question_answer 1) If in a nuclear reactor using U235 as fuel, the power output is 4.8 MW. The number of fissions per second is : [Energy released per fission of \[{{U}^{235}}=200\text{ }MeV,1\text{ }eV\]\[=1.6\times {{10}^{-19}}j\]]
A)
\[1.5\times {{10}^{17}}\]
done
clear
B)
\[3\times {{10}^{19}}\]
done
clear
C)
\[1.5\times {{10}^{25}}\]
done
clear
D)
\[3\times {{10}^{25}}\]
done
clear
View Answer play_arrow
question_answer 2) The first member of the paschen series in hydrogen spectrum of wave length is \[18800\overset{\text{o}}{\mathop{\text{A}}}\,\] The short Wavelength limit of Paschen series is:
A)
\[1215\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[6560\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[8225\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[12850\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 3) An n-type and p-type silicon can be obtained by doping pure silicon with:
A)
arsenic and phosphorous
done
clear
B)
indium and aluminium
done
clear
C)
phosphorous and indium
done
clear
D)
aluminium and boron
done
clear
View Answer play_arrow
question_answer 4) The physical quantity which has dimensional formula as that of \[\frac{\text{Energy}}{\text{Mass x length}}\] is:
A)
force
done
clear
B)
power
done
clear
C)
pressure
done
clear
D)
acceleration
done
clear
View Answer play_arrow
question_answer 5) The dimensional formula of magnetic induction is:
A)
\[[M{{T}^{-1}}{{A}^{-1}}]\]
done
clear
B)
\[[M{{T}^{-2}}{{A}^{-1}}]\]
done
clear
C)
\[[ML{{A}^{-2}}]\]
done
clear
D)
\[[M{{T}^{-2}}A]\]
done
clear
View Answer play_arrow
question_answer 6) The unit vector parallel to resultant of the vectors \[A\to =4\widehat{i}+3\widehat{j}+6\widehat{k}\] and \[B\to =-\widehat{i}+6\widehat{j}-8\widehat{k}\] is:
A)
\[\frac{1}{7}(3\widehat{i}+3\widehat{j}-2\widehat{k})\]
done
clear
B)
\[\frac{1}{7}(3\widehat{i}+6\widehat{j}-2\widehat{k})\]
done
clear
C)
\[\frac{1}{49}(3\widehat{i}+6\widehat{j}-2\widehat{k})\]
done
clear
D)
\[\frac{1}{49}(3\widehat{i}-6\widehat{j}+2\widehat{k})\]
done
clear
View Answer play_arrow
question_answer 7) The average velocity of a body moving with uniform acceleration after travelling a distance of 3.06m is \[0.34m{{s}^{-1}}.\] If the change in velocity of the body is \[0.18\text{ }m{{s}^{-}}1,\] during this time its uniform acceleration is:
A)
\[0.01m{{s}^{-2}}\]
done
clear
B)
\[0.02m{{s}^{-2}}\]
done
clear
C)
\[0.03m{{s}^{-2}}\]
done
clear
D)
\[0.04m{{s}^{-2}}\]
done
clear
View Answer play_arrow
question_answer 8) For a projectile the ratio of maximum height reached to the square of flight time is: \[(g=10m{{s}^{-2}})\]
A)
5 : 4
done
clear
B)
5 : 2
done
clear
C)
5 : 1
done
clear
D)
10 : 1
done
clear
View Answer play_arrow
question_answer 9) A stone is projected with a velocity u at angle \[\theta \] with the horizontal reaches maximum height H1, when it is projected with a velocity u at an angle \[\left( \frac{\pi }{2}-\theta \right)\] with the horizontal, it reaches to a maximum height H2. The relation between the horizontal range R of the projectile, H1 and H2 is:
A)
\[R=4\sqrt{{{H}_{1}}{{H}_{2}}}\]
done
clear
B)
\[R=4({{H}_{1}}-{{H}_{2}})\]
done
clear
C)
\[R=4({{H}_{1}}+{{H}_{2}})\]
done
clear
D)
\[R={{H}_{1}}^{2}/{{H}_{2}}^{2}\]
done
clear
View Answer play_arrow
question_answer 10) A constant force acts on a body of mass 0.9 kg at rest for 10 s. If the body moves a distance of 250 m the magnitude of the force is:
A)
3 N
done
clear
B)
3.5 N
done
clear
C)
4.0 N
done
clear
D)
4.5 N
done
clear
View Answer play_arrow
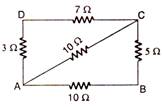
question_answer 11)
Four resistances 10 \[\Omega \], 5 \[\Omega \], 7 \[\Omega \] and 3 \[\Omega \] are connected so as to form the four sides of a rectangle AB, BC, CD and DA respectively. Another resistance of 10\[\Omega \] is connected across the diagonal AC. The equivalent between A and B is:
A)
2 \[\Omega \]
done
clear
B)
5 \[\Omega \]
done
clear
C)
7 \[\Omega \]
done
clear
D)
10 \[\Omega \]
done
clear
View Answer play_arrow
question_answer 12)
Consider the following statements A and B and identify the correct choice of given answers: A: Thermo emf is minimum at neutral temperature of a thermo couple. B: When two junctions made of two different metallic wires are maintained at different temperatures, an electric current is generated in the circuit
.
