-
question_answer1)
For the reaction \[{{H}_{2}}(g)+{{I}_{2}}(g)\]⇌\[2HI(g)\], the rate of reaction is expressed as [CBSE PMT 1997; AIEEE 2002]
A)
\[-\frac{\Delta [{{I}_{2}}]}{\Delta t}=-\frac{\Delta [{{H}_{2}}]}{\Delta t}=\frac{1}{2}\frac{\Delta [HI]}{\Delta t}\] done
clear
B)
\[\frac{\Delta [{{I}_{2}}]}{\Delta t}=\frac{\Delta [{{H}_{2}}]}{\Delta t}=\frac{\Delta [HI]}{2\Delta t}\] done
clear
C)
\[\frac{\Delta [{{H}_{2}}]}{\Delta t}=\frac{1}{2}\frac{\Delta [{{I}_{2}}]}{\Delta t}=-\frac{\Delta [HI]}{\Delta t}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer2)
If \[3A\to 2B,\] then the rate of reaction of \[+\frac{d(B)}{dt}\]is equal to [CBSE PMT 2002]
A)
\[+2\frac{d(A)}{dt}\] done
clear
B)
\[-\frac{1}{3}\frac{d(A)}{dt}\] done
clear
C)
\[-\frac{2}{3}\frac{d(A)}{dt}\] done
clear
D)
\[-\frac{3}{2}\frac{d(A)}{dt}\] done
clear
View Solution play_arrow
-
question_answer3)
The differential rate law for the reaction \[{{H}_{2}}+{{I}_{2}}\to 2HI\] is [AIEEE 2002]
A)
\[-\frac{d[{{H}_{2}}]}{dt}=-\frac{d[{{I}_{2}}]}{dt}=+\frac{1}{2}\frac{d[HI]}{dt}\] done
clear
B)
\[\frac{d[{{H}_{2}}]}{dt}=\frac{d[HI]}{dt}=\frac{1}{2}\frac{d[HI]}{dt}\] done
clear
C)
\[\frac{1}{2}\frac{d[{{H}_{2}}]}{dt}=\frac{1}{2}\frac{d[{{I}_{2}}]}{dt}=-\frac{d[HI]}{dt}\] done
clear
D)
\[-2\frac{d[{{H}_{2}}]}{dt}=-2\frac{d[{{I}_{2}}]}{dt}=+\frac{d[HI]}{dt}\] done
clear
View Solution play_arrow
-
question_answer4)
The rate law for a reaction between the substances A and B is given by, rate \[=k{{[A]}^{n}}{{[B]}^{m}}\]. On doubling the concentration of A and halving the concentration of B, the ratio of the new rate to the earlier rate of the reaction will be as [AIEEE 2003]
A)
\[\frac{1}{{{2}^{(m+n)}}}\] done
clear
B)
\[(m+n)\] done
clear
C)
\[(n-m)\] done
clear
D)
\[{{2}^{(n-m)}}\] done
clear
View Solution play_arrow
-
question_answer5)
If we plot a graph between log K and \[\frac{1}{T}\] by Arrhenius equation, the slope is [UPSEAT 2001]
A)
\[-\frac{{{E}_{a}}}{R}\] done
clear
B)
\[+\frac{{{E}_{a}}}{R}\] done
clear
C)
\[-\frac{{{E}_{a}}}{2.303\,R}\] done
clear
D)
\[+\frac{{{E}_{a}}}{2.303\,R}\] done
clear
View Solution play_arrow
-
question_answer6)
For an endothermic reaction, where \[\Delta H\] represents the enthalpy of the reaction in \[kJ/mol\], the minimum value for the energy of activation will be [IIT 1992]
A)
Less than \[\Delta H\] done
clear
B)
Zero done
clear
C)
More than \[\Delta H\] done
clear
D)
Equal to \[\Delta H\] done
clear
View Solution play_arrow
-
question_answer7)
The rate constant \[({K}')\] of one reaction is double of the rate constant \[({K}'')\] of another reaction. Then the relationship between the corresponding activation energies of the two reactions (\[{{E}_{a}}^{\prime }\] and \[{{E}_{a}}^{\prime\prime }\]) will be [MP PET 1994; UPSEAT 2001]
A)
\[{{E}_{a}}^{\prime }>{{E}_{a}}^{\prime\prime }\] done
clear
B)
\[{{E}_{a}}^{\prime }={{E}_{a}}^{\prime\prime }\] done
clear
C)
