-
question_answer1)
Which of the following hydroxide is acidic?
A)
\[Al{{\left( OH \right)}_{3}}\] done
clear
B)
\[Ca{{\left( OH \right)}_{3}}\] done
clear
C)
\[Tl{{\left( OH \right)}_{3}}\] done
clear
D)
\[B{{\left( OH \right)}_{3}}\] done
clear
View Solution play_arrow
-
question_answer2)
Al is more reactive than Fe but Al is less easily corroded than Fe because
A)
it is a noble metal done
clear
B)
oxygen forms a protective oxide layer done
clear
C)
iron undergoes reaction easily with water done
clear
D)
Fe form mono and divalent ions. done
clear
View Solution play_arrow
-
question_answer3)
Often a ground glass stopper gets stuck in the neck of a glass bottle containing \[NaOH\] solution. This is due to:
A)
The presence of dirt particles in between. done
clear
B)
The formation of solid silicate in between by the reaction of \[Si{{O}_{2}}\] of glass with \[NaOH.\] done
clear
C)
The formation of \[N{{a}_{2}}C{{\text{O}}_{3}}\] in between by the reaction of \[C{{O}_{2}}\] of air and \[NaOH.\] done
clear
D)
Glass contains a boron compound which forms a precipitate with the \[NaOH\] solution. done
clear
View Solution play_arrow
-
question_answer4)
\[\left( S{{i}_{2}}{{O}_{5}} \right)_{n}^{2n-}\] anion is obtained when:
A)
no oxygen of a \[SiO_{4}^{4-}\] 'tetrahedron is shared with another \[SiO_{4}^{4-}\] tetrahedron done
clear
B)
one oxygen of a \[SiO_{4}^{4-}\] tetrahedron is shared with another \[SiO_{4}^{4-}\] tetrahedron done
clear
C)
two oxygen of a \[SiO_{4}^{4-}\] tetrahedron are shared with another \[SiO_{4}^{4-}\] tetrahedron done
clear
D)
three oxygen of a \[SiO_{4}^{4-}\] tetrahedron are shared with another \[SiO_{4}^{4-}\] tetrahedron done
clear
View Solution play_arrow
-
question_answer5)
Boric acid is a weak monobasic acid and acts as Lewis acid
A)
By donating \[{{H}^{+}}\] done
clear
B)
By accepting \[O{{H}^{-}}\] done
clear
C)
By donating lone pair of electrons done
clear
D)
By accepting lone pair of electrons. done
clear
View Solution play_arrow
-
question_answer6)
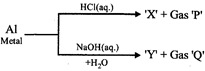
The incorrect statement regarding above reactions is:
A)
Al shows amphoteric character done
clear
B)
Gas 'P' and 'Q' are different done
clear
C)
Both X and Y are water soluble done
clear
D)
Gas Q is inflammable done
clear
View Solution play_arrow
-
question_answer7)
Ge(II)compounds are powerful reducing agents whereas Pb(IV)compounds are strong oxidants. It is because
A)
Pb is more electropositive than Ge done
clear
B)
ionization potential of lead is less than that of Ge done
clear
C)
ionic radii of \[P{{b}^{2+}}\]and \[P{{b}^{4+}}\] are larger than those of \[G{{e}^{2+}}\] and \[G{{e}^{4+}}\] done
clear
D)
of more pronounced inert pair effect in lead than in Ge done
clear
View Solution play_arrow
-
question_answer8)
Which of the following conceivable structures for \[CC{{l}_{4}}\] will have a zero dipole moment?
A)
Square planar done
clear
B)
Square pyramid (carbon at apex) done
clear
C)
Irregular tetrahedron done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer9)
Least thermally stable is -
A)
\[CC{{l}_{4}}\] done
clear
B)
\[SiC{{l}_{4}}\] done
clear
C)
\[GeC{{l}_{4}}\] done
clear
D)
\[GeB{{r}_{4}}\] done
clear
View Solution play_arrow
-
question_answer10)
To a piece of charcoal, sulphuric acid is added. Then:
A)
there is no reaction done
clear
B)
water gas is formed done
clear
C)
\[S{{O}_{2}}\] and \[C{{O}_{2}}\] are evolved done
clear
D)
CO and \[S{{O}_{2}}\] are evolved done
clear
View Solution play_arrow
-
question_answer11)
The correct statement with respect to CO is
A)
it combines with \[{{H}_{2}}O\] to give carbonic acid done
clear
B)
it reacts with haemoglobin in RBC done
clear
C)
it is powerful oxidising agent done
clear
D)
it is used to prepare aerated drinks done
clear
View Solution play_arrow
-
question_answer12)
\[C{{O}_{2}}\] and \[{{N}_{2}}\] are non-supporters of combustion. However for putting out fires \[C{{O}_{2}}\] is preferred over \[{{N}_{2}}\] because \[C{{O}_{2}}\]
A)
does not burn done
clear
B)
forms non-combustible products with burning substances done
clear
C)
is denser than nitrogen done
clear
D)
is a more reactive gas done
clear
View Solution play_arrow
-
question_answer13)
Which of the following is not correct?
