-
question_answer1)
Which of the following is not a condition for complex formation?
A)
Small atomic size done
clear
B)
High nuclear charge done
clear
C)
Variable oxidation states done
clear
D)
Availability of vacant d orbitals done
clear
View Solution play_arrow
-
question_answer2)
Zn gives \[{{H}_{2}}\] gas with \[{{H}_{2}}S{{O}_{4}}\] and \[HCl\] but not with \[HN{{O}_{3}}\] because
A)
Zn acts as an oxidising agent when react with\[HN{{O}_{3}}\]. done
clear
B)
\[HN{{O}_{3}}\] is weaker acid than \[{{H}_{2}}S{{O}_{4}}\] and\[HCl\]. done
clear
C)
In electrochemical series Zn is above hydrogen. done
clear
D)
\[NO_{3}^{-}\] ion is reduced in preference to hydroniumion. done
clear
View Solution play_arrow
-
question_answer3)
Which one of the following species is stable in aqueous solution?
A)
\[C{{r}^{2+}}\] done
clear
B)
\[MnO_{4}^{-}\] done
clear
C)
\[MnO_{4}^{3-}\] done
clear
D)
\[C{{u}^{+}}\] done
clear
View Solution play_arrow
-
question_answer4)
Which of the following is not correct about transition metals?
A)
Their melting and boiling points are high done
clear
B)
Their compounds are generally coloured done
clear
C)
They can form ionic or covalent compounds done
clear
D)
They do not exhibit variable valency done
clear
View Solution play_arrow
-
question_answer5)
Zinc and mercury do not show variable valency like d-block element because
A)
they are soft. done
clear
B)
their d-shells are complete. done
clear
C)
they have only two electrons in the outermost shell. done
clear
D)
their d-shells are incomplete. done
clear
View Solution play_arrow
-
question_answer6)
Which one of the following is diamagnetic ion?
A)
\[C{{o}^{2+}}\] done
clear
B)
\[C{{u}^{2+}}\] done
clear
C)
\[M{{n}^{2+}}\] done
clear
D)
\[S{{c}^{3+}}\] done
clear
View Solution play_arrow
-
question_answer7)
Which group contains coloured ions out of
| 1. \[C{{u}^{2+}}\] 2. \[T{{i}^{4+}}\] 3. \[C{{o}^{2+}}\] 4. \[F{{e}^{2+}}\] |
A)
1, 2, 3, 4 done
clear
B)
1, 3, 4 done
clear
C)
2, 3 done
clear
D)
1, 2 done
clear
View Solution play_arrow
-
question_answer8)
A compound of a metal ion \[{{M}^{x+}}(Z=24)\] has a spin only magnetic moment of \[\sqrt{15}\] Bohr Magnetons. The number of unpaired electrons in the compound are
A)
2 done
clear
B)
4 done
clear
C)
5 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer9)
Cuprous ion is colourless while cupric ion is coloured because
A)
both have half-filled p-and d-orbitals. done
clear
B)
cuprous ion has incomplete d-orbital and cupric ion has a complete d-orbital. done
clear
C)
both have unpaired electrons in the d-orbitals. done
clear
D)
cuprous ion has complete d-orbital and cupric ion has an incomplete d-orbital. done
clear
View Solution play_arrow
-
question_answer10)
Among the following, the compound that is both paramagnetic and coloured, is
A)
\[KMn{{O}_{4}}\] done
clear
B)
\[Cu{{F}_{4}}\] done
clear
C)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
D)
All are coloured done
clear
View Solution play_arrow
-
question_answer11)
Which of the following oxides of manganese is amphoteric?
