-
The following concentrations
were obtained for the formation of\[N{{H}_{3}}\] from \[{{N}_{2}}\] and \[{{H}_{2}}\]
at equilibrium at 500 K.
\[[{{N}_{2}}]=1.5\times {{10}^{-2}}M,[{{H}_{2}}]=3\times
{{10}^{-2}}M\]and\[[N{{H}_{3}}]=1.2\times {{10}^{-2}}M.\]Calculate the
equilibrium constant.
View Answer play_arrow
-
At equilibrium the
concentrations of
\[[{{N}_{2}}]=3\times
{{10}^{-3}}M,[{{O}_{2}}]=4.2\times {{10}^{-3}}M\]and\[[NO]=28\times
{{10}^{-3}}M\]in a sealed vessel at 800 K. What will be \[{{K}_{c}}\] for the
reaction:
\[{{N}_{2}}(g)+{{O}_{2}}(g)\rightleftharpoons 2NO(g)?\]
View Answer play_arrow
-
\[PC{{l}_{5}},PC{{l}_{3}}\]and\[C{{l}_{2}}\] are at
equilibrium at\[500K\]and having concentration 1.59 M\[PC{{l}_{3}},1.59M\,C{{l}_{2}}\]
and\[1.41\,M\,PC{{l}_{5}}\]. Calculate \[{{K}_{c}}\] for the reaction:
View Answer play_arrow
-
The value of \[{{K}_{c}}=4.24\]at
800 K for the reaction:
\[CO(g)+{{H}_{2}}O(g)\rightleftharpoons
C{{O}_{2}}(g)+{{H}_{2}}(g)\]
Calculate equilibrium concentrations of \[C{{O}_{2}}(g),{{H}_{2}}(g)\]\[CO(g)\]and
\[{{H}_{2}}O(g)\]at 800K, if only CO and \[{{H}_{2}}O\] are present initially
at concentrations of 0.1 M each.
View Answer play_arrow
-
For the equilibrium,
\[2NOCl(g)\rightleftharpoons
2NO(g)+C{{l}_{2}}(g)\]
the value of the equilibrium constant, \[{{K}_{c}}\] is\[3.75\times
{{10}^{-6}}\].Calculate the \[{{K}_{p}}\]for the reaction at this temperature.
View Answer play_arrow
-
The value of K p for the
reaction,
\[C{{O}_{2}}(g)+C(s)\rightleftharpoons 2CO(g)\]is 3 at 1000K.
If initially\[{{p}_{C{{O}_{2}}}}=0.48\]bar and \[{{p}_{CO}}=0\]bar and pure
graphite is present, calculate the equilibrium partial pressures of CO and\[C{{O}_{2}}\].
View Answer play_arrow
-
The value of \[{{K}_{c}}\]
for the reaction:
\[2A\rightleftharpoons
B+C\]is \[2\times {{10}^{-3}}\]. At a given time, the composition of reaction
mixture is:
\[[A]=[B]=[C]=3\times {{10}^{-4}}M\]
In which direction, the reaction will proceed?
View Answer play_arrow
-
13.8 g of \[{{N}_{2}}{{O}_{4}}\](g)
was placed in a 1L reaction vessel at 400 K and allowed to attain equilibrium:
\[{{N}_{2}}{{O}_{4}}(g)\rightleftharpoons 2N{{O}_{2}}(g)\]
The total pressure at equilibrium was found to be 9.15 bar.
Calculate \[{{K}_{c}},{{K}_{p}}\] and partial pressure at equilibrium.
View Answer play_arrow
-
\[3mol\] of\[PC{{l}_{5}}\]
kept in 1 L closed vessel was
allowed to attain
equilibrium at 380 K. Calculate the composition of the reaction mixture at
equilibrium. \[{{K}_{c}}=1.80\]
View Answer play_arrow
-
The value of\[\Delta {{G}^{{}^\circ }}\]for the
phosphorylation of glucose in glycolysis is 13.8 kJ/mol. Find the value of \[{{K}_{c}}\]
at 298K.
View Answer play_arrow
-
Hydrolysis of sucrose gives
Sucrose + \[{{H}_{2}}O\rightleftharpoons
\] Glucose + Fructose
Equilibrium constant \[{{K}_{c}}\] for the reaction is \[2\times
{{10}^{13}}\] at 300 K. Calculate \[\Delta G{}^\circ \]at 300 K.
View Answer play_arrow
-
What will be the
conjugate bases for the following Bronsted acids?
\[HF,{{H}_{2}}S{{O}_{4}},HCO_{3}^{-}\]
View Answer play_arrow
-
Write down the conjugate acids for the following Bronsted
bases: \[NH_{2}^{-},N{{H}_{3}}\] and\[HCO{{O}^{-}}\].
View Answer play_arrow
-
The species: \[{{H}_{2}}O,HCO_{3}^{-},HSO_{4}^{-},N{{H}_{3}}\]can
act both as Bronsted acids and bases. For each case give the corresponding
conjugate acid and conjugate base.
View Answer play_arrow
-
Classify the
following species into Lewis acids and Lewis bases and show how these act as
Lewis acid/base?
(a) \[O{{H}^{-}}\] (b)
\[{{F}^{-}}\]
(c) \[{{H}^{+}}\] (d)
\[BC{{l}_{3}}\]
View Answer play_arrow
-
The concentration of hydrogen ion in a sample of soft drink
is\[3.8\times {{10}^{-3}}M\]. What is its pH?
View Answer play_arrow
-
Calculate the pH of \[1.0\times {{10}^{-8}}MHCl\]solution.
View Answer play_arrow
-
The ionisation constant of HF is\[3.2\times {{10}^{-4}}\]. Calculate
the degree of dissociation of HF in its 0.02 M solution. Calculate the
concentration of all species present\[{{H}_{3}}{{O}^{+}},{{F}^{-}}\] and HF in
the solution and its pH.
View Answer play_arrow
-
The pH of 0.1 M monobasic acid is 4.50.Calculate the
concentration of species \[{{H}^{+}},{{A}^{-}}\] and HA at equilibrium.
Calculate the value of\[{{K}_{a}}\] and \[p{{K}_{a}}\] also.
View Answer play_arrow
-
Calculate the pH of 0.08 M solution of hypochlorous acid,\[HOCl\].
The ionization constant of the acid is\[2.5\times {{10}^{-5}}\]. Determine the
percentage dissociation of\[HOCl\].
View Answer play_arrow
-
The pH of 0.004 M hydrazine solution is 9.7. Calculate its
ionization constants \[{{K}_{b}}\]and \[p{{K}_{b}}\] .
View Answer play_arrow
-
Calculate the pH of solution formed on mixing 0.2 M \[N{{H}_{4}}Cl\]
and 0.1 M \[N{{H}_{3}}\]. The\[p{{K}_{b}}\] of ammonia solution is 4.75.
View Answer play_arrow
-
Determine the degree of ionization and \[pH\] of 0.05 M of
ammonia solution. The ionization constant of ammonia is\[1.77\times
{{10}^{-5}}\]. Also calculate the ionization constant of conjugate acid of
ammonia.
View Answer play_arrow
-
Calculate the pH of a 0.1 M
ammonia solution. Calculate the pH after 50 mL of this solution is treated with
25 mL of 0.1 M HCl. The dissociation constant of ammonia,
\[{{K}_{b}}=1.77\times {{10}^{-5}}\].
View Answer play_arrow
-
The \[p{{K}_{a}}\] of acetic acid and \[p{{K}_{b}}\] of ammonium
hydroxide are 4.76 and 4.75 respectively. Calculate the \[pH\] of ammonium
acetate solution.
View Answer play_arrow
-
Calculate the solubility of \[{{A}_{2}}{{X}_{3}}\] in pure Water,
assuming that neither kind of ion reacts with water. The solubility product of\[{{A}_{2}}{{X}_{3}},{{K}_{sp}}=1.1\times
{{10}^{-23}}\].