A)
A is false and B is true
done
clear
B)
A is true and B is false
done
clear
C)
both A and B are false
done
clear
D)
both A and B are true
done
clear
View Answer play_arrow
question_answer 13) If an electron revolves in the path of a circle of radius of \[0.5\times {{10}^{-10}}m\]at a frequency of \[5\times {{10}^{15}}\] cycle/sec. The electric current in the circle is: (Charge of an electron is\[1.6\times {{10}^{-19}}C\])
A)
0.4 mA
done
clear
B)
0.8 mA
done
clear
C)
1.2 mA
done
clear
D)
1.6 mA
done
clear
View Answer play_arrow
question_answer 14) The ratio of the longest to shortest wavelengths in Lyman series of hydrogen spectra is:
A)
\[\frac{25}{9}\]
done
clear
B)
\[\frac{17}{6}\]
done
clear
C)
\[\frac{9}{5}\]
done
clear
D)
\[\frac{4}{3}\]
done
clear
View Answer play_arrow
question_answer 15) The work functions of the metals A and B are in the ratio 1 : 2. If the light of frequencies f and 2f are incident on metal surfaces of A and B respectively. The ratio of maximum kinetic energy of photo electrons emitted is [f is greater than threshold frequency of A. If is greater than threshold frequency of B]:
A)
1 : 1
done
clear
B)
1 : 2
done
clear
C)
1 : 3
done
clear
D)
1 : 4
done
clear
View Answer play_arrow
question_answer 16)
Consider the following two statements A and B and identify the correct choice in the given answers: A: The characteristic X-rays spectrum depends on the nature of the material of the target. B: The short wavelength limit of continuous X-ray spectrum varies inversely on the potential difference applied to the X- ray tube.
A)
A is true and B is false
done
clear
B)
A is false and B is true
done
clear
C)
both A and B are true
done
clear
D)
both A and B are false
done
clear
View Answer play_arrow
question_answer 17) In hydrogen atom if the difference in the energy of the electron in n=2 and n=3 orbits is E. The ionization potential of hydrogen atom is:
A)
13.2 E
done
clear
B)
7.2 E
done
clear
C)
5.6 E
done
clear
D)
3.2 E
done
clear
View Answer play_arrow
question_answer 18) A body of mass 5 kg starts from the origin with an initial velocity \[u\to =30\widehat{i}+40\widehat{j}\,m{{s}^{-1}}.\] If a constant force \[F\to =\sqrt{6}\,\widehat{i}-0.5\widehat{j}\,N,\]acts on the body in time in which y component of the velocity becomes zero is:
A)
5 s
done
clear
B)
20 s
done
clear
C)
40 s
done
clear
D)
80 s
done
clear
View Answer play_arrow
question_answer 19) A gun of mass 10 kg fires 4 bullets per second. The mass of each bullet is 20 g and the velocity of the bullet, when it leaves the gun is \[300\text{ }m{{s}^{-1}}.\] The force required to hold the gun while firing is:
A)
6 N
done
clear
B)
8 N
done
clear
C)
24 N
done
clear
D)
240 N
done
clear
View Answer play_arrow
question_answer 20) A body of mass 5 kg rest on a rough horizontal surface of coefficient of friction 0.2. The body is pulled through a distance of 10 m by a horizontal force of 25 N. The kinetic energy acquired by it is:
A)
200 J
done
clear
B)
150 J
done
clear
C)
100 J
done
clear
D)
50 J
done
clear
View Answer play_arrow
question_answer 21) The average resisting force that must act on a 5 kg mass to reduce its speed from 65 cm/s to 15 cm/s in 0.2s is:
A)
12.5 N
done
clear
B)
25 N
done
clear
C)
50 N
done
clear
D)
100 N
done
clear
View Answer play_arrow
question_answer 22) Four particles each of mass m are placed at the corners of a square of side length l. The radius of gyration of the system about an axis perpendicular to the square and passing through its centre is:
A)
\[\frac{l}{\sqrt{2}}\]
done
clear
B)
\[{{l}_{2}}\]
done
clear
C)
\[l\]
done
clear
D)
\[\sqrt{2}l\]
done
clear
View Answer play_arrow
question_answer 23) A particle tied to a string of negligible weight and length l, is swinging in a horizontal circular path with constant angular velocity having time period T. If the string length is shortened by \[\frac{l}{2}\], while the particle is in motion, the period is:
A)
4 T
done
clear
B)
2 T
done
clear
C)
T
done
clear
D)
\[\frac{T}{4}\]
done
clear
View Answer play_arrow
