question_answer 1) Tail of comet is because of
A)
sublimation of vapour in comet
done
clear
B)
vaporisation of water in comet
done
clear
C)
vaporisation of heat in comet
done
clear
D)
cooling of water in the comet
done
clear
View Answer play_arrow
question_answer 2) The energy required to excite an electron from the ground state of hydrogen atom to the First excited state will be
A)
\[1.632\times {{10}^{-18}}\,\,V\]
done
clear
B)
\[1.612\times {{10}^{-14}}\,\,V\]
done
clear
C)
\[1.619\times {{10}^{-16}}\,\,V\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 3) Magnification of least distance of distinct vision of a simple microscope of focal length \[5\,\,cm\], will be
A)
6
done
clear
B)
5
done
clear
C)
4
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 4) The resistance of an ideal voltmeter is
A)
\[500\Omega \]
done
clear
B)
\[100\Omega \]
done
clear
C)
infinity
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 5) Two inductors each of inductance \[L\] are connected in parallel. Their equivalent inductance will be
A)
zero
done
clear
B)
\[\frac{L}{2}\]
done
clear
C)
\[2\,\,L\]
done
clear
D)
\[L\]
done
clear
View Answer play_arrow
question_answer 6) If an electron is brought towards another electron, then electric potential of the system
A)
remains same
done
clear
B)
becomes zero
done
clear
C)
decrease
done
clear
D)
increases
done
clear
View Answer play_arrow
question_answer 7) Velocity of sound in air is
A)
directly proportional to temperature
done
clear
B)
faster in dry air than in moisture
done
clear
C)
directly proportional to pressure
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 8) A cylinder contains \[10\,\,kg\] of gas at pressure of\[10\,\,N/{{m}^{2}}\]. The quantity of gas taken out of the cylinder, if final pressure is \[2.5\times {{10}^{6}}\,\,N/{{m}^{2}}\] will be (temperature of gas is constant)
A)
\[15.2\,\,kg\]
done
clear
B)
\[3.7\,\,kg\]
done
clear
C)
\[zero\]
done
clear
D)
\[7.5\,\,kg\]
done
clear
View Answer play_arrow
question_answer 9) Moment of inertia of a thin circular disc of mass \[M\] and radius \[R\] about any diameter is
A)
\[2M{{R}^{2}}\]
done
clear
B)
\[M{{R}^{2}}\]
done
clear
C)
\[\frac{M{{R}^{2}}}{2}\]
done
clear
D)
\[\frac{M{{R}^{2}}}{4}\]
done
clear
View Answer play_arrow
question_answer 10) Velocity of a body on reaching the point from which it was projected upward, will be
A)
\[v=u\]
done
clear
B)
\[v=0.5\,\,u\]
done
clear
C)
\[v=0\]
done
clear
D)
\[v=2u\]
done
clear
View Answer play_arrow
question_answer 11) Retrograde means
A)
west to east rotation of all planets
done
clear
B)
east to west rotation of venus
done
clear
C)
east to west rotation of Uranus
done
clear
D)
both [b] and [c]
done
clear
View Answer play_arrow
question_answer 12) Number of electrons, having one coulomb of charge is
A)
\[6.25\times {{10}^{18}}\]
done
clear
B)
\[5.26\times {{10}^{28}}\]
done
clear
C)
\[1.6\times {{10}^{19}}\]
done
clear
D)
\[1.6\times {{10}^{21}}\]
done
clear
View Answer play_arrow
question_answer 13) A plane convex lens is made of refractive index\[1.6\]. The radius of curvature of the curved surface is\[60\,\,cm\]. Then, the focal length of the lens will be
A)
\[400\,\,cm\]
done
clear
B)
\[200\,\,cm\]
done
clear
C)
\[100\,\,cm\]
done
clear
D)
\[50\,\,cm\]
done
clear
View Answer play_arrow
question_answer 14) If the magnet of length \[10\,\,cm\] and pole strength \[40\,\,Am\] is placed at an angle of \[{{45}^{o}}\] in an uniform induction field of intensity \[2\times {{10}^{-4}}\]tesla. The couple acting on it will be
A)
\[0.656\times {{10}^{-5}}\,\,N\text{-}m\]
done
clear
B)
\[0.656\times {{10}^{-4}}\,\,N\text{-}m\]
done
clear
C)
\[0.5656\times {{10}^{-4}}\,\,N\text{-}m\]
done
clear
D)
\[0.5656\times {{10}^{-3}}\,\,N\text{-}m\]
done
clear
View Answer play_arrow
question_answer 15) Charge \[{{q}_{2}}\] is at the centre of a circular path of radius\[r\]. Work done in carrying charge \[{{q}_{1}}\], one around this equipotential path, would be
A)
\[66\]
done
clear
B)
\[44\]
done
clear
C)
\[22\]
done
clear
D)
\[zero\]
done
clear
View Answer play_arrow
question_answer 16) When a dielectric material is introduced between plates of a charged condenser, then electric Field between the plates.
A)
remain constant
done
clear
B)
decreases
done
clear
C)
increases
done
clear
D)
first (c) then (b)
done
clear
View Answer play_arrow
question_answer 17) The frequency of rod is\[200\,\,Hz\]. If the velocity of sound in air is\[340\,\,m/s\]. Then the wavelength of the produced sound is
A)
\[6.8\,\,cm\]
done
clear
B)
\[1.7\,\,cm\]
done
clear
C)
\[6.8\,\,cm\]
done
clear
D)
\[1.7\,\,m\]
done
clear
View Answer play_arrow
question_answer 18) Density of substance at \[{{0}^{o}}C\] is \[10\,\,g/cc\] and at\[{{100}^{o}}C\], its density is\[9.7\,\,g/cc\]. The coefficient of linear expansion of the substance will be
A)
\[{{10}^{2}}\]
done
clear
B)
\[{{10}^{-2}}\]
done
clear
C)
\[{{10}^{-3}}\]
done
clear
D)
\[{{10}^{-4}}\]
done
clear
View Answer play_arrow
question_answer 19) A body of mass \[5\,\,kg\] moving with a velocity \[10\,\,m/s\] collides with another body of the mass \[20\,\,kg\] at rest and conies to rest. The velocity of second due to collision will be
A)
\[10\,\,m/s\]
done
clear
B)
\[7.5\,\,m/s\]
done
clear
C)
\[2.5\,\,m/s\]
done
clear
D)
\[5\,\,m/s\]
done
clear
View Answer play_arrow
question_answer 20) The angular speed of a fly wheel making 180 revolutions/minute will be
A)
\[6\,\pi \,\,rad/\sec \]
done
clear
B)
\[12\,\pi \,\,rad/\sec \]
done
clear
C)
\[2\,\,\pi \,\,rad/\sec \]
done
clear
D)
\[\pi \,\,rad/\sec \]
done
clear
View Answer play_arrow
question_answer 21) In case of a semi-conductor, which of the following statement is wrong?