View Answer play_arrow
-
The values of\[{{K}_{sp}}\]
of two sparingly soluble
Salt \[Ni{{(OH)}_{2}}\]
and\[AgCN\]are \[2\times {{10}^{-15}}\]and \[6\times {{10}^{-17}}\]respectively.
Which salt is more
soluble? Explain.
View Answer play_arrow
-
Calculate the molar
solubility of \[Ni{{(OH)}_{2}}\]in 0.1 M\[NaOH\]. The ionic product of\[Ni{{(OH)}_{2}}\]is\[2\times
{{10}^{-15}}\].
View Answer play_arrow
-
A liquid is in equilibrium with
its vapours in a sealed container at a fixed temperature. The volume of the container
is suddenly increased.
(a) What is the initial effect
of the change on the vapour pressure?
(b) How do the rates of
evaporation and condensation change initially?
(c) What happens when the equilibrium is restored finally and
what will be the final vapour pressure?
View Answer play_arrow
-
What is \[{{K}_{c}}\] for the
following equilibrium when the equilibrium concentration of each substance is:
\[[S{{O}_{2}}]=0.6M;[{{O}_{2}}]=0.82M;[S{{O}_{3}}]=1.90M?\] \[2S{{O}_{2}}(g)+{{O}_{2}}(g)\rightleftharpoons
2S{{O}_{3}}(g)\]
View Answer play_arrow
-
At a certain temperature and
total pressure of \[{{10}^{5}}\]Pa, iodine vapour contains 40% by volume of \[I\]
atoms.
\[{{I}_{2}}(g)\rightleftharpoons
2I(g)\]
Calculate \[{{K}_{p}}\] for the equilibrium.
View Answer play_arrow
-
Write down the
expression for the equilibrium constant,\[{{K}_{c}}\] for each of the following
reactions:
(i) \[2NOCl(g)\rightleftharpoons
2NO(g)+C{{l}_{2}}(g)\]
(ii) \[2Cu{{(N{{O}_{3}})}_{2}}(s)\rightleftharpoons
2CuO(s)+4N{{O}_{2}}(g)+{{O}_{2}}(g)\]
(iii) \[C{{H}_{3}}COO{{C}_{2}}{{H}_{5}}(aq)+{{H}_{2}}O(aq)\rightleftharpoons
C{{H}_{3}}COOH(aq)\] \[+{{C}_{2}}{{H}_{5}}OH(aq)\]
(iv)\[F{{e}^{3+}}(aq)+3O{{H}^{-}}(aq)\rightleftharpoons
Fe{{(OH)}_{2}}(s)\]
(v) \[{{I}_{2}}(s)+5{{F}_{2}}(g)\rightleftharpoons
2I{{F}_{5}}(g)\]
View Answer play_arrow
-
Find out the value ofx\[{{K}_{c}}\]
for each of the following equilibria from the value of \[{{K}_{p}}\]:
(i) \[2NOCl(g)\underset{{}}{\leftrightarrows}2NO(g)+C{{l}_{2}}(g);\]\[{{K}_{p}}=1.8\times
{{10}^{-2}}\] at 500 K
(ii) \[CaC{{O}_{3}}(s)\underset{{}}{\leftrightarrows}CaO(s)+C{{O}_{2}}(g);\]
\[{{K}_{p}}=167\] at 1073 K
View Answer play_arrow
-
For the following
equilibrium \[{{K}_{c}}=6.3\times {{10}^{14}}\]at 1000 K
\[NO(g)+{{O}_{3}}(g)\rightleftharpoons
N{{O}_{2}}(g)+{{O}_{2}}(g)\]
Both forward and backward reactions in the equilibrium are
elementary bimolecular reactions. What is the\[{{K}_{c}}\], for the reverse
reaction?
View Answer play_arrow
-
Explain, why pure liquids and solids can be ignored while
writing the equilibrium constant expression?
View Answer play_arrow
-
Reaction between \[{{N}_{2}}\]
and \[{{O}_{2}}\] takes place as follows:
\[2{{N}_{2}}(g)+{{O}_{2}}(g)\rightleftharpoons
2{{N}_{2}}O(g)\]
If a mixture of 0.482 mol \[{{N}_{2}}\] and 0.933 mol of \[{{O}_{2}}\]
is placed in a 10L reaction vessel and allowed to form \[{{N}_{2}}O\]at a
temperature for which\[{{K}_{c}}=2\times {{10}^{-37}}\], determine the composition
of equilibrium mixture.
View Answer play_arrow
-
Nitric oxide reacts with
bromine and gives nitrosyl bromide as per the reaction given below:
\[2NO(g)+B{{r}_{2}}(g)\rightleftharpoons
2NOBr(g)\]
when 0.087 mol of NO and 0.0437 mol of\[B{{r}_{2}}\] are
mixed in a closed container at constant temperature, 0.0518 mol of\[NOBr\]is
obtained at equilibrium. Calculate the equilibrium amount of nitric oxide and
bromine.
View Answer play_arrow
-
At\[450K,{{K}_{p}}=2\times
{{10}^{10}}ba{{r}^{-1}}\]for the given reaction at equilibrium,
\[2S{{O}_{2}}(g)+{{O}_{2}}(g)\rightleftharpoons
2S{{O}_{3}}(g)\]
What is \[{{K}_{c}}\] at this temperature?
View Answer play_arrow
-
A sample of HI (g) is placed in
a flask at a pressure of 0.2aim. At equilibrium the partial pressure of HI (g)
is 0.04atm. What is \[{{K}_{p}}\]for the given equilibrium?
\[2HI(g)\rightleftharpoons {{H}_{2}}(g)+{{I}_{2}}(g)\]
View Answer play_arrow
-
A mixture of 1.57 mole of \[{{N}_{2}}\]
1.92 mole of \[{{H}_{2}}\] and 8.13mole of\[N{{H}_{3}}\] is introduced into a
20 L reaction vessel at 500K. At this temperature, the equilibrium constant \[{{K}_{c}}\]
for the reaction:
\[{{N}_{2}}(g)+3{{H}_{2}}(g)\rightleftharpoons
2N{{H}_{3}}(g)\]
is\[1.7\times {{10}^{2}}\].Is the reaction mixture at
equilibrium? If not, what is the direction of the net reaction?
View Answer play_arrow
-
The equilibrium constant
expression for a gas reaction is,
\[{{K}_{c}}=\frac{{{[N{{H}_{3}}]}^{4}}{{[{{O}_{2}}]}^{5}}}{{{[NO]}^{4}}{{[{{H}_{2}}O]}^{6}}}\]
Write down the balanced chemical equation corresponding
to this expression.
View Answer play_arrow
-
At 700 K, equilibrium constant
for the reaction:
\[{{H}_{2}}(g)+{{I}_{2}}(g)\rightleftharpoons
2HI(g)\]
is 54.8. If \[0.5mol\,{{L}^{-1}}\]of\[HI\left( g \right)\]is
present at equilibrium at700 K. What is the concentration of \[{{H}_{2}}(g)\]and
\[{{I}_{2}}(g)\]assuming that we initially started with \[HI\left( g \right)\]and
allowed it to reach equilibrium at 700 K?
View Answer play_arrow
-
At 700 K, equilibrium constant
for the reaction:
\[{{H}_{2}}(g)+{{I}_{2}}(g)\rightleftharpoons
2HI(g)\]
is 54.8. If \[0.5mol\,{{L}^{-1}}\]of\[HI\left( g \right)\]is
present at equilibrium at700 K. What is the concentration of \[{{H}_{2}}(g)\]and
\[{{I}_{2}}(g)\]assuming that we initially started with \[HI\left( g \right)\]and
allowed it to reach equilibrium at 700 K?
View Answer play_arrow
-
What is the concentration of each of the substances in the equilibrium
when the initial concentration of\[~ICl\] was 0.78M?
