Using mass (M), length (L), time (T) and current [A] as fundamental quantities, the dimensional formula of permittivity is
A)
\[[M{{L}^{-2}}{{T}^{2}}A]\]
done
clear
B)
\[[{{M}^{-1}}{{L}^{-3}}{{T}^{4}}{{A}^{2}}]\]
done
clear
C)
\[[ML{{T}^{-2}}A]\]
done
clear
D)
\[[M{{L}^{2}}{{T}^{-1}}{{A}^{2}}]\]
done
clear
View Answer play_arrow
A Ge specimen is doped with Al. The concentration of acceptor atoms is\[\sim 1{{0}^{21}}atoms/{{m}^{3}}\]. Given that the intrinsic concentration of electron-hole pairs is \[\sim 1{{0}^{19}}\,/{{m}^{3}}\], the concentration of electrons in the specimen is
A)
\[1{{0}^{17}}\,/{{m}^{3}}\]
done
clear
B)
\[1{{0}^{15}}\,/{{m}^{3}}\]
done
clear
C)
\[1{{0}^{4}}\,/{{m}^{3}}\]
done
clear
D)
\[1{{0}^{2}}\,/{{m}^{3}}\]
done
clear
View Answer play_arrow
The cyclotron frequency of an electron gyrating in a magnetic field of 1 T is approximately
A)
28 MHz
done
clear
B)
280 MHz
done
clear
C)
2.8 GHz
done
clear
D)
28 GHz
done
clear
View Answer play_arrow
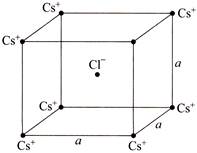
In the basic CsCl crystal structure, \[C{{s}^{+}}\] and \[C{{l}^{-}}\] ions are arranged in a bcc configuration as shown in the figure. The net electrostatic force exerted by the eight \[C{{s}^{+}}\] ions on the \[C{{l}^{-}}\] ion is
A)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\,\,\frac{4{{e}^{2}}}{3{{a}^{2}}}\]
done
clear
B)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\,\,\frac{16{{e}^{2}}}{3{{a}^{2}}}\]
done
clear
C)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\,\,\frac{32{{e}^{2}}}{3{{a}^{2}}}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
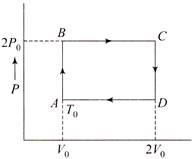
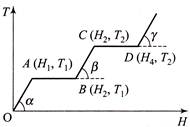
N moles of a monoatomic gas is carried round the reversible rectangular cycle ABCDA as shown in the diagram. The temperature at A is\[{{T}_{0}}\]. The thermodynamic efficiency of the cycle is
A)
\[15\,%\]
done
clear
B)
\[50\,%\]
done
clear
C)
\[20\,%\]
done
clear
D)
\[20\,%\]
done
clear
View Answer play_arrow
An organ pipe closed at one end has fundamental frequency of 1500 Hz. The maximum number of overtones generated by this pipe which a normal person can hear is
A)
4
done
clear
B)
13
done
clear
C)
6
done
clear
D)
9
done
clear
View Answer play_arrow
A monochromatic beam of light is used for the formation of fringes on the screen by illuminating the two slits in the Young's double slit interference experiment. When a thin film of mica is interposed in the path of one of the interfering beams, then
A)
the fringe width increases
done
clear
B)
the fringe width decreases
done
clear
C)
the fringe width remains the same but the pattern shifts
done
clear
D)
the fringe pattern disappears
done
clear
View Answer play_arrow
A bomb of mass 3.0 kg explodes in air into two pieces of masses 2.0 kg and 1.0 kg. The smaller mass goes at a speed of 80 m/s. The total energy imparted to the two fragments is
A)
1.07 kJ
done
clear
B)
2.14 Kj
done
clear
C)
2.4 kJ
done
clear
D)
4.8 kJ
done
clear
View Answer play_arrow
Suppose the sun expands so that its radius becomes 100 times its present radius and its surface temperature becomes half of its present value. The total energy emitted by it then will increase by a factor of
A)
\[{{10}^{4}}\]
done
clear
B)
625
done
clear
C)
256
done
clear
D)
16
done
clear
View Answer play_arrow
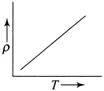
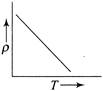
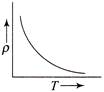
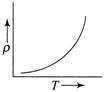
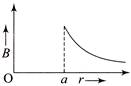
The temperature (7) dependence of resistivity \[(\rho )\] of a semiconductor is represented by:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
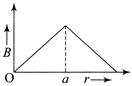
A circular coil of radius R carries an electric current. The magnetic field due to the coil at a point on the axis of the coil located at a distance r from the centre of the coil, such that \[\operatorname{r} >> R\], varies as
A)
\[1/r\]
done
clear
B)
\[1/{{r}^{3}}{{^{/}}^{2}}\]
done
clear
C)
\[1/{{r}^{2}}\]
done
clear
D)
\[1/{{r}^{3}}\]
done
clear
View Answer play_arrow
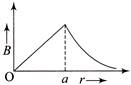
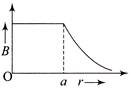
The magnetic field due to a straight conductor of uniform cross-section of radius a and carrying a steady current is represented by
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
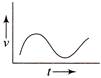
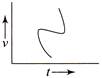
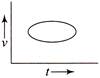
Which of the following velocity-time graphs shows a realistic situation for a body in motion?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Equipotential surfaces associated with an electric field which is increasing in magnitude along the direction are
A)
planes parallel to yz-plane
done
clear
B)
planes parallel to xy-plane
done
clear
C)
planes parallel to xz-plane
done
clear
D)
coaxial cylinders of increasing radii around the x-axis
done
clear
View Answer play_arrow
A proton and an\[\alpha -particle\], moving with the same velocity enter a uniform of their motion. The ratio of the radii of the circular paths described by the proton and a-particle is:
A)
\[1:2\]
done
clear
B)
\[1 : 4\]
done
clear
C)
\[1 : 16\]
done
clear
D)
\[4\,\,:\,\,1\]
done
clear
View Answer play_arrow
A nucleus of mass number A, originally at rest, emits an \[\alpha -particle\] with speed v. The daughter nucleus recoils with a speed
A)
\[\frac{2\,\nu }{A+4}\]
done
clear
B)
\[\frac{4\,\nu }{A+4}\]
done
clear
C)
\[\frac{4\,\nu }{A-4}\]
done
clear
D)
\[\frac{2\,\nu }{A-4}\]
done
clear
View Answer play_arrow
When an electron-positron pair annihilates, the energy released is about
A)
\[0.8 \times \,\,{{10}^{-}}^{13}\,J\]
done
clear
B)
\[1.6 \times \,\,{{10}^{-}}^{13}\,J\]
done
clear
C)
\[3.2 \,\times \,\,{{10}^{-}}^{13}\,J\]
done
clear
D)
\[4.8 \,\times \,\,{{10}^{-}}^{13}\,J\]
done
clear
View Answer play_arrow
A photon of energy 4 eV is incident on a metal surface whose work function is 2 eV. The minimum reverse potential to be applied for stopping the emission of electrons is
A)
2 V
done
clear
B)
4 V
done
clear
C)
6 V
done
clear
D)
8 V
done
clear
View Answer play_arrow
We wish to see inside an atom. Assuming the atom to have a diameter of 100 pm, this means that one must be able to resolve a width of say 10 pm. If an electron microscope is used, the minimum electron energy required is about
A)
1.5 keV
done
clear
B)
15 keV
done
clear
C)
150 keV
done
clear
D)
1.5 MeV
done
clear
View Answer play_arrow
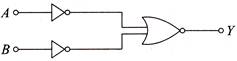
Which logic gate is represented by the following combination of logic gates?