A)
At absolute zero temperature, it behave like a conductor
done
clear
B)
Resistivity is in between that of a conductor and insulator.
done
clear
C)
Doping increases conductivity
done
clear
D)
Temperature coefficient of resistance is negative
done
clear
View Answer play_arrow
question_answer 22) The \[X-\]ray cannot be diffracted by means of ordinary grating due to
A)
large wavelength
done
clear
B)
short wavelength
done
clear
C)
high speed
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 23) The time taken by the light to cross a glass of thickness \[4\,\,mm\] and refractive index\[\mu =3\], is
A)
\[4\times {{10}^{-11}}\sec \]
done
clear
B)
\[8\times {{10}^{-10}}\sec \]
done
clear
C)
\[2\times {{10}^{-11}}\sec \]
done
clear
D)
\[1\times {{10}^{-11}}\sec \]
done
clear
View Answer play_arrow
question_answer 24) The radius of circular path of an electron when subjected to a perpendicular magnetic field, will be
A)
\[\frac{mv}{Be}\]
done
clear
B)
\[\frac{me}{B}\]
done
clear
C)
\[\frac{mE}{B}\]
done
clear
D)
\[\frac{Be}{mv}\]
done
clear
View Answer play_arrow
question_answer 25) Three capacitors of capacitance\[3\mu F\], \[10\mu F\] and \[15\mu F\] are joined in series to a voltage source of\[100\,\,V\]. The charge on \[15\mu F\] will be
A)
\[280\]
done
clear
B)
\[50\,\,\mu C\]
done
clear
C)
\[100\,\,\mu C\]
done
clear
D)
\[200\,\,\mu C\]
done
clear
View Answer play_arrow
question_answer 26) Electromotive force is the force which is able to maintain a constant
A)
potential difference
done
clear
B)
power
done
clear
C)
current
done
clear
D)
resistance
done
clear
View Answer play_arrow
question_answer 27) Two waves are approaching each other with a velocity of \[20\,\,m/s\] and frequency\[n\], then the distance between two consecutive nodes will be
A)
\[\frac{10}{n}\]
done
clear
B)
\[\frac{5}{n}\]
done
clear
C)
\[\frac{n}{5}\]
done
clear
D)
\[\frac{20}{n}\]
done
clear
View Answer play_arrow
question_answer 28) In order that the heat flow from one part of a solid to another part, the required property is
A)
uniform temperature
done
clear
B)
temperature gradient
done
clear
C)
density of radiant
done
clear
D)
density gradient
done
clear
View Answer play_arrow
question_answer 29) If the momentum of the body is increased\[100%\]. Then the percentage increase in the kinetic energy will be
A)
\[300%\]
done
clear
B)
\[150%\]
done
clear
C)
\[75%\]
done
clear
D)
\[200%\]
done
clear
View Answer play_arrow
question_answer 30) If\[|\overset{\to }{\mathop{A}}\,\times \overset{\to }{\mathop{B}}\,|=|\overset{\to }{\mathop{A}}\,.\overset{\to }{\mathop{B}}\,|\]then the angle between\[\overset{\to }{\mathop{A}}\,\]and\[\overset{\to }{\mathop{B}}\,\]is
A)
\[{{90}^{o}}\]
done
clear
B)
\[{{60}^{o}}\]
done
clear
C)
\[{{45}^{o}}\]
done
clear
D)
\[{{30}^{o}}\]
done
clear
View Answer play_arrow
question_answer 31) The potential barrier in the depletion layer is because of
A)
electrons
done
clear
B)
ions
done
clear
C)
holes
done
clear
D)
both (a) and (c)
done
clear
View Answer play_arrow
question_answer 32) A tank is filled with benzene to a height of\[120\,\,mm\]. The apparent depth of a needle lying at a bottom of the tank is measured by a microscope to be\[80\,\,mm\]. Then the refractive index of benzene will be
A)
\[4.5\]
done
clear
B)
\[3\]
done
clear
C)
\[.75\]
done
clear
D)
\[1.5\]
done
clear
View Answer play_arrow
question_answer 33) In an apparatus, the electric field was found to oscillate with an amplitude of\[18\,\,V/m\]. The magnitude of the oscillating magnetic field is
A)
\[6\times {{10}^{-8}}tesla\]
done
clear
B)
\[3\times {{10}^{-8}}tesla\]
done
clear
C)
\[12\times {{10}^{-8}}tesla\]
done
clear
D)
\[9\times {{10}^{-8}}tesla\]
done
clear
View Answer play_arrow
question_answer 34) The intensity of magnetic field is \[H\] and moment of magnet is\[M\]. Then the maximum potential energy will be
A)
\[4MH\]
done
clear
B)
\[3MH\]
done
clear
C)
\[2MH\]
done
clear
D)
\[MH\]
done
clear
View Answer play_arrow
question_answer 35) There are six identical cells each cell having an electromotive force (emf) of\[6\,\,volt\], which are joined in parallel. Then the resultant emf of the combination will be
A)
\[13\,\,V\]
done
clear
B)
\[\frac{1}{6}V\]
done
clear
C)
\[12\,\,V\]
done
clear
D)
\[6\,\,V\]
done
clear
View Answer play_arrow
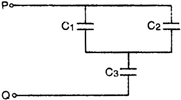
question_answer 36)
In a given network capacitor \[{{C}_{1}}=10\,\,\mu F,\,\,{{C}_{2}}=\mu F,\], and\[{{C}_{3}}=4\,\,\mu F\]. The equivalent capacitance between \[PQ\] will be
A)
\[1.6\,\,\mu F\]
done
clear
B)
\[3.2\,\,\mu F\]
done
clear
C)
\[6.4\,\,\mu F\]
done
clear
D)
\[4.5\,\,\mu F\]
done
clear
View Answer play_arrow
question_answer 37) A tuning fork produces wave in a medium. If the temperature of the medium change, then which of the following will change?