A)
\[Mn{{O}_{2}}\] done
clear
B)
\[M{{n}_{2}}{{O}_{3}}\] done
clear
C)
\[M{{n}_{2}}{{O}_{7}}\] done
clear
D)
\[MnO\] done
clear
View Solution play_arrow
-
question_answer12)
In the laboratory, manganese (II) salt is oxidised to permanganate ion in aqueous solution by
A)
hydrogen peroxide done
clear
B)
cone. nitric acid done
clear
C)
peroxy disulphate done
clear
D)
dichromate done
clear
View Solution play_arrow
-
question_answer13)
When Cu reacts with \[AgN{{O}_{3}}\] solution, the reaction takes place is
A)
oxidation of Cu done
clear
B)
reduction of Cu done
clear
C)
oxidation of \[A{{g}^{+}}\] done
clear
D)
reduction of \[NO_{3}^{-}\] done
clear
View Solution play_arrow
-
question_answer14)
\[CuS{{O}_{4}}\] reacts with KCN solution and forms
A)
\[Cu\left( CN \right)\] done
clear
B)
\[Cu{{\left( CN \right)}_{2}}\] done
clear
C)
\[{{K}_{3}}[Cu{{\left( CN \right)}_{4}}]\] done
clear
D)
\[{{K}_{4}}[Cu{{(CN)}_{6}}]\] done
clear
View Solution play_arrow
-
question_answer15)
Which of the following compounds gives red precipitate with \[AgN{{O}_{3}}\]?
A)
KI done
clear
B)
\[{{K}_{2}}Cr{{O}_{4}}\] done
clear
C)
\[NaBr\] done
clear
D)
\[NaN{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer16)
When horn silver ore is dissolved in excess of sodium cyanide solution compound formed is
A)
\[AgCN\] done
clear
B)
\[Na\left[ Ag{{\left( CN \right)}_{2}} \right]\] done
clear
C)
\[N{{a}_{2}}\left[ Ag{{\left( CN \right)}_{3}} \right]\] done
clear
D)
\[N{{a}_{4}}\left[ Ag{{\left( CN \right)}_{5}} \right]\] done
clear
View Solution play_arrow
-
question_answer17)
In the equation
| \[4M+8C{{N}^{-}}+2{{H}_{2}}O+{{O}_{2}}\to \] \[4{{[M{{\left( CN \right)}_{2}}]}^{-}}+4O{{H}^{-}}\] |
| Identify the metalM. It is |
A)
copper done
clear
B)
iron done
clear
C)
gold done
clear
D)
zinc done
clear
View Solution play_arrow
-
question_answer18)
Mercury is the only metal which is liquid at \[0{}^\circ C\]. This is due to its
A)
very high ionisation energy and weak metallic bond. done
clear
B)
low ionisation potential. done
clear
C)
high atomic weight. done
clear
D)
high vapour pressure. done
clear
View Solution play_arrow
-
question_answer19)
If \[KMn{{O}_{4}}\] is reduced by oxalic acid in an acidic medium then oxidation number of Mn changes from
A)
4 to 2 done
clear
B)
6 to 4 done
clear
C)
+7 to+2 done
clear
D)
7 to 4 done
clear
View Solution play_arrow
-
question_answer20)
In the preparation of \[KMn{{O}_{4}}\] pyrolusite \[(Mn{{O}_{2}})\] is first converted to potassium manganite\[({{K}_{2}}Mn{{O}_{4}})\]. In this conversion, the oxidation state of manganese changes from
A)
+1 to +3 done
clear
B)
+2 to +4 done
clear
C)
+3 to +5 done
clear
D)
+4 to +6 done
clear
View Solution play_arrow
-
question_answer21)
In an alkaline condition \[KMn{{O}_{4}}\] reacts as follows: \[2KMn{{O}_{4}}+2KOH\to 2{{K}_{2}}Mn{{O}_{4}}+{{H}_{2}}O+O\] Its equivalent weight is
A)
31.6 done
clear
B)
52.7 done
clear
C)
49 done
clear
D)
158 done
clear
View Solution play_arrow
-
question_answer22)
In which of the following compounds manganese has oxidation number equal to that of iodine in\[KI{{O}_{4}}\]?
