Urea done
clear
B)
Ammonium sulphate done
clear
C)
Ammonium nitrate done
clear
D)
Calcium nitrate done
clear
View Solution play_arrow
-
question_answer138)
Superphosphate of lime contains [CPMT 1984]
A)
\[C{{a}_{3}}{{(P{{O}_{4}})}_{2}}\] done
clear
B)
\[CaHP{{O}_{4}}\] done
clear
C)
\[C{{a}_{3}}{{(P{{O}_{4}})}_{2}}+{{H}_{3}}P{{O}_{4}}\] done
clear
D)
\[Ca{{({{H}_{2}}P{{O}_{4}})}_{2}}\] done
clear
View Solution play_arrow
-
question_answer139)
Thomas slag is [CPMT 1988]
A)
\[C{{a}_{3}}{{(P{{O}_{4}})}_{2}}\] done
clear
B)
\[MnSi{{O}_{3}}\] done
clear
C)
\[CaSi{{O}_{3}}\] done
clear
D)
\[FeSi{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer140)
When equal weights of the two fertilizers, urea and ammonium sulphate are taken, urea contains [KCET 1989]
A)
Less nitrogen than ammonium sulphate done
clear
B)
As much nitrogen as ammonium sulphate done
clear
C)
Twice the amount of nitrogen present in ammonium sulphate done
clear
D)
More than twice the amount of nitrogen present in ammonium sulphate done
clear
View Solution play_arrow
-
question_answer141)
Which statement is wrong for \[NO\] [DPMT 2005]
A)
It is anhydride of nitrous acid done
clear
B)
Its dipole moment in 0.22 D done
clear
C)
It forms dimer done
clear
D)
it is paramagnetic done
clear
View Solution play_arrow
-
question_answer142)
The resonance hybrid of nitrate ion is [AFMC 2002]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer143)
Blasting of TNT is done by mixing [AFMC 1993]
A)
\[N{{H}_{4}}Cl\] done
clear
B)
\[N{{H}_{4}}N{{O}_{3}}\] done
clear
C)
\[N{{H}_{4}}N{{O}_{2}}\] done
clear
D)
\[{{(N{{H}_{4}})}_{2}}S{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer144)
Sodium hydroxide solution reacts with phosphorus to give phosphine. To bring about this reaction, we need [KCET 1989]
A)
White phosphorus and dil. \[NaOH\] done
clear
B)
White phosphorus and conc. \[NaOH\] done
clear
C)
Red phosphorus and dil. \[NaOH\] done
clear
D)
Red phosphorus and conc. \[NaOH\] done
clear
View Solution play_arrow
-
question_answer145)
Which of the following exhibits highest solubility in water [MP PET 1994]
A)
\[N{{H}_{3}}\] done
clear
B)
\[P{{H}_{3}}\] done
clear
C)
\[As{{H}_{3}}\] done
clear
D)
\[Sb{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer146)
Which of the following has highest boiling point [MP PET 1994]
A)
\[N{{H}_{3}}\] done
clear
B)
\[P{{H}_{3}}\] done
clear
C)
\[As{{H}_{3}}\] done
clear
D)
\[Sb{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer147)
In the following reaction \[{{P}_{4}}+3NaOH+3{{H}_{2}}O\to P{{H}_{3}}+3Na{{H}_{2}}P{{O}_{2}}\] [MP PMT 1994]
A)
Phosphorus is oxidised done
clear
B)
Phosphorus is oxidised and reduced done
clear
C)
Phosphorus is reduced done
clear
D)
Sodium is oxidized done
clear
View Solution play_arrow
-
question_answer148)
\[HN{{O}_{3}}\] in aqueous solution yields [AMU 2000]
A)
\[NO_{3}^{-}\] and \[{{H}^{+}}\] done
clear
B)
\[NO_{3}^{-}\] and \[{{H}_{3}}{{O}^{+}}\] done
clear
C)
\[NO_{2}^{-}\] and \[O{{H}^{-}}\] done
clear
D)
\[{{N}_{2}}{{O}_{5}}\] and \[{{H}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer149)
The oxyacid of phosphorus, in which phosphorus has the lowest oxidation state, is [KCET (Med.) 2001]
A)
Hypophosphorus acid done
clear
B)
Orthophosphoric acid done
clear
C)
Pyrophosphoric acid done
clear
D)
Metaphosphoric acid done
clear
View Solution play_arrow
-
question_answer150)
Superphosphate is a mixture of [KCET (Med.) 2001]
A)
\[Ca{{({{H}_{2}}P{{O}_{4}})}_{2}}{{H}_{2}}O+CaC{{l}_{2}}\,.\,2{{H}_{2}}O\] done
clear
B)