A)
Time period
done
clear
B)
Amplitude
done
clear
C)
Frequency
done
clear
D)
Wavelength
done
clear
View Answer play_arrow
question_answer 38) A hydraulic lift is designed to lift cars of maximum mass of\[3000\,\,kg\]. The area of cross section of the piston carrying the load is\[4.25\times {{10}^{-2}}{{m}^{2}}\]. Then, the maximum pressure would the smaller piston have to bear, is
A)
\[7.82\times {{10}^{7}}\,\,N/{{m}^{2}}\]
done
clear
B)
\[6.92\times {{10}^{5}}\,\,N/{{m}^{2}}\]
done
clear
C)
\[13.76\times {{10}^{5}}\,\,N/{{m}^{2}}\]
done
clear
D)
\[9.63\,\,N/{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 39) If the mass of the earth is \[80\] times of that of planet and diameter is double that of planet and \[g\] on earth\[9.8\,\,m/{{s}^{2}}\]. Then, the value of \[g\] on the planet will be
A)
\[0.49\,\,m/{{s}^{2}}\]
done
clear
B)
\[4.9\,\,m/{{s}^{2}}\]
done
clear
C)
\[0.98\,\,m/{{s}^{2}}\]
done
clear
D)
\[9.8\,\,m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 40) The number of significant figures in \[5418000\] are
A)
\[7\]
done
clear
B)
\[6\]
done
clear
C)
\[5\]
done
clear
D)
\[4\]
done
clear
View Answer play_arrow
question_answer 41) According to Moseleys law, frequency of a spectral line in \[X-\]rays spectrum varies as
A)
fourth power of the atomic number of the element
done
clear
B)
square root of atomic number of the element
done
clear
C)
square of the atomic number of the element
done
clear
D)
atomic number of the element
done
clear
View Answer play_arrow
question_answer 42) If in Rams dens eye piece, the field lens and eye lens have focal length \[{{f}_{1}}\] and \[{{f}_{2}}\] respectively and separated by a distance \[d\] then
A)
\[{{f}_{1}}={{f}_{2}}\]and\[d=\frac{2}{3}{{f}_{1}}\]
done
clear
B)
\[{{f}_{1}}=3{{f}_{2}}\]and\[d={{f}_{1}}+{{f}_{2}}\]
done
clear
C)
\[{{f}_{1}}={{f}_{2}}\]and\[d={{f}_{1}}+{{f}_{2}}\]
done
clear
D)
\[{{f}_{1}}=\frac{2}{3}{{f}_{2}}\]and\[d=\frac{2}{3}{{f}_{1}}\]
done
clear
View Answer play_arrow
question_answer 43) The magnetic material, which moves from stronger to weaker part of the magnetic field, then it is known as
A)
anti-ferromagnetic
done
clear
B)
ferromagnetic
done
clear
C)
paramagnetic
done
clear
D)
diamagnetic
done
clear
View Answer play_arrow
question_answer 44) The magnetic field along axis at a point at a distance d of a short, bar magnet will be
A)
\[\frac{{{\mu }_{0}}}{3\pi }\times \frac{M}{{{d}^{3}}}\]
done
clear
B)
\[\frac{{{\mu }_{0}}}{\pi }\times \frac{M}{{{d}^{3}}}\]
done
clear
C)
\[\frac{{{\mu }_{0}}}{2\pi }\times \frac{M}{{{d}^{3}}}\]
done
clear
D)
\[\frac{{{\mu }_{0}}}{4\pi }\times \frac{M}{{{d}^{3}}}\]
done
clear
View Answer play_arrow
question_answer 45) Frequency range of audible sound is
A)
\[20\,\,MHz-200\,\,MHz\]
done
clear
B)
\[20\,\,kHz-20\,\,MHz\]
done
clear
C)
\[20\,\,Hz-20\,\,kHz\]
done
clear
D)
\[0\,\,Hz-200\,\,Hz\]
done
clear
View Answer play_arrow
question_answer 46) A tape-recorder records sound in the form of
A)
variable resistance on the tape
done
clear
B)
magnetic energy
done
clear
C)
magnetic field on the tape
done
clear
D)
electrical energy
done
clear
View Answer play_arrow
question_answer 47) A monoatomic gas is suddenly compressed to\[\frac{1}{8}\text{th}\]of its volume adiabatically. The ratio of first pressure of the gas to that of original pressure will be
A)
\[128\,\,times\]
done
clear
B)
\[32\,\,times\]
done
clear
C)
\[8\,\,times\]
done
clear
D)
\[16\,\,times\]
done
clear
View Answer play_arrow
question_answer 48) If in a wire of Youngs modulus\[Y\], longitudinal strain \[X\] is produced. Then the potential energy stored in its unit volume is
A)
\[0.5\,\,Y{{X}^{2}}\]
done
clear
B)
\[0.5\,\,{{Y}^{2}}X\]
done
clear
C)
\[Y{{X}^{2}}\]
done
clear
D)
\[2\,\,Y{{X}^{2}}\]
done
clear
View Answer play_arrow
question_answer 49) A boy of \[50\,\,kg\] is standing in a lift moving down with an acceleration\[9.8\,\,m/{{s}^{2}}\]. The apparent weight of the boy will be
A)
\[50/9.8\,\,N\]
done
clear
B)
\[50\times 9.8\,\,N\]
done
clear
C)
\[50\,\,N\]
done
clear
D)
\[zero\]
done
clear
View Answer play_arrow
question_answer 50) If \[C\] and \[R\] denote capacitance and resistance then the dimensions of \[CR\] are
A)
\[[{{M}^{0}}L{{T}^{0}}{{A}^{-1}}]\]
done
clear
B)
\[[ML{{T}^{6}}{{A}^{2}}]\]
done
clear
C)
\[[M{{L}^{0}}T{{A}^{-2}}]\]
done
clear
D)
\[[{{M}^{0}}{{L}^{0}}T{{A}^{0}}]\]
done
clear
View Answer play_arrow
question_answer 51) Galvanization process involves
A)
\[Zn\]
done
clear
B)
\[Cu\]
done
clear
C)
\[Ag\]
done
clear
D)
\[Fe\]
done
clear
View Answer play_arrow
question_answer 52) The number of water molecules in gypsum and plaster of Paris respectively are
A)
1/2 and 2
done
clear
B)
2 and ½
done
clear
C)
2 and 1
done
clear
D)
5 and 2
done
clear
View Answer play_arrow
question_answer 53) Which of the following will not displace hydrogen?
A)
\[Hg\]
done
clear
B)
\[Sn\]
done
clear
C)
\[Pb\]
done
clear
D)
\[Ba\]
done
clear
View Answer play_arrow
question_answer 54) A certain sample of gas has a volume of \[0.2\] litre measured at\[1\,\,atm\]. Pressure and\[{{0}^{o}}C\]. At the same pressure at \[{{273}^{o}}C\], its volume will be
A)
\[1.4\,\,litre\]
done
clear
B)
\[1.8\,\,litre\]
done
clear
C)
\[0.4\,\,litre\]
done
clear
D)
\[0.8\,\,litre\]
done
clear
View Answer play_arrow
question_answer 55) Hess law is applicable for the determination of the heat of
A)
transition
done
clear
B)
formation
done
clear
C)
reaction
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 56) The \[pH\] value of\[HCl({{10}^{-12}}M)\]is
A)
\[-7\]
done
clear
B)
\[7\]
done
clear
C)
\[12\]
done
clear
D)
\[-12\]
done
clear
View Answer play_arrow
question_answer 57) In radioactive decay, the emitted electrons, come from
A)
inner orbital of the atom
done
clear
B)
orbit having principle quantum number one
done
clear
C)
outer most orbit of the atom
done
clear
D)
nucleus of the atom
done
clear
View Answer play_arrow
question_answer 58) Hydrolysis of sucrose is called
A)
saponification
done
clear
B)
inversion
done
clear
C)
hydration
done
clear
D)
esterification
done
clear
View Answer play_arrow
question_answer 59) In carbonium ion the carbon bearing the positive charge is
A)
\[s{{p}^{2}}\] hybridized
done
clear
B)
\[s{{p}^{3}}d\] hybridized
done
clear
C)
\[sp\]hybridized
done
clear
D)
\[s{{p}^{3}}\]hybridized
done
clear
View Answer play_arrow
question_answer 60) The number of unpaired electrons in \[M{{n}^{+2}}\] is
A)
\[4\]
done
clear
B)
\[5\]
done
clear
C)
\[3\]
done
clear
D)
\[1\]
done
clear
View Answer play_arrow
question_answer 61) Which of the following is formed by the action of water on sodium peroxide?