A)
Potassium manganite done
clear
B)
Potassium permanganate done
clear
C)
Manganous chloride done
clear
D)
Manganese chloride done
clear
View Solution play_arrow
-
question_answer23)
The colour of \[CuC{{r}_{2}}{{O}_{7}}\] solution in water is green because
A)
\[C{{r}_{2}}O_{7}^{2-}\] ions are green. done
clear
B)
\[C{{u}^{2+}}\] ions are green. done
clear
C)
Both \[C{{r}_{2}}O_{7}^{2-}\] and \[C{{u}^{2+}}\] ions are green. done
clear
D)
\[C{{u}^{2+}}\] ions are blue and \[C{{r}_{2}}O_{7}^{2-}\]ions are yellow. done
clear
View Solution play_arrow
-
question_answer24)
The correct order of magnetic moments (spin only values in B.M.) among is
(Atomic nos.: Mn = 25, Fe = 26, Co = 27)
A)
\[{{\left[ Fe{{\left( CN \right)}_{6}} \right]}^{4-}}>{{\left[ MnC{{l}_{4}} \right]}^{2}}^{-}>{{\left[ CoC{{l}_{4}} \right]}^{2-}}\] done
clear
B)
\[{{[MnC{{l}_{4}}]}^{2-}}>{{[Fe{{(CN)}_{6}}]}^{4-}}>{{[CoC{{l}_{4}}]}^{2-}}\] done
clear
C)
\[{{[MnC{{l}_{4}}]}^{2-}}>{{[Co{{(Cl)}_{4}}]}^{2-}}>{{[Fe{{(CN)}_{6}}]}^{4-}}\] done
clear
D)
\[{{[Fe{{(CN)}_{6}}]}^{4-}}>{{[Co{{(Cl)}_{4}}]}^{2-}}>{{[MnC{{l}_{4}}]}^{2-}}\] done
clear
View Solution play_arrow
-
question_answer25)
Out of \[{{[Ti{{F}_{6}}]}^{2-}},{{[Co{{F}_{6}}]}^{3-}},C{{u}_{2}}C{{l}_{2}}\] and \[{{[NiC{{l}_{4}}]}^{2-}}\]of (Z of Ti = 22, Co = 27, Cu = 29, Ni = 28), the colourless species are:
A)
\[C{{u}_{2}}C{{l}_{2}}\,\]and \[{{[NiC{{l}_{4}}]}^{2-}}\] done
clear
B)
\[{{[Ti{{F}_{6}}]}^{2-}}\] and \[C{{u}_{2}}C{{l}_{2}}\,\] done
clear
C)
\[{{[Co{{F}_{6}}]}^{3-}}\] and \[{{[NiC{{l}_{4}}]}^{2-}}\] done
clear
D)
\[{{[Ti{{F}_{6}}]}^{2-}}\] and \[{{[Co{{F}_{6}}]}^{3-}}\] done
clear
View Solution play_arrow
-
question_answer26)
The basic character of the transition metal monoxides follows the order (Atomic Nos., Ti = 22, V = 23, Cr = 24, Fe = 26)
A)
\[TiO>VO>CrO>FeO\] done
clear
B)
\[VO>CrO>TiO>FeO\] done
clear
C)
\[CrO>VO>FeO>TiO\] done
clear
D)
\[TiO>FeO>VO>CrO\] done
clear
View Solution play_arrow
-
question_answer27)
Which of the following statement is incorrect?
