-
question_answer1)
For the reaction, \[C+{{O}_{2}}\to C{{O}_{2}};\,\Delta H=-393\,J\] \[2Zn+{{O}_{2}}\to 2ZnO;\,H=-412\,J\] Which one is correct?
A)
Carbon can reduce ZnO to Zn done
clear
B)
Oxidation of carbon is not feasible done
clear
C)
Oxidation of Zn is not feasible done
clear
D)
Z'n liberates more heat than carbon during oxidation done
clear
View Solution play_arrow
-
question_answer2)
An ideal gas expands from an initial volume Pinto vacuum under isothermal conditions. For this process,
A)
\[\Delta U\ne 0,\,W=0\,and\,Q\ne 0\] done
clear
B)
\[\Delta U\ne 0,\,W\ne 0\,and\,Q\ne 0\] done
clear
C)
\[\Delta U\ne 0,\,W\ne 0\,and\,Q=0\] done
clear
D)
\[\Delta U=0,\,W=0\,and\,Q=0\] done
clear
View Solution play_arrow
-
question_answer3)
Evaluate \[\Delta U\] at 500 K for the process \[Mg(s)+2HCl(g)\to MgC{{l}_{2}}(s)+{{H}_{2}}(g)\Delta H=-109\]kcal The volume change of solids to be ignored
A)
-204 kcal done
clear
B)
-108 kcal done
clear
C)
-9 kcal done
clear
D)
+9 kcal done
clear
View Solution play_arrow
-
question_answer4)
The value of enthalpy change (\[\Delta H\]) for the reaction \[{{C}_{2}}{{H}_{2}}OH(l)+3{{O}_{2}}(g)\to 2C{{O}_{2}}(g)+3{{H}_{2}}O(l)\]a \[27{}^\circ C\]is -1366.5 kJ\[mo{{l}^{-1}}\]. The value of internal energy change for the above reaction at this temperature will be
A)
-1371.5 kJ done
clear
B)
-1369.0 kJ done
clear
C)
-1364.0 kJ done
clear
D)
-1361.5 kJ done
clear
View Solution play_arrow
-
question_answer5)
If 50 calories are added to a system and system does work of 30 calories on surroimdings, the change in internal energy of system is
A)
20 cal done
clear
B)
50 cal done
clear
C)
40 cal done
clear
D)
30 cal done
clear
View Solution play_arrow
-
question_answer6)
The standard heat of formation of \[{{U}_{3}}{{O}_{8}}\] is -853.5 kcal/mol and the standard heat of the reaction \[3U{{O}_{2}}+{{O}_{2}}\to {{U}_{3}}{{O}_{8}}\] is -76.01 kcal. The standard heat of formation of \[U{{O}_{2}}\] is
A)
-1083 kJ \[mo{{l}^{-1}}\] done
clear
B)
-108.3 kJ \[mo{{l}^{-1}}\] done
clear
C)
-10.83 kJ \[mo{{l}^{-1}}\] done
clear
D)
-1.083 kJ \[mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer7)
The enthalpies of combustion of carbon and carbon monoxide are -390 kJ and -278 kJ respectively. The enthalpy of formation of CO in kJ is
A)
668 done
clear
B)
112 done
clear
C)
-112 done
clear
D)
-668 done
clear
View Solution play_arrow
-
question_answer8)
\[{{C}_{2}}{{H}_{6}}(g)+3.5{{O}_{2}}(g)\to 2C{{O}_{2}}(g)+3{{H}_{2}}O(g)\]\[\Delta {{S}_{vap}}({{H}_{2}}O,\ell )={{x}_{1}}cal\,{{K}^{-1}}\] (boiling point = \[{{T}_{1}}\]) \[\Delta {{H}_{f}}({{H}_{2}}O,\ell )={{x}_{2}}\] \[\Delta {{H}_{f}}({{C}_{2}}O,)={{x}_{2}}\] \[\Delta {{H}_{f}}({{C}_{2}}{{H}_{6}},)={{x}_{4}}\] Hence, \[\Delta H\] for the reaction is
A)
\[2{{x}_{3}}+3{{x}_{2}}-{{x}_{4}}\] done
clear
B)
\[2{{x}_{3}}+3{{x}_{2}}-{{x}_{4}}+3{{x}_{1}}{{T}_{1}}\] done
clear
C)
\[2{{x}^{+}}+3{{x}_{2}}-{{x}_{4}}+3{{x}_{1}}{{T}_{1}}\] done
clear
D)
\[{{x}_{1}}{{T}_{1}}+{{x}_{2}}+{{x}_{3}}-{{x}_{4}}\] done
clear
View Solution play_arrow
-
question_answer9)
An ideal monatomic gas is taken round the cycle ABCD as shown in the figure. The work done during the cycle is
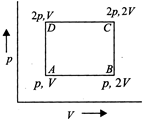
A)
\[pV\] done
clear
B)
\[2pV\] done
clear
C)
\[\frac{1}{2}pV\] done
clear
D)
zero done
clear
View Solution play_arrow
-