\[C{{a}_{3}}{{(P{{O}_{4}})}_{2}}\,.\,{{H}_{2}}O+CaC{{l}_{2}}\,.\,2{{H}_{2}}O\] done
clear
C)
\[C{{a}_{3}}{{(P{{O}_{4}})}_{2}}\,.\,{{H}_{2}}O+2CaS{{O}_{4}}\,.\,2{{H}_{2}}O\] done
clear
D)
\[Ca{{({{H}_{2}}P{{O}_{4}})}_{2}}.\,{{H}_{2}}O+2CaS{{O}_{4}}\,.\,2{{H}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer151)
Solid \[PC{{l}_{5}}\] exits as [JIPMER 2002]
A)
\[PC{{l}_{5}}\] done
clear
B)
\[PCl_{4}^{+}\] done
clear
C)
\[PCl_{6}^{-}\] done
clear
D)
\[PCl_{4}^{+}\] and \[PCl_{6}^{-}\] done
clear
View Solution play_arrow
-
question_answer152)
In the reaction, \[{{P}_{2}}{{O}_{5}}+3CaO\to C{{a}_{3}}{{(P{{O}_{4}})}_{2}};\,{{P}_{2}}{{O}_{5}}\] acts as...... [Orissa JEE 2002]
A)
Acidic flux done
clear
B)
Basic flux done
clear
C)
Basic impurity done
clear
D)
Acidic impurity done
clear
View Solution play_arrow
-
question_answer153)
Atoms in a \[{{P}_{4}}\] molecule of white phosphorus are arranged regularly in space in which of the following way [Kerala (Engg.) 2002]
A)
At the corners of tetrahedron done
clear
B)
At the corners of a cube done
clear
C)
At the corners of a four membered ring done
clear
D)
At the centre and corners of an equilateral triangle done
clear
View Solution play_arrow
-
question_answer154)
The most common minerals of phosphorus are [Kerala (Med.) 2002]
A)
Hydroxy apatite and kernite done
clear
B)
Colemanite and fluoraptite done
clear
C)
Borax and fluoraptite done
clear
D)
Hydroxy apatite and colemanite done
clear
E)
Hydroxy apatite and fluorapatile done
clear
View Solution play_arrow
-
question_answer155)
The three important oxidation states of phosphorus are [Kerala (Med.) 2002]
A)
\[-3,\,+3\] and \[+5\] done
clear
B)
\[-3,\,+3\] and \[-5\] done
clear
C)
\[-3,\,+4\] and \[-4\] done
clear
D)
\[-3,\,+3\] and \[+4\] done
clear
View Solution play_arrow
-
question_answer156)
In case of nitrogen, \[NC{{l}_{3}}\] is possible but not \[NC{{l}_{5}}\] while in case of phosphorous, \[PC{{l}_{3}}\] as well as \[PC{{l}_{5}}\]are possible. It is due to [AIEEE 2002]
A)
Availability of vacant d-orbital in P but not in N done
clear
B)
Lower electronegativity of P than N done
clear
C)
Lower tendency of H bond formation in P than N done
clear
D)
Occurrence of P in solid while N in gaseous state at room temperature done
clear
View Solution play_arrow
-
question_answer157)
When ammonia is passed over heated copper oxide, the metallic copper is obtained. the reaction shows that ammonia is [MP PET 2002]
A)
A dehydrating agent done
clear
B)
An oxidising agent done
clear
C)
A reducing agent done
clear
D)
A nitrating agent done
clear
View Solution play_arrow
-
question_answer158)
Urea is preferred to ammonium sulphate as a nitrogeneous fertilizer because [KCET 2003]
A)
It is more soluble in water done
clear
B)
It is cheaper than ammonium sulphate done
clear
C)
It is quite stable done
clear
D)
It does not cause acidity in the soil done
clear
View Solution play_arrow
-
question_answer159)
Liquid ammonia is used for refrigeration because [MP PET 2002]
A)
It has a high dipole moment done
clear
B)
It has a high heat of vapourisation done
clear
C)
It is basic done
clear
D)
It is a stable compound done
clear
View Solution play_arrow
-
question_answer160)
Action of concentrated nitric acid \[(HN{{O}_{3}})\] on metallic tin produces [BHU 2002]
A)
Stannic nitrate done
clear
B)
Stannous nitrate done
clear
C)
Stannous nitrite done
clear
D)
Meta stannic acid done
clear
View Solution play_arrow
-
question_answer161)
How can you synthesize nitric oxide in the laboratory [Orissa JEE 2003]
A)
Zinc with cold and dilute \[HN{{O}_{3}}\] done