A)
OR
done
clear
B)
NAND
done
clear
C)
AND
done
clear
D)
NOR
done
clear
View Answer play_arrow
A particle of mass m is moving in a horizontal circle of radius r under a centripetal force equal to\[-K/{{r}^{2}}\], where K is a constant. The total energy of the particle is
A)
\[\frac{K}{2r}\]
done
clear
B)
\[-\frac{K}{2r}\]
done
clear
C)
\[-\frac{K}{r}\]
done
clear
D)
\[\frac{K}{r}\]
done
clear
View Answer play_arrow
A ray of light is incident at \[50{}^\circ \] on the middle, of one of the two mirrors arranged at an angle of \[60{}^\circ \] between them. The ray then touches the second mirror, get reflected back to the first mirror, making an angle of incidence of
A)
\[50{}^\circ \]
done
clear
B)
\[60{}^\circ \]
done
clear
C)
\[70{}^\circ \]
done
clear
D)
\[80{}^\circ \]
done
clear
View Answer play_arrow
A point object is placed at a distance of 30 cm from a convex mirror of focal length 30 cm. The image will form at
A)
Infinity
done
clear
B)
Focus
done
clear
C)
Pole
done
clear
D)
15 cm behind the mirror
done
clear
View Answer play_arrow
The graph shows the variation of temperature (T) of one kilogram of a material with the heat (H) supplied to it. At O, the substance is in the solid state. From the graph, we can conclude that
A)
\[{{T}_{2}}\] is the melting point of the solid
done
clear
B)
BC represents the change of state from solid to liquid
done
clear
C)
\[\left( {{H}_{2}}-{{H}_{1}} \right)\] represents the latent heat of fusion of the substance
done
clear
D)
\[\left( {{H}_{3}}-{{H}_{1}} \right)\] represents the latent heat of vaporization of the liquid
done
clear
View Answer play_arrow
A disc and a ring of same mass are rolling and if their kinetic energies are equal, then the ratio of their velocities will be
A)
\[\sqrt{4}\,\,:\,\,\sqrt{3}\]
done
clear
B)
\[\sqrt{3}\,\,:\,\,\sqrt{4}\]
done
clear
C)
\[\sqrt{3}\,\,:\,\,\sqrt{2}\]
done
clear
D)
\[\sqrt{2}\,\,:\,\,\sqrt{3}\]
done
clear
View Answer play_arrow
Select the pair whose dimensions are the same
A)
Pressure and stress
done
clear
B)
Stress and strain
done
clear
C)
Pressure and force
done
clear
D)
Power and force
done
clear
View Answer play_arrow
A body is moving from rest under constant acceleration and let \[{{S}_{1}}\] be the displacement in the first \[(p-1)\] sec and \[{{S}_{2}}\] be the displacement in the first p sec. The displacement in \[\left( {{p}^{2}}-p +1 \right)th\] sec will be
A)
\[{{\operatorname{S}}_{1}}+{{S}_{2}}\]
done
clear
B)
\[{{\operatorname{S}}_{1}}{{S}_{2}}\]
done
clear
C)
\[{{\operatorname{S}}_{1}}-{{S}_{2}}\]
done
clear
D)
\[{{\operatorname{S}}_{1}}/{{S}_{2}}\]
done
clear
View Answer play_arrow
If a particle of mass m is moving in a horizontal circle of radius r with a centripetal force \[\left( -k/{{r}^{2}} \right)\]the total energy is
A)
\[-\frac{k}{2r}\]
done
clear
B)
\[-\frac{k}{r}\]
done
clear
C)
\[-\frac{2\,k}{r}\]
done
clear
D)
\[-\frac{4\,k}{r}\]
done
clear
View Answer play_arrow
A machine gun is mounted on a 2000 kg car on a horizontal frictionless surface. At some instant the gun fires bullets of mass 10 gm with a velocity of 500 m/sec with respect to the car. The number of bullets fired per second is ten. The average thrust on the system is
A)
550 N
done
clear
B)
50 N
done
clear
C)
250 N
done
clear
D)
250 dyne
done
clear
View Answer play_arrow
A projectile fired with initial velocity u at some jl angle \[\theta \] has a range R. If the initial velocity be j doubled at the same angle of projection, then the range will be
A)
2R
done
clear
B)
R/2
done
clear
C)
R
done
clear
D)
4R
done
clear
View Answer play_arrow
When a 1.0 kg mass hangs attached to a spring of length 50 cm, the spring stretches by 2 cm. The mass is pulled down until the length of the spring becomes 60 cm. What is the amount of elastic energy stored in the spring in this condition, if \[\operatorname{g}= 10 m/{{s}^{2}}\]
A)
1.5 Joule
done
clear
B)
2.0 Joule
done
clear
C)
2.5 Joule
done
clear
D)
3.0 Joule
done
clear
View Answer play_arrow
A body weighs 700 gm wt on the surface of the earth. How much will it weigh on the surface of a planet whose mass is 1/7 and radius is half that of the earth?