A)
Across a period from Sc to Cu the densities increase with increasing atomic number. done
clear
B)
The melting point of transition elements rise to a maximum from Sc to Cr and then decreases from Fe to Zn. done
clear
C)
Transition elements have high enthalpies of atomization and in 3d series increase regularly from Sc to Cu. done
clear
D)
On going down a group from 3d to 6d series the stability of higher oxidation state increases with increasing atomic number. done
clear
View Solution play_arrow
-
question_answer28)
On the basis of data given below,
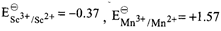 |
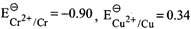 |
| Which of the following statements is incorrect? |
A)
\[S{{c}^{3+}}\] has good stability due of \[\left[ Ar \right]3{{d}^{0}}4{{s}^{0}}\] configuration. done
clear
B)
\[M{{n}^{3+}}\] is more stable than \[M{{n}^{2+}}\]. done
clear
C)
\[C{{r}^{2+}}\] is reducing in nature. done
clear
D)
Copper does not give \[{{H}_{2}}\] on reaction with dil. \[{{H}_{2}}S{{O}_{4}}\]. done
clear
View Solution play_arrow
-
question_answer29)
A compound of iron exists as a dimer in vapour state. It is hygroscopic in nature and dissolves in water giving brown acidic solution. The compound is
A)
\[F{{e}_{3}}{{O}_{4}}\] done
clear
B)
\[FeS{{O}_{4}}\] done
clear
C)
\[FeC{{l}_{3}}\] done
clear
D)
\[Fe{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer30)
A metal M and its compound can give the following observable changes in a consequence of reactions

A)
Mg done
clear
B)
Pb done
clear
C)
Zn done
clear
D)
Sn done
clear
View Solution play_arrow
-
question_answer31)
| \[\left( X \right)+{{K}_{2}}C{{O}_{3}}+Air\xrightarrow{heat}\left( Y \right)\] |
| \[\left( Y \right)+C{{l}_{2}}\xrightarrow{{}}\left( Z \right)Pink\] |
| Which of the following is correct? |
A)
\[X=black,\text{ }Mn{{O}_{2}},Y=Blue,\] \[{{K}_{2}}Cr{{O}_{4}},Z=KMn{{O}_{4}}\] done
clear
B)
\[X=green,\text{ }C{{r}_{2}}{{O}_{3}},Y=Yellow,\] \[{{K}_{2}}Cr{{O}_{4}},\,\,Z={{K}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
C)
\[X=black,\text{ }Mn{{O}_{2}}\text{, }Y=green,\] \[{{K}_{2}}Mn{{O}_{4}},Z=KMn{{O}_{4}}\] done
clear
D)
\[X=black,B{{i}_{2}}{{O}_{3}},\,\,Y=colourless\] \[KBi{{O}_{2}},Z=KBi{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer32)
\[CuS{{O}_{4}}(aq.)\xrightarrow{{{H}_{2}}S\uparrow }M\downarrow \xrightarrow[of\,KCN]{Excess}N+O\] Then final products N and O are respectively.
A)
\[{{[Cu{{(CN)}_{4}}]}^{3-}},{{(CN)}_{2}}\] done
clear
B)
\[CuCN,{{(CN)}_{2}}\] done
clear
C)
\[{{[Cu{{(CN)}_{4}}]}^{2-}},{{(CN)}_{2}}\] done
clear
D)
\[Cu{{\left( CN \right)}_{2}},{{K}_{2}}S\] done
clear
View Solution play_arrow
-
question_answer33)
Which of the following statements is false?
A)
\[N{{a}_{2}}C{{r}_{2}}{{O}_{7}}\] is less soluble than \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]. done
clear
B)
\[N{{a}_{2}}C{{r}_{2}}{{O}_{7}}\] is primary standard in volumetry. done
clear
C)
\[CrO{{_{4}^{2-}}^{~}}\] is tetrahedral in shape. done
clear
D)
\[C{{r}_{2}}{{O}_{7}}^{2-}\] has a \[Cr-O-Cr\] bond. done
clear
View Solution play_arrow
-
question_answer34)
A red solid is insoluble in water. However it becomes soluble if some KI is added to water. Heating the red solid in a test tube results in liberation of some violet coloured fumes and droplets of a metal appear on the cooler parts of the test tube. The red solid is
A)
\[Hg{{I}_{2}}\] done
clear
B)
\[HgO\] done
clear
C)
\[P{{b}_{3}}{{O}_{4}}\] done
clear
D)
\[{{(N{{H}_{4}})}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
View Solution play_arrow
-
question_answer35)
Of Cr (VI) as \[C{{r}_{2}}O{{_{7}^{2-}}^{~}}\] and \[CrO{{_{4}^{2-}}^{~}}\], which is better oxidising agent?