clear
B)
Zinc with concentrated \[HN{{O}_{3}}\] done
clear
C)
Copper with cold and dilute \[HN{{O}_{3}}\] done
clear
D)
Heating \[N{{H}_{4}}N{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer162)
What would happen when a solution of potassium chromate is treated with an excess of dilute nitric acid [AIEEE 2003]
A)
\[C{{r}^{3+}}\] and \[C{{r}_{2}}O_{7}^{2-}\] are formed done
clear
B)
\[C{{r}_{2}}O_{7}^{2-}\]and \[{{H}_{2}}O\] are formed done
clear
C)
\[CrO_{4}^{2-}\] is reduced to \[+3\] state of \[Cr\] done
clear
D)
\[CrO_{4}^{2-}\] is oxidized to \[+7\] state of \[Cr\] done
clear
View Solution play_arrow
-
question_answer163)
The pentavalence in phosphorus is more stable as compared to that of nitrogen even though they belong to the same group. It is due to [KCET 2002]
A)
Inert nature of nitrogen done
clear
B)
Reactivity of phosphorus done
clear
C)
Larger size of phosphorus atom done
clear
D)
Dissimilar electronic configuration done
clear
View Solution play_arrow
-
question_answer164)
A neutral fertilizer among the following compounds is [KCET 2002]
A)
Urea done
clear
B)
Ammonium nitrate done
clear
C)
Ammonium sulphate done
clear
D)
Calcium ammonium nitrate done
clear
View Solution play_arrow
-
question_answer165)
Which is true with regard to the properties of \[P{{H}_{3}}\] [BHU 2000]
A)
\[P{{H}_{3}}\] is not much stable done
clear
B)
\[P{{H}_{3}}\] is neutral towards litmus done
clear
C)
\[P{{H}_{3}}\] has fishy smell done
clear
D)
\[P{{H}_{3}}\] is insoluble in water done
clear
View Solution play_arrow
-
question_answer166)
Nitrogen is obtained when \[NaN{{O}_{2}}\] reacts with [UPSEAT 2003]
A)
\[N{{H}_{4}}Cl\] done
clear
B)
\[N{{H}_{4}}N{{O}_{3}}\] done
clear
C)
\[{{(N{{H}_{4}})}_{2}}C{{O}_{3}}\] done
clear
D)
\[N{{H}_{4}}OH\] done
clear
View Solution play_arrow
-
question_answer167)
\[{{N}_{2}}\] combines with metal to form [JIPMER 2000]
A)
Nitride done
clear
B)
Nitrate done
clear
C)
Nitrite done
clear
D)
Nitrosyl chloride done
clear
View Solution play_arrow
-
question_answer168)
The number of \[P-O-P\] bridges in the structure of phosphorous pentoxide and phosphorous trioxide are respectively [AIIMS 2005]
A)
6, 6 done
clear
B)
5, 5 done
clear
C)
5, 6 done
clear
D)
6, 5 done
clear
View Solution play_arrow
-
question_answer169)
Sodium pyrophosphate is [CPMT 2003]
A)
\[N{{a}_{2}}{{P}_{2}}{{O}_{7}}\] done
clear
B)
\[N{{a}_{4}}{{P}_{2}}{{O}_{7}}\] done
clear
C)
\[NaP{{O}_{4}}\] done
clear
D)
\[N{{a}_{2}}P{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer170)
Which of the following is solid in nature [UPSEAT 2003; AFMC 2004]
A)
\[NO\] done
clear
B)
\[{{N}_{2}}O\] done
clear
C)
\[{{N}_{2}}{{O}_{3}}\] done
clear
D)
\[{{N}_{2}}{{O}_{5}}\] done
clear
View Solution play_arrow
-
question_answer171)
Which of the following is a cyclic phosphate [KCET 1996]
A)
\[{{H}_{5}}{{P}_{3}}{{O}_{10}}\] done
clear
B)
\[{{H}_{6}}{{P}_{4}}{{O}_{13}}\] done
clear
C)
\[{{H}_{5}}{{P}_{5}}{{O}_{15}}\] done
clear
D)
\[{{H}_{7}}{{P}_{5}}{{O}_{16}}\] done
clear
View Solution play_arrow
-
question_answer172)
The reaction, which forms nitric oxide, is [KCET (Med.) 2001]
A)
C and \[{{O}_{2}}\] done
clear
B)
\[Cu\] and \[{{N}_{2}}O\] done
clear
C)
\[Na\] and \[N{{H}_{3}}\] done
clear
D)
\[Cu\] and \[HN{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer173)
Which one of the following can be used as an anaesthetic [EAMCET 1998]
A)
\[{{N}_{2}}O\] done
clear
B)
\[NO\] done
clear
C)
\[NC{{l}_{3}}\] done
clear
D)
\[N{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer174)