A)
200 gm wt
done
clear
B)
400gmwt
done
clear
C)
50 gm wt
done
clear
D)
300 gm wt
done
clear
View Answer play_arrow
Increase in length of a wire is 1 mm when suspended by a weight. If the same weight is suspended on a wire of double its length and double its radius, the increase in length will be
A)
2 mm
done
clear
B)
0.5 mm
done
clear
C)
4 mm
done
clear
D)
0.25 mm
done
clear
View Answer play_arrow
Energy needed in breaking a drop of radius R into n drops of radii r is given by
A)
\[4\pi T\left( n{{r}^{2}}-{{R}^{2}} \right)\]
done
clear
B)
\[\frac{4}{3}\pi \left( {{r}^{3}}n-{{R}^{2}} \right)\]
done
clear
C)
\[4\pi T\left( {{R}^{2}}-{{n}^{2}} \right)\]
done
clear
D)
\[4\pi T\left( n{{r}^{2}}+{{R}^{2}} \right)\]
done
clear
View Answer play_arrow
The volume of an air bubble becomes three times as it rises from the bottom of a lake to its surface. Assuming atmospheric pressure to be 75 cm of Hg and the density of water to be 1/10 J of the density of mercury, the depth of the lake is
A)
5 m
done
clear
B)
10 m
done
clear
C)
15 m
done
clear
D)
20 m
done
clear
View Answer play_arrow
A pendulum clock keeps correct time at\[0{}^\circ C\]. Its mean coefficient of linear expansions is \[\alpha /{}^\circ C\], then the loss in seconds per day by the clock if the temperature rises by \[t{}^\circ C\] is
A)
\[\frac{\frac{1}{2}\alpha t\times 864000}{1-\frac{\alpha t}{2}}\]
done
clear
B)
\[\frac{1}{2}\alpha t\times 86400\]
done
clear
C)
\[\frac{\frac{1}{2}\alpha t\times 86400}{{{\left( 1-\frac{\alpha t}{2} \right)}^{2}}}\]
done
clear
D)
\[\frac{\frac{1}{2}\alpha t\times 86400}{1-\frac{\alpha t}{2}}\]
done
clear
View Answer play_arrow
One mole of a monoatomic ideal gas is mixed with one mole of a diatomic ideal gas. The molar specific heat of the mixture at constant volume is
A)
8
done
clear
B)
\[\frac{3}{2}\,R\]
done
clear
C)
2R
done
clear
D)
2.5 R
done
clear
View Answer play_arrow
A system is provided with 200 cal of heat and the work done by the system on the surrounding is 40 J. Then its internal energy
A)
Increases by 600 J
done
clear
B)
Decreases by 800 J
done
clear
C)
Increases by 800 J
done
clear
D)
Decreases by 50 J
done
clear
View Answer play_arrow
If the radius and length of a copper rod are both doubled, the rate of flow of heat along the rod increases
A)
4 times
done
clear
B)
2 times
done
clear
C)
8 times
done
clear
D)
16 times
done
clear
View Answer play_arrow
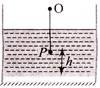
A plane mirror is placed at the bottom of the tank containing a liquid of refractive index\[\mu \]. P is a small object at a height h above the mirror. An observer O-vertically above P outside the liquid see P and its image in the mirror. The apparent distance between these two will be
A)
\[2\mu h\]
done
clear
B)
\[\frac{2h}{\mu }\]
done
clear
C)
\[\frac{2h}{\mu -1}\]
done
clear
D)
\[h\left( 1+\frac{1}{\mu } \right)\]
done
clear
View Answer play_arrow
Three rods each of length L and mass M are placed along X, Y and Z-axes in such a way that one end of each of the rod is at the origin. The moment of inertia of this system about Z axis is
A)
\[\frac{2\,M{{L}^{2}}}{3}\]
done
clear
B)
\[\frac{4\,M{{L}^{2}}}{3}\]
done
clear
C)
\[\frac{5\,M{{L}^{2}}}{3}\]
done
clear
D)
\[\frac{M{{L}^{2}}}{3}\]
done
clear
View Answer play_arrow
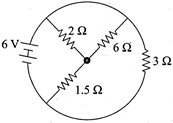
The total current supplied to the circuit by the battery is
A)
1 A
done
clear
B)
2 A
done
clear
C)
4 A
done
clear
D)
6 A
done
clear
View Answer play_arrow
A short magnet is allowed to fall along the axis of a horizontal metallic ring. Starting from rest, the distance fallen by the magnet in one second may be
A)
4 m
done
clear
B)
5 m
done
clear
C)
6 m
done
clear
D)
7 m
done
clear
View Answer play_arrow
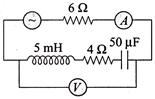
In the circuit shown in the figure, the ac source gives a voltage\[\operatorname{V}=20 cos \left( 2000 t \right)\]. Neglecting source resistance, the voltmeter and ammeter reading will be
A)
0 V, 0.47 A
done
clear
B)
1.68 V, 0.47 A
done
clear
C)
0 V, 1.4 A
done
clear
D)
5.6 V, 1.4 A
done
clear
View Answer play_arrow
Which of the transitions in hydrogen atom emits a photon of lowest frequency (n = quantum number)
A)
\[n= 2 to\,\,n =1\]
done
clear
B)
\[n =4 to\,\,n = 3\]
done
clear
C)
\[n= 3 \,to\,\,n = 1\]
done
clear
D)
\[n = 4\,\,to\,\,n = 2\]
done
clear
View Answer play_arrow
The vapour pressures of ethanol and methanol are 42.0 mm and 88.5 mm Hg respectively. An ideal solution is formed at the same temperature by mixing 46.0 g of ethanol with 16.0 g of methanol. The mole fraction of methanol in the vapour is:
A)
0.467
done
clear
B)
0.502
done
clear
C)
0.513
done
clear
D)
0.556
done
clear
View Answer play_arrow
Faraday's laws of electrolysis are related to the
A)
Atomic number of cation
done
clear
B)
Atomic number of anion
done
clear
C)
Equivalent weight of the electrolyte
done
clear
D)
Speed of the cation
done
clear
View Answer play_arrow
The decomposition of \[C{{l}_{2}}{{O}_{7}}\] at 400 K in the gas phase to \[C{{l}_{2}}\] and \[{{O}_{2}}\] is a first-order reaction. After 55 seconds at 400 K the pressure of \[C{{l}_{2}}{{O}_{7}}\] falls from 0.062 to 0.044 atm. The rate constant and pressure of \[C{{l}_{2}}{{O}_{7}}\] after 100 sec of decomposition at this temperature are
A)
\[5.2 \times 1{{0}^{-}}^{4}se{{c}^{-}}^{1}; \,0.05 atm\]
done
clear
B)
\[6.2 \times 1{{0}^{-}}^{3}se{{c}^{-}}^{1}; \,0.033 atm\]
done
clear
C)
\[5.8 \times 1{{0}^{-}}^{3}se{{c}^{-}}^{1}; \,0.44 atm\]
done
clear
D)
\[4.6 \times 1{{0}^{-}}^{3}se{{c}^{-}}^{1}; \,0.005 atm\]
done
clear
View Answer play_arrow
The boiling point of water \[\left( 100{}^\circ C \right)\] becomes\[100.52{}^\circ C\], if 3 grams of a nonvolatile solute is dissolved in 200 ml of water. The molecular weight of solute is (\[{{K}_{b}}\], for water is 0.6 K/m)
A)
\[12.2\,\,g\,mo{{l}^{-1}}\]
done
clear
B)
15.4 g mol
done
clear
C)
\[17.3\,\,g\,mo{{l}^{-1}}\]
done
clear
D)
20.4 g mol
done
clear
View Answer play_arrow
The limiting radius ratio of the complex \[{{\left[ Ni\left( C{{N}_{4}} \right) \right]}^{2-}}\]
A)
0.225-0.414
done
clear
B)
0.414-0.732
done
clear
C)
0.155-0.225
done
clear
D)
None
done
clear
View Answer play_arrow
Which of the following plots represents the behaviour of an ideal binary liquid solution?
A)
Plot of \[{{P}_{total}}\,vs\,{{Y}_{A}}\] (mole fraction of A in vapour phase) is linear
done
clear
B)
Plot of \[{{P}_{total}}\,vs\,{{Y}_{B}}\] is linear
done
clear
C)
Plot of \[1/{{P}_{total}}\,vs\,{{Y}_{A}}\] is linear
done
clear
D)
Plot of \[1/{{P}_{total}}\,vs\,{{Y}_{B}}\] 1s non-linear
done
clear
View Answer play_arrow
Normal boiling point of water is 373 K (at 760 mm). Vapour pressure of water at 298 K is 23 mm. If y the enthalpy of evaporation is 40.656 kJ/mole, the boiling point of water at 23 mm pressure will be
A)
250 K
done
clear
B)
294 K
done
clear
C)
51.6 K
done
clear
D)
12.5 K
done
clear
View Answer play_arrow
The required charge for one equivalent weight n U of silver deposit on cathode is
A)
\[9.65 \times 1{{0}^{7}}C~\]
done
clear
B)
\[9.65 \times 1{{0}^{4}}C~\]
done
clear
C)
\[9.65 \times 1{{0}^{3}}C~\]
done
clear
D)
\[9.65 \times 1{{0}^{5}}C~\]
done
clear
View Answer play_arrow
A substance having a half-life period of 30 minutes decomposes according to the first-order rate law. The fraction is decomposed, and the balance remaining after 1.5 hours and time for \[60%\] decomposition on its doubling the initial concentration will be
A)
87.4; 0.126; 39.7 mins
done
clear
B)
80.6; 0.135; 40.8 mins
done
clear
C)
90.5; 0.144; 2829 mins
done
clear
D)
802; 0.135; 26.6 mins
done
clear
View Answer play_arrow
Which of the following electrolytes is most effective in the coagulation of gold solution?