\[{{E}_{a}}^{\prime }<{{E}_{a}}^{\prime\prime }\] done
clear
D)
\[{{E}_{a}}^{\prime }=4{{E}_{a}}^{\prime\prime }\] done
clear
View Solution play_arrow
-
question_answer8)
The rate constant, the activation energy and the arrhenius parameter of a chemical reaction at \[{{25}^{o}}C\] are \[3.0\times {{10}^{-4}}{{s}^{-1}}\], \[104.4\,kJ\,mo{{l}^{-1}}\] and \[6.0\times {{10}^{14}}{{s}^{-1}}\] respectively. The value of the rate constant as \[T\to \infty \] is [IIT 1996]
A)
\[2.0\times {{10}^{18}}{{s}^{-1}}\] done
clear
B)
\[6.0\times {{10}^{14}}{{s}^{-1}}\] done
clear
C)
Infinity done
clear
D)
\[3.6\times {{10}^{30}}{{s}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer9)
The \[\Delta H\] value of the reaction \[{{H}_{2}}+C{{l}_{2}}\]⇌ \[2HCl\] is \[-44.12\,kcal\]. If \[{{E}_{1}}\] is the activation energy of the products, then for the above reaction [EAMCET 1997]
A)
\[{{E}_{1}}>{{E}_{2}}\] done
clear
B)
\[{{E}_{1}}<{{E}_{2}}\] done
clear
C)
\[{{E}_{1}}={{E}_{2}}\] done
clear
D)
\[\Delta H\] is not related to \[{{E}_{1}}\] and \[{{E}_{2}}\] done
clear
E)
None is correct done
clear
View Solution play_arrow
-
question_answer10)
The temperature dependence of rate constant \[(k)\] of a chemical reaction is written in terms of Arrhenius equation, \[K=A.{{e}^{-E*/RT}}\]. Activation energy \[({{E}^{*}})\] of the reaction can be calculated by plotting [CBSE PMT 2003]
A)
\[\log \,k\,vs\,\frac{1}{\log \,T}\] done
clear
B)
\[k\,vs\,T\] done
clear
C)
\[k\,vs\,\frac{1}{\log \,T}\] done
clear
D)
\[\log \,k\,vs\,\frac{1}{T}\] done
clear
View Solution play_arrow
-
question_answer11)
Activation energy of a chemical reaction can be determined by [CBSE PMT 1998; AFMC 1999; BHU 2000]
A)
Changing concentration of reactants done
clear
B)
Evaluating rate constant at standard temperature done
clear
C)
Evaluating rate constants at two different temperatures done
clear
D)
Evaluating velocities of reaction at two different temperatures done
clear
View Solution play_arrow
-
question_answer12)
The activation energy for a reaction is \[9.0\,K\,cal/mol.\] The increase in the rate constant when its temperature is increased from 298K to 308K is [JIPMER 2000]
A)
63% done
clear
B)
50% done
clear
C)
100% done
clear
D)
10% done
clear
View Solution play_arrow
-
question_answer13)
Which of the following is the fastest reaction [Pb. CET 2002]
A)
\[C+\frac{1}{2}{{O}_{2}}\xrightarrow{250{}^\circ C}CO\] done
clear
B)
\[C+\frac{1}{2}{{O}_{2}}\xrightarrow{500{}^\circ C}CO\] done
clear
C)
\[C+\frac{1}{2}{{O}_{2}}\xrightarrow{750{}^\circ C}CO\] done
clear
D)
\[C+\frac{1}{2}{{O}_{2}}\xrightarrow{1000{}^\circ C}CO\] done
clear
View Solution play_arrow
-
question_answer14)
The rate constant k, for the reaction \[{{N}_{2}}{{O}_{5}}(g)\to \] \[2N{{O}_{2}}(g)+\frac{1}{2}{{0}_{2}}(g)\] is \[2.3\times {{10}^{-2}}{{s}^{-1}}\]. Which equation given below describes the change of \[[{{N}_{2}}{{O}_{5}}]\] with time? \[{{[{{N}_{2}}{{O}_{5}}]}_{0}}\] and \[{{[{{N}_{2}}{{O}_{5}}]}_{t}}\]correspond to concentration of \[{{N}_{2}}{{O}_{5}}\]initially and at time, t [AIIMS 2004]