A)
\[{{N}_{2}}\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[{{H}_{2}}\]
done
clear
D)
\[{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 62) The total structural isomers of \[{{C}_{4}}{{H}_{10}}O\] are
A)
\[2\]
done
clear
B)
\[7\]
done
clear
C)
\[5\]
done
clear
D)
\[4\]
done
clear
View Answer play_arrow
question_answer 63) The bond order of nitrogen molecule is:
A)
\[4\]
done
clear
B)
\[1\]
done
clear
C)
\[7\]
done
clear
D)
\[3\]
done
clear
View Answer play_arrow
question_answer 64) In laboratory silicon can be prepared by the reaction of
A)
by heating carbon in electric furnace
done
clear
B)
by heating potassium fluosilicate with potassium
done
clear
C)
silica with magnesium
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 65) The average kinetic energy of an ideal gas, per molecule at \[{{25}^{o}}C\] will be
A)
\[6.17\times {{10}^{-21}}J\]
done
clear
B)
\[6.17\times {{10}^{-20}}J\]
done
clear
C)
\[61.7\times {{10}^{-21}}J\]
done
clear
D)
\[7.16\times {{10}^{-20}}J\]
done
clear
View Answer play_arrow
question_answer 66) In a gas phase reaction: \[{{C}_{2}}{{H}_{4}}+{{H}_{2}}={{C}_{2}}{{H}_{6}}\], equilibrium constant can be expressed in
A)
\[mole.litr{{e}^{-1}}\]
done
clear
B)
\[mol{{e}^{2}}.litr{{e}^{-2}}\]
done
clear
C)
\[litre.mol{{e}^{-1}}\]
done
clear
D)
\[litr{{e}^{-1}}.mol{{e}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 67) The frequency of a wave of light is\[12\times {{10}^{14}}{{s}^{-1}}\]. The wave number associated with this light is.
A)
\[5\times {{10}^{-7}}m\]
done
clear
B)
\[4\times {{10}^{-8}}c{{m}^{-1}}\]
done
clear
C)
\[2\times {{10}^{-7}}{{m}^{-1}}\]
done
clear
D)
\[4\times {{10}^{4}}c{{m}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 68) Crystalline solids are
A)
plastic
done
clear
B)
sugar
done
clear
C)
rubber
done
clear
D)
glass
done
clear
View Answer play_arrow
question_answer 69) Which of the following is the example of chain growth polymer?
A)
Bakclite
done
clear
B)
Teflon
done
clear
C)
Terylene
done
clear
D)
Nylon\[-66\]
done
clear
View Answer play_arrow
question_answer 70) Which one of the following statements is true for a electrochemical cell?
A)
\[{{H}_{2}}\] is cathode and \[Cu\] is anode
done
clear
B)
Oxidation occurs at \[Cu\] electrode
done
clear
C)
\[{{H}_{2}}\] is anode and \[Cu\] is cathode
done
clear
D)
reduction occurs at \[{{H}_{2}}\] electrode
done
clear
View Answer play_arrow
question_answer 71) Leucine amino acids is the
A)
non-essential
done
clear
B)
aromatic
done
clear
C)
basic
done
clear
D)
essential
done
clear
View Answer play_arrow
question_answer 72) Electronic configuration\[1{{s}^{2}},\,\,2{{s}^{2}}2{{p}_{x}}^{1}2{{p}_{y}}^{1}2{{p}_{z}}^{1}\]represents which of the following elements?
A)
Hydrogen
done
clear
B)
Oxygen
done
clear
C)
Fluorine
done
clear
D)
Nitrogen
done
clear
View Answer play_arrow
question_answer 73) If the compound contains\[C,\,\,H\]and halogen. When \[C\] and \[H\] are to be estimated the combustion tube at the exist should contain a
A)
copper spiral
done
clear
B)
silver spiral
done
clear
C)
iron spiral
done
clear
D)
lead spiral
done
clear
View Answer play_arrow
question_answer 74) Which decolorize aqueous bromine and gives white fumes of \[HCl\] on reaction with\[PC{{l}_{5}}\]
A)
\[C{{H}_{3}}COC{{H}_{2}}CH=C{{H}_{2}}\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}CH\]
done
clear
C)
\[C{{H}_{3}}CH=CHC{{H}_{2}}C{{H}_{2}}OH\]
done
clear
D)
\[C{{H}_{3}}OC{{H}_{2}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 75) In\[C{{H}_{2}}C{{l}_{2}}\], the oxidation number of \[C\] is
A)
\[+2\]
done
clear
B)
\[-4\]
done
clear
C)
\[0\]
done
clear
D)
\[+4\]
done
clear
View Answer play_arrow
question_answer 76) Alcohols of low molecular weight are
A)
soluble in water
done
clear
B)
soluble, in all solvents
done
clear
C)
insoluble in all solvents
done
clear
D)
soluble in water on heating
done
clear
View Answer play_arrow
question_answer 77) The glucose is an example of
A)
aldohexose
done
clear
B)
ketohexose
done
clear
C)
disaccharide
done
clear
D)
non-reducing sugar
done
clear
View Answer play_arrow
question_answer 78) A piece of wood was found to have \[{{C}^{14}}/{{C}^{12}}\] ratio \[0.7\] times that in a living plant. The time period when the plant died is (half-life of\[{{C}^{14}}=5760\,\,yr\])
A)
\[2980\,\,yr\]
done
clear
B)
\[3070\,\,yr\]
done
clear
C)
\[2966\,\,yr\]
done
clear
D)
\[2770\,\,yr\]
done
clear
View Answer play_arrow
question_answer 79) The rate of a reaction depends upon the
A)
concentration of reactant
done
clear
B)
pressure
done
clear
C)
volume
done
clear
D)
force
done
clear
View Answer play_arrow
question_answer 80) The rate constant of a first order reaction is \[3\times {{10}^{-6}}\] per second. If the initial concentration is \[0.10\,\,M\], the initial rate of reaction is
A)
\[3\times {{10}^{-6}}\,\,M{{s}^{-1}}\]
done
clear
B)
\[3\times {{10}^{-8}}\,\,M{{s}^{-1}}\]
done
clear
C)
\[3\times {{10}^{-5}}\,\,M{{s}^{-1}}\]
done
clear
D)
\[3\times {{10}^{-7}}\,\,M{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 81) Which of the following electronic configurations corresponds to an elements with the lowest ionization energy?