Solution of sodium metal in liquid ammonia is strongly reducing due to the presence in the solution of the following [KCET 2000; MP PMT 2001]
A)
Sodium hydride done
clear
B)
Sodium amide done
clear
C)
Sodium atoms done
clear
D)
Solvated electrons done
clear
View Solution play_arrow
-
question_answer175)
What may be expected to happen when phosphine gas is mixed with chlorine gas [AIEEE 2003]
A)
The mixture only cools down done
clear
B)
\[PC{{l}_{3}}\] and HCl are formed and the mixture warms up done
clear
C)
\[PC{{l}_{5}}\] and HCl are formed and the mixture cools down done
clear
D)
\[P{{H}_{3}}.\,C{{l}_{2}}\] is formed with warming up done
clear
View Solution play_arrow
-
question_answer176)
\[SO_{4}^{2-}\] is not used with to dry \[N{{H}_{3}}\]gas because [KCET 2001]
A)
\[{{P}_{4}}{{O}_{10}}\] is basic and \[N{{H}_{3}}\] is acidic done
clear
B)
\[{{P}_{4}}{{O}_{10}}\] is acidic and \[N{{H}_{3}}\]is basic done
clear
C)
\[{{P}_{4}}{{O}_{10}}\] is not a drying agent done
clear
D)
\[{{P}_{4}}{{O}_{10}}\] reacts with moisture in \[N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer177)
When ammonia reacts with sodium hypochlorite, product containing nitrogen is [AFMC 2000]
A)
\[{{N}_{2}}\] done
clear
B)
\[{{N}_{2}}O\] done
clear
C)
\[N{{H}_{2}}OH\] done
clear
D)
\[{{H}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer178)
\[{{P}_{2}}{{O}_{5}}\] is used extensively as a [BVP 2003]
A)
Reducing agent done
clear
B)
Oxidising agent done
clear
C)
Dehydrating agent done
clear
D)
Preservative done
clear
View Solution play_arrow
-
question_answer179)
Inertness of \[{{N}_{2}}\] gas is due to [DCE 2000; MP PET 2001]
A)
No vacant d-orbital done
clear
B)
High dissociation energy done
clear
C)
High electronegativity done
clear
D)
None done
clear
View Solution play_arrow
-
question_answer180)
Which show maximum valency [CPMT 2003]
A)
Phosphorus done
clear
B)
Tin done
clear
C)
Antimony done
clear
D)
Bismuth done
clear
View Solution play_arrow
-
question_answer181)
Which is used in the Haber process for the manufacture of \[N{{H}_{3}}\] [MH CET 2001]
A)
\[Pt\] done
clear
B)
\[Fe+Mo\] done
clear
C)
\[CuO\] done
clear
D)
\[A{{l}_{2}}{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer182)
On adding excess of ammonium hydroxide to a copper chloride solution. [MH CET 1999]
A)
A deep blue solution is obtained done
clear
B)
No change is observed done
clear
C)
Blue precipitate of copper hydroxide is obtained done
clear
D)
Black precipitate of copper oxide is obtained done
clear
View Solution play_arrow
-
question_answer183)
The product obtained by heating\[{{(N{{H}_{4}})}_{2}}S{{O}_{4}}\]and \[KCNO\]is [DPMT 2000]
A)
Hydrocyanic acid done
clear
B)
Ammonia done
clear
C)
Ammonium cyanide done
clear
D)
Urea done
clear
View Solution play_arrow
-
question_answer184)
The number of \[P-O-P\] bonds in cyclic metaphosphoric acid is [IIT-JEE (Screening) 2000]
A)
Zero done
clear
B)
Two done
clear
C)
Three done
clear
D)
Four done
clear
View Solution play_arrow
-
question_answer185)
When \[HN{{O}_{3}}\] is dropped into the palm and washed with water, it turns into yellow. It shows the presence of [CPMT 1997]
A)
\[N{{O}_{2}}\] done
clear
B)
\[{{N}_{2}}O\] done
clear
C)
\[NO\] done
clear
D)
\[{{N}_{2}}{{O}_{5}}\] done
clear
View Solution play_arrow
-
question_answer186)
Which of the following is nitrogenous fertilizers [CPMT 1999]
A)
Bone meal done
clear
B)
Thomas meal done
clear
C)
Nitro phosphate done
clear
D)
Ammonium sulphate done
clear
View Solution play_arrow
-
question_answer187)