question_answer 24) The angular velocity of the earth with which it has to rotate so that, acceleration due to gravity on 60° latitude becomes zero is: (Radius of earth = 6400 km, at the poles\[g=10\text{ }m{{s}^{-2}}\])
A)
\[2.5\times {{10}^{-3}}rad/s\]
done
clear
B)
\[5.0\times {{10}^{-3}}rad/s\]
done
clear
C)
\[10\times {{10}^{-3}}rad/s\]
done
clear
D)
\[7.8\times {{10}^{-2}}rad/s\]
done
clear
View Answer play_arrow
question_answer 25) When a satellite going round the earth in a circular orbit of radius r and speed v loses some of its energy, then r and v change as:
A)
r and v both will increase
done
clear
B)
r and v both will decrease
done
clear
C)
r will decrease and v will increase
done
clear
D)
r will increase and v will decrease
done
clear
View Answer play_arrow
question_answer 26) A particle is executing SHM with an amplitude of 4 cm. At the mean position, the velocity of the particle is 10 cm/s. The distance of the particle from mean position, when its speed becomes 5 cm/s is:
A)
\[\sqrt{3}\] cm
done
clear
B)
\[\sqrt{5}\] cm
done
clear
C)
\[2\sqrt{3}\] cm
done
clear
D)
\[2\sqrt{5}\] cm
done
clear
View Answer play_arrow
question_answer 27) A particle executes SHM with a period of T and amplitude A. The shortest time it takes to reach point \[\frac{A}{\sqrt{2}}\]m from its mean position in second is:
A)
T
done
clear
B)
\[\frac{T}{2}\]
done
clear
C)
\[\frac{T}{8}\]
done
clear
D)
\[\frac{T}{16}\]
done
clear
View Answer play_arrow
question_answer 28) When a tension F is applied the elongation produced in uniform wire of length l and radius r is e. When tension 2f is applied the elongation produced in another uniform wire of length 21 and radius 2r made of same material is:
A)
0.5 e
done
clear
B)
1.0 e
done
clear
C)
1.5 e
done
clear
D)
2.0 e
done
clear
View Answer play_arrow
question_answer 29) The surface energy of a liquid film on a ring of area \[0.15\text{ }{{m}^{2}}\]is : (Surface tension of liquid is\[=\text{ }5\text{ }N{{m}^{-1}}\])
A)
0.75 J
done
clear
B)
1.5 J
done
clear
C)
2.25 J
done
clear
D)
3.0 J
done
clear
View Answer play_arrow
question_answer 30) The two metal rods A and B are having their initial lengths in the ratio 2 :3 and coefficient of linear expansion in the ratio 3 :4. When they are heated through same temperature difference, the ratio of linear expansions is:
A)
1 : 2
done
clear
B)
2 : 3
done
clear
C)
3 : 4
done
clear
D)
4 : 3
done
clear
View Answer play_arrow
question_answer 31) The length of a metal rod at \[0{}^\circ C\] is 0.5 m. When it is heated, its length increases by 2.7mm. The final temperature of the rod is: (Coefficient of linear expansion of the metal\[=90\times {{10}^{-6}}/{}^\circ C\])
A)
\[20{}^\circ C\]
done
clear
B)
\[30{}^\circ C\]
done
clear
C)
\[40{}^\circ C\]
done
clear
D)
\[60{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 32) A closed copper vessel contains water equal to half of its volume. When the temperature of the vessel is raised to \[447{}^\circ C\]. The pressure of steam in the vessel is: (Treat steam as an ideal gas. Universal gas constant = 8310 J/mol-K, mole-density of water is \[1000\text{ }kg/{{m}^{3}},\] molecular weight of water = 18)
A)
\[33.24\times {{10}^{7}}Pa\]
done
clear
B)
\[16.62\times {{10}^{7}}Pa\]
done
clear
C)
\[10.31\times {{10}^{7}}Pa\]
done
clear
D)
\[8.31\times {{10}^{7}}Pa\]
done
clear
View Answer play_arrow
question_answer 33) The pressure and temperature of an ideal gas in a closed vessel are 720 kPa and \[40{}^\circ C\] respectively. If \[\frac{1}{4}\]th of the gas is released from the vessel and the temperature of the remaining gas is raised to \[353{}^\circ C\], the final pressure of the gas is:
A)
1440 kPa
done
clear
B)
1920 kPa
done
clear
C)
720 kPa
done
clear
D)
540 kPa
done
clear
View Answer play_arrow
question_answer 34) A steel ball of mass 0.1 kg falls freely from a height of 10 m and bounces to a height of 5.4 m from the ground. If the dissipated energy in this process is absorbed by the ball. the rise in its temperature is: (The specific heat of steel is \[460\text{ }J/\text{ }kg\text{ }{}^\circ C\])
A)
\[0.01\text{ }{}^\circ C\]
done
clear
B)
\[0.1\text{ }{}^\circ C\]
done
clear
C)
\[7\text{ }{}^\circ C\]
done
clear
D)