A)
\[1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}}\]
done
clear
B)
\[1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}},\,\,3{{s}^{1}}\]
done
clear
C)
\[1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{5}}\]
done
clear
D)
\[1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{3}}\]
done
clear
View Answer play_arrow
question_answer 82) The number of electrons and neutrons of an element is \[18\] and \[20\] respectively. Its mass number is
A)
\[37\]
done
clear
B)
\[38\]
done
clear
C)
\[9\]
done
clear
D)
\[22\]
done
clear
View Answer play_arrow
question_answer 83) The bond length between the hybridised carbon atom and other carbon atom is minimum in
A)
butane
done
clear
B)
propyne
done
clear
C)
propene
done
clear
D)
propane
done
clear
View Answer play_arrow
question_answer 84) Bleaching powder is obtained by treating chlorine with.
A)
\[CaC{{O}_{3}}\]
done
clear
B)
\[CaS{{O}_{4}}\]
done
clear
C)
\[CaO\]
done
clear
D)
\[Ca{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 85) The oxide which gives hydrogen peroxide \[({{H}_{2}}{{O}_{2}})\] on the treatment with a dilute acid \[({{H}_{2}}S{{O}_{4}})\] is
A)
\[Mn{{O}_{2}}\]
done
clear
B)
\[Pb{{O}_{2}}\]
done
clear
C)
\[N{{a}_{2}}{{O}_{2}}\]
done
clear
D)
\[Ti{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 86) Stainless steel contains iron and
A)
\[Fe+Cr+Ni\]
done
clear
B)
\[Cr+Ni\]
done
clear
C)
\[Cr+Zn\]
done
clear
D)
\[Zn+Pb\]
done
clear
View Answer play_arrow
question_answer 87) Highest percentage of carbon is found in
A)
anthracite
done
clear
B)
peat
done
clear
C)
lignite
done
clear
D)
bituminous
done
clear
View Answer play_arrow
question_answer 88) Which of the following has minimum freezing point?
A)
\[0.1\,\,M\,\,{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]
done
clear
B)
\[0.1\,\,M\,\,N{{H}_{4}}Cl\]
done
clear
C)
\[0.1\,\,M\,\,BaS{{O}_{4}}\]
done
clear
D)
\[0.1\,\,M\,\,A{{l}_{2}}{{(S{{O}_{4}})}_{3}}\]
done
clear
View Answer play_arrow
question_answer 89) Tyndall effect is shown by
A)
solution
done
clear
B)
precipitation
done
clear
C)
sol
done
clear
D)
plasma
done
clear
View Answer play_arrow
question_answer 90) The oxidation number of chromium in potassium dichromate \[({{K}_{2}}C{{r}_{2}}{{O}_{7}})\], is
A)
\[+2\]
done
clear
B)
\[+6\]
done
clear
C)
\[+4\]
done
clear
D)
\[+8\]
done
clear
View Answer play_arrow
question_answer 91) \[AlC{{l}_{3}}\] acts as Lewis acid because it is
A)
an electron donor
done
clear
B)
proton donor
done
clear
C)
proton acceptor
done
clear
D)
electron pair acceptor
done
clear
View Answer play_arrow
question_answer 92) Acetylene hydrocarbons are acidic because
A)
acetylene contains least number of hydrogen atoms among the possible.
done
clear
B)
acetylene has only one hydrogen atom at each carbon atom
done
clear
C)
acetylene belongs to the class of alkynes with formula\[{{C}_{n}}{{H}_{2\,\,n-2}}\]
done
clear
D)
Sigma electron density of\[C-H\] bond in acetylene is nearer a carbon which has \[50%\,\,S-\]character
done
clear
View Answer play_arrow
question_answer 93) \[n-\]propyl alcohol and isopropyl alcohol are
A)
position isomers
done
clear
B)
chain isomers
done
clear
C)
tautomers
done
clear
D)
geometrical isomers
done
clear
View Answer play_arrow
question_answer 94) \[C{{H}_{3}}COCl\]reacts with
A)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\]
done
clear
B)
salicylic acid
done
clear
C)
\[{{C}_{6}}{{H}_{5}}OH\]
done
clear
D)
all
done
clear
View Answer play_arrow
question_answer 95) \[x\xrightarrow{{}}\]Benzotrichloride\[+y,\,\,x\]and\[y\] respectively.
A)
benzene, benzaldehyde
done
clear
B)
toluene, benzaldehyde
done
clear
C)
toluene, benzoic acid
done
clear
D)
benzene, benzonic acid
done
clear
View Answer play_arrow
question_answer 96) Chemically soap is
A)
salt
done
clear
B)
acid
done
clear
C)
none
done
clear
D)
base
done
clear
View Answer play_arrow
question_answer 97) The process, in which no heat enters or leaves the system, is termed as
A)
isobaric
done
clear
B)
isochoric
done
clear
C)
adiabatic
done
clear
D)
isothermal
done
clear
View Answer play_arrow
question_answer 98) Which of the following information can be obtained on the basis of Le Chateliers principle?
A)
Dissociation constant of a weak acid
done
clear
B)
Entrophy change in a reaction
done
clear
C)
Shift in equilibrium position on changing value of a constant
done
clear
D)
Equilibrium constant of a chemical reaction
done
clear
View Answer play_arrow
question_answer 99) If enthalpies of formation for \[{{C}_{2}}{{H}_{4}}(g),\,\,C{{O}_{2}}(g)\]and\[{{H}_{2}}O\,\,(l)\]at\[{{25}^{o}}C\] and \[1\,\,atm\] pressure are\[52,\] \[-394\] and\[-286\,\,kJ/mol\] respectively, then total enthalpy of combustion is
A)
\[+1412\,\,kJ/mol\]
done
clear
B)
\[+141.2\,\,kJ/mol\]
done
clear
C)
\[-141.2\,\,kJ/mol\]
done
clear
D)
\[-14.2\,\,kJ/mol\]
done
clear
View Answer play_arrow
question_answer 100) Formaldehyde gives an additive product with methyl megnesium iodide which on aqueous hydrolysis gives?
A)
methyl alcohol
done
clear
B)
isopropyl alcohol
done
clear
C)
propyl alcohol
done
clear
D)
ethyl alcohol
done
clear
View Answer play_arrow
question_answer 101) Penicillium belongs to class
A)
Basidiomycetes
done
clear
B)
Ascomycetes
done
clear
C)
Duteromycetes
done
clear
D)
Phycomycetes
done
clear
View Answer play_arrow
question_answer 102) Atmospheric \[{{N}_{3}}\] fixation is carried on by
A)
Anabaena
done
clear
B)
Funaria
done
clear
C)
Chlamydomonas
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 103) The protein coat of virus is known as
A)
chromomere
done
clear
B)
capsid
done
clear
C)
chromophore
done
clear
D)
capsomere
done
clear
View Answer play_arrow
question_answer 104) In mitochondria cristae act as sites for
A)
lipid synthesis
done
clear
B)
protein synthesis
done
clear
C)
fat synthesis
done
clear
D)
oxidation reduction reaction
done
clear
View Answer play_arrow
question_answer 105) During \[DNA\] replication, the term leading strand is applied to the one which replicates in
A)
\[5\to 3\]direction discontinuously
done
clear
B)
\[3\to 5\]direction continuously
done
clear
C)
\[5\to 3\]direction continuously
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 106) Insectivorous plants eat the insects for
A)
phosphorus
done
clear
B)
calcium
done
clear
C)
nitrogen
done
clear
D)
sulphur
done
clear
View Answer play_arrow
question_answer 107) Which pigment responsible for photomorphogenetic response?