Which compound is related to Haber's process [RPET 1999]
A)
\[C{{O}_{2}}\] done
clear
B)
\[{{H}_{2}}\] done
clear
C)
\[N{{O}_{2}}\] done
clear
D)
\[N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer188)
Ammonia is dried over [CPMT 2002; JIPMER 2002]
A)
Quick lime done
clear
B)
Slaked lime done
clear
C)
Anhy.\[CaC{{l}_{2}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer189)
Which of the following compounds is sparingly soluble in ammonia [JIPMER 1999]
A)
\[AgI\] done
clear
B)
\[AgBr\] done
clear
C)
\[AgCl\] done
clear
D)
\[CuC{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer190)
The carbonate which does not leave a residue on heating is [JIPMER 1999; DCE 1999]
A)
\[N{{a}_{2}}C{{O}_{3}}\] done
clear
B)
\[A{{g}_{2}}C{{O}_{3}}\] done
clear
C)
\[CuC{{O}_{3}}\] done
clear
D)
\[{{(N{{H}_{4}})}_{2}}C{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer191)
Which of the following has the highest dipole moment [CBSE PMT 1997]
A)
\[N{{H}_{3}}\] done
clear
B)
\[P{{H}_{3}}\] done
clear
C)
\[Sb{{H}_{3}}\] done
clear
D)
\[As{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer192)
The structural formula of hypophosphorus acid is [CBSE PMT 1997; AIIMS 2001; BCECE 2005; Pb. CET 2002]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer193)
Repeated use of which of the following fertilizers would increase the acidity of the soil [CBSE PMT 1998]
A)
Urea done
clear
B)
Potassium nitrate done
clear
C)
Ammonium sulphate done
clear
D)
Superphosphate of lime done
clear
View Solution play_arrow
-
question_answer194)
Aqua-regia is [Orissa JEE 2005]
A)
1 : 3 conc. \[HN{{O}_{3}}\] and conc. \[HCl\] done
clear
B)
1 : 2 conc. \[HN{{O}_{3}}\] and conc. \[HCl\] done
clear
C)
3 : 1 conc. \[HN{{O}_{3}}\] and conc. \[HCl\] done
clear
D)
2 : 1 conc. \[HN{{O}_{3}}\] and conc. \[HCl\] done
clear
View Solution play_arrow
-
question_answer195)
Which oxide of nitrogen is obtained on heating ammonium nitrate at \[{{250}^{o}}C\] [AIIMS 1999]
A)
Nitric oxide done
clear
B)
Nitrous oxide done
clear
C)
Nitrogen dioxide done
clear
D)
Dinitrogen oxide done
clear
View Solution play_arrow
-
question_answer196)
The oxidation number of phosphorus vary from [Kurukshetra CEE 1998; DCE 2001]
A)
-1 to +3 done
clear
B)
-3 to +3 done
clear
C)
-3 to +5 done
clear
D)
-5 to +1 done
clear
View Solution play_arrow
-
question_answer197)
Inorganic graphite is [KCET 2003]
A)
\[{{B}_{3}}{{N}_{3}}{{H}_{6}}\] done
clear
B)
\[{{B}_{3}}{{N}_{3}}\] done
clear
C)
\[SiC\] done
clear
D)
\[Fe{{(CO)}_{5}}\] done
clear
View Solution play_arrow
-
question_answer198)
Conc.\[HN{{O}_{3}}\] can be stored in container of [MH CET 2001]
A)
Al done
clear
B)
Sn done
clear
C)
Cu done
clear
D)
Zn done
clear
View Solution play_arrow
-
question_answer199)
Which of the following compounds does not exist [JIPMER 1997]
A)
\[SbC{{l}_{3}}\] done
clear
B)
\[BiC{{l}_{5}}\] done
clear
C)
\[SbC{{l}_{5}}\] done
clear
D)
\[AsC{{l}_{5}}\] done
clear
View Solution play_arrow
-
question_answer200)
The formula of an oxyacid of phosphorus is \[{{H}_{3}}P{{O}_{4}}\]. It is a [MP PMT 1996; CPMT 1999; J & K CET 2005]
A)
Dibasic acid done
clear
B)
Monobasic acid done
clear
C)
Tribasic acid done
clear
D)
Tetrabasic acid done
clear
View Solution play_arrow
-
question_answer201)
Which salt can be classified as an acid salt [CPMT 1993]
A)
\[N{{a}_{2}}S{{O}_{4}}\] done
clear
B)
\[BiOCl\] done
clear
C)
\[Pb(OH)Cl\] done
clear
D)
\[N{{a}_{2}}HP{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer202)
In \[N{{H}_{3}}\] and \[P{{H}_{3}}\], the common is [AFMC 1995]
A)
Odour done
