question_answer 1) In Youngs experiment, the wavelength of red light is\[7800\overset{o}{\mathop{\text{A}}}\,\] and that of blue light is\[5200\overset{o}{\mathop{\text{A}}}\,\]. The value of n for which\[(n+1)th\] blue band coincides with nth red band is
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 2) The bubble constant has the dimensions of
A)
time
done
clear
B)
\[tim{{e}^{-1}}\]
done
clear
C)
length
done
clear
D)
mass
done
clear
View Answer play_arrow
question_answer 3) A boy has 60 kg weight. He wants to swim in a river with the help of a wooden log. If relative density of wood is 0.6. What is the minimum volume of wooden log? (Density of river water is\[1000\text{ }kg/{{m}^{3}}\])
A)
\[0.66\,{{m}^{3}}\]
done
clear
B)
\[150\,{{m}^{3}}\]
done
clear
C)
\[\frac{3}{1}\,{{m}^{3}}\]
done
clear
D)
\[\frac{3}{20}\,{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 4) When a triode is used as an amplifier the phase difference between the input signal voltage and output is
A)
zero
done
clear
B)
\[\pi \]
done
clear
C)
\[\frac{\pi }{2}\]
done
clear
D)
\[\frac{\pi }{3}\]
done
clear
View Answer play_arrow
question_answer 5) For Balmer series that lies in the visible region, the shortest wavelength corresponds to quantum number
A)
\[n=1\]
done
clear
B)
\[n=2\]
done
clear
C)
\[n=3\]
done
clear
D)
\[n=\infty \]
done
clear
View Answer play_arrow
question_answer 6) A double convex lens\[(\mu ~=3/2)\]of focal length 20 cm is totally immersed in water\[(\mu ~=4/2)\]. Its focal length now will be
A)
20 cm
done
clear
B)
30 cm
done
clear
C)
40cm
done
clear
D)
10cm
done
clear
View Answer play_arrow
question_answer 7) Under which of the following conditions is the law\[pV=RT\]obeyed most closely by a real gas?
A)
High pressure and high temperature
done
clear
B)
Low pressure and low temperature
done
clear
C)
Low pressure and high temperature
done
clear
D)
High pressure and low temperature
done
clear
View Answer play_arrow
question_answer 8) An artificial satellite revolves around the earth in a circular orbit with a speed\[v\]. If m is the mass of the satellite, its total energy is
A)
\[\frac{1}{2}m{{v}^{2}}\]
done
clear
B)
\[-\frac{1}{2}m{{v}^{2}}\]
done
clear
C)
\[-m{{v}^{2}}\]
done
clear
D)
\[\frac{3}{2}m{{v}^{2}}\]
done
clear
View Answer play_arrow
question_answer 9) When the intermolecular distance decreases due to compressive force, there is
A)
zero resultant force between molecules
done
clear
B)
repulsive force between molecules
done
clear
C)
attractive force between molecules
done
clear
D)
no force between molecules
done
clear
View Answer play_arrow
question_answer 10) A 0.5 kg ball moves in a circle of radius 0.4 m at a velocity of 4 m/s. The centripetal force on the ball is
A)
10 N
done
clear
B)
20 N
done
clear
C)
40 N
done
clear
D)
80 N
done
clear
View Answer play_arrow
question_answer 11) In the unmagnetized state, magnetic domains of a magnetic substance are oriented at
A)
\[60{}^\circ \]
done
clear
B)
\[90{}^\circ \]
done
clear
C)
randomly
done
clear
D)
\[150{}^\circ \]
done
clear
View Answer play_arrow
question_answer 12) If the radius of the earth were to shrink by 1%, its mass remaining the same, the acceleration due to gravity on the earths surface would
A)
decrease by 1%
done
clear
B)
remain unchanged
done
clear
C)
increase by 1%
done
clear
D)
increase by 2%
done
clear
View Answer play_arrow
question_answer 13) One-fourth length of a spring of force constant \[k\]is cut away. The force constant of the remaining spring will be
A)
\[\frac{3}{4}k\]
done
clear
B)
\[\frac{4}{3}k\]
done
clear
C)
\[k\]
done
clear
D)
\[4k\]
done
clear
View Answer play_arrow
question_answer 14) A particle is executing simple harmonic motion with amplitude of 0.1 m. At a certain instant when its displacement is 0.02 and its acceleration is\[0.5\text{ }m/{{s}^{2}}\]. The maximum velocity of the particle is (in m/s)
A)
0.01
done
clear
B)
0.05
done
clear
C)
0.5
done
clear
D)
0.25
done
clear
View Answer play_arrow
question_answer 15) The area of cross-section of a steel wire \[(Y=2.0\times {{10}^{11}}N/{{m}^{2}})\]is\[0.1\text{ }c{{m}^{2}}\]. The force required to double its length will be
A)
\[2\times {{10}^{12}}N\]
done
clear
B)
\[2\times {{10}^{11}}N\]
done
clear
C)
\[2\times {{10}^{10}}N\]
done
clear
D)
\[2\times {{10}^{6}}N\]
done
clear
View Answer play_arrow
question_answer 16) If a glass rod is dipped in mercury and withdrawn out, the mercury does not wet the rod because
A)
angle of contact is acute
done
clear
B)
cohesion force is more
done
clear
C)
adhesion force is more
done
clear
D)
density of mercury is more
done
clear
View Answer play_arrow
question_answer 17) Gas exerts pressure on the walls of the container because gas
A)
has weight
done
clear
B)
molecules have momentum
done
clear
C)
molecules collide with each other
done
clear
D)
molecules collide with the walls of the container
done
clear
View Answer play_arrow
question_answer 18) The bulk modulus of an ideal gas at constant temperature is
A)
equal to its pressure
done
clear
B)
equal to its volume
done
clear
C)
equal to p/2
done
clear
D)
cannot be determined
done
clear
View Answer play_arrow
question_answer 19) If in an isothermal process the volume of ideal gas is halved, then we can say that
A)
internal energy of the system decreases
done
clear
B)
infernal energy of the system increases
done
clear
C)
work done by the gas is negative
done
clear
D)
work done by the gas is positive
done
clear
View Answer play_arrow
question_answer 20) X-ray beam of intensity\[{{I}_{0}}\]passes through an absorption plate of thickness d. If absorption coefficient of material of plate is\[\mu ,\]the correct statement regarding the transmitted intensity\[I\]of X-ray is
A)
\[I={{I}_{0}}(I-{{e}^{-\mu d}})\]
done
clear
B)
\[I={{I}_{0}}{{e}^{-\mu d}}\]
done
clear
C)
\[I={{I}_{0}}(I-{{e}^{d/\mu }})\]
done
clear
D)
\[I={{I}_{0}}{{e}^{-\mu /e}}\]
done
clear
View Answer play_arrow
question_answer 21) A\[2\mu F\]capacitor is charged to 100 V and then its plates are connected by a conducting wire, the heat produced is
A)
\[1\,J\]
done
clear
B)
\[0.1\,J\]
done
clear
C)
\[0.01\,J\]
done
clear
D)
\[0.00\,J\]
done
clear
View Answer play_arrow
question_answer 22) Gauss is unit of which quantity?