A)
Phytochrome
done
clear
B)
Chlorophyll-a
done
clear
C)
Chlorophyll-b
done
clear
D)
Xanthophyll
done
clear
View Answer play_arrow
question_answer 108) Phosphorus is a structural element in
A)
nucleotide
done
clear
B)
fat
done
clear
C)
lipid
done
clear
D)
starch
done
clear
View Answer play_arrow
question_answer 109) The sun loving plants are called
A)
xerophyte
done
clear
B)
hydrophyte
done
clear
C)
heliophytic
done
clear
D)
halophytes
done
clear
View Answer play_arrow
question_answer 110) Anaerobic breakdown of biomass produced biogas by methanogenic bacteria. It is a
A)
two-step process
done
clear
B)
one step process
done
clear
C)
four step process
done
clear
D)
three step process
done
clear
View Answer play_arrow
question_answer 111) The name of Temin and Baltimore is associated with
A)
DNA replication
done
clear
B)
reverse transcription
done
clear
C)
photorespiration
done
clear
D)
RNA synthesis
done
clear
View Answer play_arrow
question_answer 112) The technique of growing plants, in water culture, is called
A)
tissue culture
done
clear
B)
aero culture
done
clear
C)
hydroponics
done
clear
D)
cell culture
done
clear
View Answer play_arrow
question_answer 113) What sound level is considered hazardous noise pollution?
A)
Above\[80\,\,dB\]
done
clear
B)
Below\[60\,\,dB\]
done
clear
C)
Above\[130\,\,dB\]
done
clear
D)
Above\[140\,\,dB\]
done
clear
View Answer play_arrow
question_answer 114) The soil erosion can be prevented by
A)
deforestation
done
clear
B)
afforestation
done
clear
C)
over grazing
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 115) In plant disease control, the latest trend is
A)
chemical control
done
clear
B)
mechanical control
done
clear
C)
biological control
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 116) Which type of DNA is found in bacteria?
A)
Helical DNA
done
clear
B)
Membrane bound DNA
done
clear
C)
Straight DNA
done
clear
D)
Circular free DNA
done
clear
View Answer play_arrow
question_answer 117) Hargovind Khorana was awarded the Nobel Prize for
A)
gene synthesis
done
clear
B)
discovery of bacteria
done
clear
C)
genetic code
done
clear
D)
protein synthesis
done
clear
View Answer play_arrow
question_answer 118) The enzyme that converts glucose to glucose 6-phosphate is
A)
phosphorylase
done
clear
B)
phosphatase
done
clear
C)
hexokinase
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 119) Photophosphorylation was discovered by
A)
Hill
done
clear
B)
Arnon
done
clear
C)
Jolly
done
clear
D)
Dixon
done
clear
View Answer play_arrow
question_answer 120) Which type of RNA molecule is essential for protein synthesis?
A)
\[s-RNA\]
done
clear
B)
\[t-RNA\]
done
clear
C)
\[m-RNA\]
done
clear
D)
\[r-RNA\]
done
clear
View Answer play_arrow
question_answer 121) The sphere of living matter together with water, air and soil on earths surface is called
A)
lithosphere
done
clear
B)
xerosphere
done
clear
C)
biosphere
done
clear
D)
hydrosphere
done
clear
View Answer play_arrow
question_answer 122) Water pollution is caused due to
A)
industrial effluents
done
clear
B)
sewage and other wastes
done
clear
C)
agricultural discharges
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 123) The ecosystem of a pond is referred as
A)
xeric
done
clear
B)
lotic
done
clear
C)
lentic
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 124) The pollutants released by jet planes are
A)
smog
done
clear
B)
fog
done
clear
C)
aerosols
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 125) Which is the importance of molybdenum in plants metabolism?
A)
Sulphur reduction
done
clear
B)
Carbon assimilation
done
clear
C)
Nitrate reduction
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 126) Tyiakoids are present in
A)
chloroplast
done
clear
B)
nucleus
done
clear
C)
mitochondria
done
clear
D)
peroxisome
done
clear
View Answer play_arrow
question_answer 127) Which type of enzyme is present in lysosome?
A)
ATPase
done
clear
B)
Hydrolytic
done
clear
C)
Lyase
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 128) Which is a single membrane structure?
A)
Chloroplast
done
clear
B)
Lysosomes
done
clear
C)
Mitochondria
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 129) Which plant virus has DNA in it?
A)
Tomato mosaic virus
done
clear
B)
Cauliflower mosaic virus
done
clear
C)
Tobacco mosaic virus
done
clear
D)
Potato mosaic virus
done
clear
View Answer play_arrow
question_answer 130) Rough E. R. differs from smooth E.R. due to the presence of
A)
golgi body
done
clear
B)
ribosomes
done
clear
C)
nucleus
done
clear
D)
DNA
done
clear
View Answer play_arrow
question_answer 131) Mycorrhiza helps in absorption of
A)
nutrient
done
clear
B)
calcium
done
clear
C)
water
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 132) The real force responsible for the movement of water from one cell to other cell, is mainly through
A)
\[W.P.\]
done
clear
B)
\[T.P.\]
done
clear
C)
\[O.P.\]
done
clear
D)
\[D.P.D.\]
done
clear
View Answer play_arrow
question_answer 133) Embryo sac is also known as
A)
microsporangium
done
clear
B)
micro gametophyte
done
clear
C)
megagametophyte
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 134) Which is a social foresting species?
A)
Leucaena leucocephala
done
clear
B)
Tectona grandis
done
clear
C)
Mangifera indica
done
clear
D)
Rosa indica
done
clear
View Answer play_arrow
question_answer 135) If a dry wooden stakes driven into a small crack in a rock and then soaked, can develop enough pressure to split the rock. Such pressure is built up through the phenomenon of
A)
plasmolysis
done
clear
B)
imbibition
done
clear
C)
deplasmolysis
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 136) Which phase is responsible for DNA replication?
A)
\[S-\]phase
done
clear
B)
\[{{G}_{2}}-\]phase
done
clear
C)
\[{{G}_{1}}-\]phase
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 137) Fungus, without any mycelium is
A)
Saccharomyces
done
clear
B)
Puccinia
done
clear
C)
Agariciis
done
clear
D)
Altemaria
done
clear
View Answer play_arrow
question_answer 138) Mesarch xylem is commonly found in
A)
fungi
done
clear
B)
ferns
done
clear
C)
bryophytes
done
clear
D)
algae
done
clear
View Answer play_arrow
question_answer 139) The process of cytokinesis shows the division of
A)
chromatids
done
clear
B)
cytoplasm
done
clear
C)
nucleus
done
clear
D)
chromosomes
done
clear
View Answer play_arrow
question_answer 140) Turgor pressure becomes equal to the wall pressure when
A)
water enters the cell
done
clear
B)
water leaves the cell
done
clear
C)
no exchange of water takes place
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 141) Which family have diadelphous condition?