clear
B)
Combustibility done
clear
C)
Basic nature done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer203)
Which one of the following hydrides is least stable
A)
\[As{{H}_{3}}\] done
clear
B)
\[Sb{{H}_{3}}\] done
clear
C)
\[N{{H}_{3}}\] done
clear
D)
\[P{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer204)
Which element from V group, gives most basic compound with hydrogen [MP PMT 1995]
A)
Nitrogen done
clear
B)
Bismuth done
clear
C)
Arsenic done
clear
D)
Phosphorus done
clear
View Solution play_arrow
-
question_answer205)
The acid which forms two series of salts is [KCET 1996]
A)
\[{{H}_{3}}P{{O}_{4}}\] done
clear
B)
\[{{H}_{3}}P{{O}_{3}}\] done
clear
C)
\[{{H}_{3}}B{{O}_{3}}\] done
clear
D)
\[{{H}_{3}}P{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer206)
Which gas is obtained when urea is heated with \[HN{{O}_{2}}\] [CPMT 1996]
A)
\[{{N}_{2}}\] done
clear
B)
\[{{H}_{2}}\] done
clear
C)
\[{{O}_{2}}\] done
clear
D)
\[N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer207)
Atomic number of \[N\] is 7. The atomic number of IIIrd member of nitrogen family is [CPMT 1996]
A)
23 done
clear
B)
15 done
clear
C)
33 done
clear
D)
43 done
clear
View Solution play_arrow
-
question_answer208)
Which of the following have least covalent \[P-H\] bond [CPMT 1996]
A)
\[P{{H}_{3}}\] done
clear
B)
\[{{P}_{2}}{{H}_{6}}\] done
clear
C)
\[{{P}_{2}}{{H}_{5}}\] done
clear
D)
\[PH_{6}^{+}\] done
clear
View Solution play_arrow
-
question_answer209)
Sides of match box have coating of [BHU 1995]
A)
Potassium chlorate, red lead done
clear
B)
Potassium chlorate, antimony sulphide done
clear
C)
Antimony sulphide, red phosphorus done
clear
D)
Antimony sulphide, red lead done
clear
View Solution play_arrow
-
question_answer210)
Which of the following compound is tribasic acid [AIIMS 2002]
A)
\[{{H}_{3}}P{{O}_{2}}\] done
clear
B)
\[{{H}_{3}}P{{O}_{3}}\] done
clear
C)
\[{{H}_{3}}P{{O}_{4}}\] done
clear
D)
\[{{H}_{4}}{{P}_{2}}{{O}_{7}}\] done
clear
View Solution play_arrow
-
question_answer211)
Which of the following is manufactured from the molecular nitrogen by bacteria [MP PET 1999]
A)
\[N{{O}_{3}}\] done
clear
B)
\[N{{O}_{2}}\] done
clear
C)
Amino acids done
clear
D)
Ammonia done
clear
View Solution play_arrow
-
question_answer212)
One mole of calcium phosphide on reaction with excess water gives [IIT-JEE 1999]
A)
One mole of phosphine done
clear
B)
Two moles of phosphoric acid done
clear
C)
Two moles of phosphine done
clear
D)
One mole of phosphorous pentoxide done
clear
View Solution play_arrow
-
question_answer213)
On heating ammonium dichromate, the gas evolved is [IIT-JEE 1999]
A)
Oxygen done
clear
B)
Ammonia done
clear
C)
Nitrous oxide done
clear
D)
Nitrogen done
clear
View Solution play_arrow
-
question_answer214)
In compounds of type \[EC{{l}_{3}}\], where \[E=B,\] P, As or \[Bi\], the angles \[Cl-E-Cl\] for different E are in the order [IIT-JEE 1999]
A)
\[B>P=As=Bi\] done
clear
B)
\[B>P>As>Bi\] done
clear
C)
\[B<P=As=Bi\] done
clear
D)
\[B<P<As<Bi\] done
clear
View Solution play_arrow
-
question_answer215)
Ammonia on reaction with hypochlorite anion, can form [IIT-JEE 1999]
A)
\[NO\] done
clear
B)
\[N{{H}_{4}}Cl\] done
clear
C)
\[{{N}_{2}}{{H}_{4}}\] done
clear
D)
\[HN{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer216)
Orthophosphoric acid represents the molaysis condition due to [CPMT 1993]
A)
Hydrogen bonding done
clear
B)
Phosphorous group done
clear
C)
Maximum oxygen group done
clear
D)
Tribasicity done
clear
View Solution play_arrow
-
question_answer217)