View Answer play_arrow
-
\[{{K}_{p}}=0.04\]atm
at 899 K for the quilibrium shown below. What is the equilibrium concentration
(pressure) of \[{{C}_{2}}{{H}_{6}}\]when it is placed in flask at 4 atm
pressure and allowed to come to equilibrium?
\[{{C}_{2}}{{H}_{6}}(g)\rightleftharpoons
{{C}_{2}}{{H}_{4}}(g)+{{H}_{2}}(g)\]
View Answer play_arrow
-
The ester, ethyl acetate, is
formed by the reaction of ethanol and acetic acid and the equilibrium is
represented as:
\[C{{H}_{3}}COOH(l)+{{C}_{2}}{{H}_{5}}OH(l)\rightleftharpoons
C{{H}_{3}}COO{{C}_{2}}{{H}_{5}}(l)+{{H}_{2}}O(l)\](i) Write the
concentration ratio, Qfor this reaction. Note that water is not in excess and
is not a solvent in this reaction.
(ii) At 293 K, if
one starts with 1.000 mol of acetic acid and 0.180 mol of ethanol, there is
0.171 mol of ethyl acetate in the final equilibrium mixture. Calculate the equilibrium
constant.
(iii) Starting with 0.50 mol of ethanol and 1.000 mol of acetic
acid and maintaining it at 293 K, 0.214 mol of ethyl acetate is found after
some time. Has equilibrium been reached?
View Answer play_arrow
-
A sample of pure \[PC{{l}_{5}}\] was introduced into an
evacuated vessel at 473 K. After equilibrium was reached, the concentration of\[PC{{l}_{5}}\]was
found to be\[0.5\times {{10}^{-1}}mol\,{{L}^{-1}}\]. If \[{{K}_{c}}\] is \[8.3\times
{{10}^{-3}}\], what are the concentrations of\[PC{{l}_{3}}\] and\[C{{l}_{2}}\] at the equilibrium?
View Answer play_arrow
-
One of the reaction that takes
place in producing steel from iron ore is the reduction of iron (II) oxide by
carbon monoxide to give iron metal and \[C{{O}_{2}}(g)\]
\[FeO(s)+CO(g)\rightleftharpoons
FeO(s)+CO(g)\]
\[{{K}_{p}}\]=
0.265 atm at 1050 K.
What is the
equilibrium partial pressure of CO and \[C{{O}_{2}}\] at1050 K if the initial
partial pressure is:
\[{{p}_{CO}}=1.4\]atm and \[{{p}_{C{{O}_{2}}}}=0.80\]atm?
View Answer play_arrow
-
Equilibrium constant \[{{K}_{c}}\] for the
reaction, \[{{N}_{2}}(g)+3{{H}_{2}}(g)\rightleftharpoons 2N{{H}_{3}}(g)\] at 500 K is 0.061. At particular
time, the analysis shows that the composition of the reaction mixture is: \[3.0mol{{L}^{-1}}\]of\[{{N}_{2}};\,\,2.0mol\,{{L}^{-1}}\]of
\[{{H}_{2}};0.50mol{{L}^{-1}}\] of\[N{{H}_{3}}\] . Is the reaction at
equilibrium? If not, in which direction does the reaction tend to proceed to reach
the equilibrium?
View Answer play_arrow
-
Bromine monochloride \[\left(
BrCl \right)\]decomposes into bromine and chlorine and reaches the equilibrium:
\[2BrCl(g)\rightleftharpoons
B{{r}_{2}}(g)+C{{l}_{2}}(g)\]
The value of\[{{K}_{c}}\] is 32 at 500 K. If initially pure \[BrCl\]
is present at a concentration of\[3.3\times {{10}^{-3}}mol{{L}^{-1}}\], what is
its molar concentration in the mixture at equilibrium?
View Answer play_arrow
-
At 1127 K and 1 atm pressure, a
gaseous mixture of CO and\[C{{O}_{2}}\]in equilibrium with solid carbon has
90.55% CO by mass.
\[C(s)+C{{O}_{2}}(g)\rightleftharpoons
2CO(g)\]
Calculate \[{{K}_{c}}\] for this reaction.
View Answer play_arrow
-
Calculate (a)\[\Delta
{{G}^{{}^\circ }}\]and (b) the equilibrium constant for the formation of \[N{{O}_{2}}\]
from NO and \[{{O}_{2}}\]at \[298\,K\]
\[NO(g)+\frac{1}{2}{{O}_{2}}(g)\rightleftharpoons
N{{O}_{2}}(g)\]where\[{{\Delta }_{f}}{{G}^{{}^\circ
}}(N{{O}_{2}})=52kJ/mol;\,\]\[{{\Delta }_{f}}{{G}^{{}^\circ }}(NO)\]
\[=87kJ/mol;\]\[{{\Delta }_{f}}{{G}^{{}^\circ
}}({{O}_{2}})=0kJ/mol\]
View Answer play_arrow
-
Does the number of
moles of reaction products increase, decrease or remain same when each of the
following equilibria is subjected to a decrease in pressure by increasing the
volume?
(i)\[PC{{l}_{5}}(g)\rightleftharpoons
PC{{l}_{3}}(g)+C{{l}_{2}}(g)\]
(ii) \[CaO(s)+C{{O}_{2}}(g)\rightleftharpoons
CaC{{O}_{3}}(s)\]
(iii)\[3Fe(s)+4{{H}_{2}}O(g)\rightleftharpoons
F{{e}_{3}}{{O}_{4}}(s)+4{{H}_{2}}(g)\]
View Answer play_arrow
-
Which of the
following reactions will get affected by increasing the pressure? Also, mention
whether change will cause the reaction to go into forward or backward direction?
(i) \[COC{{l}_{2}}(g)\rightleftharpoons
CO(g)+C{{l}_{2}}(g)\]
(ii) \[C{{H}_{4}}(g)+2{{S}_{2}}(s)\rightleftharpoons
C{{S}_{2}}(g)+2{{H}_{2}}S(g)\]
(iii) \[C{{O}_{2}}(g)+C(s)\rightleftharpoons
2CO(g)\]
(iv)\[2{{H}_{2}}(g)+CO(g)\rightleftharpoons
C{{H}_{3}}OH(g)\]
(v) \[CaC{{O}_{3}}(s)\rightleftharpoons
Cao(s)+C{{O}_{2}}(g)\]
(vi)\[4N{{H}_{3}}(g)+5{{O}_{2}}(g)\rightleftharpoons
4NO(g)+6{{H}_{2}}O(g)\]
View Answer play_arrow
-
The equilibrium constant for
the following reaction is\[1.6\times {{10}^{5}}\]at\[1024\,K.\]
\[{{H}_{2}}(g)+B{{r}_{2}}(g)\rightleftharpoons
2HBr(g)\]
Find the equilibrium pressure of all gases if 10 bar of\[HBr\]is
introduced into a sealed container at 1024 K.
View Answer play_arrow
-
Dihydrogen gas is obtained from
the natural gas by partial oxidation with steam as per following endothermic reaction:
\[C{{H}_{4}}(g)+{{H}_{2}}O(g)\rightleftharpoons
CO(g)+3{{H}_{2}}(g)\]
(a) Write
the expression for \[{{K}_{p}}\]for the above reaction.
(b) How will
the value of \[{{K}_{p}}\]and composition of equilibrium mixture be affected
by:
(i) increasing the
pressure (ii) increasing the temperature
(iii) using a catalyst?
View Answer play_arrow
-
What is the effect
of:
(i) addition
of \[{{H}_{2}}\]
(ii) addition
of \[C{{H}_{3}}OH\]
(iii) removal
of \[CO\]
(iv) removal
of \[C{{H}_{3}}OH\]
on the
equilibrium
\[2{{H}_{2}}(g)+CO(g)\rightleftharpoons
C{{H}_{3}}OH(g)?\]
View Answer play_arrow
-
At 473 K, the
equilibrium constant \[{{K}_{c}}\] for the decomposition of phosphorus
pentachloride \[(PC{{l}_{5}})\]is\[8.3\times {{10}^{-3}}\]. If decomposition
proceeds as:\[PC{{l}_{5}}(g)\rightleftharpoons PC{{l}_{3}}(g)+C{{l}_{2}}(g);\] \[\Delta
H=+124.0kJ\,mo{{l}^{-1}}\]
(a) Write
an expression for \[{{K}_{c}}\] for the reaction.