A)
H
done
clear
B)
B
done
clear
C)
\[\phi \]
done
clear
D)
\[I\]
done
clear
View Answer play_arrow
question_answer 23) A body starts to fall freely under gravity. The distances covered by it in first, second and third seconds are in ratio
A)
\[1:3:5\]
done
clear
B)
\[1:2:3\]
done
clear
C)
\[1:4:9\]
done
clear
D)
\[1:5:6\]
done
clear
View Answer play_arrow
question_answer 24) Two bodies of masses m and 2 m have same momentum. Their respective kinetic energies\[{{K}_{1}}\]and\[{{K}_{2}}\]are in the ratio
A)
\[1:2\]
done
clear
B)
\[2:1\]
done
clear
C)
\[1:\sqrt{2}\]
done
clear
D)
\[1:4\]
done
clear
View Answer play_arrow
question_answer 25) If the velocity of projection is increased by 1% (other things remaining constant) the horizontal range will increase by
A)
1%
done
clear
B)
2%
done
clear
C)
4%
done
clear
D)
8%
done
clear
View Answer play_arrow
question_answer 26) Light of frequency\[v\]is incident on a substance of threshold frequency\[{{v}_{0}}({{v}_{0}}<v)\]. The energy of the emitted photoelectron will be
A)
\[h(v-{{v}_{0}})\]
done
clear
B)
\[h/v\]
done
clear
C)
\[he(v-{{v}_{0}})\]
done
clear
D)
\[h/{{v}_{0}}\]
done
clear
View Answer play_arrow
question_answer 27) Line spectrum contains information about the
A)
atoms of the prism
done
clear
B)
atoms of the source
done
clear
C)
molecules of the source
done
clear
D)
atoms as well as molecules of the source
done
clear
View Answer play_arrow
question_answer 28) A radioactive material has a half-life of 8 yr. The activity of the material will decrease to about\[\frac{1}{8}\]th of its original value in
A)
256 yr
done
clear
B)
128 yr
done
clear
C)
64 yr
done
clear
D)
24 yr
done
clear
View Answer play_arrow
question_answer 29) The ratio of forward bias to reverse bias resistance of p-n junction diode is
A)
\[{{10}^{-1}}:1\]
done
clear
B)
\[{{10}^{-2}}:1\]
done
clear
C)
\[{{10}^{-3}}:1\]
done
clear
D)
\[{{10}^{-4}}:1\]
done
clear
View Answer play_arrow
question_answer 30) The time of revolution of an electron around a nucleus of charge\[Ze\]in nth Bohrs orbit is irectly proportional to
A)
\[n\]
done
clear
B)
\[\frac{{{n}^{3}}}{{{Z}^{2}}}\]
done
clear
C)
\[\frac{{{n}^{2}}}{Z}\]
done
clear
D)
\[\frac{Z}{n}\]
done
clear
View Answer play_arrow
question_answer 31) The average kinetic energy of thermal neutron is of the order of
A)
0.03eV
done
clear
B)
3 eV
done
clear
C)
3 keV
done
clear
D)
3 MeV (Boltzmanns constant\[{{k}_{B}}=8\times {{10}^{-5}}eV/K\])
done
clear
View Answer play_arrow
question_answer 32) If the velocity of sound in air is 336 m/s. The maximum length of a closed pipe that would produce a just audible sound will be
A)
3.2 cm
done
clear
B)
4.2 m
done
clear
C)
4.2 cm
done
clear
D)
3.2 m
done
clear
View Answer play_arrow
question_answer 33) The phenomenon of rotation of plane polarized light is called
A)
Kerr effect
done
clear
B)
double refraction
done
clear
C)
optical activity
done
clear
D)
dichroism
done
clear
View Answer play_arrow
question_answer 34) Infrared radiation was discovered in 1800 by
A)
William Wallaston
done
clear
B)
William Herschel
done
clear
C)
Wilhelm Roentgen
done
clear
D)
Thomas Young
done
clear
View Answer play_arrow
question_answer 35) White light is passed through a dilute solution of potassium permagnate. The spectrum produced by the emergent light is
A)
band emission spectrum
done
clear
B)
line emission spectrum
done
clear
C)
band absorption spectrum
done
clear
D)
line absorption spectrum
done
clear
View Answer play_arrow
question_answer 36) The ionization energy of\[L{{i}^{2+}}\]is equal to
A)
\[9~hcR\]
done
clear
B)
\[6~hcR\]
done
clear
C)
\[2~hcR\]
done
clear
D)
\[~hcR\]
done
clear
View Answer play_arrow
question_answer 37) The error in the measurement of radius of sphere is 0.3%, what is percentage error in the measurement of its volume?
A)
0.3%
done
clear
B)
0.6%
done
clear
C)
0.9%
done
clear
D)
\[\frac{4}{3}\pi {{(0.3)}^{3}}\]
done
clear
View Answer play_arrow
question_answer 38)
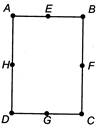
In a rectangle\[ABCD\text{ }(BC=2AB),\]the moment of inertia along axis will be minimum through
A)
BC
done
clear
B)
BD
done
clear
C)
HF
done
clear
D)
EG
done
clear
View Answer play_arrow
question_answer 39) A child is sitting on a swing. Its minimum and maximum heights from the ground are 0.75 m and 2 m respectively, its maximum speed will be
A)
5 m/s
done
clear
B)
8 m/s
done
clear
C)
10 m/s
done
clear
D)
15 m/s
done
clear
View Answer play_arrow
question_answer 40) An oscillator is basically an amplifier with gain
A)
less than unity
done
clear
B)
more than unity
done
clear
C)
zero
done
clear
D)
0.5
done
clear
View Answer play_arrow
question_answer 41) Which one of the following represents simple harmonic motion?
A)
\[{{x}^{2}}=a+bv\]
done
clear
B)
\[x=\sqrt{a+b{{v}^{2}}}\]
done
clear
C)
\[x=a-bv\]
done
clear
D)
\[x=\sqrt{a-b{{v}^{2}}}\]
done
clear
View Answer play_arrow
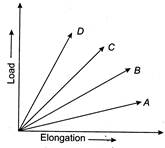
question_answer 42)
The load versus elongation graph of four wires of same length and of the same material is shown in figure. The thinnest wire is represented by the line
A)
OA
done
clear
B)
OB
done
clear
C)
OC
done
clear
D)
OD
done
clear
View Answer play_arrow
question_answer 43) An ideal choke of 10 H is joined in series with resistance of 5 0. and a battery of 5 V. The current in the circuit in 2 s after joining in ampere will be
A)
\[{{e}^{-1}}\]
done
clear
B)
\[1-{{e}^{-1}}\]
done
clear
C)
\[1-e\]
done
clear
D)
\[e\]
done
clear
View Answer play_arrow
question_answer 44) Generally semiconductor can be used safely between the temperatures
A)
\[-75{}^\circ C\]and\[200{}^\circ C\]
done
clear
B)
\[0{}^\circ C\] and\[75{}^\circ C\]
done
clear
C)
\[-25{}^\circ C\]and\[300{}^\circ C\]
done
clear
D)
\[-40{}^\circ C\]and\[1000{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 45) In a given process on an ideal gas\[dW=0\]and \[dQ<0,\]then for the gas
A)
the temperature will decrease
done
clear
B)
the volume will increase
done
clear
C)
the pressure will remain constant
done
clear
D)
the temperature will increase
done
clear
View Answer play_arrow
question_answer 46) The wavelength of light in air is\[6000\overset{o}{\mathop{\text{A}}}\,\]and in medium its value is\[4000\overset{o}{\mathop{\text{A}}}\,\]. It means that the refractive index of that medium with respect to air is
A)
1.2
done
clear
B)
2.4
done
clear
C)
0.66
done
clear
D)
1.5
done
clear
View Answer play_arrow
question_answer 47) A hollow prism is filled with water and placed in air. It will deviate the incident rays
A)
towards the base
done
clear
B)
away from base
done
clear
C)
parallel to base
done
clear
D)
towards or away from base depending on the location
done
clear
View Answer play_arrow
question_answer 48) A uniform electric field and a uniform magnetic field are produced, pointed in the same direction. An electron is projected with its velocity pointing in the same direction
A)
the electron will turn to its left
done
clear
B)
the electron will turn on its right
done
clear
C)
the electron velocity will increase in magnitude
done
clear
D)
the electron velocity will decrease in magnitude
done
clear
View Answer play_arrow
question_answer 49) In the case of constant a and P of a transistor
A)
\[\alpha \beta =1\]
done
clear
B)
\[\beta >1,\alpha >1\]
done
clear
C)
\[\alpha =\beta \]
done
clear
D)
\[\beta <1,\alpha >1\]
done
clear
View Answer play_arrow
question_answer 50) A cup of tea cools from\[80{}^\circ C\]to\[60{}^\circ C\]in 1 min. The ambient temperature is\[30{}^\circ C\]. In next 1 min its temperature will be
A)
\[40{}^\circ C\]
done
clear
B)
\[45{}^\circ C\]
done
clear
C)
\[48{}^\circ C\]
done
clear
D)
\[42{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 51) A hydrogen atom is paramagnetic. A hydrogen molecule is
A)
diamagnefic
done
clear
B)
paramagnetic
done
clear
C)
ferromagnetic
done
clear
D)
antiferromagnetic
done
clear
View Answer play_arrow