A)
\[NaN{{O}_{3}}\]
done
clear
B)
\[{{K}_{4}}[Fe(C{{N}_{6}})]\]
done
clear
C)
\[N{{a}_{3}}P{{O}_{4}}\]
done
clear
D)
\[MaC{{l}_{2}}\]
done
clear
View Answer play_arrow
Extraction of Ag from sulphide ore and removal of unreacted silver bromide from photographic plate involve complexes:
A)
\[{{\left[ Ag{{({{S}_{2}}{{O}_{3}})}_{2}} \right]}^{3-}}\,\,in\,\,both\]
done
clear
B)
\[{{[Ag{{(CN)}_{2}}]}^{-}}\,\,in\,\,both\]
done
clear
C)
\[{{[Ag{{({{S}_{2}}{{O}_{3}})}_{2}}]}^{3-}},\,\,{{[Ag{{(CN)}_{2}}]}^{-}}\,\,respectively\]
done
clear
D)
\[{{[Ag{{(CN)}_{2}}]}^{-}}\,,\,\,{{[Ag{{({{S}_{2}}{{O}_{3}})}_{2}}]}^{3-}}\,\,respectively\]
done
clear
View Answer play_arrow
Close packing is maximum in the crystal which is
A)
Simple cube
done
clear
B)
bcc
done
clear
C)
fcc
done
clear
D)
none
done
clear
View Answer play_arrow
A catalyst is used in a reaction to
A)
Change the nature of reaction products
done
clear
B)
Increase the reaction yield
done
clear
C)
Decrease the need for reactants
done
clear
D)
Decrease the time required for the reaction
done
clear
View Answer play_arrow
Which of the following has maximum thermo-dynamic spontaneity at \[400{}^\circ C\]?
A)
\[C+{{O}_{2}}\,\,\xrightarrow{{}}\,\,C{{O}_{2}}\]
done
clear
B)
\[2C\,\,+\,\,{{O}_{2}}\,\,\xrightarrow{{}}\,\,2CO\]
done
clear
C)
\[2CO+{{O}_{2}}\,\,\xrightarrow{{}}\,\,2C{{O}_{2}}\]
done
clear
D)
All have same
done
clear
View Answer play_arrow
The boiling points of the following hydrides follow the order of
A)
\[N{{H}_{3}} > As{{H}_{3}} > P{{H}_{3}} > Sb{{H}_{3}}\]
done
clear
B)
\[Sb{{H}_{3}} > As{{H}_{3}} > P{{H}_{3}} > N{{H}_{3}}\]
done
clear
C)
\[Sb{{H}_{3}} > N{{H}_{3}} > As{{H}_{3}} > P{{H}_{3}}\]
done
clear
D)
\[N{{H}_{3}} > P{{H}_{3}} > As{{H}_{3}} > Sb{{H}_{3}}\]
done
clear
View Answer play_arrow
Potassium permanganate acts as an oxidant in neutral, alkaline as well as acidic media. The final products obtained from it in the three conditions are, respectively,
A)
\[Mn{{O}_{2}},\,\,Mn{{O}_{2}},\,\,M{{n}^{2+}}\]
done
clear
B)
\[M{{n}^{2-}}_{4},\,\,M{{n}^{3+}},\,\,M{{n}^{2+}}\]
done
clear
C)
\[Mn{{O}_{2}},\,\,Mn{{O}^{2-}},\,\,M{{n}^{3+}}\]
done
clear
D)
\[MnO,\,\,Mn{{O}_{4}},\,\,M{{n}^{2+}}\]
done
clear
View Answer play_arrow
Which of the following oxyacids of phosphorus is a reducing agent and monobasic?
A)
\[{{\operatorname{H}}_{3}}P{{O}_{2}}\]
done
clear
B)
\[{{\operatorname{H}}_{3}}P{{O}_{3}}\]
done
clear
C)
\[{{\operatorname{H}}_{3}}P{{O}_{4}}\]
done
clear
D)
\[{{H}_{4}}{{P}_{2}}{{O}_{6}}~~\]
done
clear
View Answer play_arrow
In acidic medium potassium dichromate acts as an oxidant according to the equation, \[{{\operatorname{CrO}}_{7}}^{2-}+\,\,14{{H}^{+}}+6{{e}^{-}}\,\,\to \,\,2C{{r}^{3+}}\,+\,\,7{{H}_{2}}O\]. What is the equivalent weight of\[{{\operatorname{K}}_{2}}C{{r}_{2}}{{O}_{7}}\]?\[(\operatorname{mol}. wt.=M)\]
A)
M
done
clear
B)
M/2
done
clear
C)
M/3
done
clear
D)
M/6
done
clear
View Answer play_arrow
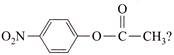
2-bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is
A)
Pent-1-ene
done
clear
B)
cis-pent-2-ene
done
clear
C)
trans-pent-2-ene
done
clear
D)
2-ethoxypentane
done
clear
View Answer play_arrow
In the process of extraction of gold, Roasted gold ore
\[{{\operatorname{CN}}^{-}}+{{H}_{2}}O\,\,\xrightarrow{{{O}_{2}}} [X]\,\,+\,\,O{{H}^{-}}\] \[[X]\,\,+\,\,Zn\,\,\to \,\,[Y]\,\,+\,\,Au\] Identify the complexes [X] and [Y]
A)
\[\operatorname{X}={{\left[ Au{{\left( CN \right)}_{2}} \right]}^{-}},\,\,Y={{\left[ Zn{{\left( CN \right)}_{4}} \right]}^{2-}}\]
done
clear
B)
\[\operatorname{X}={{\left[ Au{{\left( CN \right)}_{4}} \right]}^{3-}},\,\,Y={{\left[ Zn{{\left( CN \right)}_{4}} \right]}^{2-}}\]
done
clear
C)
\[\operatorname{X}={{\left[ Au{{\left( CN \right)}_{2}} \right]}^{-}},\,\,Y={{\left[ Zn{{\left( CN \right)}_{6}} \right]}^{4-}}\]
done
clear
D)
\[\operatorname{X}={{\left[ Au{{\left( CN \right)}_{4}} \right]}^{-}},\,\,Y={{\left[ Zn{{\left( CN \right)}_{4}} \right]}^{2-}}\]
done
clear
View Answer play_arrow
In which alkyl halide, \[{{S}_{N}}2\] mechanism is favoured maximum by
A)
\[C{{H}_{3}}Cl\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}Cl\]
done
clear
C)
\[{{\left( C{{H}_{3}} \right)}_{2}}CHCl\]
done
clear
D)
\[{{\left( C{{H}_{3}} \right)}_{3}}C-Cl\]
done
clear
View Answer play_arrow
The \[-OH\] group of methyl alcohol cannot be replaced by chlorine by the action of
A)
Chlorine
done
clear
B)
Hydrogen chloride
done
clear
C)
Phosphorus trichloride
done
clear
D)
Phosphorus pentachloride
done
clear
View Answer play_arrow
Which of the following will not give a precipitate with\[AgN{{O}_{3}}\]?