(b) What is
the value of \[{{K}_{c}}\] for the reverse reaction at the same temperature?
(c) What would be
the effect on \[{{K}_{c}}\]if
(i) More of\[PC{{l}_{5}}\] is added (ii) Temperature is
increased?
View Answer play_arrow
-
Dihydrogen gas used in Haber's
process is produced by reacting methane from natural gas with high temperature steam.
The first stage of two stage reaction involves the formation of CO and\[{{H}_{2}}\].
In second stage, CO formed in first stage is reacted with more steam in water
gas shift reaction,
\[CO(g)+{{H}_{2}}O(g)\rightleftharpoons
C{{O}_{2}}(g)+{{H}_{2}}(g)\]
If a
reaction vessel at 400C is charged with an equimolar mixture of CO and steam,
such that,
\[{{p}_{CO}}={{p}_{{{H}_{2}}O}}=4bar\]be
the partial pressure of
\[{{H}_{2}}\]at equilibrium? \[{{K}_{p}}\] = 0.1 at 400C
View Answer play_arrow
-
Predict which of the following
will have appreciable concentration of reactants and products?
(a) \[C{{l}_{2}}(g)\rightleftharpoons
2Cl(g);{{K}_{c}}=5\times {{10}^{-39}}\]
(b)\[C{{l}_{2}}(g)+2NO(g)\rightleftharpoons
2NOCl(g);\,\,\,{{K}_{c}}=3.7\times {{10}^{8}}\]
(c)\[C{{l}_{2}}(g)+2N{{O}_{2}}(g)\rightleftharpoons
2N{{O}_{2}}Cl(g);\,\,\,\,{{K}_{c}}=1.8\]
View Answer play_arrow
-
The value of \[{{K}_{c}}\] for the reaction \[3{{O}_{2}}(g)\rightleftharpoons
2{{O}_{3}}(g)\]is\[2\times {{10}^{-50}}\]at\[{{250}^{{}^\circ }}C\]. If the
equilibrium concentration of \[{{O}_{2}}\]in air at\[25{{\,}^{o}}C\,is1.6\text{
}\times \text{ }{{10}^{-2}}M\], what is the concentration of\[{{O}_{3}}?\]
View Answer play_arrow
-
The
reaction,
\[CO(g)+3{{H}_{2}}(g)\rightleftharpoons
C{{H}_{4}}(g)+{{H}_{2}}O(g)\]
is at equilibrium at 1300Kin a 1 L flask. It also contains 0.3mole of CO, 0.1 mole
of \[{{H}_{2}}\] and 0.02 mole of \[{{H}_{2}}O\]and an unknown amount of in the
flask. Determine the concentration of\[C{{H}_{4}}(g)\] in the mixture. The
equilibrium constant, \[{{K}_{c}}\] for the reaction at a given temperature, is
3.90.
View Answer play_arrow
-
What
is meant by the conjugate acid-base pair? Find the conjugate acid/base for the
following species:
\[HN{{O}_{3}},\overset{-}{\mathop{C}}\,N,HCl{{O}_{4}},{{F}^{-}},O{{H}^{-}},CO_{3}^{2-},{{S}^{2-}}\]
View Answer play_arrow
-
Which
of the following are Lewis acids?
\[{{H}_{2}}O,B{{F}_{3}},{{H}^{+}}\]and\[NH_{4}^{+}\]
View Answer play_arrow
-
Which
of the following are Lewis acids?
\[{{H}_{2}}O,B{{F}_{3}},{{H}^{+}}\]and\[NH_{4}^{+}\]
View Answer play_arrow
-
Write
the conjugate acids for the following Bronsted bases:
\[NH_{2}^{-},N{{H}_{3}},HCO{{O}^{-}}\]
View Answer play_arrow
-
The species \[{{H}_{2}}O,HCO_{3}^{-},HSO_{4}^{-},N{{H}_{3}}\]
can act both as Bronsted acids and bases. For each case give the corresponding
conjugate acid and base.
View Answer play_arrow
-
Classify the following species into Lewis acids and Lewis bases
and show how these act as Lewis acid/base:
(a) \[O{{H}^{-}}\] (b) \[{{F}^{-}}\]
(c) \[{{H}^{+}}\] (d)
\[BC{{l}_{3}}\]
View Answer play_arrow
-
The concentration of hydrogen ion
in a sample of soft drink is \[3.8\text{ }\times \text{ }{{10}^{-3}}M\]. What
is its \[pH\]?
View Answer play_arrow
-
The pH of a sample of vinegar is 3.76. Calculate the concentration of hydrogen ion in it.
View Answer play_arrow
-
The ionisation constant of HF,
HCOOH and HCN at 298 K are \[6.8\text{ }\times
\text{ }{{10}^{-4}},\text{ }1.8\text{ }\times \text{ }{{10}^{-4}}\] and\[4.8\times
{{10}^{-9}}\]respectively. Calculate the ionisation constants of the corresponding
conjugate base.
View Answer play_arrow
-
The ionisation constant of phenol
is\[1\times {{10}^{-10}}\]. What is the concentration of phenolate
ion in 0.05 M solution of phenol? What will be
its degree of ionisation if the solution is also 0.01 M in sodium phenolate?
View Answer play_arrow
-
The first ionization constant of\[{{H}_{2}}S\]is\[9.1\times {{10}^{-8}}\]. Calculate the concentration of\[H{{S}^{-}}ions\] in its 0.1M solution and how will this concentration be affected if the solution is 0.1 M in HCl also? If the second dissociation constant of \[{{H}_{2}}S\] is \[1.2\times {{10}^{-13}}\], calculate the concentration of \[{{S}^{2-}}\] under both conditions.
View Answer play_arrow
-
The ionisation constant of acetic
acid is\[1.74\times {{10}^{-5}}\]. Calculate
the degree of dissociation of acetic acid in its 0.05 M solution. Calculate the
concentration of acetate ion in the solution and its\[pH\].
View Answer play_arrow
-
It has been found that the pH of a 0.01 M solution of organic acid is 4.15. Calculate the
concentration of the onion, the ionisation constant of the acid and its\[p{{K}_{a}}\].
View Answer play_arrow
-
Assuming
complete dissociation, calculate the pH of following solutions:
(i) 0.003 M HCl (b) 0.005 M \[NaOH\](c)
0.002 M \[HBr\](d) 0.002 M KOH.
View Answer play_arrow
-
Calculate the pH of the following solutions:
(a) 2 g of \[TlOH\] dissolved in water to give 2 litre of
solution
(b) 0.3 g of\[Ca{{(OH)}_{2}}\] dissolved in water to give
500 mL solution
(c) 0.3 g of \[NaOH\]dissolved
in water to give 200 mL of solution
(d) 1 mL of 13.6 M \[HCl\]
is diluted with water to give 1 litre of \[HCl\]solution.
View Answer play_arrow
-
The degree of ionization of 0.1 M bromoacetic acid solution is 0.132. Calculate the pH of
the solution and the \[p{{K}_{a}}\] of bromoacetic acid.
View Answer play_arrow
-
The pH of 0.005 M codeine (\[{{C}_{18}}{{H}_{21}}N{{O}_{3}}\])
solution is 9.95.Calculate its ionization constant and\[p{{K}_{b}}\].
View Answer play_arrow
-
What is the pH of 0.001 M aniline solution? The ionization constant of aniline
is\[4.27\times {{10}^{-10}}\]. Calculate the degree of ionization of aniline in
the solution. Also calculate the ionization of the conjugate acid of aniline.
View Answer play_arrow
-
Calculate the degree of ionization
of 0.05 M acetic acid if its \[p{{K}_{a}}\] value is 4.74. How is the degree of dissociation affected when its
solution also contains (a) 0.01 M HCl (b) 0.1 M HCl?
View Answer play_arrow
-
The ionization constant of dimethylamine is\[5.4\times
{{10}^{-4}}\]. Calculate its degree of
ionization in its 0.02 M solution. What percentage of dimethylamine is ionized
if the solution is also 0.1 M\[NaOH\]?