question_answer 52) A DC circuit contains\[10\,\Omega \]. of resistance in series with 10 H coil. The impedance of the circuit is
A)
\[10\,\Omega \]
done
clear
B)
\[20\,\Omega \]
done
clear
C)
\[1\,\Omega \]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 53) Critical temperature of\[C{{O}_{2}}\]is\[31.2{}^\circ C\]. In summer, the room temperature is\[40{}^\circ C\]
A)
\[C{{O}_{2}}\]cannot be liquefied
done
clear
B)
can be liquefied with increase of pressure
done
clear
C)
can be liquefied with decrease of pressure
done
clear
D)
can be liquefied if temperature of\[C{{O}_{2}}\]is decreased below\[31.2{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 54) In a meter bridge with a standard resistance of \[5\,\Omega ,\]in the left gap, the ratio of balancing lengths on the meter bridge wire is\[2:3\]. The unknown resistance is
A)
\[3.3\,\Omega \]
done
clear
B)
\[7.5\,\Omega \]
done
clear
C)
\[10\,\Omega \]
done
clear
D)
\[15\,\Omega \]
done
clear
View Answer play_arrow
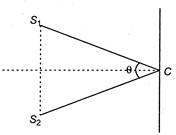
question_answer 55)
Youngs double slit experimental arrangement is shown in figure. If\[\lambda \]is the wavelength of light used and\[<{{S}_{1}}C{{S}_{2}}=\theta ,\]then the fringe width will be
A)
\[\frac{\lambda }{\theta }\]
done
clear
B)
\[\frac{\lambda }{2\theta }\]
done
clear
C)
\[\lambda \theta \]
done
clear
D)
\[\frac{2\lambda }{\theta }\]
done
clear
View Answer play_arrow
question_answer 56) If at the same temperature and pressure, the densities of two diatomic gases are\[{{d}_{1}}\]and\[{{d}_{2}}\]respectively, the ratio of mean kinetic energy per molecule of gases will be
A)
\[1:1\]
done
clear
B)
\[{{d}_{1}}:{{d}_{2}}\]
done
clear
C)
\[\sqrt{{{d}_{1}}}:\sqrt{{{d}_{2}}}\]
done
clear
D)
\[\sqrt{{{d}_{2}}}:\sqrt{{{d}_{1}}}\]
done
clear
View Answer play_arrow
question_answer 57) In AC circuit a resistance of\[R\,\Omega ,\]is connected in series with an inductance L. If the phase difference between the current and voltage is \[45{}^\circ ,\]the inductive reactance will be
A)
\[R/2\]
done
clear
B)
\[R/4\]
done
clear
C)
R
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 58) A metallic wire of density d of floats in water. The maximum radius of the wire, so that it may not sink is
A)
\[\sqrt{2\pi dgT}\]
done
clear
B)
\[\sqrt{\frac{2T}{\pi dg}}\]
done
clear
C)
\[\sqrt{\frac{\pi dg}{2T}}\]
done
clear
D)
\[\sqrt{\frac{2Tg}{\pi d}}\]
done
clear
View Answer play_arrow
question_answer 59) Musical interval between two notes of frequencies 320 and 240 is
A)
1.33
done
clear
B)
80
done
clear
C)
7
done
clear
D)
1.78
done
clear
View Answer play_arrow
question_answer 60) A cubical copper block has each side 2.0 cm. It is suspended by a string and submerged in oil of density\[820\text{ }kg/{{m}^{3}}\]. The tension in the string is (density of copper\[8920\text{ }kg/{{m}^{3}},\text{ }g=10\text{ }m/{{s}^{2}}\])
A)
0.648 N
done
clear
B)
0.712 N
done
clear
C)
0.066 N
done
clear
D)
1.37 N
done
clear
View Answer play_arrow
question_answer 61) Bond polarity of diatomic molecule is because\[o\]
A)
difference in electron affinities of two atoms
done
clear
B)
difference in electro negativities of two atoms
done
clear
C)
difference in ionisation potentials
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 62) The structure of\[P{{F}_{5}}\]molecule is
A)
square planar
done
clear
B)
tetrahedral
done
clear
C)
trigonal bipyramidal
done
clear
D)
pentagonal bipyramidal
done
clear
View Answer play_arrow
question_answer 63) A solid is made of two elements X and Z. The atoms Z are in CCP arrangement while the atoms X occupy all the tetrahedral sites. What is the formula of the compound?
A)
\[XZ\]
done
clear
B)
\[X{{Z}_{2}}\]
done
clear
C)
\[{{X}_{2}}Z\]
done
clear
D)
\[{{X}_{2}}{{Z}_{3}}\]
done
clear
View Answer play_arrow
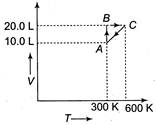
question_answer 64)
This graph expresses the various steps of the system containing 1 mole of gas. Which type of process system has when it moves from C to A?
A)
Isochoric
done
clear
B)
Isobaric
done
clear
C)
Isothermal
done
clear
D)
Cyclic
done
clear
View Answer play_arrow
question_answer 65) During the evaporation of liquid
A)
the temperature of the liquid will rise
done
clear
B)
the temperature of the liquid will fall
done
clear
C)
may rise or fall depending on the nature
done
clear
D)
the temperature remains unaffected
done
clear
View Answer play_arrow
question_answer 66) A system absorb 600 J of heat and work equivalent to 300 J on its surroundings. The change in internal energy is
A)
300 J
done
clear
B)
400 J
done
clear
C)
500 J
done
clear
D)
600 J
done
clear
View Answer play_arrow
question_answer 67) The enthalpies of combustion of carbon and carbon monoxide are\[-393.5\]and\[-283\text{ }kJ\] \[mo{{l}^{-1}}\]respectively. The enthalpy of formation of carbon monoxide per mole is
A)
\[-676.9\text{ }kJ\]
done
clear
B)
\[676.5\text{ }kJ\]
done
clear
C)
\[110.5\text{ }kJ\]
done
clear
D)
\[-110.5\text{ }kJ\]
done
clear
View Answer play_arrow
question_answer 68) A reversible chemical reaction having two reactants in equilibrium. If the concentrations of the reactants are doubled, then the equilibrium constant will
A)
be halved
done
clear
B)
also be doubled
done
clear
C)
reamins the same
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 69) For which order half-life period is independent of initial concentration?
A)
Zero
done
clear
B)
First
done
clear
C)
Second
done
clear
D)
Third
done
clear
View Answer play_arrow
question_answer 70) Ammonia gas dissolves in water to form\[N{{H}_{4}}OH\]. In this reaction water acts as
A)
a conjugate base
done
clear
B)
a non-polar solvent
done
clear
C)
an acid
done
clear
D)
a base
done
clear
View Answer play_arrow
question_answer 71) If acetic acid is mixed with sodium acetate then \[{{H}^{+}}\]ion concentration will be
A)
increased
done
clear
B)
decreased
done
clear
C)
remains unchanged
done
clear
D)
pH decreased
done
clear
View Answer play_arrow
question_answer 72) A weak acid HX has the dissociation constant\[1\times {{10}^{-5}}M\]. It forms a salt\[NaX\]on reaction with alkali. The degree of hydrolysis of 0.1 M solution of\[NaX\]is
A)
0.0001%
done
clear
B)
0.01%
done
clear
C)
0.1%
done
clear
D)
0.15%
done
clear
View Answer play_arrow
question_answer 73) The electron affinity of halogens are\[F=322,\] \[Cl=349,\text{ }Br=324,l=295\text{ }kJ\text{ mo}{{\text{l}}^{-1}}\]. The higher value for\[Cl\]as compared to that of F is due to
A)
weaker electron-electron repulsion in \[Cl\]
done
clear
B)
higher atomic radius of F
done
clear
C)
smaller electronegativity of F
done
clear
D)
more vacant p-subshell in \[Cl\]
done
clear
View Answer play_arrow
question_answer 74) The first ionization potential of\[Na,Mg,Al\] and \[Si\]are in the order,
A)
\[Na>Mg>Al>Si\]
done
clear
B)
\[Na>Mg>Al<Si\]
done
clear
C)
\[Na<Al<Mg<Si\]
done
clear
D)
\[Na<Mg<Al>Si\]
done
clear
View Answer play_arrow
question_answer 75) \[NaCN\]is sometimes added in the froth floatation process as a depressant when\[ZnS\] and\[PbS\] minerals are expected because
A)
\[ZnS\]forms soluble complex\[N{{a}_{2}}[Zn{{(CN)}_{4}}]\]while\[PbS\] forms froth
done
clear
B)
\[Pb{{(CN)}_{2}}\]is precipitated while no effect on \[ZnS\]
done
clear
C)
\[PbS\]forms soluble complex\[N{{a}_{2}}[Pb{{(CN)}_{4}}]\] while\[ZnS\]form froth
done
clear
D)
\[NaCN\]is never added in froth floatation Process
done
clear
View Answer play_arrow
question_answer 76) Which of the following ore is called malachite?
A)
\[C{{u}_{2}}S\]
done
clear
B)
\[CuC{{O}_{3}}\]
done
clear
C)
\[CuC{{O}_{3}}.Cu{{(OH)}_{2}}\]
done
clear
D)
\[C{{u}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 77) White phosphorus\[({{P}_{4}})\]has
A)
four\[P-P\]single bonds
done
clear
B)
four lone pair of electrons
done
clear
C)
PPP angle of\[60{}^\circ \]
done
clear
D)
light\[P-P\]single bonds
done
clear
View Answer play_arrow
question_answer 78) Which of the following is most polarised?