A)
\[Ge{{(OH)}_{2}}\] is amphoteric done
clear
B)
\[GeC{{l}_{2}}\] is more stable than \[GeC{{l}_{4}}\] done
clear
C)
\[Ge{{O}_{2}}\] is weakly acidic done
clear
D)
\[GeC{{l}_{4}}\] in \[HCl\] forms \[{{[GeC{{l}_{2}}]}^{2-}}\] ion done
clear
View Solution play_arrow
-
question_answer14)
Which of the following is similar to graphite?
A)
B done
clear
B)
BN done
clear
C)
\[{{B}_{2}}{{H}_{6}}\] done
clear
D)
\[{{B}_{4}}C\] done
clear
View Solution play_arrow
-
question_answer15)
In the following sets of reactants which two sets best exhibit the amphoteric characters of
| \[A{{l}_{2}}{{O}_{3}}.x{{H}_{2}}O~\] |
| Set 1: \[A{{l}_{2}}{{O}_{3}}.x{{H}_{2}}O(s)\,and\,O{{H}^{-}}(aq)\] |
| Set 2: \[A{{l}_{2}}{{O}_{3}}.x{{H}_{2}}O(s)\,and\,{{H}_{2}}O(l)\] |
| Set 3: \[A{{l}_{2}}{{O}_{3}}.x{{H}_{2}}O(s)\,and\,{{H}^{+}}(aq)\] |
| Set 4: \[A{{l}_{2}}{{O}_{3}}.x{{H}_{2}}O(s)\,and\,N{{H}_{3}}(aq)\] |
A)
1 and 2 done
clear
B)
1 and 3 done
clear
C)
2 and 4 done
clear
D)
3 and 4 done
clear
View Solution play_arrow
-
question_answer16)
The gas evolved on heating \[Ca{{F}_{2}}\] and \[Si{{O}_{2}}\]with concentrated \[{{H}_{2}}S{{O}_{4}}\] on hydrolysis gives a white gelatinous precipitate. The precipitate is:
A)
hydrofluosilicic acid done
clear
B)
silica gel done
clear
C)
silicic acid done
clear
D)
calciumfluorosilicate done
clear
View Solution play_arrow
-
question_answer17)
On adding ammonium hydroxide solution to \[A{{l}_{2}}{{(S{{O}_{4}})}_{3}}(aq)\]:
A)
A precipitate is formed which does not dissolve in excess of ammonium hydroxide done
clear
B)
A precipitate is formed which dissolves in excess of ammonia solution done
clear
C)
No precipitate is formed done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer18)
Which of the following statements are correct?
| (i) Aluminium forms \[{{[Al{{F}_{6}}]}^{3-}}\] ion while boron forms only \[{{[B{{F}_{4}}]}^{-}}\] ion due to presence of d-orbitals in aluminium. |
| (ii) The first member of a group differs from the heavier members in its ability to form \[p\pi -p\pi \] multiple bonds to itself and to other second row elements. While heavier member forms \[d\pi -p\pi \] bonds. |
| (iii) d-orbitals contribute more to the overall stability of molecules than \[p\pi -p\pi \] bonding of second row elements. |
A)
(i) (ii) (iii) done
clear
B)
(i) (iii) done
clear
C)
(i) (ii) done
clear
D)
(ii) (iii) done
clear
View Solution play_arrow
-
question_answer19)
An aqueous solution of \[FeS{{O}_{4}},A{{l}_{2}}{{(S{{O}_{4}})}_{3}}\] and chrome alum is heated with excess of \[N{{a}_{2}}{{O}_{2}}\] and filtered. The materials obtained are:
A)
a colourless filtrate and a green residue done
clear
B)
a yellow filtrate and a green residue done
clear
C)
a yellow filtrate and a brown residue done
clear
D)
a green filtrate and a brown residue done
clear
View Solution play_arrow
-
question_answer20)
Which statement is not true about potash alum?