View Answer play_arrow
-
Calculate the hydrogen ion concentration in the following biological
fluids whose pH are given below:
(a) Human muscle-fluid,
6.83 (b) Human stomach, 1.2
(c) Human bloody 7.38 (d)
Human saliva, 6.4.
View Answer play_arrow
-
The pH of milk black coffee,
tomato juice, lemon juice and white egg are
6.8, 5, 4.2, 2.2 and 7.8 respectively. Calculate corresponding hydrogen ion
concentration of each.
View Answer play_arrow
-
If
0.561 g of KOH is dissolved in water to give 200 mL of solution at 298 K. Calculate
the concentrations of potassium, hydrogen and hydroxyl ions. What is the pH of solution?
View Answer play_arrow
-
The solubility of\[Sr{{(OH)}_{2}}\]at 298 K is 19.23 g/L. Calculate the concentration of
strontium, hydroxyl ions and the Ph of the solutions.
View Answer play_arrow
-
The ionization constant of propanoic
acid is\[1.32\times {{10}^{-5}}\].Calculate
the degree of ionization of the acid in its 0.05 M solution and also its\[pH\].
What will be its degree of ionization if the solution is 0.01 M in HCl also?
View Answer play_arrow
-
The pH of 0.1 M solution of HCNO (cyanic acid) is 2.34. Calculate the ionization constant of the acid and its
degree of ionization in the solution.
View Answer play_arrow
-
The ionisation constant of nitrous
acid is\[4.5\times {{10}^{-4}}\].Calculate the
pH of 0.04 M sodium nitrite solution and also its
degree of hydrolysis.
View Answer play_arrow
-
A 0.02 M solution of pyridinium
hydrochloride has pH = 3.44. Calculate the ionisation constant of pyridine.
View Answer play_arrow
-
Predict
if the solutions of the following salts are neutral acidic or basic:
\[NaCl,KBr,NaCN,N{{H}_{4}}N{{O}_{3}},NaN{{O}_{2}}\]and\[KF\]
View Answer play_arrow
-
Ionisation constant of
chloroacetic acid is\[1.35\times {{10}^{-3}}\].What
will be the pH of 0.1 M acid and 0.1 M salt solution?
View Answer play_arrow
-
Ionic product of water at 310 K is\[2.7\times {{10}^{-14}}\]. What is the pH of
neutral water at this temperature?
View Answer play_arrow
-
Calculate the pH of the resultant mixtures:
(a)\[10\,mL\,of\,0.2MCa{{(OH)}_{2}}+25mL\,of0.1M\,HCl\](b) \[10mL\,of\,0.1\,M\,{{H}_{2}}S{{O}_{4}}+10mL\,of\,0.01M\,Ca{{(OH)}_{2}}\]
(c)\[10mL\,of\,0.1\,M\,{{H}_{2}}S{{O}_{4}}+10mL\,of\,0.1M\,KOH\]
View Answer play_arrow
-
Determine the solubilities of silver chromate, barium chromate,
ferric hydroxide, lead chloride and mercurous iodide at 298 K from the solubility product constant. Determine the
molarities of individual ions.
\[{{K}_{sp}}A{{g}_{2}}Cr{{O}_{4}}=1.1\times {{10}^{-12}}\]
\[{{K}_{sp}}Fe{{(OH)}_{3}}=1\times {{10}^{-38}}\]
\[{{K}_{sp}}PbC{{l}_{2}}=1.6\times {{10}^{-5}}\]
\[{{K}_{sp}}H{{g}_{2}}{{I}_{2}}=4.5\times {{10}^{-29}}\]
View Answer play_arrow
-
The solubility product constant of\[A{{g}_{2}}Cr{{O}_{4}}\]
and \[AgBr\] are \[1.1\times {{10}^{-12}}\]and \[5\times {{10}^{-13}}\]respectively.
Calculate the ratio of molarities of their saturated solutions.
View Answer play_arrow
-
Equal
volume of 0.002 M solutions of sodium iodate
and cupric chlorate are mixed together. Will it lead to precipitation of copper
iodate? (For cupric iodate
\[{{K}_{sp}}=7.4\times
{{10}^{-8}})\]
View Answer play_arrow
-
The ionization constant of benzoic
acid is\[6.46\times {{10}^{-5}}\]and\[{{K}_{sp}}\] for silver benzoate is\[2.5\times
{{10}^{-13}}\]. How many times the silver benzoate
is more soluble in buffer of pH 3.19 compared to its solubility in pure water?
View Answer play_arrow
-
What
is the maximum concentration of equimolar solution of ferrous sulphate and
sodium sulphide so that when mixed in equal volumes, there is no precipitation
of iron sulphide? (For iron sulphide,
\[{{K}_{sp}}=6.3\times
{{10}^{-18}})\]
View Answer play_arrow
-
What is the minimum volume of Water
required to dissolve 1g calcium sulphate at\[298K?\]For \[CaS{{O}_{4}}\],\[{{K}_{sp}}\]
is\[9.1\times {{10}^{-6}}\].
View Answer play_arrow
-
The
concentration of sulphide ion in \[0.1\,M\,HCl\] solution saturated with
hydrogen sulphide is \[1\times {{10}^{-19}}M.\] If 10 mL of this is added to 5
mL of 0.04 M solution of the following :
\[FeS{{O}_{4}},\,MnC{{l}_{2}},\,ZnC{{l}_{2}}\,and\,CdC{{l}_{2}}.\]
In which of these solutions precipitation will take place?
Given \[{{K}_{sp}}FeS=6.3\times
{{10}^{-18}};\,\,{{K}_{sp}}MnS=2.5\times {{10}^{-13}}\]
\[{{K}_{sp}}ZnS=1.6\times
{{10}^{-24}};\,\,\,{{K}_{sp}}CdS=8\times {{10}^{-27}}\]
View Answer play_arrow
-
question_answer102)
In the following questions only one
option is correct.
We know that the relationship
between \[{{K}_{c}}\] and\[{{K}_{p}}\] is\[{{K}_{p}}={{K}_{c}}{{(RT)}^{\Delta
n}}\]. What would be the value of An for the reaction \[N{{H}_{4}}Cl(s)\underset{{}}{\leftrightarrows}N{{H}_{3}}(g)+HCl(g)?\]
(a) 1 (b)
0.5 (c) 1.5 (d)
2
View Answer play_arrow
-
question_answer103)
For the reaction\[{{H}_{2}}(g)+{{I}_{2}}(g)\underset{{}}{\leftrightarrows}2HI(g),\]
the standard free energy is\[\Delta {{G}^{O-}}>0.\] The equilibrium constant
(K) would be:
(a)\[K=0\] (b)
\[K>1\] (c) \[K=1\] (d) \[K<1\]
View Answer play_arrow
-
question_answer104)
Which of the following is not a
general characteristic of equilibrium involving physical processes?
(a) Equilibrium is possible only
in a closed system at a given temperature.
(b) All measurable properties of
the system remain constant.
(c) All the physical processes
stop at equilibrium.
(d) The opposing processes occur
at the same rate and there is dynamic but stable condition
View Answer play_arrow
-
question_answer105)
\[PC{{l}_{5}},PC{{l}_{3}}\] and \[C{{l}_{2}}\]
are at equilibrium at 500 K in a closed container and their concentrations are\[0.8\times
{{10}^{-3}}mol{{L}^{-1}}\], \[1.2\times {{10}^{-3}}mol{{L}^{-1}}\] and \[1.2\times
{{10}^{-3}}mol{{L}^{-1}}\] respectively. The value of\[~{{K}_{c}}\] for the
reaction \[PC{{l}_{5}}(g)\underset{{}}{\leftrightarrows}PC{{l}_{3}}(g)+C{{l}_{2}}(g)\]will
be:
(a) \[1.8\times
{{10}^{3}}mol\,{{L}^{-1}}\] (b) \[1.8\times
{{10}^{-3}}mol{{L}^{-1}}\] (c) \[1.8\times
{{10}^{-3}}L\,mo{{l}^{-1}}\] (d) \[0.55\times {{10}^{4}}mol{{L}^{-1}}\]
View Answer play_arrow
-
question_answer106)
Which of the following
statements is incorrect?