A)
\[Kr\]
done
clear
B)
\[Ar\]
done
clear
C)
\[He\]
done
clear
D)
\[Xe\]
done
clear
View Answer play_arrow
question_answer 79) On dissolving moderate amount of sodium metal in liquid\[N{{H}_{3}}\]at low temperature, which one of the following does not occur?
A)
\[N{{a}^{+}}\]ions are formed in the solution
done
clear
B)
Blue coloured solution is obtained
done
clear
C)
Liquid\[N{{H}_{3}}\]becomes good conductor of electricity
done
clear
D)
Liquid ammonia remains diamagnetic
done
clear
View Answer play_arrow
question_answer 80) Which of the following has highest ionic radii?
A)
\[F{{e}^{3+}}\]
done
clear
B)
\[C{{r}^{3+}}\]
done
clear
C)
\[M{{n}^{3+}}\]
done
clear
D)
\[C{{o}^{3+}}\]
done
clear
View Answer play_arrow
question_answer 81) The basic character of the transition metal monoxides follows the order (At no. of \[Ti=2%V=23,Cr=24,Fe=26\])
A)
\[TiO>VO>CrO>FeO\]
done
clear
B)
\[VO>CrO>TiO>FeO\]
done
clear
C)
\[CrO>VO>FeO>TiO\]
done
clear
D)
\[TiO>FeO>VO>CrO\]
done
clear
View Answer play_arrow
question_answer 82) Which of the following is not an element?
A)
22 carat gold
done
clear
B)
Graphite
done
clear
C)
Diamond
done
clear
D)
Rhombic sulphur
done
clear
View Answer play_arrow
question_answer 83) Which of the following weights less when weighted in magnetic field?
A)
\[SrC{{l}_{3}}\]
done
clear
B)
\[FeC{{l}_{3}}\]
done
clear
C)
\[TiC{{l}_{3}}\]
done
clear
D)
\[VC{{l}_{3}}\]
done
clear
View Answer play_arrow
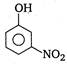
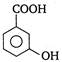
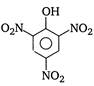
question_answer 84) Picric acid is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 85) The alcohol that produces turbidity immediately with Lucas reagent at room temperature
A)
1-hydroxy butane
done
clear
B)
2-hydroxy butane
done
clear
C)
2-hydroxy-2-methyl propane
done
clear
D)
1-hydroxy-2-methyl propane
done
clear
View Answer play_arrow
question_answer 86) \[A\xrightarrow[{}]{HCN}B\xrightarrow[{}]{{{H}_{3}}{{O}^{+}}}\]Lactic acid. Identify A
A)
\[HCHO\]
done
clear
B)
\[C{{H}_{3}}CHO\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}CHO\]
done
clear
D)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 87) Which one of the following undergoes reaction with 50% sodium hydroxide solution to give the corresponding alcohol and acid?
A)
Phenol
done
clear
B)
Benzaldehyde
done
clear
C)
Butanal
done
clear
D)
Benzoic acid
done
clear
View Answer play_arrow
question_answer 88) Catalyst\[SnC{{l}_{2}}/HCl\]is used in
A)
Stephens reduction
done
clear
B)
Cannizzaros reaction
done
clear
C)
Clemmensen reduction
done
clear
D)
Rosenmunds reduction
done
clear
View Answer play_arrow
question_answer 89) The number of ions formed when cuprammonium sulphate is dissolved in water is
A)
zero
done
clear
B)
1
done
clear
C)
2
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 90) An example of double salt is
A)
potash alum
done
clear
B)
hypo
done
clear
C)
\[{{K}_{4}}[Fe{{(CN)}_{6}}]\]
done
clear
D)
bleaching powder
done
clear
View Answer play_arrow
question_answer 91) In\[Fe{{(CO)}_{5}},\]the\[Fe-C\]bond possesses
A)
\[\pi \]character only
done
clear
B)
both\[\sigma \]and\[\pi \]characters
done
clear
C)
ionic character
done
clear
D)
\[\sigma \]character only
done
clear
View Answer play_arrow
question_answer 92) Formic acid and acetic acid are distinguished by
A)
\[NaHC{{O}_{3}}\]
done
clear
B)
\[FeC{{l}_{3}}\]
done
clear
C)
Victor Mayer test
done
clear
D)
Toilers reagent
done
clear
View Answer play_arrow
question_answer 93) The main product obtained in the reaction of acetamide and\[HN{{O}_{2}}\]is
A)
\[C{{H}_{3}}CN\]
done
clear
B)
\[C{{H}_{3}}NC\]
done
clear
C)
\[C{{H}_{3}}N{{H}_{2}}\]
done
clear
D)
\[C{{H}_{3}}COOH\]
done
clear
View Answer play_arrow
question_answer 94) \[Ca{{C}_{2}}+{{H}_{2}}O\xrightarrow[{}]{{}}A\xrightarrow[{}]{{{H}_{2}}S{{O}_{4}}/HgS{{O}_{4}}}B\] Identify A and B in the given reaction
A)
\[{{C}_{2}}{{H}_{2}}\]and\[C{{H}_{3}}CHO\]
done
clear
B)
\[C{{H}_{4}}\]and\[HCOOH\]
done
clear
C)
\[{{C}_{2}}{{H}_{4}}\]and\[C{{H}_{3}}COOH\]
done
clear
D)
\[{{C}_{2}}{{H}_{2}}\] and\[C{{H}_{3}}COOH\]
done
clear
View Answer play_arrow
question_answer 95) Which one of the following has the minimum boiling point?
A)
\[n-\]butane
done
clear
B)
\[1-\]butyne
done
clear
C)
\[1-\]butene
done
clear
D)
Isobutene
done
clear
View Answer play_arrow
question_answer 96) Which of these do not form Grignard reagent?
A)
\[C{{H}_{3}}Cl\]
done
clear
B)
\[C{{H}_{3}}F\]
done
clear
C)
\[C{{H}_{3}}Br\]
done
clear
D)
\[C{{H}_{3}}I\]
done
clear
View Answer play_arrow
question_answer 97) In alkaline hydrolysis of a tertiary halide by aqueous alkali if concentration of alkali is doubled, then the reaction
A)
will be doubled
done
clear
B)
will be halved
done
clear
C)
will remain constant
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 98) The functional group which is found in amino acid is
A)
\[-COOH\]
done
clear
B)
\[-N{{H}_{2}}\] group
done
clear
C)
\[-C{{H}_{3}}\]group
done
clear
D)
Both [a] and [b]
done
clear
View Answer play_arrow
question_answer 99) Glucose and manose are
A)
anomers
done
clear
B)
epimers
done
clear
C)
ketohexoses
done
clear
D)
disaccharides
done
clear
View Answer play_arrow
question_answer 100) The antiseptic present in Dettol is
A)
qodine
done
clear
B)
chloroxylenol
done
clear
C)
bithional
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 101) Reduction- of nitrobenzene in the presence of \[Zn/N{{H}_{4}}Cl\]gives
A)
hydrazobenzene
done
clear
B)
aniline
done
clear
C)
azobenzene
done
clear
D)
N-phenyl hydroxylamme
done
clear
View Answer play_arrow
question_answer 102) Amongst the following the most basic compound is
A)
\[p-\]nitroaniline
done
clear
B)
acetahilide
done
clear
C)
aniline
done
clear
D)
benzylamine
done
clear
View Answer play_arrow
question_answer 103) Reaction of aniline with acetyl chloride in the presence of\[NaOH\]gives
A)
acetanilide
done
clear
B)
\[p-\]chloroaniline
done
clear
C)
a red dye
done
clear
D)
aniline hydrochloride
done
clear
View Answer play_arrow
question_answer 104) A metal oxide has the formula\[{{A}_{2}}{{O}_{3}}\]. It can be reduced by hydrogen to give free metal and water. 0.1596 g of this metal oxide requires 6 mg of hydrogen for complete reduction. What is the atomic weight of metal?
A)
\[52.3\]
done
clear
B)
\[57.5\]
done
clear
C)
\[55.8\]
done
clear
D)
\[59.3\]
done
clear
View Answer play_arrow
question_answer 105) An electron moves away from the nucleus, its potential energy
A)
increases
done
clear
B)
decreases
done
clear
C)
remains constant
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 106) An\[f-\]shell containing 6 unpaired electrons can exchange
A)
6 electrons
done
clear
B)
9 electrons
done
clear
C)
12 electrons
done
clear
D)
15 electrons
done
clear
View Answer play_arrow
question_answer 107) The electronic configuration of P in\[{{H}_{3}}P{{O}_{4}}\]is
A)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}\]
done
clear
B)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}\]
done
clear
C)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}\]
done
clear
D)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{1}}\]
done
clear
View Answer play_arrow
question_answer 108) Underlined carbon is\[s{{p}^{3}}\]hydridised in
A)
\[C{{H}_{3}}\underline{C}H=C{{H}_{2}}\]
done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-N{{H}_{2}}\]
done
clear
C)
\[C{{H}_{3}}\underline{C}ON{{H}_{2}}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}\underline{C}N\]
done
clear
View Answer play_arrow
question_answer 109) Acetaldehyde is the rearrangement product of
A)
methyl alcohol
done
clear
B)
allyl alcohol
done
clear
C)
vinyl alcohol
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 110) Which of the following does, not show\[{{S}_{{{N}_{2}}}}\]reaction?