question_answer10)
Work for the following process ABCD on a monoatomic gas is
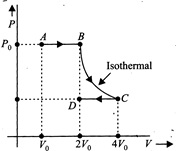
A)
\[W=-2\,{{P}_{0}}{{V}_{0}}ln\,2\] done
clear
B)
\[W=-2\,{{P}_{0}}{{V}_{0}}(1+ln\,2)\] done
clear
C)
\[W=-\,{{P}_{0}}{{V}_{0}}(1+ln\,2)\] done
clear
D)
\[W=-\,{{P}_{0}}{{V}_{0}}ln\,2\] done
clear
View Solution play_arrow
-
question_answer11)
Select the correct set of statement/s:
| I. Work done by the surrounding in case of infinite stage expansion is more than single stage expansion |
| II. Irreversible work is always greater than reversible work (with sign) |
| III. On an ideal gas in case of single stage expansion and compression system as well as surrounding are restored back to their original states |
| IV. If gas is in thermodynamic equilibrium the expansion is taken from state A to state B, by four successive single stage expansions. Then we can plot 4 points on the P-V indicator diagram. |
A)
II done
clear
B)
I, II, III, IV done
clear
C)
II, IV done
clear
D)
I, II, IV done
clear
View Solution play_arrow
-
question_answer12)
If the internal energy of a system decreases by 128 J and at the same time it absorbs 56 J of heat, then
A)
work is done by the system equal to +71 J done
clear
B)
work is done by the system equal to -184 J done
clear
C)
work is done by the system equal to -71 J done
clear
D)
work is done by the system equal to 179 J done
clear
View Solution play_arrow
-
question_answer13)
A vessel contains 100 litres of a liquid X. Heat is supplied to the liquid in such a fashion that heat given = change in enthalpy. The volume of the liquid increases by.2 litres. If the external pressure is 1 ami and 202.6 joules of heat were supplied, then [U= total internal energy]
A)
\[\Delta U=0,\,\Delta H=0\] done
clear
B)
\[\Delta U=+202.6J,\,\Delta H=+202.6J\] done
clear
C)
\[\Delta U=-202.6J,\,\Delta H=-202.6J\] done
clear
D)
\[\Delta U=0,\,\Delta H=+202.6J\] done
clear
View Solution play_arrow
-
question_answer14)
In an irreversible process taking place at constant P and T in which only pressure-volume work is being done by the system, the change in Gibbs energy (dG) and change in entropy (dS) satisfy the criterion
A)
\[{{(dS)}_{v,u}}=0,\,{{(dG)}_{T,P}}=0\] done
clear
B)
\[{{(dS)}_{v,u}}=0,\,{{(dG)}_{T,P}}=+ve\] done
clear
C)
\[{{(dS)}_{v,u}}=-ve,\,{{(dG)}_{T,P}}=-ve\] done
clear
D)
\[{{(dS)}_{v,u}}=+ve,\,{{(dG)}_{T,P}}=-ve\] done
clear
View Solution play_arrow
-
question_answer15)
The enthalpy change states for the following processes are listed below:
|
\[C{{l}_{2}}(g)\to 2Cl(g);\]
|
\[242.3kJ\text{ }mo{{l}^{-1}}\]
|
|
\[{{I}_{2}}(g)\to 2I(g);\]
|
\[151.0kJ\text{ }mo{{l}^{-1}}\]
|
|
\[ICl(g)\to I(g)+Cl(g);\]
|
\[211.3kJ\text{ }mo{{l}^{-1}}\]
|
|
\[{{I}_{2}}(s)\to {{I}_{2}}(g);\]
|
\[62.76kJ\text{ }mo{{l}^{-1}}\]
|
Given that the standard states for iodine chlorine are \[{{I}_{2}}(s)\] and\[C{{l}_{2}}(g)\], the standard enthalpy o formation for ICl (g) is:
A)
+244.8 kJ \[mo{{l}^{-1}}\] done
clear
B)
-14.6 kJ \[mo{{l}^{-1}}\] done
clear
C)
-16.8kJ \[mo{{l}^{-1}}\] done
clear
D)
+ 16.8kJ \[mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer16)
An ideal gas is allowed to expand both reversibly and irreversibly in an isolated system. If \[{{T}_{i}}\] is the initial temperature and\[{{T}_{f}}\]is the final temperature, which of following statements is correct?