(a) In equilibrium mixture of
ice and water kept in perfectly insulated flask mass of ice and water does not
change with time.
(b) The intensity of red colour
increases when oxalic acid is added to a solution containing iron(III) nitrate
and potassium thiocyanate.
(c) On addition of catalyst the
equilibrium constant value is not affected.
(d) Equilibrium constant for a
reaction with negative \[\Delta H\]value decreases as the temperature
increases.
View Answer play_arrow
-
question_answer107)
When hydrochloric acid is added
to cobalt nitrate solution at room temperature, the following reaction takes
place and the reaction mixture becomes blue. On cooling the mixture it becomes
pink. On the basis of this information mark the correct answer.
\[\underset{(pink)}{\mathop{{{[Co{{({{H}_{2}}O)}_{6}}]}^{2+}}}}\,(aq)+4C{{l}^{-}}(aq)\underset{{}}{\leftrightarrows}\underset{(blue)}{\mathop{{{[CoC{{l}_{4}}]}^{2-}}}}\,(aq)\]\[+6{{H}_{2}}O(l)\]
(a) \[\Delta H>0\]for the
reaction
(b) \[\Delta H<0\]for the
reaction
(c)\[\Delta H=0\] for the
reaction
(d) The sign of \[\Delta H\]
cannot be predicted on the basis of this information
View Answer play_arrow
-
question_answer108)
The pH of neutral water at \[25{}^\circ
C\]is 7.0. As the temperature increases, ionisation of water increases,
however, the concentration of \[{{H}^{+}}\] ions and \[O{{H}^{-}}\] ions are
equal. What will be the pH of pure water at 60°C?
(a) Equal to 7.0 (b)
Greater than 7.0 (c) Less than 7.0 (d) Equal to zero
View Answer play_arrow
-
question_answer109)
The ionisation constant of an
acid,\[{{K}_{a}}\], is the measure of strength of an acid. The \[{{K}_{a}}\]
values of acetic acid, hypochlorous acid and formic acid are \[1.74\times
{{10}^{-5}}\],\[3.0\times {{10}^{-8}}\]and \[1.8\times {{10}^{-4}}\]respectively.
Which of the following orders of pH of 0.1 mol \[d{{m}^{-3}}\] solutions of
these acids is correct?
(a) Acetic acid > hypochlorous
acid > formic acid
(b) Hypochlorous acid >
acetic acid > formic acid
(c) Formic acid > hypochlorous
acid > acetic acid
(d) Formic acid > acetic acid
>hypochlorous acid
View Answer play_arrow
-
question_answer110)
\[{{K}_{{{a}_{1}}}},{{K}_{{{a}_{2}}}}\]
and \[{{K}_{{{a}_{3}}}}\] are the respective ionisation constants for the
following reactions:
\[{{H}_{2}}S\underset{{}}{\leftrightarrows}{{H}^{+}}+H{{S}^{-}}\]
\[H{{S}^{-}}\underset{{}}{\leftrightarrows}{{H}^{+}}+{{S}^{2-}}\]
\[{{H}_{2}}S\underset{{}}{\leftrightarrows}2{{H}^{+}}+{{S}^{2-}}\]
The correct relationship between
\[{{K}_{{{a}_{1}}}},{{K}_{{{a}_{2}}}}\] and \[{{K}_{{{a}_{3}}}}\] is:
(a) \[{{K}_{{{a}_{3}}}}={{K}_{{{a}_{1}}}}\times
{{K}_{{{a}_{2}}}}\] (b) \[{{K}_{{{a}_{3}}}}={{K}_{{{a}_{1}}}}+{{K}_{{{a}_{2}}}}\] (c)
\[{{K}_{{{a}_{3}}}}={{K}_{{{a}_{1}}}}-{{K}_{{{a}_{2}}}}\] (d) \[{{K}_{{{a}_{3}}}}={{K}_{{{a}_{1}}}}/{{K}_{{{a}_{2}}}}\]
View Answer play_arrow
-
question_answer111)
Acidity of \[B{{F}_{3}}\] can be
explained on the basis of which of the following concepts?
(a) Arrhenius concept (b)
Bronsted Lowry concept
(c) Lewis concept (d)
Bronsted Lowry as well as Lewis concept
View Answer play_arrow
-
question_answer112)
Which of the following will
produce a buffer solution when mixed in equal volumes?
(a) \[0.1mol\,d{{m}^{-3}}N{{H}_{4}}OH\,and\,0.1mol\,d{{m}^{-3}}HCl\] (b)
\[0.05mol\,d{{m}^{-3}}N{{H}_{4}}OH\,and\,0.1mol\,d{{m}^{-3}}HCl\]
(c) \[0.1mol\,d{{m}^{-3}}N{{H}_{4}}OH\,and\,0.05mol\,d{{m}^{-3}}HCl\] (d)
\[0.1mol\,d{{m}^{-3}}C{{H}_{4}}COONa\,and\,0.1mol\,d{{m}^{-3}}NaOH\]
View Answer play_arrow
-
question_answer113)
In which of the following
solvents is silver chloride most soluble?
(a) \[0.1\text{ }mol\text{
}d{{m}^{-3}}AgN{{O}_{3}}\] solution (b) \[0.1\text{
}mol\text{ }d{{m}^{-3}}\text{ }HCl\]solution
(c) \[{{H}_{2}}O\] (d)
Aqueous ammonia
View Answer play_arrow
-
question_answer114)
What will be the value of pH of
0.1 mol \[0.1mol\,d{{m}^{3}}C{{H}_{3}}COOH\]
(a) 3.4 (b)
3.6 (c) 3.9 (d) 3.0
View Answer play_arrow
-
question_answer115)
\[{{K}_{a}}\]for \[C{{H}_{3}}COOH\]
is \[1.8\times {{10}^{-5}}\] and \[{{K}_{b}}\] for \[N{{H}_{4}}OH\] is\[1.8\times
{{10}^{-5}}\]. The pH of ammonium acetate will be:
(a) 7.005 (b)
4.75 (c) 7.0 (d)
between 6 and 7
View Answer play_arrow
-
question_answer116)
Which of the following options
will be correct for the stage of half completion of the reaction\[A\underset{{}}{\leftrightarrows}B\]?
(a)\[\Delta {{G}^{O-}}=0\] (b)
\[\Delta {{G}^{O-}}>0\] (c) \[\Delta
{{G}^{O-}}<0\] (d) \[\Delta {{G}^{O-}}=-RT\ln
2\]
View Answer play_arrow
-
question_answer117)
On increasing the pressure, in which
direction will the gas phase reaction proceed to re-establish equilibrium, is predicted
by applying the Le Chatelier's principle. Consider the reaction
\[{{N}_{2}}(g)+3{{H}_{2}}(g)\underset{{}}{\leftrightarrows}2N{{H}_{3}}(g)\]
Which of the following is correct, if
the total pressure at which the equilibrium is established, is increased
without changing the temperature?
(a) K will remain same
(b) K will decrease
(c) K will increase
(d) K will increase initially
and decrease when pressure is very high
View Answer play_arrow
-
question_answer118)
What will be the correct order
of vapour pressure of water, acetone and ether at\[30{}^\circ C\]? Given that
among these compounds, water has maximum boiling point and ether has minimum
boiling point?
(a) Water < ether <
acetone (b) Water < acetone < ether
(c) Ether < acetone <
water (d) Acetone < ether < water
View Answer play_arrow
-
question_answer119)
At 500 K, equilibrium constant, \[{{K}_{c}}\],
for the following reaction is 5:
\[\frac{1}{2}{{H}_{2}}(g)+\frac{1}{2}{{I}_{2}}(g)\underset{{}}{\leftrightarrows}HI(g)\]
What would be the equilibrium
constant \[{{K}_{c}}\]for the reaction?
\[2HI(g)\underset{{}}{\leftrightarrows}{{H}_{2}}(g)+{{I}_{2}}(g)\]
(a) 0.04 (b)
0.4 (c) 25 (d) 2.5
View Answer play_arrow
-
question_answer120)
In which of the following
reactions, the equilibrium remains unaffected on addition of small amount of
argon at constant volume?