A)
Vinylic halide,
done
clear
B)
Allyl chloride,\[C{{H}_{2}}=CH-C{{H}_{2}}Cl\]
done
clear
C)
Chloro benzene.\[{{C}_{6}}{{H}_{5}}Cl\]
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 111) Meso tartaric acid is optically inactive due to the presence of
A)
molecular symmetry
done
clear
B)
molecular asymmetry
done
clear
C)
external compensation
done
clear
D)
two asymmetric C-atoms
done
clear
View Answer play_arrow
question_answer 112) IUPAC name of\[C{{H}_{3}}O{{C}_{2}}{{H}_{5}}\]is
A)
ethoxy methane
done
clear
B)
methoxy ethane
done
clear
C)
Both [a] and [b]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 113) Which of the following applies in the reaction? \[C{{H}_{3}}CHBrC{{H}_{2}}C{{H}_{3}}\xrightarrow[{}]{Alco.\,KOH}?\] (I)\[C{{H}_{2}}CH=CHC{{H}_{3}}\](major product) (II) \[C{{H}_{2}}=CHC{{H}_{2}}C{{H}_{3}}\] (minor product)
A)
Hofmanns rule
done
clear
B)
Saytzeffs rule
done
clear
C)
Kharasch effect
done
clear
D)
Markownikoffs rule
done
clear
View Answer play_arrow
question_answer 114) In which case Raoults law is not applicable?
A)
\[1\text{ }M\text{ }NaCl\]
done
clear
B)
1 M-urea
done
clear
C)
1M glucose
done
clear
D)
1M sucrose
done
clear
View Answer play_arrow
question_answer 115) The freezing point of one molal\[NaCl\]solution. assuming\[NaCl\]to be 100% dissociated in water is (molal depression constant is 1.86)
A)
\[-2.72{}^\circ C\]
done
clear
B)
\[-3.72{}^\circ C\]
done
clear
C)
\[2.72{}^\circ C\]
done
clear
D)
\[3.72{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 116) On passing 3 A of electricity for 50 min, 1.8 g metal deposits. The equivalent mass of metal is
A)
9.3
done
clear
B)
19.3
done
clear
C)
38.3
done
clear
D)
39.9
done
clear
View Answer play_arrow
question_answer 117) The standard emf for the given cell reaction, \[Zn+C{{u}^{2+}}\xrightarrow[{}]{{}}Cu+Z{{n}^{2+}}\]is 1.10 V at\[25{}^\circ C\]. The emf for the cell reaction, when\[0.1\text{ }M\text{ }C{{u}^{2+}}\]and solutions are used at \[25{}^\circ C\] is
A)
1.10V
done
clear
B)
\[-1.10V\]
done
clear
C)
2.20V
done
clear
D)
\[-2.20V\]
done
clear
View Answer play_arrow
question_answer 118) Purple of cassius is
A)
colloidal solution of Au
done
clear
B)
colloidal solution of \[Pt\]
done
clear
C)
colloidal solution of Ag
done
clear
D)
colloidal solution of As
done
clear
View Answer play_arrow
question_answer 119) Adsorption is accompanied by the evolution of heat, so, according to Le-Chatelier principle, the amount of substance adsorbed should
A)
increase with decrease in T
done
clear
B)
increase with increase in T
done
clear
C)
decrease with decrease in T
done
clear
D)
decrease with increase in T
done
clear
View Answer play_arrow
question_answer 120) The oxidation number of iron in\[F{{e}_{3}}{{O}_{4}}\]is
A)
\[+2\]
done
clear
B)
\[+3\]
done
clear
C)
\[\frac{8}{3}\]
done
clear
D)
\[\frac{2}{3}\]
done
clear
View Answer play_arrow
question_answer 121) Chance favours the trained mind. This statement was made by
A)
lan Wilmut
done
clear
B)
Robert Koch
done
clear
C)
Louis Pasteur
done
clear
D)
James D Watson
done
clear
View Answer play_arrow
question_answer 122) In agar plate medium having bacteriophages and bacteria, there are clear transparent area called
A)
transport parts
done
clear
B)
holes
done
clear
C)
bacteriophages
done
clear
D)
plaques
done
clear
View Answer play_arrow
question_answer 123) Species is
A)
population of one type
done
clear
B)
a group of interbreeding populations
done
clear
C)
a group of individuals inhabiting geographical area
done
clear
D)
population of individuals having same genotypes and phenotypes
done
clear
View Answer play_arrow
question_answer 124) Most of the broad spectrum antibiotics have been obtained from
A)
Actinomycetes
done
clear
B)
bacilli
done
clear
C)
spirochaetes
done
clear
D)
archaebacteria
done
clear
View Answer play_arrow
question_answer 125) Which is a wrong statement?
A)
Plasmids of cyanobacteria have been used in biotechnology
done
clear
B)
DNA of cyanobacteria is circular and double-stranded
done
clear
C)
Cyanobacteria possess single linkage group
done
clear
D)
Like bacteria, they also exhibit genetic recombination
done
clear
View Answer play_arrow
question_answer 126) Quinine, the remedy for malaria is extracted from
A)
stem of Hevea
done
clear
B)
bark of Cinchona
done
clear
C)
bark of Cinnamon
done
clear
D)
leaves of Ocimum
done
clear
View Answer play_arrow
question_answer 127) Which part of Ephedra yields ephedrine?
A)
Flowers
done
clear
B)
Stem or whole plant
done
clear
C)
Leaves
done
clear
D)
Roots
done
clear
View Answer play_arrow
question_answer 128) Which is not a larva of sponge?
A)
Tornaria
done
clear
B)
Parenchymula
done
clear
C)
Stereogastrula
done
clear
D)
Amphiblastula
done
clear
View Answer play_arrow
question_answer 129) Which of these phenomenon is found in Hydra?
A)
Metamerism
done
clear
B)
Metabolism
done
clear
C)
Metamorphosis
done
clear
D)
Sexual dimorphism
done
clear
View Answer play_arrow
question_answer 130) Complete metamorphosis occurs in
A)
bedbug
done
clear
B)
silverfish
done
clear
C)
grasshopper
done
clear
D)
moths and mosquitoes
done
clear
View Answer play_arrow
question_answer 131) Equus rests on
A)
one digit
done
clear
B)
three digits
done
clear
C)
four digits
done
clear
D)
five digits
done
clear
View Answer play_arrow
question_answer 132) Which of the following stains is used for determination of cytochrome oxidase activity in the cell?
A)
Eosin
done
clear
B)
Neutral red
done
clear
C)
Janus green B
done
clear
D)
Methylene blue
done
clear
View Answer play_arrow
question_answer 133) The cell theory is not applicable to
A)
algae
done
clear
B)
fungi
done
clear
C)
viruses
done
clear
D)
lichens
done
clear
View Answer play_arrow
question_answer 134) \[N{{a}^{+}}-{{K}^{+}}\]pump is found in the membranes of many cells, like nerve cells. It works against electrochemical gradient and involve an integral protein ATPase. For each molecule of ATP used
A)
3 ions of\[N{{a}^{+}}\]are pumped out and\[2{{K}^{+}}\]are taken in
done
clear
B)
3 ions of\[N{{a}^{+}}\]are taken in and\[2{{K}^{+}}\]are pumped out.
done
clear
C)
2 ions of\[N{{a}^{+}}\]are thrown out and\[3{{K}^{+}}\]are absorbed
done
clear
D)
3 ions of\[{{K}^{+}}\]are absorbed and\[3N{{a}^{+}}\]are pumped out
done
clear
View Answer play_arrow
question_answer 135) Which one is not protein?
A)
Actin
done
clear
B)
Myosin
done
clear
C)
Albumin
done
clear
D)
Haematin
done
clear
View Answer play_arrow
question_answer 136) Maximum number of enzymes are found in
A)
herbivores
done
clear
B)
carnivores
done
clear
C)
omnivores
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 137) A gene whose phenotypic effect kills the bearer is called
A)
lethal
done
clear
B)
pleiotropic
done
clear
C)
supplementary
done
clear
D)
complementary
done
clear
View Answer play_arrow
question_answer 138) Balbiani rings are the structural features of
A)
allosomes
done
clear
B)
autosomes
done
clear
C)
polytene chromosomes
done
clear
D)
Lampbrush chromosomes
done
clear
View Answer play_arrow
question_answer 139) Which of the following statements regarding a double helical molecule of DNA is true?