Which of the following elements forms a strongly acidic oxide
A)
\[P\] done
clear
B)
\[As\] done
clear
C)
\[Sb\] done
clear
D)
\[Bi\] done
clear
View Solution play_arrow
-
question_answer218)
In group V-A of the periodic table nitrogen forms only a trihalide but other elements form pentahalides also. The reason is
A)
Nitrogen has less affinity towards halogens done
clear
B)
Nitrogen halides are covalent done
clear
C)
Nitrogen undergoes \[{{d}^{2}}s{{p}^{3}}\] hybridization done
clear
D)
Nitrogen does not have d-orbitals done
clear
View Solution play_arrow
-
question_answer219)
In the nitrogen family the \[H-M-H\] bond angle in the hydrides \[M{{H}_{3}}\] gradually becomes closer to \[{{90}^{o}}\] on going from N to Sb. This shows that gradually [MP PET/PMT 1998; MP PMT 2000]
A)
The basic strength of hydrides increases done
clear
B)
Almost pure p-orbitals are used for \[M-H\] bonding done
clear
C)
The bond energies of \[M-H\] bond increase done
clear
D)
The bond pairs of electrons become nearer to the central atom done
clear
View Solution play_arrow
-
question_answer220)
An element (X) forms compounds of the formula \[XC{{l}_{3}},\] \[{{X}_{2}}{{O}_{5}}\] and \[C{{a}_{3}}{{X}_{2}},\] but does not form \[XC{{l}_{5}}\], which of the following is the element \[X\] [MP PET 1997]
A)
\[B\] done
clear
B)
\[Al\] done
clear
C)
\[N\] done
clear
D)
\[P\] done
clear
View Solution play_arrow
-
question_answer221)
Which of the following tendencies remains unchanged on going down in the nitrogen family (Group-VA)? [MP PMT 1997]
A)
Highest oxidation state done
clear
B)
Non-metallic character done
clear
C)
Stability of hydrides done
clear
D)
Physical state done
clear
View Solution play_arrow
-
question_answer222)
Which of the following oxy acids of phosphorus is a reducing agent and monobasic [DCE 2004]
A)
\[{{H}_{3}}P{{O}_{2}}\] done
clear
B)
\[{{H}_{3}}P{{O}_{3}}\] done
clear
C)
\[{{H}_{3}}P{{O}_{4}}\] done
clear
D)
\[{{H}_{4}}{{P}_{2}}{{O}_{6}}\] done
clear
View Solution play_arrow
-
question_answer223)
Bone black is a polymorphic form of [DCE 2003]
A)
Phosphorus done
clear
B)
Sulphur done
clear
C)
Carbon done
clear
D)
Nitrogen done
clear
View Solution play_arrow
-
question_answer224)
Nitrous oxide is known as [AFMC 2004]
A)
Breathing gas done
clear
B)
Laughing gas done
clear
C)
exercising gas done
clear
D)
Laboratory gas done
clear
View Solution play_arrow
-
question_answer225)
When lead nitrate is heated, it gives [MH CET 2003]
A)
\[N{{O}_{2}}\] done
clear
B)
\[NO\] done
clear
C)
\[{{N}_{2}}{{O}_{5}}\] done
clear
D)
\[{{N}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer226)
Which element exist as a solid at \[25{}^\circ C\] and 1 atmospheric pressure among the following [DCE 2003]
A)
Br done
clear
B)
Cl done
clear
C)
Hg done
clear
D)
P done
clear
View Solution play_arrow
-
question_answer227)
In the reaction \[HN{{O}_{3}}+{{P}_{4}}{{O}_{10}}\to 4HP{{O}_{3}}+x\], the product x is [MH CET 2003; DPMT 2004]
A)
\[{{N}_{2}}{{O}_{3}}\] done
clear
B)
\[{{N}_{2}}{{O}_{5}}\] done
clear
C)
\[N{{O}_{2}}\] done
clear
D)
\[{{H}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer228)
The number of hydrogen atom (s) attached to phosphorus atom in hypophosphorous acid is [AIEEE 2005]
A)
Zero done
clear
B)
Two done
clear
C)
One done
clear
D)
Three done
clear
View Solution play_arrow
-
question_answer229)
Which blue liquid is obtained on reacting equimolar amounts of two gases at \[-30{}^\circ C\] [IIT-JEE (Screening) 2005]
A)
\[{{N}_{2}}O\] done
clear
B)
\[{{N}_{2}}{{O}_{3}}\] done
clear
C)