(a) \[{{H}_{2}}(g)+{{I}_{2}}(g)\underset{{}}{\leftrightarrows}HI(g)\]
(b) \[PC{{l}_{5}}(g)\underset{{}}{\leftrightarrows}PC{{l}_{3}}(g)+C{{l}_{2}}(g)\]
(c) \[{{N}_{2}}(g)3{{H}_{2}}(g)\underset{{}}{\leftrightarrows}2N{{H}_{3}}(g)\]
(d) The equilibrium will remain
unaffected in all the three cases.
View Answer play_arrow
-
question_answer121)
In the following questions
two or more options may be correct.
For the reaction, \[{{N}_{2}}{{O}_{4}}(g)\underset{{}}{\leftrightarrows}2N{{O}_{2}}(g),\]
the value of K is 50 at 400 K and 1700 at 500 K. Which of the following options
is correct?
(a) The reaction is endothermic
(b) The reaction is exothermic
(c) If \[N{{O}_{2}}(g)\] and \[{{N}_{2}}{{O}_{4}}(g)\]
are mixed at 400 K at partial pressures 20 bar and 2 bar respectively, more \[{{N}_{2}}{{O}_{4}}(g)\]will
be formed
(d) The entropy of the system
increases
View Answer play_arrow
-
question_answer122)
At a particular temperature and
atmospheric pressure, the solid and liquid phases of a pure substance can exist
in equilibrium. Which of the following term defines this temperature?
(a) Normal melting point (b)
Equilibrium temperature
(c) Boiling point (d)
Freezing point
View Answer play_arrow
-
question_answer123)
The ionization of hydrochloric
acid in water is given below:
\[HCl(aq)+{{H}_{2}}O(l)\underset{{}}{\leftrightarrows}{{H}_{3}}{{O}^{+}}(aq)+C{{l}^{-}}(aq)\]
Label two conjugate acid-base
pairs in this ionization.
View Answer play_arrow
-
question_answer124)
The aqueous solution of sugar
does not conduct electricity. However, when sodium chloride is added to water,
it conducts electricity. How will you explain this statement on the basis of
ionisation and how is it affected by concentration of sodium chloride?
View Answer play_arrow
-
question_answer125)
\[B{{F}_{3}}\]does not have
proton but still acts as an acid and reacts with \[N{{H}_{3}}\]. Why is it so?
What type of bond is formed between the two?
View Answer play_arrow
-
question_answer126)
Ionisation constant of a weak
base MOH, is given by the expression
\[{{K}_{b}}=\frac{[{{M}^{+}}][O{{H}^{-}}]}{[MOH]}\]
Values of ionisation constant of
some weak bases at a particular temperature are given below:
|
Base
|
Dimethylamine
|
Urea
|
Pyridine
|
Ammonia
|
|
\[{{K}_{b}}\]
|
\[5.4\times
{{10}^{-4}}\]
|
\[1.3\times
{{10}^{-14}}\]
|
\[1.77\times {{10}^{-9}}\]
|
\[1.77\times {{10}^{-5}}\]
|
Arrange the bases in decreasing
order of the extent of their ionisation at equilibrium. Which of the above base
is the strongest?
View Answer play_arrow
-
question_answer127)
Conjugate acid of a weak base is
always stronger. What will be the decreasing order of basic strength of the
following conjugate bases?
\[O{{H}^{-}},R{{O}^{-}}C{{H}_{3}}CO{{O}^{-}},C{{l}^{-}}\]
View Answer play_arrow
-
question_answer128)
Arrange the following in increasing
order of pH:
\[KN{{O}_{3}}(aq),C{{H}_{3}}COONa(aq),N{{H}_{4}}Cl(aq),\]\[{{C}_{6}}{{H}_{5}}COON{{H}_{4}}(aq)\]
View Answer play_arrow
-
question_answer129)
The value of \[{{K}_{c}}\] for
the reaction \[2HI(g)\underset{{}}{\leftrightarrows}{{H}_{2}}(g)+{{I}_{2}}(g)\]
is \[1\times {{10}^{-4}}\]. At a given time, the composition of reaction mixture
is:
\[[HI]=2\times
{{10}^{5}}mol,[{{H}_{2}}]=1\times {{10}^{-5}}mol\] and \[[{{I}_{2}}]=1\times
{{10}^{-5}}mol\]
In which direction will the
reaction proceed?
View Answer play_arrow
-
question_answer130)
On the basis of the equation \[pH=-\log
[{{H}^{+}}]\], the pH of \[{{10}^{-8}}mol\,d{{m}^{-3}}\] solution of \[HCl\]
should be 8. However, it is observed to be less than 7.0. Explain the reason.
View Answer play_arrow
-
question_answer131)
pH of a solution of a strong
acid is 5.0. What will be the pH of the solution obtained after dilution the
given solution a100 times?
View Answer play_arrow
-
question_answer132)
A sparingly soluble salt gets
precipitated only when the product of concentration of its ions in the solution
\[({{Q}_{sp}})\]becomes greater than its solubility product. If the solubility
of \[BaS{{O}_{4}}\] in water is\[8\times {{10}^{-4}}mol\,d{{m}^{-3}}\].
Calculate its solubility in \[0.01\,mol\,d{{m}^{-3}}\] of\[{{H}_{2}}S{{O}_{4}}\].
View Answer play_arrow
-
question_answer133)
pH of \[0.08mol\,d{{m}^{-3}}HOCl\]
solution is 2.85. Calculate its ionization constant.
View Answer play_arrow
-
question_answer134)
Calculate the pH of a solution formed
by mixing equal volumes of two solutions A and B of a strong acid having pH = 6
and pH = 4 respectively.
View Answer play_arrow
-
question_answer135)
The solubility product of \[Al{{(OH)}_{3}}\]
is\[2.7\times {{10}^{-11}}\].
Calculate its solubility in \[g{{L}^{-1}}\]
and also find out pH of this solution. (Atomic mass of Al = 27 u)
View Answer play_arrow
-
question_answer136)
Calculate the volume of water
required to dissolve 0.1 g\[PbC{{l}_{2}}\] to get a saturated solution. \[({{K}_{sp}}PbC{{l}_{2}}=3.2\times
{{10}^{-8}},\] Atomic mass of \[Pb=207amu\]
View Answer play_arrow
-
question_answer137)
A reaction between ammonia and
boron trifluoride is given below:
\[B{{F}_{3}}+\,_{\centerdot
}^{\centerdot }N{{H}_{3}}\to {{H}_{3}}N_{\centerdot }^{\centerdot }B{{F}_{3}}\]
Identify the acid and base in this
reaction. Which theory explains it? What is the hybridization of B and N in the
reactants?
View Answer play_arrow
-
question_answer138)
Following data is given for the reaction
:
\[CaC{{O}_{3}}(s)\to CaO(s)+C{{O}_{2}}(g)\]
\[{{\Delta }_{f}}{{H}^{O-}}[CaO(s)]=-635.1kJ\,mo{{l}^{-1}}\]
\[{{\Delta
}_{f}}{{H}^{O-}}[C{{O}_{2}}(s)]=-393.5kJ\,mo{{l}^{-1}}\]
\[{{\Delta
}_{f}}{{H}^{O-}}[CaC{{O}_{3}}(s)]=-1206.9kJ\,mo{{l}^{-1}}\]
Predict
the effect of temperature on the equilibrium constant of the above reaction.
View Answer play_arrow
-
question_answer139)
In some of the following questions,
one option of left column may be correlated to more than one option in the
right column.