A)
\[Cr{{O}_{4}}^{2-}\], basic medium done
clear
B)
\[C{{r}_{2}}{{O}_{7}}^{2-}\], basic medium done
clear
C)
\[C{{r}_{2}}{{O}_{7}}^{2-}\], acidic medium done
clear
D)
\[Cr{{O}_{3}}\] basic medium done
clear
View Solution play_arrow
-
question_answer36)
A blue colouration is not obtained when
A)
ammonium hydroxide dissolves in copper sulphate. done
clear
B)
copper sulphate solution reacts with \[{{K}_{4}}\left[ Fe{{\left( CN \right)}_{6}} \right].\] done
clear
C)
ferric chloride reacts with sod. ferrocyanide. done
clear
D)
anhydrous \[CuS{{O}_{4}}\] is dissolved in water. done
clear
View Solution play_arrow
-
question_answer37)
Which of the statements is not true?
A)
On passing \[{{H}_{2}}S\] through acidified \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] solution, a milky colour is observed. done
clear
B)
\[N{{a}_{2}}C{{r}_{2}}{{O}_{7}}\] is preferred over \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] in volumetric analysis. done
clear
C)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]solution in acidic medium is orange. done
clear
D)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]solution becomes yellow on increasing the pH beyond 7. done
clear
View Solution play_arrow
-
question_answer38)
The blue complex formed on addition of cone. \[N{{H}_{4}}OH\] solution to a \[C{{u}^{2+}}\] salt solution has the structure?
A)
\[{{[Cu{{(N{{H}_{4}})}_{4}}]}^{2+}}\] done
clear
B)
\[{{[Cu{{(N{{H}_{3}})}_{2}}]}^{2+}}\] done
clear
C)
\[{{[Cu{{(N{{H}_{3}})}_{4}}]}^{2+}}\] done
clear
D)
\[{{[Cu{{(N{{H}_{4}})}_{2}}]}^{2+}}\] done
clear
View Solution play_arrow
-
question_answer39)
Identify the incorrect statement
A)
\[CuS{{O}_{4}}\] reacts with \[KCl\] in aqueous solution to give \[C{{u}_{2}}C{{l}_{2}}\]. done
clear
B)
\[CuS{{O}_{4}}\]reacts with KI in aqueous solution to give \[C{{u}_{2}}{{I}_{2}}\]. done
clear
C)
\[CuS{{O}_{4}}\] react with \[NaOH\] and glucose in aqueous medium to give \[C{{u}_{2}}O\]. done
clear
D)
\[CuS{{O}_{4}}\] on strong heating gives CuO. done
clear
View Solution play_arrow
-
question_answer40)
The black compound formed during the reaction between sodium thiosulphate and silver nitrate is
A)
silver thiosulphate \[(A{{g}_{2}}{{S}_{2}}{{O}_{3}})\] done
clear
B)
silver sulphide \[(A{{g}_{2}}{{S}_{2}})\] done
clear
C)
silver sulphate \[(A{{g}_{2}}S{{O}_{4}})\] done
clear
D)
silver sulphite \[(A{{g}_{2}}S{{O}_{3}})\] done
clear
View Solution play_arrow
-
question_answer41)
When \[KMn{{O}_{4}}\] is added to oxalic acid, the decolourisation is slow in the beginning but becomes instantaneous after sometime because
A)
\[M{{n}^{2+}}\] acts as autocatalyst. done
clear
B)
\[C{{O}_{2}}\] is formed as the product. done
clear
C)
Reaction is exothermic. done