A)
\[\left[ Co{{\left( N{{H}_{3}} \right)}_{3}}C{{l}_{3}} \right]\]
done
clear
B)
\[\left[ Co{{\left( N{{H}_{3}} \right)}_{4}}C{{l}_{2}} \right]Cl\]
done
clear
C)
\[\left[ Co{{\left( N{{H}_{3}} \right)}_{5}}Cl \right]C{{l}_{2}}\]
done
clear
D)
\[\left[ Co{{\left( N{{H}_{3}} \right)}_{6}} \right]C{{l}_{3}}\]
done
clear
View Answer play_arrow
The alcohol which easily reacts with cone. HCl is
A)
\[C{{H}_{3}}CHOHC{{H}_{2}}C{{H}_{3}}\]
done
clear
B)
\[{{\left( C{{H}_{3}} \right)}_{3}}COH\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}{{C}_{2}}C{{H}_{2}}OH\]
done
clear
D)
\[{{\left( C{{H}_{3}} \right)}_{3}}CHC{{H}_{2}}OH\]
done
clear
View Answer play_arrow
How will you distinguish the products obtained by acidic hydrolysis of I
II
A)
Fehling's solution
done
clear
B)
\[NaHS{{O}_{3}}\]
done
clear
C)
Brady?s reagent
done
clear
D)
Lucas test
done
clear
View Answer play_arrow
Mendius reaction involves the
A)
reduction of aldehydes to give alcohols
done
clear
B)
Reduction of nitriles with sodium and ethanol
done
clear
C)
Oxidation of nitrites
done
clear
D)
Hydrolysis of cyanides
done
clear
View Answer play_arrow
The two polypeptide chains of insulin are cross- linked at
A)
two places
done
clear
B)
three places
done
clear
C)
four places
done
clear
D)
five places
done
clear
View Answer play_arrow
Which of the following amines reacts most rapidly with
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
\[\underset{(A)}{\mathop{PhCONH}}\,\xrightarrow[KOBr]{}\,\,(B)\,\,\xrightarrow[MeOH]{}\,(C)\] Product [c] is
A)
\[PhN{{H}_{2}}\]
done
clear
B)
\[PhNHCOOMe\]
done
clear
C)
\[PhNHCOOPh\]
done
clear
D)
None
done
clear
View Answer play_arrow
Which one of the following is employed as a tranquillizer?
A)
Naproxen
done
clear
B)
Tetracycline
done
clear
C)
Chlorpheniramine
done
clear
D)
Equanil
done
clear
View Answer play_arrow
0.5 gm of fuming \[{{H}_{2}}S{{O}_{4}}\] (oleum) is diluted with water. This solution is completely neutralised by 26.7 ml of 0.4 N NaOH. The percentage of free 803 in the sample is
A)
\[30.6\,%\]
done
clear
B)
\[40.6\,%\]
done
clear
C)
\[20.6\,%\]
done
clear
D)
\[50\,%\]
done
clear
View Answer play_arrow
Which one of the following is considered as the main postulates of Bohr?s model of atom?
A)
Protons are present in the nucleus
done
clear
B)
Electrons are revolving around the nucleus
done
clear
C)
Centrifugal force produced due to the revolving electrons balances the force of attraction between the electron and the protons
done
clear
D)
Angular momentum of electron is an integral multiple of \[\frac{h}{2\pi }\]
done
clear
View Answer play_arrow
The number of moles present in \[1\,{{m}^{3}}\] of any gas At NTP is
A)
32.4
done
clear
B)
54.6
done
clear
C)
44.6 g
done
clear
D)
28.2
done
clear
View Answer play_arrow
Styrene at room temperature is
A)
Solid
done
clear
B)
Liquid
done
clear
C)
Gas
done
clear
D)
Colloidal solution
done
clear
View Answer play_arrow
Rutherford?s scattering experiment is related to the size of the
A)
Nucleus
done
clear
B)
Atom
done
clear
C)
Electron
done
clear
D)
Neutron
done
clear
View Answer play_arrow
The elements that occupy the peaks of ionisation energy curve are
A)
Na, K, Rb, Cs
done
clear
B)
Na, Mg, Cl, I
done
clear
C)
Cl, Br, I, F
done
clear
D)
He, Ne, Ar, Kr
done
clear
View Answer play_arrow
The number of S - S bonds in sulphur trioxide trimer \[{{S}_{3}}{{O}_{9}}\] is
A)
Three
done
clear
B)
Two
done
clear
C)
One
done
clear
D)
Zero
done
clear
View Answer play_arrow
In which of the following the central atom does A not use \[s{{p}^{3}}\] hybrid orbitals in its bonding
A)
\[BeF_{3}^{-}\]
done
clear
B)
\[OH_{3}^{+}\]
done
clear
C)
\[NH_{2}^{{}}\]
done
clear
D)
\[N{{F}_{3}}\]
done
clear
View Answer play_arrow
The first ionization potentials in electron volts of nitrogen and oxygen atoms are respectively given by
A)
14.6, 13.6
done
clear
B)
13.6, 14.6
done
clear
C)
13.6, 13.6
done
clear
D)
14.6, 14.6
done
clear
View Answer play_arrow
What is the value of \[\Delta E\] (heat change at constant volume) for reversible isothermal evaporation of 90 g water\[100{}^\circ C\]. Assuming water vapour behaves as an ideal gas and\\[{{\left( \Delta {{H}_{vap}} \right)}_{water}}=540\,cal\,\,{{g}^{-1}}\].
A)
\[9\times {{10}^{3}}\,cals\]
done
clear
B)
\[6\times {{10}^{3}}\,cals\]
done
clear
C)
4.49 cals
done
clear
D)
none of these
done
clear
View Answer play_arrow
Dalton' s law of partial pressure is not applicable to, at normal conditions
A)
\[{{H}_{2}}\,and\,\,{{N}_{3}}\,\,mixture\]
done
clear
B)
\[{{H}_{2}}\,and\,\,C{{l}_{2}}\,\,mixture\]
done
clear
C)
\[{{H}_{2}}\,and\,\,C{{O}_{2}}\,\,mixture\]
done
clear
D)
\[{{H}_{2}}\,and\,\,{{O}_{2}}\,\,mixture\]
done
clear
View Answer play_arrow
Calculate \[\Delta G{}^\circ \] for conversion of oxygen to (ozone\[3/2\,{{O}_{2}}(g)\to at\,\,298\,\,K\], at 298 K, if \[{{K}_{p}}\] for this conversion is\[2.47 \times 1{{0}^{-}}^{29}\].