A)
Each strand is identical
done
clear
B)
Each strand replicates itself
done
clear
C)
Bases are perpendicular to the axis
done
clear
D)
All hydroxyl groups of pentose are involved; in linkage
done
clear
View Answer play_arrow
question_answer 140) The central dogma is not applicable in the case of
A)
retroviruses
done
clear
B)
all prokaryotes
done
clear
C)
all animal viruses
done
clear
D)
all plant viruses
done
clear
View Answer play_arrow
question_answer 141) A gene which synthesises a represser protein is
A)
operator gene
done
clear
B)
promoter gene
done
clear
C)
structural gene
done
clear
D)
regulator gene
done
clear
View Answer play_arrow
question_answer 142) The immediate product of transcription in eukaryotes will be
A)
\[hn-RNA\]
done
clear
B)
\[mRNA\]
done
clear
C)
\[c-DNA\]
done
clear
D)
\[Sn-RNA\]
done
clear
View Answer play_arrow
question_answer 143) Mr. Sharma has Bb autosomal gene pair and d allele sex-linked. What will be the proportion of\[Bd\]in sperms?
A)
0
done
clear
B)
\[1/4\]
done
clear
C)
\[1/2\]
done
clear
D)
\[1/8\]
done
clear
View Answer play_arrow
question_answer 144) The category of molecules produced by the Miller-Urey experiment was
A)
organic polymers
done
clear
B)
inorganic polymers
done
clear
C)
organic monomers
done
clear
D)
inorganic monomers
done
clear
View Answer play_arrow
question_answer 145) In mammals, Melanocyte Stimulating Hormone (MSH) is
A)
steroid hormone
done
clear
B)
vestigial hormone
done
clear
C)
effective hormone
done
clear
D)
protein hormone
done
clear
View Answer play_arrow
question_answer 146) Birbal Sahni worked on
A)
algae
done
clear
B)
bryophytes
done
clear
C)
fossil plants
done
clear
D)
angiosperms
done
clear
View Answer play_arrow
question_answer 147) Modem man differs from apes in
A)
protruding eyes
done
clear
B)
thick body hairs
done
clear
C)
wearing of clothes
done
clear
D)
arms shorter than legs
done
clear
View Answer play_arrow
question_answer 148) Orobanche grow in
A)
wheat field
done
clear
B)
paddy field
done
clear
C)
mustard field
done
clear
D)
dense tropical forest
done
clear
View Answer play_arrow
question_answer 149) Coconut fruit is dispersed by
A)
man
done
clear
B)
wind
done
clear
C)
birds
done
clear
D)
water
done
clear
View Answer play_arrow
question_answer 150) Outermost part of bark consisting of dead cells refers to
A)
rhytidome
done
clear
B)
phellem
done
clear
C)
phellogen
done
clear
D)
phelloderm
done
clear
View Answer play_arrow
question_answer 151) In hypertonic solution, water potential of cell
A)
increases
done
clear
B)
decreases
done
clear
C)
first increases and then decreases
done
clear
D)
remains unchanged
done
clear
View Answer play_arrow
question_answer 152) The stage of ornithine cycle at which arginase enzyme used is
A)
Ornithine\[\xrightarrow[{}]{{}}\]citrulline
done
clear
B)
Arginine\[\xrightarrow[{}]{{}}\] ornithine
done
clear
C)
Citrulline\[\xrightarrow[{}]{{}}\]Argino succinic acid
done
clear
D)
Ornithine\[\xrightarrow[{}]{{}}\]Urea
done
clear
View Answer play_arrow
question_answer 153) Oxygen produced in photosynthesis comes from\[Hp\]was shown by
A)
Robert Mayer
done
clear
B)
Ruben and Kamen
done
clear
C)
Calvin
done
clear
D)
Robert Hill
done
clear
View Answer play_arrow
question_answer 154) The RQ of\[{{C}_{39}}{{H}_{72}}{{O}_{6}}\]is
A)
2.71
done
clear
B)
1.34
done
clear
C)
0.718
done
clear
D)
3.250
done
clear
View Answer play_arrow
question_answer 155)
Match the following columns. Column I Column II A. Skeletal tissue 1. Between muscles B. Blood 2. Vascular tissue C. Areolar tissue 3. Sweat glands D. Serous glands 3. Cartilage
A)
A- 4 B- 2 C-1 D-3
done
clear
B)
A-1 B-2 C-3 D-1
done
clear
C)
A-2 B-1 C-3 D-4
done
clear
D)
A-4 B- 2 C-1 D-3
done
clear
View Answer play_arrow
question_answer 156)
Compare the statement A and B. Statement A Liver is 3-lobed in frog, 5-lobed in rabbit and 4-lobed in man. Statement B Liver is ectodermat in origin select the correct description.
A)
Both the statements A and B are correct and B is the correct explanation of A
done
clear
B)
Both the statements A and B are correct and B is not correct explanation of A.
done
clear
C)
Statement A is correct and B is wrong
done
clear
D)
Statement A is wrong and B is correct
done
clear
View Answer play_arrow
question_answer 157) Which of these are never present in frogs ovary?
A)
Oogonia
done
clear
B)
Corpus luteum
done
clear
C)
Ovarian follicles
done
clear
D)
Germinal epithelium
done
clear
View Answer play_arrow
question_answer 158) Amylopsin acts upon
A)
polypeptides
done
clear
B)
polysaccharide in any medium
done
clear
C)
polysaccharide in acidic medium
done
clear
D)
polysaccharide in alkaline medium
done
clear
View Answer play_arrow
question_answer 159) The medullary respiratory centre is directly affected
A)
chemically
done
clear
B)
physically
done
clear
C)
neuronally
done
clear
D)
None-of these
done
clear
View Answer play_arrow
question_answer 160) Blood of which vessel in mammals carries least percentage of urea?
A)
Renal vein
done
clear
B)
Dorsal aorta
done
clear
C)
Renal artery
done
clear
D)
Posterior vena caya
done
clear
View Answer play_arrow
question_answer 161)
Match the following columns. Column I Column II A. Polyuria 1. WBCs pus in urine B. Pyuria 2. High level of uric acid in blood C. Gout 3. Excess of urine output D. Haematuria 3. Presence of blood (RBCs) in urine
A)
A-3 B-1 C-2 D-4
done
clear
B)
A-2 B-3 C- 1 D-4
done
clear
C)
A- 1 B-2 C-3 D-4
done
clear
D)
A-4 B- 3 C-2 D-1
done
clear
View Answer play_arrow
question_answer 162) Brush border is a characteristic of
A)
proximal convoluted tubule
done
clear
B)
neck of nephron
done
clear
C)
collecting tube
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 163) Which of the following is not a sesamoid bone?
A)
Radius
done
clear
B)
Patella
done
clear
C)
Fibulla
done
clear
D)
Pisciform
done
clear
View Answer play_arrow
question_answer 164) Rigidity that develops in the muscle after death is known as
A)
twitch
done
clear
B)
treppe
done
clear
C)
tetanus
done
clear
D)
Rigor Mortrs
done
clear
View Answer play_arrow
question_answer 165) Acetylcholine is
A)
neural messenger
done
clear
B)
antistress hormone
done
clear
C)
chemical messenger
done
clear
D)
chemical transmitter
done
clear
View Answer play_arrow
question_answer 166) Heariny is controlled by
A)
cerebellum
done
clear
B)
diencephalon
done
clear
C)
frontal lobe of cerebrum
done
clear
D)
temporal lobe of cerebrum
done
clear
View Answer play_arrow
question_answer 167) When a person suffers from a marked fall in blood pressure, it is helpful to administer to him the following hormone
A)
GH
done
clear
B)
insulin
done
clear
C)
thyroxine
done
clear
D)
adrenaline
done
clear
View Answer play_arrow
question_answer 168) Nurse tissue technique is applied in
A)
pollen culture
done
clear
B)
embryo culture
done
clear
C)
ovule culture
done
clear
D)
ovary culture
done
clear
View Answer play_arrow
question_answer 169)
Compare the statement A and B. Statement: A Ethylene is a gas which acts as growth regulator of plants. Statement: B It is the most simple plant hormone.
A)
Both the statements A and B are correct and B is the correct explanation of A
done
clear
B)
Both the statements A and B are correct and B is not correct explanation of A.
done
clear
C)
Statement A is correct and B is wrong
done
clear
D)
Statement A is wrong and B is correct
done
clear
View Answer play_arrow
question_answer 170) Maximum amount of growth in root occurs
A)
in the presence of light
done
clear
B)
at its apex
done
clear
C)
behind the apex
done
clear
D)
in the presence of soil
done
clear
View Answer play_arrow
question_answer 171) Which of the following is found inside Graafian follicle?