A)
On heating it melts and loses its water of crystallization. done
clear
B)
It's aqueous solution is basic in nature. done
clear
C)
It is used in dyeing industries. done
clear
D)
It's empirical formula is \[KAl{{\left( S{{O}_{4}} \right)}_{2}}.12{{H}_{2}}O.\] done
clear
View Solution play_arrow
-
question_answer21)
Anhydrous aluminium chloride \[(A{{l}_{2}}C{{l}_{6}})\] is covalent compound and soluble in water giving:
A)
\[A{{l}^{3+}}\] and \[C{{l}^{-}}\]ions done
clear
B)
\[{{[Al{{({{H}_{2}}O)}_{6}}]}^{3+}}\] and \[C{{l}^{-}}\] ions done
clear
C)
\[{{[AlC{{l}_{2}}{{({{H}_{2}}O)}_{4}}]}^{3+}}\] and \[{{[AlC{{l}_{4}}{{({{H}_{2}}O)}_{2}}]}^{-}}\] ions done
clear
D)
none of the above done
clear
View Solution play_arrow
-
question_answer22)
Borax is converted into crystalline boron by the following steps: \[Borax\xrightarrow{X}{{H}_{3}}B{{O}_{3}}\xrightarrow{\Delta }{{B}_{2}}{{O}_{3}}\xrightarrow[\Delta ]{Y}B\] X and Y are respectively:
A)
\[HCl,Mg\] done
clear
B)
\[HCl,C\] done
clear
C)
\[C,Al\] done
clear
D)
\[HCl,Al\] done
clear
View Solution play_arrow
-
question_answer23)
On controlled hydrolysis and condensation, \[{{R}_{3}}SiCl\] yields
A)
\[{{R}_{3}}Si-O-Si{{R}_{3}}\] done
clear
B)
\[-({{R}_{3}}Si-O-Si{{R}_{3}}){{-}_{n}}\] done
clear
C)
\[{{R}_{3}}SiOH\] done
clear
D)
View Solution play_arrow
-
question_answer24)
\[B{{(OH)}_{3}}+NaOH\to NaB{{O}_{2}}+Na[B{{(OH)}_{4}}+{{H}_{2}}O\] How can this reaction is made to proceed in forward direction?
A)
addition of cis 1,2-diol done
clear
B)
addition of borax done
clear
C)
addition of trans 1,2-diol done
clear
D)
addition of \[N{{a}_{2}}HP{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer25)
Anhydrous \[AlC{{l}_{3}}\] cannot be obtained from which of the following reactions?
A)
Heating \[AlC{{l}_{3}}.6{{H}_{2}}O\] done
clear
B)
By passing dry \[HCl\] over hot aluminium powder done
clear
C)
By passing dry \[C{{l}_{2}}\] over hot aluminium powder done
clear
D)
By passing dry \[C{{l}_{2}}\] over a hot mixture of alumina and coke done
clear
View Solution play_arrow
-
question_answer26)
The role of fluorspar \[(Ca{{F}_{2}})\] which is added in small quantities in the electrolytic reduction of alumina dissolved in fused cryolite \[(N{{a}_{3}}Al{{F}_{6}})\] is
A)
as a catalyst done
clear
B)
to make the fused mixture very conducting done
clear
C)
to increase the temperature of the melt. done
clear
D)
to decrease the rate of oxidation of carbon at the anode. done
clear
View Solution play_arrow
-
question_answer27)
The correct order of increasing \[C-O\] bond length of \[CO,C{{O}_{2}}\] and \[CO_{3}^{2-}\] is:
A)
\[CO_{3}^{2-}~<C{{O}_{2}}<CO\] done
clear
B)
\[C{{O}_{2}}<CO_{3}^{2-}<CO\] done
clear
C)
\[CO<CO_{3}^{2-}<C{{O}_{2}}\] done
clear
D)
\[CO<C{{O}_{2}}<CO_{3}^{2-}\] done
clear
View Solution play_arrow
-
question_answer28)
In silicon dioxide
A)
there are double bonds between silicon and oxygen atoms done
clear
B)
silicon atom is bonded to two oxygen atoms done
clear
C)
each silicon atom is surrounded by two oxygen atoms and each oxygen atom is bonded to two silicon atoms done
clear
D)