A)
Gramineae
done
clear
B)
Rosaceae
done
clear
C)
Liliaceae
done
clear
D)
Papilionaceae
done
clear
View Answer play_arrow
question_answer 142) Tyioses thickenings are seen in
A)
ray parenchyma and xylem cells
done
clear
B)
sclerenchyma
done
clear
C)
ray parenchyma only
done
clear
D)
collenchyma
done
clear
View Answer play_arrow
question_answer 143) Which becterium is associated with the roots of legumes?
A)
Spirogyra
done
clear
B)
Rhizobium
done
clear
C)
Clostridium
done
clear
D)
Nostoc
done
clear
View Answer play_arrow
question_answer 144) Binomial system of nomenclature was given by
A)
Linnaeus
done
clear
B)
Theophrastus
done
clear
C)
Hooker
done
clear
D)
Bentham
done
clear
View Answer play_arrow
question_answer 145) In Cycas coralloid roots has
A)
Clostridium
done
clear
B)
Rhizobium
done
clear
C)
Mycorrhiza
done
clear
D)
Anabaena
done
clear
View Answer play_arrow
question_answer 146) Sucking roots are present in the plant
A)
Betel
done
clear
B)
Cuscuta
done
clear
C)
Mangifera
done
clear
D)
Solarium
done
clear
View Answer play_arrow
question_answer 147) In gram positive bacteria cell wall is made up of
A)
cellulose
done
clear
B)
protein
done
clear
C)
murein
done
clear
D)
lipid
done
clear
View Answer play_arrow
question_answer 148) For ascent of sap, most acceptable theory is
A)
atmospheric theory
done
clear
B)
cohesion and transpiration pull theory
done
clear
C)
root pressure theory
done
clear
D)
pulsation theory
done
clear
View Answer play_arrow
question_answer 149) In Datura the fruit is
A)
septicidal capsule
done
clear
B)
porous capsule
done
clear
C)
septifragal capsule
done
clear
D)
loculicidal capsule
done
clear
View Answer play_arrow
question_answer 150) The bread wheat is
A)
diploid
done
clear
B)
triploid
done
clear
C)
tetraploid
done
clear
D)
hexaploid
done
clear
View Answer play_arrow
question_answer 151) If the rate of addition of new members increases with respect to the individual host of the same population, then the graph obtained has
A)
declined growth
done
clear
B)
exponential growth
done
clear
C)
zero population growth
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 152) Infection of Enterobius is caused to man is by
A)
inoculation
done
clear
B)
contamination
done
clear
C)
piercing
done
clear
D)
flying
done
clear
View Answer play_arrow
question_answer 153) Microfilaria is found in the peripheral blood of man during
A)
day time
done
clear
B)
night time
done
clear
C)
morning
done
clear
D)
evening
done
clear
View Answer play_arrow
question_answer 154) The infective stage of Entamoeba histolytica is
A)
cysi
done
clear
B)
egg
done
clear
C)
spore
done
clear
D)
trophozoite
done
clear
View Answer play_arrow
question_answer 155) Which of the following is a communicable disease?
A)
Diabetes
done
clear
B)
Hypertension
done
clear
C)
Kwasiorkor
done
clear
D)
Diphtheria
done
clear
View Answer play_arrow
question_answer 156) The disease filaria is transmitted by
A)
tse-tse fly
done
clear
B)
sandfly
done
clear
C)
Anopheles
done
clear
D)
Culex
done
clear
View Answer play_arrow
question_answer 157) In Entamoeba histolytica, the presence of chromatid bodies is characteristic of
A)
precystic stage
done
clear
B)
trophozoite stage
done
clear
C)
mature quadrinucleate cyst
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 158) Euploidy is best explained by
A)
exact multiple of a haploid set of chromosomes
done
clear
B)
one chromosome less than the haploid set of chromosomes
done
clear
C)
one chromosome more than the haploid set of chromosomes
done
clear
D)
one chromosome more than the diploid set of chromosomes
done
clear
View Answer play_arrow
question_answer 159) A man known to be victim of haemophilia marries a normal women whose father was known to be a bleeder. Then it is expected that
A)
all their children will be bleeders
done
clear
B)
one-fourth of their children will be bleeders
done
clear
C)
half of their children will be bleeders
done
clear
D)
one-tenth of their children will be bleeders
done
clear
View Answer play_arrow
question_answer 160) If a boys father has haemophilia and his mother has one gene for haemophilia, what is the chance that the boy will inherit the disease?
A)
\[100%\]
done
clear
B)
\[75%\]
done
clear
C)
\[50%\]
done
clear
D)
done
clear
View Answer play_arrow
question_answer 161) The adaptive similarities in different animals, found in the same habitat, is called
A)
adaptive radiation
done
clear
B)
convergent evolution
done
clear
C)
parallel evolution
done
clear
D)
retrogressive evolution
done
clear
View Answer play_arrow
question_answer 162) Arboreal mammals have
A)
flying character
done
clear
B)
burrowing character
done
clear
C)
climbing character
done
clear
D)
jumping character
done
clear
View Answer play_arrow
question_answer 163) The egg of a rabbit is
A)
microlecithal
done
clear
B)
megalccithal
done
clear
C)
lelolecithal
done
clear
D)
alccithal
done
clear
View Answer play_arrow
question_answer 164) The hormone that regulates the calcium level of blood is
A)
parathormone
done
clear
B)
thyroxine
done
clear
C)
insulin
done
clear
D)
glucagon
done
clear
View Answer play_arrow
question_answer 165) Hormone, which helps in metamorphosis in insects, is
A)
pheromone
done
clear
B)
thyroxine
done
clear
C)
ecdysone
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 166) In the development of the human body, the ectoderm is responsible for the formation of
A)
sweat glands
done
clear
B)
nervous system
done
clear
C)
lens of the eye
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 167) In human zygote, the male sex is determined by whether
A)
mother gets good nutrition
done
clear
B)
father is stronger than mother
done
clear
C)
strength of male chromosome
done
clear
D)
required composition of chromosomes
done
clear
View Answer play_arrow
question_answer 168) Polar bodies are produced during the formation of
A)
sperm
done
clear
B)
oogonium
done
clear
C)
spermatocytes
done
clear
D)
secondary oocyte
done
clear
View Answer play_arrow
question_answer 169) Melanocyte stimulating hormone (MSH) is secreted by
A)
anterior lobe of pituitary
done
clear
B)
posterior lobe of pituitary
done
clear
C)
intermediate lobe of pituitary
done
clear
D)
not by any lobe of pituitary
done
clear
View Answer play_arrow
question_answer 170) The two lateral ventricles, in the brain of frog, communicate with the third ventricle throught
A)
rhinocoel
done
clear
B)
sylvius
done
clear
C)
foramen magnum
done
clear
D)
foramen of monro
done
clear
View Answer play_arrow
question_answer 171) The respiratory centre, which regulates respiration, is located in
A)
cerebellum
done
clear
B)
the vagus nerve
done
clear
C)
cerebral peduncle
done
clear
D)
medulla oblongata
done
clear
View Answer play_arrow
question_answer 172) The unit of photoreception, in a compound eye of cockroach and other insects, is
A)
ctenidium
done
clear
B)
osphradium
done
clear
C)
ommatidium
done
clear
D)
rhabdome
done
clear
View Answer play_arrow
question_answer 173) The skull of a bird is
A)
dicondylic
done
clear
B)
mono condylic
done
clear
C)
amphicondylic
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 174) Astragalus and calcanium are present in
A)
fore limb
done
clear
B)
hind limb
done
clear
C)
scapula
done
clear
D)
clavicle
done
clear
View Answer play_arrow
question_answer 175) In micturition
A)
urethra relaxes
done
clear
B)
ureter contracts
done
clear
C)
ureter relaxes
done
clear
D)
urethra contracts
done
clear
View Answer play_arrow
question_answer 176) Two examples in which the nitrogenous wastes are excreted from body in the form of uric acid are
A)
birds and/lizards
done
clear
B)
mammals and mollusk
done
clear
C)
insects and bony fishes
done
clear
D)
frogs and cartilaginous fishes
done
clear
View Answer play_arrow
question_answer 177) Water reabsorption in kidney is controlled by
A)
GH
done
clear
B)
ADH
done
clear
C)
oxytocin
done
clear
D)
aldosterone
done
clear
View Answer play_arrow
question_answer 178) The abiogenesis occured about how many billion years ago?