clear
D)
\[MnO_{4}^{-}\] catalyses the reaction. done
clear
View Solution play_arrow
-
question_answer42)
Number of electrons transfered in each case when \[KMn{{O}_{4}}\] acts as an oxidising agent to give \[Mn{{O}_{2}},M{{n}^{2+}},Mn{{(OH)}_{3}}\] and \[MnO_{4}^{2-}\] are respectively
A)
3, 5, 4 and 1 done
clear
B)
4, 3, 1 and 5 done
clear
C)
1, 3, 4 and 5 done
clear
D)
5, 4, 3 and 1 done
clear
View Solution play_arrow
-
question_answer43)
Chloro compound of Vanadium has only spin magnetic moment of 1.73 BM. This Vanadium chloride has the formula:
A)
\[VC{{l}_{2}}\] done
clear
B)
\[VC{{l}_{4}}\] done
clear
C)
\[VC{{l}_{3}}\] done
clear
D)
\[VC{{l}_{5}}\] done
clear
View Solution play_arrow
-
question_answer44)
Which pair of elements with the given atomic numbers is expected to have similar properties?
A)
40, 72 done
clear
B)
20, 36 done
clear
C)
10, 28 done
clear
D)
11, 12 done
clear
View Solution play_arrow
-
question_answer45)
When concentrated \[HCl\] is added to an aqueous solution of \[CoC{{l}_{2}}\] its colour changes from reddish pink to deep blue. Which complex ion gives blue colour in this reaction?
A)
\[{{[CoC{{l}_{4}}]}^{2-}}\] done
clear
B)
\[{{[CoC{{l}_{6}}]}^{3-}}\] done
clear
C)
\[{{[CoC{{l}_{6}}]}^{4-}}\] done
clear
D)
\[{{[Co{{({{H}_{2}}O)}_{6}}]}^{2+}}\] done
clear
View Solution play_arrow
-
question_answer46)
Which of the following is not formed when \[{{H}_{2}}S\] reacts with acidic \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] solution?
A)
\[CrS{{O}_{4}}\] done
clear
B)
\[C{{r}_{2}}{{(S{{O}_{4}})}_{3}}\] done
clear
C)
\[{{K}_{2}}S{{O}_{4}}\] done
clear
D)
S done
clear
View Solution play_arrow
-
question_answer47)
Potassium dichromate when heated with concentrated sulphuric acid and a soluble chloride, gives brown-red vapours of:
A)
\[Cr{{O}_{3}}\] done
clear
B)
\[CrC{{l}_{3}}\] done
clear
C)
\[Cr{{O}_{2}}C{{l}_{2}}\] done
clear
D)
\[C{{r}_{2}}{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer48)
When a small amount of \[KMn{{O}_{4}}\] is added to concentrated \[{{H}_{2}}S{{O}_{4}}\] a green oily compound is obtained which is highly explosive in nature. Compound may be:
A)
\[MnS{{O}_{4}}\] done
clear
B)
\[M{{n}_{2}}{{\text{O}}_{7}}\] done
clear
C)
\[Mn{{O}_{2}}\] done
clear
D)
\[M{{n}_{2}}{{\text{O}}_{3}}\] done
clear
View Solution play_arrow
-
question_answer49)
Identify incorrect statement:
A)
\[C{{u}_{2}}O\] is colourless. done
clear
B)
Copper (I) compounds are colourless except when colour results from charge transfer. done
clear
C)
Copper (I) compounds are diamagnetic. done
clear
D)
\[C{{u}_{2}}S\] is black. done
clear
View Solution play_arrow
-
question_answer50)
Which of the following compounds has colour but no unpaired electrons?