each silicon atom is surrounded by four oxygen atoms and each oxygen atom is bonded to two silicon atoms. done
clear
View Solution play_arrow
-
question_answer29)
Example of a three-dimensional silicate is:
A)
Zeolites done
clear
B)
Ultramarines done
clear
C)
Feldspars done
clear
D)
Beryls done
clear
View Solution play_arrow
-
question_answer30)
Identify the incorrect statement:
A)
In \[{{\left( S{{i}_{3}}{{O}_{9}} \right)}^{6-}},\] tetrahedral \[Si{{O}_{4}}\] units share two oxygen atoms. done
clear
B)
Trialkylchlorosilane on hydrolysis gives \[{{R}_{3}}SiOH.\] done
clear
C)
\[SiC{{l}_{4}}\] undergoes hydrolysis to give \[{{H}_{4}}Si{{O}_{4}}\] done
clear
D)
\[{{\left( S{{i}_{3}}{{O}_{9}} \right)}^{6-}}\] has cyclic structure. done
clear
View Solution play_arrow
-
question_answer31)
Aluminium chloride exists as dimer, \[A{{l}_{2}}C{{l}_{6}}\] in solid state as well as in solution of non-polar solvents such as benzene. When dissolved in water, it gives
A)
\[{{[Al{{(OH)}_{6}}]}^{3-}}+3HCl\] done
clear
B)
\[{{[Al{{({{H}_{2}}O)}_{6}}]}^{3+}}+3C{{l}^{-}}\] done
clear
C)
\[A{{l}^{3+}}+3C{{l}^{-}}\] done
clear
D)
\[A{{l}_{2}}{{O}_{3}}+6HCl\] done
clear
View Solution play_arrow
-
question_answer32)
When Al is added to \[NaOH\] solution
A)
No action takes place done
clear
B)
\[NaAl{{O}_{2}}\] is formed and \[{{H}_{2}}\] is evolved done
clear
C)
\[Al{{\left( OH \right)}_{3}}\] is formed and \[{{H}_{2}}\] is evolved done
clear
D)
\[N{{a}_{2}}Al{{O}_{2}}\] is formed and\[{{H}_{2}}\]is evolved done
clear
View Solution play_arrow
-
question_answer33)
Which of the following statement(s) is / are incorrect for\[C{{O}_{2}}\]?
| (i) In laboratory \[C{{O}_{2}}\] is prepared by the action of dilute \[HCl\] on calcium carbonate |
| (ii) Carbon dioxide is a poisonous gas |
| (iii) Increase in carbon dioxide content in atmosphere lead to increase in green house effect. |
| (iv) \[C{{O}_{2}}\] as dry ice is used as a refrigerant for ice cream and frozen food. |
A)
(i) and (ii) done
clear
B)
Only (ii) done
clear
C)
(i),(ii) and (iii) done
clear
D)
(ii) and (iii) done
clear
View Solution play_arrow
-
question_answer34)
A group 14 element is oxidised to form corresponding oxide which is gaseous in nature, when dissolved in water pH of the water decreases further addition of group 2 hydroxides leads to precipitation. This oxide can be
A)
\[Ge{{O}_{2}}\] done
clear
B)
CO done
clear
C)
\[C{{O}_{2}}\] done
clear
D)
\[Sn{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer35)
\[Pb{{F}_{4}},PbC{{l}_{4}}\] exist but \[PbB{{r}_{4}}\] and \[Pb{{I}_{4}}\] do not exist because of
A)
large size of \[B{{r}^{-}}\] and \[{{I}^{-}}\] done
clear
B)
strong oxidising character of \[P{{b}^{4+}}\] done
clear
C)
strong reducing character of \[P{{b}^{4+}}\] done
clear
D)
low electronegativity of \[B{{r}^{-}}\] and \[{{I}^{-}}\]. done
clear
View Solution play_arrow
-
question_answer36)
Which one of the following is a correct set for \[Si{{O}_{2}}\]?
A)
Linear acidic done
clear
B)
Linear, basic done
clear
C)
Tetrahedral, acidic done
clear
D)
Angular, basic. done
clear
View Solution play_arrow
-
question_answer37)
Which is not the use of orthoboric acid?