A)
\[1.63 kJ mo{{l}^{-1}}\]
done
clear
B)
\[2.4\,\,\times \,\,{{10}^{2}}\,kJ\,mo{{l}^{\text{-}1}}\]
done
clear
C)
\[1.63\,\,kJ\,mo{{l}^{\text{-}1}}\]
done
clear
D)
\[2.8\,\,\times \,\,{{106}^{2}}\,kJ\,mo{{l}^{\text{-}1}}\]
done
clear
View Answer play_arrow
\[\Delta G{}^\circ \left( HI, g \right) \cong +1.7 kJ\]. What is the equilibrium constant at \[25{}^\circ C\] for\[2HI\,(g)\,\,\rightleftharpoons \,\,{{H}_{2}}(g)\,\,+\,\,{{I}_{2}}\,(g)\].
A)
24.0
done
clear
B)
3.9
done
clear
C)
2.0
done
clear
D)
0.5
done
clear
View Answer play_arrow
Helium atom is two times heavier than a hydrogen molecule. At 298 K, the average kinetic energy of a Helium atom is
A)
two times that of hydrogen molecule
done
clear
B)
same as that of a hydrogen molecule
done
clear
C)
four times that of a hydrogen molecule
done
clear
D)
half that of a hydrogen molecule
done
clear
View Answer play_arrow
Which of the statement is correct?
A)
Slope of adiabatic \[P-V\] curve will be the same as that of isothermal one.
done
clear
B)
Slope of adiabatic \[P-V\] curve is smaller than that in isothermal one.
done
clear
C)
Slope of adiabatic \[P-V\] curve is larger than that in isothermal one.
done
clear
D)
Slope of adiabatic \[P-V\] curve may be smaller or larger depending on the value V.
done
clear
View Answer play_arrow
If Mendel might have studied 7 pairs of characters in a plant with 12 chromosomes instead of 14 then
A)
He could not discovered independent assortment
done
clear
B)
He might have discovered linkage
done
clear
C)
He might have discovered crossing over
done
clear
D)
He might have not observed dominance
done
clear
View Answer play_arrow
Contraction in gall bladder stimulated by
A)
CCK
done
clear
B)
PZ
done
clear
C)
Secretin
done
clear
D)
Enterogastrone
done
clear
View Answer play_arrow
Water is essential for bryophyta
A)
For fertilization and homosporos nature
done
clear
B)
Water should be filled in archegonium for fertilization
done
clear
C)
Water is necessary for movement of sperm
done
clear
D)
For dissemination of spores
done
clear
View Answer play_arrow
Which of the following yields citric acid?
A)
Penicillium citrinum
done
clear
B)
Aspergillus niger
done
clear
C)
Saccharomyces
done
clear
D)
Azospirillum
done
clear
View Answer play_arrow
Saccharomyces cerevisiae is used in the formation of
A)
Ethanol
done
clear
B)
Methanol
done
clear
C)
Acetic acid
done
clear
D)
Antibiotics
done
clear
View Answer play_arrow
AA Bb Cc genotypes form how many types of gametes
A)
4
done
clear
B)
8
done
clear
C)
2
done
clear
D)
6
done
clear
View Answer play_arrow
Indicator of water pollution
A)
E. coli
done
clear
B)
Chlorella
done
clear
C)
Beggiatoa
done
clear
D)
Ulothrix
done
clear
View Answer play_arrow
DNA of E. coli
A)
ds circular
done
clear
B)
ss circular
done
clear
C)
ds linear
done
clear
D)
ss linear
done
clear
View Answer play_arrow
Nucleic acid in HIV
A)
ss RNA
done
clear
B)
ds RNA
done
clear
C)
ss DNA
done
clear
D)
ds DNA
done
clear
View Answer play_arrow
Knife of DNA
A)
DNA-ligase
done
clear
B)
Restriction endonuclease
done
clear
C)
Exonuclease
done
clear
D)
Peptidase
done
clear
View Answer play_arrow
Genetic engineering involves
A)
Use of restriction endonuclease on bacterial DNA and formation of new traits
done
clear
B)
Use of ligase for cutting DNA
done
clear
C)
Developing instruments
done
clear
D)
Use of statistic in genetics
done
clear
View Answer play_arrow
Which is wrong for cytochrome P-450?
A)
It contains Fe
done
clear
B)
It concerns with oxidation
done
clear
C)
It is a pigment
done
clear
D)
It is a coloured cell
done
clear
View Answer play_arrow
Enamel of teeth is secreted by
A)
Ameloblast
done
clear
B)
Odontoblast
done
clear
C)
Osteoblast
done
clear
D)
Osteoclast
done
clear
View Answer play_arrow
If a female having gene for haemophilia and colour blindness on its one X-chromosome marries a normal male, then what are the chances in their offspring
A)
\[50\,%\] son are diseased and \[50\,%\] are normal
done
clear
B)
All normal offspring
done
clear
C)
\[100%\] daughters are carrier
done
clear
D)
\[100%\] son are diseased
done
clear
View Answer play_arrow
First child of a normal male and female is albino, what are the chances of second child to be albino
A)
\[25\,%\]
done
clear
B)
\[50\,%\]
done
clear
C)
\[75\,%\]
done
clear
D)
\[100\,%\]
done
clear
View Answer play_arrow
Species separated by geographical barriers are called
A)
Allopatric
done
clear
B)
Sympatric
done
clear
C)
Sibling
done
clear
D)
Endemic
done
clear
View Answer play_arrow
Point mutation induced by
A)
Adenine
done
clear
B)
Guanine
done
clear
C)
3-cytosine
done
clear
D)
Bromouracil
done
clear
View Answer play_arrow
Reason for trisomy in Down's syndrome
A)
Non-disjunction during sperm formation
done
clear
B)
Non-disjunction during egg formation
done
clear
C)
Non-disjunction at the time of egg or sperm formation
done
clear
D)
Addition of one extra chromosome during mitosis
done
clear
View Answer play_arrow
Multivalent chromosome form by
A)
Inversion
done
clear
B)
Deletion
done
clear
C)
Reciprocal translocation
done
clear
D)
Point mutation
done
clear
View Answer play_arrow
A cup has 105 bacterial cells. Each bacterial cell divides into 35 minutes. What shall be the number of bacteria after 175 min?
A)
\[2\,\,\times \,\,{{10}^{5}}\]
done
clear
B)
\[5\,\,\times \,\,{{10}^{5}}\]
done
clear
C)
\[32\,\,\times \,\,{{10}^{5}}\]
done
clear
D)
\[16\,\,\times \,\,{{10}^{5}}\]
done
clear
View Answer play_arrow
Deficiency of protein leads to
A)
Rickets
done
clear
B)
Scurvy
done
clear
C)
Kwashiorkor
done
clear
D)
Carotenemia
done
clear
View Answer play_arrow
Lactose is composed of
A)
Glucose + galactose
done
clear
B)
Glucose + fructose
done
clear
C)
Glucose + glucose
done
clear
D)
Glucose + mannose
done
clear
View Answer play_arrow
True statement for cellulose molecule
A)
\[\beta \] -1-4 linkage, unbranched
done
clear
B)
\[\beta \]-1-4 linkage, branched
done
clear
C)
\[\beta \] -1-4 linkage, branched
done
clear
D)
\[\beta \] -1-6 linkage, unbranched
done
clear
View Answer play_arrow
True statement for Ulothrix
A)
Filamentous thallus and flagellated reproductive structures
done
clear
B)
Branched thallus
done
clear
C)
Flagellated cells absent
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Which of the following exercise a control over transcription?