A)
\[KMn{{O}_{4}}\] done
clear
B)
\[{{K}_{2}}Mn{{O}_{4}}\] done
clear
C)
\[MnS{{O}_{4}}\] done
clear
D)
\[MnC{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer51)
Which one of the following exhibits the large number of oxidation states?
A)
Ti (22) done
clear
B)
V (23) done
clear
C)
Cr (24) done
clear
D)
Mn (25) done
clear
View Solution play_arrow
-
question_answer52)
The element with which of the following outer electron configuration may exhibit the largest number of oxidation states in its compounds:
A)
\[3{{d}^{5}}4{{s}^{2}}\] done
clear
B)
\[3{{d}^{8}}4{{s}^{2}}\] done
clear
C)
\[3{{d}^{7}}4{{s}^{2}}\] done
clear
D)
\[3{{d}^{6}}4{{s}^{2}}\] done
clear
View Solution play_arrow
-
question_answer53)
Because of lanthanoid contraction, which of the following pairs of elements have nearly same atomic radii? (Numbers in the parenthesis are atomic numbers).
A)
\[Zr\left( 40 \right)\] and \[Nb\left( 41 \right)\] done
clear
B)
\[Zr\left( 40 \right)\] and\[Hf\left( 72 \right)\] done
clear
C)
\[Zr\left( 40 \right)\] and \[Ta\left( 73 \right)\] done
clear
D)
\[Ti\left( 22 \right)\] and \[Zr\left( 40 \right)\] done
clear
View Solution play_arrow
-
question_answer54)
Which is not the correct statement? (At. nos. Ce = 58, Lu = 71, La = 57, Yb= 70)
A)
Colour of \[Y{{b}^{3+}}\] ion is pink. done
clear
B)
\[L{{a}^{3+}}\] is diamagnetic. done
clear
C)
\[C{{e}^{4+}}\] has \[{{f}^{0}}\] configuration. done
clear
D)
\[L{{a}^{3+}}\]had \[{{f}^{14}}\] configuration. done
clear
View Solution play_arrow
-
question_answer55)
Magnetic moment of \[G{{d}^{3+}}\] ion (Z = 64) is
A)
3.62 BM done
clear
B)
9.72 BM done
clear
C)
7.9 BM done
clear
D)
10.60 BM done
clear
View Solution play_arrow
-
question_answer56)
Which of the following forms stable + 4 oxidation state?
A)
La (Z=57) done
clear
B)
Eu (Z=63) done
clear
C)
Ce (Z=58) done
clear
D)
Gd (Z=64) done
clear
View Solution play_arrow
-
question_answer57)
The number of unpaired electrons in Gadolinium [Z=64] is
A)
3 done
clear
B)
8 done
clear
C)
6 done
clear
D)
2 done
clear
View Solution play_arrow
-
question_answer58)
Which one of the following elements shows maximum number of different oxidation states in its compounds?
A)
Eu done
clear
B)
La done
clear
C)
Gd done
clear
D)
Am done
clear
View Solution play_arrow
-
question_answer59)
The radius of \[L{{a}^{3+}}\] (Atomic number of La = 57) is \[1.06\overset{\text{o}}{\mathop{\text{A}}}\,\]. Which one of the following given values will be closest to the radius of \[L{{a}^{3+}}\] (Atomic number of Lu= 71)?
A)
\[1.40\overset{\text{o}}{\mathop{\text{A}}}\,\] done
clear
B)
\[1.06\overset{\text{o}}{\mathop{\text{A}}}\,\] done
clear
C)
\[0.85\overset{\text{o}}{\mathop{\text{A}}}\,\] done
clear
D)
\[1.60\overset{\text{o}}{\mathop{\text{A}}}\,\] done
clear
View Solution play_arrow
-
question_answer60)
The electronic configuration of gadolinium (At. No. 64) is
A)
\[[Xe]4{{f}^{8}}5{{d}^{1}}6{{s}^{2}}\] done
clear
B)
\[[Xe]4{{f}^{7}}5{{d}^{1}}6{{s}^{2}}\] done
clear
C)
\[[Xe]4{{f}^{3}}5{{d}^{5}}6{{s}^{2}}\] done
clear
D)
\[[Xe]4{{f}^{6}}5{{d}^{2}}6{{s}^{2}}\] done
clear
View Solution play_arrow
-
question_answer61)
In which of the following lanthanoids oxidation state +2 is most stable?