A)
As an antiseptic and eye wash. done
clear
B)
In glass industry. done
clear
C)
In glazes for pottery. done
clear
D)
In borax - bead test. done
clear
View Solution play_arrow
-
question_answer38)
The factor responsible for weak acidic nature of B-F bonds in \[B{{F}_{3}}\] is
A)
large electronegativity of fluorine done
clear
B)
three centred two electron bonds in \[B{{F}_{3}}\] done
clear
C)
\[p\pi -d\pi \] back bonding done
clear
D)
\[p\pi -p\pi \] back bonding done
clear
View Solution play_arrow
-
question_answer39)
Which of the following has the minimum heat of dissociation:
A)
\[{{\left( C{{H}_{3}} \right)}_{3}}N:\to B{{F}_{3}}\] done
clear
B)
\[{{(C{{H}_{3}})}_{3}}N:\to B{{(C{{H}_{3}})}_{2}}F\] done
clear
C)
\[{{(C{{H}_{3}})}_{3}}N:\to B{{(C{{H}_{3}})}_{3}}\] done
clear
D)
\[{{\left( C{{H}_{3}} \right)}_{3}}N:\to \text{ }B\left( C{{H}_{3}} \right){{F}_{2}}\] done
clear
View Solution play_arrow
-
question_answer40)
Lead is not affected by dil. \[HCl\] in cold because
A)
Pb is less electronegative than H done
clear
B)
PbO film is formed which resists chemical attack by acid done
clear
C)
\[PbC{{l}_{2}}\] protective coating gets formed on Pb surface done
clear
D)
\[Pb{{O}_{2}}\] film is always present on Pb surface, which resist chemical attack done
clear
View Solution play_arrow
-
question_answer41)
The catenation tendency of C, Si and Ge is in the order \[Ge<Si<C.\] The bond energies (in\[kJmo{{l}^{-1}}\]) of \[C-C,\] \[Si-Si\] and \[Ge-Ge\] bonds are respectively
A)
348, 297, 260 done
clear
B)
297, 348, 260 done
clear
C)
348, 260, 297 done
clear
D)
260, 297, 348 done
clear
View Solution play_arrow
-
question_answer42)
The melting pt. of group 13 follows the order
A)
\[B>Al>Ga>In>Tl\] done
clear
B)
\[B>Al<Ga>In>Tl\] done
clear
C)
\[B>Al>Tl>In>Ga\] done
clear
D)
\[B>Al<Ga<In<Tl\] done
clear
View Solution play_arrow
-
question_answer43)
\[Al{{I}_{3}},\] when reacts with \[CC{{l}_{4}}\] gives
A)
\[AlC{{l}_{3}}\] done
clear
B)
\[C{{I}_{4}}\] done
clear
C)
\[A{{l}_{4}}{{C}_{3}}\] done
clear
D)
both [a] and [b] done
clear
View Solution play_arrow
-
question_answer44)
Which of the following properties of aluminium makes it useful for food packaging?
A)
Good electrical conductivity done
clear
B)
Good thermal conductivity done
clear
C)
Low density done
clear
D)
Non toxicity done
clear
View Solution play_arrow
-
question_answer45)
A metal, M forms chlorides in its +2 and +4 oxidation states. Which of the following statements about these chlorides is correct?
A)
\[MC{{l}_{2}}\] is more ionic than \[MC{{l}_{4}}\] done
clear
B)
\[MC{{l}_{2}}\] is more easily hydrolysed than \[MC{{l}_{4}}\] done
clear
C)
\[MC{{l}_{2}}\] is more volatile than \[MC{{l}_{4}}\] done
clear
D)
\[MC{{l}_{2}}\] is more soluble in anhydrous ethanol than \[MC{{l}_{4}}\] done
clear
View Solution play_arrow
-
question_answer46)
Among the following substituted silanes the one which will give rise to cross linked silicone polymer on hydrolysis is
A)
\[{{R}_{4}}Si\] done
clear
B)
\[{{R}_{2}}SiC{{l}_{2}}\] done
clear
C)
\[RSiC{{l}_{3}}\] done
clear
D)
\[{{R}_{3}}SiCl\] done
clear
View Solution play_arrow
-
question_answer47)
Which of the following statements is false?
A)
Water gas is a mixture of hydrogen and carbon monoxide done
clear
B)
Producer gas is a mixture of CO and nitrogen done
clear
C)
Water gas is a mixture of water vapour and hydrogen done
clear
D)
Natural gas consists of methane, ethane and gaseous hydrocarbons. done
clear
View Solution play_arrow
-
question_answer48)
Lead pipes are not suitable for drinking water because
A)
lead forms basic lead carbonate done
clear
B)
lead reacts with water containing air to form \[Pb{{(OH)}_{2}}\] done
clear
C)
a layer of lead dioxide is deposited over pipes done
clear
D)
lead reacts with air to form litharge done
clear
View Solution play_arrow
-
question_answer49)
Choose the correct sequence for the geometry of the given molecules Borazone, Borazole, \[{{B}_{3}}O_{6}^{3-}\] ['P' stands for planar and 'N' stands for non- planar]
A)
NP, NP, NP done
clear
B)
P, P, NP done
clear
C)
NP, P, NP done
clear
D)
NP, P, P done
clear
View Solution play_arrow
-
question_answer50)
Which out of the following compounds does not exist?