A)
Operator
done
clear
B)
Regulator
done
clear
C)
Promoter
done
clear
D)
Recon
done
clear
View Answer play_arrow
Vitamin which induces maturation of RBC
A)
B1
done
clear
B)
A
done
clear
C)
B12
done
clear
D)
D
done
clear
View Answer play_arrow
Lower jaw is composed of
A)
Dentary
done
clear
B)
Maxilla
done
clear
C)
Premaxilla
done
clear
D)
Palatine
done
clear
View Answer play_arrow
Number of bones in hind limb of man is
A)
14
done
clear
B)
24
done
clear
C)
26
done
clear
D)
30
done
clear
View Answer play_arrow
Which of the following stimulates the secretion of gastric juice?
A)
Gastrin
done
clear
B)
Enterogastrone
done
clear
C)
Secretin
done
clear
D)
Hepatocrinin
done
clear
View Answer play_arrow
Age of Dryopithecus is
A)
2.46 crore years
done
clear
B)
2.46 lakh year
done
clear
C)
1 lakh year
done
clear
D)
1 crore year
done
clear
View Answer play_arrow
Which of the following statement is true?
A)
Homo erectus is direct ancestor of Homo sapiens
done
clear
B)
Neanderthal man is direct ancestor of modem man
done
clear
C)
Australopithecus is direct ancestor of modem man
done
clear
D)
Fossils of Cro-Magnon man first found in Ethiopia
done
clear
View Answer play_arrow
Which statement is wrong for Cycas?
A)
Xylems have vessels
done
clear
B)
Female flowers are well developed
done
clear
C)
It has coralloid roots
done
clear
D)
Circinate ptyxis
done
clear
View Answer play_arrow
Evolution of heart from one to two, three and four chambered proves
A)
Biogenetic law of Haeckel
done
clear
B)
Lamarckism
done
clear
C)
Hardy-Weinberg?s law
done
clear
D)
Neo-Darwinism
done
clear
View Answer play_arrow
What is necessary for ripening of fruits?
A)
\[80\,%\] of ethylene
done
clear
B)
Abscisic acid
done
clear
C)
2, 4 D
done
clear
D)
A.M.O.-16
done
clear
View Answer play_arrow
Which of the following induces morphogeness in tissue culture?
A)
Gibberellins
done
clear
B)
Cytokmin
done
clear
C)
IAA
done
clear
D)
Ethylene
done
clear
View Answer play_arrow
Which weedicide can defoliate the complete forest?
A)
2, 4-D
done
clear
B)
AMO-1618
done
clear
C)
MH
done
clear
D)
ABA
done
clear
View Answer play_arrow
Heterosis (Hybrid vigour) desirable in vegetatively propagated plants because
A)
Heterosis is maintained for a longer duration
done
clear
B)
These plants are easy to cultivate
done
clear
C)
Vegetative reproduction help to multiply fast
done
clear
D)
It is due to homozygosity
done
clear
View Answer play_arrow
What is correct for stages of Puccinia?
A)
Telia and aecia on wheat
done
clear
B)
Telia and uredo stage on wheat
done
clear
C)
Telia and aecia on barberry
done
clear
D)
None
done
clear
View Answer play_arrow
Typhoid is caused by
A)
Rickettsiae
done
clear
B)
Chlamydia
done
clear
C)
Salmonella typhi
done
clear
D)
Mycobacterium
done
clear
View Answer play_arrow
Agent orange is
A)
Biodegradable insecticide
done
clear
B)
Di auxin (2-4, D and 2, 4, 5 T) weedicide
done
clear
C)
Biofertilizer
done
clear
D)
Biopesticide
done
clear
View Answer play_arrow
Largest sperm is of
A)
Pinus
done
clear
B)
Cycas
done
clear
C)
Ephedra
done
clear
D)
Sequoia
done
clear
View Answer play_arrow
Hollow air filled bones (pneumatic bones) occur in
A)
Mammals
done
clear
B)
Reptiles
done
clear
C)
Urodela
done
clear
D)
Aves
done
clear
View Answer play_arrow
Non-symbiotic nitrogen fixing bacteria are
A)
Rhizobium
done
clear
B)
Azospirillum
done
clear
C)
Azotobacter
done
clear
D)
Nitrosomonas
done
clear
View Answer play_arrow
Extrastellar secondary growth takes place by
A)
Vascular cambium
done
clear
B)
Phellogen
done
clear
C)
Phellem
done
clear
D)
Phelloderm
done
clear
View Answer play_arrow
If \[C{{O}_{2}}\] is absent in atmosphere of earth, then
A)
temperature will decrease
done
clear
B)
temperature will increase
done
clear
C)
plants will flourish well
done
clear
D)
no effect
done
clear
View Answer play_arrow
Acacia, Prosopis and Capparis belong to
A)
Deciduous forest
done
clear
B)
Tropical forest
done
clear
C)
Thorn forest
done
clear
D)
Evergreen forest
done
clear
View Answer play_arrow
Animals of desert are
A)
Arboreal
done
clear
B)
Fossorial
done
clear
C)
Crepuscular
done
clear
D)
Nocturnal
done
clear
View Answer play_arrow
Which part does not have only involuntary muscles?
A)
Urethra
done
clear
B)
Irish
done
clear
C)
Heart muscles
done
clear
D)
Blood vessels
done
clear
View Answer play_arrow
Solenocytes occur in
A)
Platyhelminthes
done
clear
B)
Arthropoda
done
clear
C)
Annelida
done
clear
D)
Aschelminthes
done
clear
View Answer play_arrow
Which characteristic is true for Obelia?
A)
Metagenesis
done
clear
B)
Morphogenesis
done
clear
C)
Apolysis
done
clear
D)
Pedogeny
done
clear
View Answer play_arrow
In angiosperm, characters of flowers are used in classification because
A)
Characters of flowers are conservative
done
clear
B)
Flowers are large
done
clear
C)
Flowers are attractive
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Transport of gases in alveoli takes place by
A)
Active transport
done
clear
B)
Passive transport
done
clear
C)
Simple diffusion
done
clear
D)
None
done
clear
View Answer play_arrow
Oral contraceptives contain
A)
Progesterone
done
clear
B)
LH
done
clear
C)
Oxytocin
done
clear
D)
Sterols
done
clear
View Answer play_arrow
In S-phase, DNA is replicated in a medium containing radioactive thymidine, radioactivity will be observed in
A)
Euchromatin
done
clear
B)
Heterochromatin
done
clear
C)
Both
done
clear
D)
Nucleolus
done
clear
View Answer play_arrow
CO is harmful because
A)
It forms stable compound with hemoglobin
done
clear
B)
It blocks mitosis
done
clear
C)
It is mutagenic
done
clear
D)
It causes defoliation
done
clear
View Answer play_arrow
Function of thyrocalcitonin
A)
To reduce the calcium level in blood
done
clear
B)
To increase the calcium level in blood
done
clear
C)
Oppose the action of thyroxine
done
clear
D)
Maturation of gonads
done
clear
View Answer play_arrow
Osmotic potential and water potential of pure water respectively
A)
0 and 0
done
clear
B)
0 and 1
done
clear
C)
100 and 0
done
clear
D)
100 and 100
done
clear
View Answer play_arrow
A normal leaf cell have how many genomes
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Contractile protein is
A)
Actin
done
clear
B)
Myosin
done
clear
C)
Troponin
done
clear
D)
Tropomyosin
done
clear
View Answer play_arrow
Unit of contraction is
A)
Sarcomere
done
clear
B)
Muscle fiber
done
clear
C)
Actin
done
clear
D)
None
done
clear
View Answer play_arrow
Oxidation of palmitic acid yields
A)
129ATP
done
clear
B)
132ATP
done
clear
C)
36ATP
done
clear
D)
76ATP
done
clear
View Answer play_arrow
Total amount of energy trapped by green plants in food is called
A)
Gross primary production
done
clear
B)
Net primary production
done
clear
C)
Standing crop
done
clear
D)
Standing state
done
clear
View Answer play_arrow
Role of microtubules
A)
To help in cell division
done
clear
B)
Cell membrane formation
done
clear
C)
Respiration
done
clear
D)
Pinocytosis
done
clear
View Answer play_arrow
Difference between eukaryotes and prokaryotes
A)
ss circular DNA in prokaryotes
done
clear
B)
Histone with prokaryotic DNA
done
clear
C)
Operon in eukaryotes
done
clear
D)
Membrane bound organelles in eukaryotes
done
clear
View Answer play_arrow
According to five kingdom system blue green algae belongs to
A)
Metaphyta
done
clear
B)
Monera
done
clear
C)
Protista
done
clear
D)
Algae
done
clear
View Answer play_arrow
Bacteria are essential in carbon cycle as
A)
Decomposer
done
clear
B)
Synthesizer
done
clear
C)
Consumer
done
clear
D)
Pri. Producer
done
clear
View Answer play_arrow
What occurs in crossing over?