\[{{N}_{2}}{{O}_{4}}\] done
clear
D)
\[{{N}_{2}}{{O}_{5}}\] done
clear
View Solution play_arrow
-
question_answer230)
Which is the most thermodynamically stable allotropic form of phosphorus [IIT-JEE (Screening) 2005]
A)
Red done
clear
B)
White done
clear
C)
Black done
clear
D)
Yellow done
clear
View Solution play_arrow
-
question_answer231)
\[{{(N{{H}_{4}})}_{2}}C{{r}_{2}}{{O}_{7}}\] on heating liberates a gas. The same gas will be obtained by [IIT JEE (Screening) 2004; BVP 2004]
A)
Heating \[N{{H}_{4}}N{{O}_{2}}\] done
clear
B)
Heating \[N{{H}_{4}}N{{O}_{3}}\] done
clear
C)
Treating \[{{H}_{2}}{{O}_{2}}\] with \[NaN{{O}_{2}}\] done
clear
D)
Treating \[M{{g}_{3}}{{N}_{2}}\] with \[{{H}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer232)
The element which forms oxides in all oxidation states \[+I\] to \[+V\] is [AIIMS 2004]
A)
N done
clear
B)
P done
clear
C)
As done
clear
D)
Sb done
clear
View Solution play_arrow
-
question_answer233)
The boiling points of the following hydrides follow the order of [DPMT 2004]
A)
\[N{{H}_{3}}>As{{H}_{3}}>P{{H}_{3}}>Sb{{H}_{3}}\] done
clear
B)
\[Sb{{H}_{3}}>As{{H}_{3}}>P{{H}_{3}}>N{{H}_{3}}\] done
clear
C)
\[Sb{{H}_{3}}>N{{H}_{3}}>As{{H}_{3}}>P{{H}_{3}}\] done
clear
D)
\[N{{H}_{3}}>P{{H}_{3}}>As{{H}_{3}}>Sb{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer234)
Which of the following halides is most acidic [MP PMT 2004]
A)
\[PC{{l}_{3}}\] done
clear
B)
\[BiC{{l}_{3}}\] done
clear
C)
\[SbC{{l}_{3}}\] done
clear
D)
\[CC{{l}_{4}}\] done
clear
View Solution play_arrow
-
question_answer235)
In the electrothermal process, the compound displaced by silica from calcium phosphate is [KCET 2004]
A)
Calcium done
clear
B)
Phosphine done
clear
C)
Phosphorus done
clear
D)
Phosphorus pentoxide done
clear
View Solution play_arrow
-
question_answer236)
Which of the following compound show sublimation [AFMC 1995; Pb. CET 2000]
A)
\[N{{H}_{4}}Cl\] done
clear
B)
\[CaC{{O}_{3}}\] done
clear
C)
\[BaS{{O}_{4}}\] done
clear
D)
\[CaHP{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer237)
Number of \[P-O\]bonds in \[{{P}_{4}}{{O}_{10}}\]is [DCE 2002]
A)
17 done
clear
B)
16 done
clear
C)
15 done
clear
D)
6 done
clear
View Solution play_arrow
-
question_answer238)
Most acidic oxide is [Pb. CET 2004]
A)
\[N{{a}_{2}}O\] done
clear
B)
\[ZnO\] done
clear
C)
MgO done
clear
D)
\[{{P}_{2}}{{O}_{5}}\] done
clear
View Solution play_arrow
-
question_answer239)
Which of the following is a mixed anhydride [Pb. CET 2003]
A)
\[NO\] done
clear
B)
\[N{{O}_{2}}\] done
clear
C)
\[{{N}_{2}}{{O}_{5}}\] done
clear
D)
\[{{N}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer240)
Oxidation number of As in \[{{H}_{2}}AsO_{4}^{-}\] is [CPMT 2001]
A)
6 done
clear
B)
7 done
clear
C)
5 done
clear
D)
9 done
clear
View Solution play_arrow
-
question_answer241)
When plants and animals decay, the organic nitrogen is converted into inorganic nitrogen. The inorganic nitrogen is in the form of [KCET 2005]
A)
Ammonia done
clear
B)
Elements of nitrogen done
clear
C)
Nitrates done
clear
D)
Nitrides done
clear
View Solution play_arrow
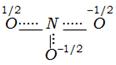 done
clear
done
clear
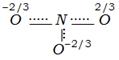 done
clear
done
clear
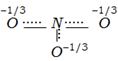 done
clear
done
clear
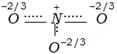 done
clear
done
clear
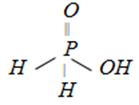 done
clear
done
clear
 done
clear
done
clear
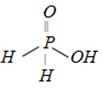 done
clear
done
clear
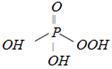 done
clear
done
clear