A)
\[{{T}_{f}}\] and \[{{T}_{i}}\] or both reversible and irreversible processes done
clear
B)
\[{{({{T}_{f}})}_{irrev}}>{{({{T}_{i}})}_{rev}}\] done
clear
C)
\[{{T}_{f}}\] > \[{{T}_{i}}\] for irreversible process but \[{{T}_{f}}\] and \[{{T}_{i}}\] for irreversible process done
clear
D)
\[{{({{T}_{f}})}_{rev}}>{{({{T}_{i}})}_{irrev}}\] done
clear
View Solution play_arrow
-
question_answer17)
The incorrect expression among the following is
A)
\[\frac{\Delta {{G}_{system}}}{\Delta {{S}_{total}}}=-T\] done
clear
B)
in isothermal process,\[{{W}_{reversible}}=-nRTIn\frac{{{V}_{f}}}{{{V}_{i}}}\] done
clear
C)
\[In\,K=\frac{\Delta H-T\Delta {{S}^{o}}}{RT}\] done
clear
D)
\[K={{e}^{-\Delta {{G}^{o}}/RT}}\] done
clear
View Solution play_arrow
-
question_answer18)
For which change \[\Delta H\ne AE:\]
A)
\[{{H}_{2}}(g)+{{I}_{2}}(g)\rightleftarrows 2HI(g)\] done
clear
B)
\[HCl+NaOH\to NaCl+{{H}_{2}}O\] done
clear
C)
\[C(s)+{{O}_{2}}(g)\rightleftarrows C{{O}_{2}}(g)\] done
clear
D)
\[{{N}_{2}}(g)+3{{H}_{2}}(g)\to 2N{{H}_{3}}(g)\] done
clear
View Solution play_arrow
-
question_answer19)
Which one of the following statement is false?
A)
Work is a state function. done
clear
B)
Temperature is a state function. done
clear
C)
Change in the state is completely defined when the initial and final states are specified. done
clear
D)
Work appears at the boundary of the system. done
clear
View Solution play_arrow
-
question_answer20)
For the process \[{{H}_{2}}O(I)\to {{H}_{2}}O(g)\] At \[T=100{}^\circ C\]and 1 atmosphere pressure, the correct choice is
A)
\[\Delta {{S}_{system}}>0\,and\,\Delta {{S}_{surroundings}}>0\] done
clear
B)
\[\Delta {{S}_{system}}>0\,and\,\Delta {{S}_{surroundings}}<0\] done
clear
C)
\[\Delta {{S}_{system}}<0\,and\,\Delta {{S}_{surroundings}}>0\] done
clear
D)
\[\Delta {{S}_{system}}<0\,and\,\Delta {{S}_{surroundings}}<0\] done
clear
View Solution play_arrow
-
question_answer21)
64 grams of methanol at a temperature of \[60{}^\circ C\]are poured into 128 gram of methanol kept at \[20{}^\circ C\]in an insulated vessel. Calculate the net change in entropy that occurs. (give your answer in J/K and in the form of nearest whole number) \[Cp\] (methanol) =81.6 J/K-mol [log(0.9198) =-0.036; log (1.045) = 0.0193]
View Solution play_arrow
-
question_answer22)
For the reaction, \[{{N}_{2}}(g)=3{{H}_{2}}(g)\to 2N{{H}_{3}}(g)\] Heat of reaction at constant volume exceeds the heat of reaction at constant pressure by the value of xRT. The value of x is ______.
View Solution play_arrow
-
question_answer23)
Heat supplied to a Carnot engine is 2 x \[{{10}^{3}}\] J. If the engine work between 300 K and 600 K. The useful work obtained from the Camot engine is ......... kJ.
View Solution play_arrow
-
question_answer24)
\[AH{}^\circ \]and AS0 for the reaction \[{{A}_{2}}+{{B}_{2}}\rightleftarrows 2AB\] at 298 K are 29.3 KJ \[mo{{l}^{-1}}\] and 104.1 \[J{{K}^{-1}}\] \[mo{{l}^{-1}}\], respectively. The equilibrium constant for the reaction is
View Solution play_arrow
-
question_answer25)
For the reaction; \[A(s)\to B(g)+C(g)\] \[{{\Delta }_{r}}{{G}^{o}}=(1000-50T\,In\,T)\]in kJ/mol \[{{\Delta }_{r}}{{C}^{o}}_{p}\] of the above reaction in kJ/mol is 10 A". Find the value of X.
View Solution play_arrow