Match the following equilibria
with the corresponding condition.
|
|
Column I
|
|
Column II
|
|
(i)
|
Liquid \[\underset{{}}{\leftrightarrows}\] Vapour
|
(a)
|
Saturated
solutions
|
|
(ii)
|
Solid \[\underset{{}}{\leftrightarrows}\]Liquid
|
(b)
|
Boiling point
|
|
(iii)
|
Solid \[\underset{{}}{\leftrightarrows}\]Vapour
|
(c)
|
Sublimation point
|
|
(iv)
|
Solute (s)\[\underset{{}}{\leftrightarrows}\]Solute
(Solution)
|
(d)
|
Melting point
|
|
|
|
(e)
|
Unsaturated solutions
|
View Answer play_arrow
-
question_answer140)
For the reaction:
\[{{N}_{2}}(g)+3{{H}_{2}}(g)\underset{{}}{\leftrightarrows}2N{{H}_{3}}(g)\]
Equilibrium constant \[{{K}_{c}}=\frac{{{[N{{H}_{3}}]}^{2}}}{[{{N}_{2}}]{{[{{H}_{2}}]}^{3}}}\]
Some reactions are written below
in Column I and their equilibrium constants in terms of \[{{K}_{c}}\] are
written in Column II. Match the following reactions with the corresponding equilibrium
constant.
|
Column l
(Reaction)
|
Column II
(Equilibrium constant)
|
|
(i) \[2{{N}_{2}}(g)+6{{H}_{2}}(g)\underset{{}}{\leftrightarrows}4N{{H}_{3}}(g)\]
|
(a) \[2{{K}_{c}}\]
|
|
(ii) \[2N{{H}_{3}}(g)\underset{{}}{\leftrightarrows}{{N}_{2}}(g)+3{{H}_{2}}(g)\]
|
(b) \[K_{c}^{\frac{1}{2}}\]
|
|
(iii) \[\frac{1}{2}{{N}_{2}}(g)+\frac{3}{2}{{H}_{2}}(g)\underset{{}}{\leftrightarrows}N{{H}_{3}}(g)\]
|
(c) \[\frac{1}{{{K}_{c}}}\]
|
|
|
(d) \[K_{c}^{2}\]
|
|
|
|
View Answer play_arrow
-
question_answer141)
Match standard free energy of
the reaction with to corresponding equilibrium constant.
|
Column l
|
Column II
|
|
(i) \[\Delta {{G}^{O-}}>0\]
|
(a) \[K>1\]
|
|
(ii) \[\Delta {{G}^{O-}}<0\]
|
(b) \[K=1\]
|
|
(iii) \[\Delta {{G}^{O-}}=0\]
|
(c) \[K=0\]
|
|
|
(d) \[K<1\]
|
View Answer play_arrow
-
question_answer142)
Match the following species with
the corresponding conjugate acid.
|
Species
|
Conjugate acid
|
|
(i) \[N{{H}_{3}}\]
|
(a) \[CO_{3}^{2-}\]
|
|
(ii) \[HCO_{3}^{-}\]
|
(b) \[NH_{4}^{+}\]
|
|
(iii) \[{{H}_{2}}O\]
|
(c) \[{{H}_{3}}{{O}^{+}}\]
|
|
(iv) \[HSO_{4}^{-}\]
|
(d) \[{{H}_{2}}S{{O}_{4}}\]
|
|
|
(e) \[{{H}_{2}}C{{O}_{3}}\]
|
View Answer play_arrow
-
question_answer143)
Match the following graphical
variation with their description.
|
Column l
|
Column II
|
(i) 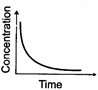
|
(a) Variation in product concentration
with time
|
(ii) 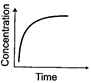
|
(b) Reaction at equilibrium
|
(iii) 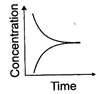
|
(c) Variation in reactant
concentration with time
|
View Answer play_arrow
-
question_answer144)
Match Column (I) with Column
(II).
|
Column l
|
Column II
|
|
(i) Equilibrium
|
(a) \[\Delta G>0,K<1\]
|
|
(ii) Spontaneous reaction
|
(b) \[\Delta G=0\]
|
|
(iii)Non spontaneous reaction
|
(c) \[\Delta {{G}^{O-}}=0\]
|
|
|
(d) \[\Delta G<0,K>1\]
|
View Answer play_arrow
-
question_answer145)
In the following questions a
statement of Assertion (A) followed by a statement of Reason (R) is given.
Choose the correct option out of the choices given below each questions.
(a) A and R both
are true and R is the correct explanation of A.
(b) A and R both
are true but R is not the correct explanation of A.
(c) A is true but R
is false.
(d) A and R both
are false.
Assertion (A): Increasing
order of acidity of hydrogen halides is\[HF<HCl<HBr<HI\].
Reason (R): While
comparing acids formed by the elements belonging to the same group of periodic
table, H-A bond strength is a more important factor in determining acidity of
an acid than the polar nature of the bond.
View Answer play_arrow
-
question_answer146)
Assertion (A): A solution
containing a mixture of acetic acid and sodium acetate maintains a constant
value of pH on addition of small amounts of acid or alkali.
Reason (R): A solution
containing a mixture of acetic acid and sodium acetate acts as a buffer
solution around pH 4.75.
View Answer play_arrow
-
question_answer147)
Assertion (A): The
ionisation of hydrogen sulphide in water is low in the presence of hydrochloric
acid.
Reason (R): Hydrogen
sulphide is a weak acid.
View Answer play_arrow
-
question_answer148)
Assertion (A): For any
chemical reaction at a particular temperature, the equilibrium constant is
fixed and is a characteristic property.
Reason (R): Equilibrium
constant is independent of temperature.
View Answer play_arrow
-
question_answer149)
Assertion (A): Aqueous
solution of ammonium carbonate is basic.
Reason (R): Acidic/basic
nature of a salt solution of a salt of weak acid and weak base depends on \[{{K}_{a}}\]
and \[{{K}_{b}}\] value of the acid and the base forming it.
View Answer play_arrow
-
question_answer150)
Assertion (A): An aqueous
solution of ammonium acetate can act as a buffer.
Reason (R): Acetic acid
is a weak acid and \[N{{H}_{4}}OH\] is a weak base.
View Answer play_arrow
-
question_answer151)
Assertion (A): In the
dissociation of \[PC{{l}_{5}}\] at constant pressure and temperature addition
of helium at equilibrium increases the dissociation of\[PC{{l}_{5}}\].
Reason (R): Helium
removes \[C{{l}_{2}}\] from the field of action.
View Answer play_arrow
-
question_answer152)
How will you predict the
following stages of a reaction by comparing the value of \[{{K}_{c}}\]and \[{{Q}_{c}}\]
?
(i) Net reaction proceeds in
forward direction
(ii) Net reaction proceeds in
backward direction
(iii) No net reaction occurs
View Answer play_arrow
-
question_answer153)
On the basis of Le Chatelier's
principle explain how the temperature and pressure can be adjusted to increase
the yield of ammonia in following reaction:
\[{{N}_{2}}(g)+3{{H}_{2}}(g)\rightleftharpoons
2N{{H}_{3}}(g)\]
\[\Delta
H=-92.38kJ\,mo{{l}^{-1}}\]
What will be the effect of
addition of argon to the above reaction mixture at constant volume?
View Answer play_arrow
-
question_answer154)
A sparingly soluble salt having
general formula \[{{A}_{x}}{{B}_{y}}\]and molar solubility s is in equilibrium
with its saturated solution. Derive a relationship between the solubility and solubility
product for such salt.
View Answer play_arrow
-
question_answer155)
Write a relation between \[\Delta
G\] and Q and define the meaning of each term and answer the following:
(a) Why a reaction proceeds
forward when Q < K and no net reaction occurs when \[Q=K?\]
(b) Explain the effect of
increase in pressure in terms of the reaction quotient Q for the reaction:
\[CO(g)+3{{H}_{2}}(g)\underset{{}}{\leftrightarrows}C{{H}_{4}}(g)+{{H}_{2}}O(l)\]
View Answer play_arrow



