Electronic Displacement In Covalent Bonds
Category : JEE Main & Advanced
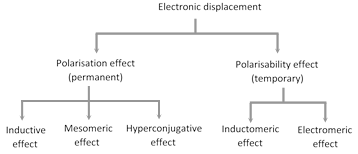
It is observed that most of the attacking reagents always possess either a positive or a negative charge, therefore for a reaction to take place on the covalent bond the latter must possess oppositely charged centres. This is made possible by displacement (partial or complete) of the bonding electrons. The electronic displacement in turn may be due to certain effects, some of which are permanent and others are temporary. The former effects are permanently operating in the molecule and are known as polarisation effects, while the latter are brought into play by the attacking reagent and as soon as the attacking reagent is removed, the electronic displacement disappears; such effects are known as the polarisability effects.
You need to login to perform this action.
You will be redirected in
3 sec