A)
\[B{{F}_{3}}\] done
clear
B)
\[TIC{{l}_{3}}\] done
clear
C)
\[TIC{{l}_{5}}\] done
clear
D)
Both [b] and [c] done
clear
View Solution play_arrow
-
question_answer51)
Anhydrous \[AlC{{l}_{3}}\] is prepared from
A)
cone. \[HCl\] and Al metal done
clear
B)
aluminium and \[C{{l}_{\text{2}}}\] done
clear
C)
dry \[HCl\] gas + heated Al metal done
clear
D)
dil. \[HCl\] and Al metal done
clear
View Solution play_arrow
-
question_answer52)
Boric acid is polymeric due to
A)
its acidic nature done
clear
B)
the presence of hydrogen bonds done
clear
C)
its monobasic .nature done
clear
D)
its geometry done
clear
View Solution play_arrow
-
question_answer53)
\[A{{l}_{2}}{{O}_{3}}\] can be converted to anhydrous \[AlC{{l}_{3}}\] by heating
A)
\[A{{l}_{2}}{{O}_{3}}\] with \[NaCl\] in solid state done
clear
B)
a mixture of \[A{{l}_{2}}{{O}_{3}}\] and carbon in dry \[C{{l}_{2}}\] gas done
clear
C)
\[A{{l}_{2}}{{O}_{3}}\] wither gas done
clear
D)
\[A{{l}_{2}}{{O}_{3}}\] with \[HCl\] gas done
clear
View Solution play_arrow
-
question_answer54)
In which of the following, a salt of the type \[KM{{O}_{2}}\] is obtained?
A)
\[{{B}_{2}}{{H}_{6}}+KOH\,(aq)\to \] done
clear
B)
\[Al+KOH\left( aq \right)\to \] done
clear
C)
Both done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer55)
Bauxite ore is generally contaminated with impurity of oxides of two elements X and Y. Which of the following statement is correct?
A)
X is a non-metal and belongs to the third period while Y is a metal and belongs to the fourth period. done
clear
B)
One of two oxides has three-dimensional polymeric structure. done
clear
C)
Both [a] and [b] are correct. done
clear
D)
None of the above. done
clear
View Solution play_arrow
-
question_answer56)
A solid element (symbol Y) conducts electricity and forms two chlorides \[YC{{l}_{n}}\] (colourless volatile liquid) and \[YC{{l}_{n-2}}\] (a colourless solid). To which one of the following groups of the periodic table does Y belong?
A)
13 done
clear
B)
14 done
clear
C)
15 done
clear
D)
16 done
clear
View Solution play_arrow
-
question_answer57)
The structure and hybridization of \[Si(C{{H}_{3}})\] is
A)
Bent, sp done
clear
B)
Trigonal, \[s{{p}^{2}}\] done
clear
C)
Octahedral, \[{{d}^{2}}s{{p}^{3}}\] done
clear
D)
Tetrahedral, \[s{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer58)
When steam reacts with red hot coke to form \[C{{O}_{2}}\] and hydrogen:
A)
Water acts as an oxidising agent. done
clear
B)
Water acts as a reducing agent. done
clear
C)
Carbon acts as an oxidising agent. done
clear
D)
There is no oxidation or reduction. done
clear
View Solution play_arrow
-
question_answer59)
Which of the following is/are not correctly matched?
| (i) \[Ge{{O}_{2}}\] - Acidic |
| (ii) \[Pb{{O}_{2}}\] - Amphoteric |
| (iii) CO - Neutral |
| (iv) \[Si{{O}_{2}}\] - Amphoteric |
A)
(i) and (iv) done
clear
B)
(iv) only done
clear
C)
(ii) only done
clear
D)
(iii) only done
clear
View Solution play_arrow
-
question_answer60)
The shape of gaseous \[SnC{{l}_{2}}\] is
A)
tetrahedral done
clear
B)
linear done
clear
C)
angular done
clear
D)
T-shaped done
clear
View Solution play_arrow
 The incorrect statement regarding above reactions is:
The incorrect statement regarding above reactions is:  done
clear
done
clear