A)
Recombination
done
clear
B)
Mutation
done
clear
C)
Independent assortment
done
clear
D)
None
done
clear
View Answer play_arrow
Histamine secreted by
A)
Mast cells
done
clear
B)
Fibroblast
done
clear
C)
Histiocytes
done
clear
D)
Plasma cells
done
clear
View Answer play_arrow
Arterial blood pressure in human beings is
A)
120 and 80 mm Hg
done
clear
B)
150 and 100 mm Hg
done
clear
C)
50 and 100 mm Hg
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which of the following survives a temperature of\[104\text{ }to\text{ }106{}^\circ C\]?
A)
Marine archaebacteria
done
clear
B)
Hot water spring thermophiles
done
clear
C)
Seeds of angiosperms
done
clear
D)
Eubacteria
done
clear
View Answer play_arrow
Mental retardation in man occurs due to
A)
Loss of one X chromosome
done
clear
B)
Addition of one X chromosome
done
clear
C)
Slight growth in Y
done
clear
D)
Overgrowth in Y
done
clear
View Answer play_arrow
Symptoms of lathyrism
A)
Bone deformation
done
clear
B)
Muscular dystrophy and paralysis
done
clear
C)
Asphyxia
done
clear
D)
Cardiac arrest
done
clear
View Answer play_arrow
A cell 'A' with \[\operatorname{D}.P.D. = 8\] is surrounded by three cells 'B', 'C' and 'D' with D.P.D. respectively 4, 6 and 5. What shall be the direction of water movement?
A)
done
clear
B)
\[A\to B\to C\to D\]
done
clear
C)
\[D\to C\to B\to A\]
done
clear
D)
\[A\to B\to C\to D\]
done
clear
View Answer play_arrow
What change occurs during conversion of proto chlorophyll to chlorophyll?
A)
Addition of 2H in one pyrrole ring
done
clear
B)
Loss of 2H
done
clear
C)
Addition of Mg
done
clear
D)
Loss of Mg
done
clear
View Answer play_arrow
Transduction in bacteria carried out by
A)
Bacteriophage
done
clear
B)
B.G.A.
done
clear
C)
Mycoplasma
done
clear
D)
Rickettsiae
done
clear
View Answer play_arrow
Which of the following is most used in genetic engineering?
A)
E. coli and Agrobacterium
done
clear
B)
Mycobacteria and Salmonella
done
clear
C)
Aspergillus
done
clear
D)
Penicillium
done
clear
View Answer play_arrow
Variations in proteins are due to
A)
Sequence of amino acids
done
clear
B)
Number of amino acids
done
clear
C)
R-group
done
clear
D)
None of these
done
clear
View Answer play_arrow
Genetic drift in Mendelian population takes place in
A)
Small population
done
clear
B)
Large population
done
clear
C)
Oceanic population
done
clear
D)
Never occurs
done
clear
View Answer play_arrow
Embryo of sunflower has
A)
Two cotyledons
done
clear
B)
One cotyledons
done
clear
C)
Eight cotyledons
done
clear
D)
Cotyledons absent
done
clear
View Answer play_arrow
Effect of light and dark rhythm on plants is
A)
Photonasty
done
clear
B)
Phototropism
done
clear
C)
Photoperiodism
done
clear
D)
Photomorphogenesis
done
clear
View Answer play_arrow
ABO blood group has
A)
Two codominants and one recessive allele
done
clear
B)
Two codominants and two recessive alleles
done
clear
C)
Two incompletely dominant genes
done
clear
D)
Two pseudo alleles
done
clear
View Answer play_arrow
Walking fem name of Adiantum is due to
A)
Dispersal by animals
done
clear
B)
Reproduction by spores
done
clear
C)
Vegetative reproduction
done
clear
D)
Power of locomotion
done
clear
View Answer play_arrow
Modem farmers can increase the yield of paddy up to \[50\,%\] by the use of
A)
Cyanobacteria
done
clear
B)
Rhizobium
done
clear
C)
Cyanobacteria in Azolla pinnata
done
clear
D)
Farm yard manure
done
clear
View Answer play_arrow
Which of the following destroys the acetyl choline esterase
A)
Malathion
done
clear
B)
CO
done
clear
C)
KCN
done
clear
D)
Colchicine
done
clear
View Answer play_arrow
Growth of leaf primordia is
A)
First apical then marginal
done
clear
B)
Only apical
done
clear
C)
Only marginal
done
clear
D)
Lateral
done
clear
View Answer play_arrow
Reason for elimination of wild life is
A)
Deforestation
done
clear
B)
Forest fire
done
clear
C)
Floods
done
clear
D)
Less Rain fall
done
clear
View Answer play_arrow
Besides \[{{\operatorname{CH}}_{4}}\,\,and\,\,C{{O}_{2}}\], other greenhouse gas from agriculture area is
A)
\[S{{O}_{2}}\]
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[N{{O}_{2}}\]
done
clear
D)
CFC
done
clear
View Answer play_arrow
In which biome a new plant may adapt soon
A)
Tropical rain forest
done
clear
B)
Desert
done
clear
C)
Mangrove
done
clear
D)
Sea island
done
clear
View Answer play_arrow
In present times the origin of life is not possible from inorganic compounds due to
A)
Raw material not available
done
clear
B)
High cone of \[{{O}_{2}}\] in atmosphere
done
clear
C)
Decrease in temperature
done
clear
D)
Excess of pollution
done
clear
View Answer play_arrow
On Galapagos island Darwin observed variation in beaks of birds (Darwin's finches) and he concluded
A)
Interspecies variation
done
clear
B)
Intraspecies variation
done
clear
C)
Natural selection according to food
done
clear
D)
Inheritance of acquired characters
done
clear
View Answer play_arrow