A)
Cortex
done
clear
B)
Medulla
done
clear
C)
Corpus luteum
done
clear
D)
Membrane follicle
done
clear
View Answer play_arrow
question_answer 172) Human embryo will be called as a foetus after
A)
two months
done
clear
B)
six months
done
clear
C)
four months
done
clear
D)
seven months
done
clear
View Answer play_arrow
question_answer 173) Functional kidney of frogs tadpole is
A)
pronephros
done
clear
B)
mesonephros
done
clear
C)
holonephros
done
clear
D)
metanephros
done
clear
View Answer play_arrow
question_answer 174) Growth hormone activity is
A)
increased by thyroxine
done
clear
B)
unaffected by thyroxine
done
clear
C)
decreased by thyroxine
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 175) Helical contractile sheath is found in
A)
virus
done
clear
B)
bacterium
done
clear
C)
fungus
done
clear
D)
bacteriophage
done
clear
View Answer play_arrow
question_answer 176) The process in which DNA of a bacterial cell is transferred into another bacterial cell by a virus is known as
A)
conjugation
done
clear
B)
transduction
done
clear
C)
reproduction
done
clear
D)
transformation
done
clear
View Answer play_arrow
question_answer 177) With which one of the following organism a bacterium resembles most?
A)
Yeast
done
clear
B)
Virus
done
clear
C)
Amoeba
done
clear
D)
Anabaena
done
clear
View Answer play_arrow
question_answer 178) Mycoplasma can multiply
A)
in culture media
done
clear
B)
in body of living host only
done
clear
C)
in bacterial cells
done
clear
D)
on dead and decaying organic matter
done
clear
View Answer play_arrow
question_answer 179) The disease caused by Entamoeba gingivalis is spread through
A)
air
done
clear
B)
kissing
done
clear
C)
housefly
done
clear
D)
Anopheles
done
clear
View Answer play_arrow
question_answer 180) Gill of mushroom are meant for
A)
reproduction
done
clear
B)
respiration
done
clear
C)
assimilation
done
clear
D)
nutrition
done
clear
View Answer play_arrow
question_answer 181) How many microsporangia are found in a monothecous anther?
A)
Only one
done
clear
B)
Two
done
clear
C)
Four
done
clear
D)
Many
done
clear
View Answer play_arrow
question_answer 182) Resin or terpentine oil is obtained from
A)
Pinus
done
clear
B)
Cedrus
done
clear
C)
Cycas
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 183) The unit used for the measurement of size of cell is
A)
\[nm\]
done
clear
B)
\[mm\]
done
clear
C)
\[\overset{o}{\mathop{\text{A}}}\,\]
done
clear
D)
\[\mu m\]
done
clear
View Answer play_arrow
question_answer 184) Which one is a prokaryote?
A)
Spirogyra
done
clear
B)
Agaricus
done
clear
C)
Bacteriophage
done
clear
D)
Streptococcus
done
clear
View Answer play_arrow
question_answer 185) The primary function of intermediate filament is
A)
generate movement
done
clear
B)
provide mechanical stability
done
clear
C)
transport organelles within the cell
done
clear
D)
stabilize microtubules against disassembly
done
clear
View Answer play_arrow
question_answer 186) Mitochondria will be found in abundance in cells of tissues having
A)
minimum activity
done
clear
B)
average activity
done
clear
C)
maximum activity
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 187) The blood cell which shows phagocytosis is
A)
platelet
done
clear
B)
basophil
done
clear
C)
monocyte
done
clear
D)
eosinophil
done
clear
View Answer play_arrow
question_answer 188) Zygotic meiosis occurs in
A)
Pinus
done
clear
B)
Marchantia
done
clear
C)
Dryopteris
done
clear
D)
Chlamydomonas
done
clear
View Answer play_arrow
question_answer 189) Variations occur mostly due to
A)
Linkage
done
clear
B)
nutrition
done
clear
C)
segregation
done
clear
D)
crossing over
done
clear
View Answer play_arrow
question_answer 190) The ovum of human female has autosomes
A)
22
done
clear
B)
22 pairs
done
clear
C)
23 pairs
done
clear
D)
44 pairs
done
clear
View Answer play_arrow
question_answer 191) Which of the following enzymes is not required for DNA synthesis?
A)
Ligase
done
clear
B)
\[DNAse\]
done
clear
C)
DNA polymerase
done
clear
D)
RNA polymerase
done
clear
View Answer play_arrow
question_answer 192) A potent inhibitor of protein synthesis that acts as an analogue of aminoacyle\[-tRNA\] is
A)
rifampicin
done
clear
B)
puromycin
done
clear
C)
mitomyocin
done
clear
D)
streptomycin
done
clear
View Answer play_arrow
question_answer 193) Loreal pit in vipers act as which receptor?
A)
Photo
done
clear
B)
Thermo
done
clear
C)
Auditory
done
clear
D)
Gustatory
done
clear
View Answer play_arrow
question_answer 194) Milk glands are characteristics of
A)
all vertebrates
done
clear
B)
only mammals
done
clear
C)
only placental mammals
done
clear
D)
only ruminants
done
clear
View Answer play_arrow
question_answer 195) Irregular flowers are
A)
asymmetrical
done
clear
B)
symmetrical
done
clear
C)
achlamydous
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 196) Funnel-shaped style and stigma develops in
A)
Crocus sativus
done
clear
B)
Hibiscus
done
clear
C)
Helianthus
done
clear
D)
Gloriosa
done
clear
View Answer play_arrow
question_answer 197) Papain (a proteolytic enzyme) is found in the latex of
A)
Carica
done
clear
B)
Ficus
done
clear
C)
Nerium
done
clear
D)
Euphorbia
done
clear
View Answer play_arrow
question_answer 198) Root pressure is measured by
A)
potometer
done
clear
B)
barometer
done
clear
C)
manometer
done
clear
D)
auxanometer
done
clear
View Answer play_arrow
question_answer 199) Photolysis of water is caused by
A)
\[PS-I\]
done
clear
B)
\[PS-II\]
done
clear
C)
\[PS-I\]and\[PS-II\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 200) When 100% carbon is oxidized to \[C{{O}_{3}},\]the efficiency of such respiration is
A)
40%
done
clear
B)
60%
done
clear
C)
90%
done
clear
D)
100%
done
clear
View Answer play_arrow
question_answer 201) According to Mahatma Gandhi, education is
A)
a medium through which people are taught to become sensitive to the realities around them
done
clear
B)
a tool to develop their conscience and Constitution
done
clear
C)
a tool to develop their understanding of the Constitution of society
done
clear
D)
an instrument to develop their society Consciously
done
clear
View Answer play_arrow
question_answer 202) The National Policy on Education provided
A)
guidelines for the comprehension of education in the country
done
clear
B)
guidance material to develop education in the country
done
clear
C)
a comprehensive plan for the development. of education in the country
done
clear
D)
comprehensive development of education in the country
done
clear
View Answer play_arrow
question_answer 203) According to the passage, critics and educationists are worried that
A)
the education of girls is one of the objectives of NPE
done
clear
B)
the objectives of NPE have not been fulfilled
done
clear
C)
non-fulfilment of NPE leads to the education of girls
done
clear
D)
one of the objectives of NPE has not been fulfilled
done
clear
View Answer play_arrow
question_answer 204) According to the passage, a strong gender focus means
A)
a focus on the strength of gender
done
clear
B)
a focus on strong gender
done
clear
C)
a strong focus on gender bias
done
clear
D)
a focus on male-female ratio
done
clear
View Answer play_arrow
question_answer 205) In the passage, the author makes a plea for
A)
free education
done
clear
B)
universal education
done
clear
C)
the education of men
done
clear
D)
the education of women
done
clear
View Answer play_arrow
question_answer 206) Directions: Choose the alternative which can be substituted for the given group of words. A person who maliciously destroys by fire.
A)
Antagonist
done
clear
B)
Activist
done
clear
C)
Terrorist
done
clear
D)
Incendiary
done
clear
View Answer play_arrow
question_answer 207) Directions: Choose the alternative which can be substituted for the given group of words. A house for storing grains.
A)
Cellar
done
clear
B)
Store
done
clear
C)
Godown
done
clear
D)
Granary
done
clear
View Answer play_arrow
question_answer 208) Directions: Choose the alternative which can be substituted for the given group of words. A person very hard to please.
A)
Obstinate
done
clear
B)
Unconquerable
done
clear
C)
Fastidious
done
clear
D)
Invincible
done
clear
View Answer play_arrow
question_answer 209) Directions: Choose the alternative which can be substituted for the given group of words. A person claiming to be superior in culture and intellect to others.