A)
1.2 billion
done
clear
B)
1.5 billion
done
clear
C)
2.5 billion
done
clear
D)
3.5 billion
done
clear
View Answer play_arrow
question_answer 179) Development of patagia in animals is an
A)
cave adaptation
done
clear
B)
volant adaptation
done
clear
C)
aquatic adaptation
done
clear
D)
arboreal adaptation
done
clear
View Answer play_arrow
question_answer 180) Binomial nomenclature indicates
A)
genus and family
done
clear
B)
genus and species
done
clear
C)
phylum and genus
done
clear
D)
species and sub-species
done
clear
View Answer play_arrow
question_answer 181) The site of EMP pathway in cell is
A)
nucleus
done
clear
B)
peroxisome
done
clear
C)
cytoplasm
done
clear
D)
mitochondria
done
clear
View Answer play_arrow
question_answer 182) The mode of DNA replication is semi conservative. It was first demonstrated by
A)
Tayler
done
clear
B)
Khorana
done
clear
C)
Meselson and Stahl
done
clear
D)
Watson and Crick
done
clear
View Answer play_arrow
question_answer 183) How many base pairs are present in one turn of DNA?
A)
10
done
clear
B)
9
done
clear
C)
11
done
clear
D)
12
done
clear
View Answer play_arrow
question_answer 184) The induced fit theory to enzyme action was given by
A)
Koshland
done
clear
B)
Sumner
done
clear
C)
Fischer
done
clear
D)
Hershey and Chase
done
clear
View Answer play_arrow
question_answer 185) Several buds may occur in a single mother Hydra, at a time. Then
A)
all of them are equally old
done
clear
B)
the one towards the oral end is older
done
clear
C)
the one towards the aboral end is older
done
clear
D)
the one towards the aboral end is younger
done
clear
View Answer play_arrow
question_answer 186) Hermit crab and sea anemone relationship is
A)
mutualism
done
clear
B)
symbiosis
done
clear
C)
commensalism
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 187) How are Annelida advanced over Nematoda?
A)
Closed circulation
done
clear
B)
True coelom
done
clear
C)
Metameric segmentation
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 188) The type of vertebrae in sub-order ophidia is
A)
amphicoelous
done
clear
B)
acoelous
done
clear
C)
haterocoelous
done
clear
D)
procoelous
done
clear
View Answer play_arrow
question_answer 189) Which of the following is the burrowing marsupial?
A)
Thylacinus
done
clear
B)
Notoryctus
done
clear
C)
Myrmecobius
done
clear
D)
Phascolarctes
done
clear
View Answer play_arrow
question_answer 190) Ambulacral grooves are absent in the living forms of the class
A)
Crinoidea
done
clear
B)
Ophiuroidea
done
clear
C)
Asteroidea
done
clear
D)
Echinodermata
done
clear
View Answer play_arrow
question_answer 191) The transgenic animals are those which have
A)
foreign RNA in all its cells
done
clear
B)
foreign DNA in all its cells
done
clear
C)
foreign DNA in some of its cells
done
clear
D)
both a and c
done
clear
View Answer play_arrow
question_answer 192) Fats are richly found in
A)
alveolar tissue
done
clear
B)
lymph glands
done
clear
C)
adipose tissue
done
clear
D)
liver cells
done
clear
View Answer play_arrow
question_answer 193) The muscles associated with the heart of insects are
A)
alary
done
clear
B)
radial
done
clear
C)
striped
done
clear
D)
pericardial
done
clear
View Answer play_arrow
question_answer 194) The endothelium of blood vessel is composed of
A)
cuboidal epithelium
done
clear
B)
squamous epithelium
done
clear
C)
columnar epithelium
done
clear
D)
ciliated epithelium
done
clear
View Answer play_arrow
question_answer 195) What is common among amylase, rennin and trypsin?
A)
These all are proteins
done
clear
B)
These act at a pH lower than 7
done
clear
C)
These are produced in stomach
done
clear
D)
These all are proteolytic enzymes
done
clear
View Answer play_arrow
question_answer 196) The release of pancreatic juice from the pancreas, in a mammal, is stimulated by
A)
trypsinogen
done
clear
B)
secretin
done
clear
C)
entero kinase
done
clear
D)
choleocystokinin
done
clear
View Answer play_arrow
question_answer 197) The largest quantity of air that can be expired, after maximal inspiration effort, is called
A)
tidal volume
done
clear
B)
vital capacity
done
clear
C)
residual volume
done
clear
D)
total lung volume
done
clear
View Answer play_arrow
question_answer 198) Regarding blood circulation, it may by said that in Pheretima the dorsal vessel is a
A)
collecting vessel in first, two segments and distributing vessel in other
done
clear
B)
distributing vessel in first, five segments and distributing vessel in other
done
clear
C)
collecting vessel in first thirteen segments and distributing vessel in intestinal region
done
clear
D)
distributing vessel in first thirteen segments and collecting vessel in intestinal region
done
clear
View Answer play_arrow
question_answer 199) At high altitudes, the RBCs in human blood
A)
increases in size
done
clear
B)
decreases in size
done
clear
C)
increases in number
done
clear
D)
decreases in number
done
clear
View Answer play_arrow
question_answer 200) Each spinal nerve in a mammal arises from the spinal cord by two roots, a dorsal and a ventral. Of these the ventral root is composed of
A)
somatic motor and vesceral motor fibres
done
clear
B)
somatic sensory and vesceral motor fibres
done
clear
C)
somatic motor and visceral sensory fibres
done
clear
D)
somatic sensory and visceral sensory fibres
done
clear
View Answer play_arrow