A)
\[{{[{{N}_{2}}{{O}_{5}}]}_{t}}={{[{{N}_{2}}{{O}_{5}}]}_{0}}+kt\] done
clear
B)
\[{{[{{N}_{2}}{{O}_{5}}]}_{0}}={{[{{N}_{2}}{{O}_{5}}]}_{t}}{{e}^{kt}}\] done
clear
C)
\[{{\log }_{10}}{{[{{N}_{2}}{{O}_{5}}]}_{t}}={{\log }_{10}}{{[{{N}_{2}}{{O}_{5}}]}_{0}}-kt\] done
clear
D)
\[\text{In}\frac{{{\text{ }\!\![\!\!\text{ }{{\text{N}}_{\text{2}}}{{O}_{5}}]}_{0}}}{{{\text{ }\!\![\!\!\text{ }{{\text{N}}_{\text{2}}}{{O}_{5}}]}_{t}}}=kt\] done
clear
View Solution play_arrow
-
question_answer15)
For the reaction \[C{{H}_{3}}COOC{{H}_{3}}+{{H}_{2}}O\xrightarrow{{{H}^{+}}}\] \[C{{H}_{3}}COOH+C{{H}_{3}}OH\]The progress of the process of reaction is followed by
A)
Finding the amount of methanol formed at different intervals done
clear
B)
Finding the amount of acetic acid formed at different intervals done
clear
C)
Using a voltmeter done
clear
D)
Using a polarimeter done
clear
View Solution play_arrow
-
question_answer16)
Half life of a reaction is found to be inversely proportional to the cube of its initial concentration. The order of reaction is [KCET 2002]
A)
2 done
clear
B)
5 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer17)
The integrated rate equation is \[Rt=\log \ {{C}_{0}}-\log {{C}_{t}}\]. The straight line graph is obtained by plotting [AIEEE 2002]
A)
\[\text{time}\ \text{v/s}\ \text{log}{{\text{C}}_{\text{t}}}\] done
clear
B)
\[\frac{1}{\text{time}}\text{v}/\text{s}\ {{\text{C}}_{\text{t}}}\] done
clear
C)
\[\text{time}\ \text{v}/\text{s}\ {{\text{C}}_{\text{t}}}\] done
clear
D)
\[\frac{1}{\text{time}}\text{v}/\text{s}\ \frac{1}{{{\text{C}}_{\text{t}}}}\] done
clear
View Solution play_arrow
-
question_answer18)
For which order reaction a straight line is obtained along with x?axis by plotting a graph between half life \[({{t}_{1/2}})\]and initial concentration 'a' [RPET 2003]
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
0 done
clear
View Solution play_arrow
-
question_answer19)
The reaction, \[X\to \]product follows first order kinetics. In 40 minutes the concentration of X changes from 0.1 M to 0.025 M Then the rate of reaction when concentration of X is 0.01 M
A)
\[1.73\times {{10}^{-4}}M\ {{\min }^{-1}}\] done
clear
B)
\[3.47\times {{10}^{-5}}M\ {{\min }^{-1}}\] done
clear
C)
\[3.47\times {{10}^{-4}}M\ {{\min }^{-1}}\] done
clear
D)
\[1.73\times {{10}^{-5}}M\ {{\min }^{-1}}\] done
clear
View Solution play_arrow
-
question_answer20)
A Substance undergoes first order decomposition. The decomposition follows two parallel first order reactions as
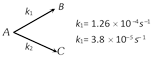
The percentage distribution of B and C are [Kerala PMT 2004]
A)
75% B and 25% C done
clear
B)
80% B and 20% C done
clear
C)
60% B and 40% C done
clear
D)
90% B and 10% C done
clear
E)
76.83% B and 23.17% C done
clear
View Solution play_arrow