A)
Intellectual
done
clear
B)
Aristocrat
done
clear
C)
Elite
done
clear
D)
Highbrow
done
clear
View Answer play_arrow
question_answer 210) Directions: Choose the most suitable alternative to fill in the blank. If a Universal language really existed, people like tourists and businessmen would find it easier to ......... with foreigners.
A)
transact
done
clear
B)
communicate
done
clear
C)
deal
done
clear
D)
exchange
done
clear
View Answer play_arrow
question_answer 211) Directions: Choose the most suitable alternative to fill in the blank. On account of his humiliating defeat in the recent elections, he appeared unusually ......... when I called on him the other day.
A)
oppressed
done
clear
B)
repressed
done
clear
C)
depressed
done
clear
D)
suppressed
done
clear
View Answer play_arrow
question_answer 212) Directions: Choose the most suitable alternative to fill in the blank. You need ......... shoes for walking in the hills.
A)
good
done
clear
B)
comfortable
done
clear
C)
satisfactory
done
clear
D)
sturdy
done
clear
View Answer play_arrow
question_answer 213) Directions: Choose the most suitable alternative to fill in the blank. Amongst the two brothers, Sammeer being worthier often ......... the younger Deepak.
A)
dominates
done
clear
B)
eclipses
done
clear
C)
subdues
done
clear
D)
overshadows
done
clear
View Answer play_arrow
question_answer 214) Directions: Choose the alternative which is an improvement upon the italicized part of the sentence. If the sentence is correct as it is, choose as your answer. He enjoys to tell stories to children.
A)
how to tell stories
done
clear
B)
telling stories
done
clear
C)
to narrate stories
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 215) Directions: Choose the alternative which is an improvement upon the italicized part of the sentence. If the sentence is correct as it is, choose as your answer. Galileo said that the earth revolved around the Sun.
A)
has revolved
done
clear
B)
has been revolving
done
clear
C)
revolves
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 216) Directions: Choose the alternative which is an improvement upon the italicized part of the sentence. If the sentence is correct as it is, choose as your answer. The matter must be considered in every point of view.
A)
with
done
clear
B)
from
done
clear
C)
at
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 217) Directions: Choose the alternative which is nearest in meaning to the word given in capital letters. FOSTER
A)
Encourage
done
clear
B)
Fabricate
done
clear
C)
Foment
done
clear
D)
Nurture
done
clear
View Answer play_arrow
question_answer 218) Directions: Choose the alternative which is nearest in meaning to the word given in capital letters. ENIGMA
A)
Elusive
done
clear
B)
Clear
done
clear
C)
Puzzle
done
clear
D)
Praise
done
clear
View Answer play_arrow
question_answer 219) Directions: Choose the alternative which is nearest in meaning to the word given in capital letters. FILTHY
A)
Healthy
done
clear
B)
Ugly
done
clear
C)
Dirty
done
clear
D)
Angry
done
clear
View Answer play_arrow
question_answer 220) Directions: Choose the alternative which is nearest in meaning to the word given in capital letters. NOSTALGIC
A)
Soothing
done
clear
B)
Homesick
done
clear
C)
Diseased
done
clear
D)
Indolent
done
clear
View Answer play_arrow
question_answer 221) Directions: In each of the following questions, there occurs a specific relations. Fill the vacant space (?) question mark according to that relation. Door : Bang : : Chain :?
A)
Clank
done
clear
B)
Tinkle
done
clear
C)
Thunder
done
clear
D)
Clinch
done
clear
View Answer play_arrow
question_answer 222) Directions: In each of the following questions, there occurs a specific relations. Fill the vacant space (?) question mark according to that relation. Bread : Wheat: : Brick :?
A)
Cement
done
clear
B)
Building
done
clear
C)
Clay
done
clear
D)
Fire
done
clear
View Answer play_arrow
question_answer 223) Directions: In each of the following questions, there occurs a specific relations. Fill the vacant space (?) question mark according to that relation. Mouse : Cat: : Fly :?
A)
Horse
done
clear
B)
Spider
done
clear
C)
Rat
done
clear
D)
Animal
done
clear
View Answer play_arrow
question_answer 224) Directions: In each of the following questions, there occurs a specific relations. Fill the vacant space (?) question mark according to that relation. Mirror : Reflection : : Water :?
A)
Immersion
done
clear
B)
Conduction
done
clear
C)
Refraction
done
clear
D)
Dispersion
done
clear
View Answer play_arrow
question_answer 225) Directions: In each of the following questions, there occurs a specific relations. Fill the vacant space (?) question mark according to that relation. Tennis : Court: : Boxing :?
A)
Ring
done
clear
B)
Course
done
clear
C)
Pool
done
clear
D)
Arena
done
clear
View Answer play_arrow
question_answer 226) Directions: In the following questions, choose the option which shows common feature in the relationship given in each question. Sarnath : Kapilavastu : Sanchi
A)
These have ancient universities
done
clear
B)
These are places having massive pillars
done
clear
C)
These are linked with Lord Buddha
done
clear
D)
These are famous for stone caves
done
clear
View Answer play_arrow
question_answer 227) Directions: In the following questions, choose the option which shows common feature in the relationship given in each question. Ebony : Rosewood : Mahogany
A)
These are hardwood trees,
done
clear
B)
These are coniferous trees
done
clear
C)
These yield good for fuel
done
clear
D)
These are trees of temperature regions
done
clear
View Answer play_arrow
question_answer 228) Directions: In the following questions, choose the option which shows common feature in the relationship given in each question. Arjun : Uddhav : Sudama
A)
They were all princes
done
clear
B)
They were friends of Krishna
done
clear
C)
They were Pandavas
done
clear
D)
They were great warriors
done
clear
View Answer play_arrow
question_answer 229) Directions: In the following questions, choose the option which shows common feature in the relationship given in each question. Sherlock Holmes : James Bond : Hercules Poirot
A)
They are the only detective agents
done
clear
B)
They are private detectives
done
clear
C)
They are agents of CBI
done
clear
D)
They are characters from detective fiction
done
clear
View Answer play_arrow
question_answer 230) Directions: In the following questions, choose the option which shows common feature in the relationship given in each question. Goose : Duck : Stork
A)
They are white
done
clear
B)
They are water birds
done
clear
C)
These species are disappearing
done
clear
D)
They migrate to India from Siberia.
done
clear
View Answer play_arrow
question_answer 231) Who is named as the flying Sikh of India?
A)
Mohinder Singh
done
clear
B)
Ajit Pal Singh
done
clear
C)
Joginder Singh
done
clear
D)
Milkha Singh
done
clear
View Answer play_arrow
question_answer 232) The members of Rajya Sabha are elected for a term of
A)
two years
done
clear
B)
four years
done
clear
C)
six years
done
clear
D)
five years
done
clear
View Answer play_arrow
question_answer 233) Who was the first Governor-General of Bengal?
A)
Robert Clive
done
clear
B)
Warren Hastings
done
clear
C)
William Bentinck
done
clear
D)
Cornwallis
done
clear
View Answer play_arrow
question_answer 234) Jnanpith Award is conferred to those in the field of
A)
Literature
done
clear
B)
History
done
clear
C)
Drama
done
clear
D)
Dance
done
clear
View Answer play_arrow
question_answer 235) What was Lala Lajpat Rai demonstrating against when he succumbed to police brutality?
A)
Rowlatt Act
done
clear
B)
Minto-Morley Reforms
done
clear
C)
Simon Commission
done
clear
D)
Pitts India Act
done
clear
View Answer play_arrow
question_answer 236) The present Indian monetary system is based on
A)
Gold Reserve System
done
clear
B)
Proportional Reserve System
done
clear
C)
Convertible Currency System
done
clear
D)
Minimum Reserve System
done
clear
View Answer play_arrow
question_answer 237) Garba is a dance form of
A)
Gujarat
done
clear
B)
Rajasthan
done
clear
C)
Odisha
done
clear
D)
Asom
done
clear
View Answer play_arrow
question_answer 238) ATM stands for
A)
Automatic Teller Machine
done
clear
B)
Automated Teller Machine
done
clear
C)
Automatic Tally Machine
done
clear
D)
Automated Tally Machanism
done
clear
View Answer play_arrow
question_answer 239) The first African National to become Secretary General of UNO was
A)
Kofi Annan
done
clear
B)
Burros Gali
done
clear
C)
Nelson Mandela
done
clear
D)
Winni Mandela
done
clear
View Answer play_arrow
question_answer 240) Who was the first Indian woman to scale Mt Everest?
A)
Bachendri Pal
done
clear
B)
Fu Dorji
done
clear
C)
Aun Sang Suu Kyi
done
clear
D)
Yoko Ono
done
clear
View Answer play_arrow