A)
Ce done
clear
B)
Eu done
clear
C)
Tb done
clear
D)
Dy done
clear
View Solution play_arrow
-
question_answer62)
Which of the following lanthanoid ions is diamagnetic? (At nos. Ce = 58, Sm = 62, Eu = 63, Yb = 70)
A)
\[S{{m}^{2+}}\] done
clear
B)
\[E{{u}^{2+}}\] done
clear
C)
\[Y{{b}^{2+}}\] done
clear
D)
\[C{{e}^{2+}}\] done
clear
View Solution play_arrow
-
question_answer63)
Lanthanoid contraction can be observed in
A)
At done
clear
B)
Gd done
clear
C)
Ac done
clear
D)
Lw done
clear
View Solution play_arrow
-
question_answer64)
Lanthanoides and actinoides resemble in
A)
Electronic configuration done
clear
B)
Oxidation state done
clear
C)
lonization energy done
clear
D)
Formation of complexes done
clear
View Solution play_arrow
-
question_answer65)
The Ce (Z = 58) belongs to IIIrd group of periodic table. If it furnish one \[\alpha \] particle to form an element 'X', then X belongs to
A)
IIIrd group done
clear
B)
IInd group done
clear
C)
Ist group done
clear
D)
zero group done
clear
View Solution play_arrow
-
question_answer66)
In aqueous solutions \[E{{u}^{2+}}\] acts as
A)
an oxidising agent. done
clear
B)
a reducing agent. done
clear
C)
can act either of these. done
clear
D)
can act as redox agent. done
clear
View Solution play_arrow
-
question_answer67)
Arrange (I) \[C{{e}^{3+}}\], (II) \[L{{a}^{3+}}\], (III) \[P{{m}^{3+}}\], and (IV) \[Y{{b}^{3+}}\] in increasing order of their ionic radii.
A)
IV<III<I<II done
clear
B)
I<IV<III<II done
clear
C)
IV<III<II<I done
clear
D)
III<II<I<IV done
clear
View Solution play_arrow
-
question_answer68)
Which of the following statement is not correct?
A)
\[La{{\left( OH \right)}_{3}}\] is less basic than \[Lu{{\left( OH \right)}_{3}}\]. done
clear
B)
In lanthanide series ionic radius of \[L{{n}^{3+}}\] ions decreases. done
clear
C)
La is actually an element of transition series rather than lanthanide series. done
clear
D)
Atomic radii of Zr and Hf are same because of lanthanide contraction. done
clear
View Solution play_arrow
-
question_answer69)
The reason for the stability of \[G{{d}^{3+}}\] ion is
A)
4f subshell - half filled. done
clear
B)
4f subshell - completely filled. done
clear
C)
Possesses the general electronic configuration of noble gases. done
clear
D)
4f-subshell empty. done
clear
View Solution play_arrow
-
question_answer70)
Knowing that the chemistry of lanthanoids(Ln) is dominated by its + 3 oxidation state, which of the following statements is incorrect?
A)
The ionic size of Ln (III) decrease in general with increasing atomic number. done
clear
B)
Ln (III) compounds are generally colourless. done
clear
C)
Ln (III) hydroxide are mainly basic in character. done
clear
D)
Because of the large size of the Ln (III) ions the bonding in its compounds is predominantly ionic in character. done
clear
View Solution play_arrow
![]()
![]()