\[1.1\text{ }{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 35) 50 g of copper is heated to increase its temperature by \[10{}^\circ C\]. If the same quantity of heat is given to 10 g of water, the rise in its temperature is:
A)
\[5{}^\circ C\]
done
clear
B)
\[6{}^\circ C\]
done
clear
C)
\[7{}^\circ C\]
done
clear
D)
\[8{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 36) One end of a metal rod of length 1.0 m and area of cross-section 1 m2 is maintained at \[100{}^\circ C\]. If the other end of the rod is maintained at \[0{}^\circ C\], the quantity of heat transmitted through the rod per minute is:
A)
\[3\times {{10}^{3}}J\]
done
clear
B)
\[6\times {{10}^{3}}J\]
done
clear
C)
\[9\times {{10}^{3}}J\]
done
clear
D)
\[12\times {{10}^{3}}J\]
done
clear
View Answer play_arrow
question_answer 37) When the temperature of an ideal gas is increased by 600 K, the velocity of sound in the gas becomes \[\sqrt{3}\]times the initial velocity in it. The initial temperature of the gas is:
A)
\[-73{}^\circ C\]
done
clear
B)
\[27{}^\circ C\]
done
clear
C)
\[127{}^\circ C\]
done
clear
D)
\[327{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 38) A man is standing between two parallel cliffs and fires a gun. If he hears first and second echoes after 1.5 s and 3.5 s respectively the distance between the cliffs is : (Velocity of sound in air\[=340m{{s}^{-1}}\])
A)
190 m
done
clear
B)
850 m
done
clear
C)
591 m
done
clear
D)
510 m
done
clear
View Answer play_arrow
question_answer 39) The fundamental frequency of a closed pipe is 220 Hz. If \[\frac{1}{4}\] th of the pipe is filled with water the frequency of the first overtone of the pipe now is:
A)
220 Hz
done
clear
B)
440 Hz
done
clear
C)
880 Hz
done
clear
D)
1760 Hz
done
clear
View Answer play_arrow
question_answer 40) When a ray of light is incident normally on refracting surface of an equilateral prism: (Refractive index of the material of the prism =1.5)
A)
emerging ray is deviated by \[30{}^\circ \]
done
clear
B)
emerging ray is deviated by \[45{}^\circ \]
done
clear
C)
emerging ray just grazes second refracting surface
done
clear
D)
the ray undergoes total internal reflection at the second refracting surface
done
clear
View Answer play_arrow
question_answer 41) In Foucaults rotating mirror experiment for determining the velocity of light, the distance between the rotating mirror and the convex lens is negligible, when compared to the radius of curvature of the concave mirror. If the radius of curvature of the concave mirror is doubled, the image shift to:
A)
halved
done
clear
B)
doubled
done
clear
C)
zero
done
clear
D)
independent of radius of curvature
done
clear
View Answer play_arrow
question_answer 42)
Consider the following two statements A and B and identify the correct choice in the given answer: A: The curved surfaces of plano- convex lenses in Ramsdens eye piece face each other. B: The focal length of field lens is 3 times the focal length of eye lens in Ransdens eyepiece.
A)
both A and B are true
done
clear
B)
both A and B are false
done
clear
C)
A is true and B is false
done
clear
D)
A is false and B is true
done
clear
View Answer play_arrow
question_answer 43) Two short bar magnets having magnetic moments in the ratio 27: 8, when placed on opposite sides of a deflection magnetometer. It shows no deflection. If the distance of the weaker magnet is 0.12 m from the centre of deflection magnetometer, the distance of the stronger magnet from the centre is:
A)
0.06m
done
clear
B)
0.08m
done
clear
C)
0.12m
done
clear
D)
0.18m
done
clear
View Answer play_arrow
question_answer 44) A long straight wire carries an electric current of 2A. The magnetic induction at a perpendicular distance of 5 m from the wire is: \[\left[ {{\mu }_{0}}=4\pi \times {{10}^{-7}}T \right]\]
A)
\[4\times {{10}^{-8}}T\]
done
clear
B)
\[8\times {{10}^{-8}}T\]
done
clear
C)
\[12\times {{10}^{-8}}T\]
done
clear
D)
\[16\times {{10}^{-8}}T\]
done
clear
View Answer play_arrow
question_answer 45) A galvanometer of 25\[\Omega \]resistance can read a maximum current of 6 mA. It can be used as voltmeter to measure maximum of 6. V by connecting a resistance to the galvanometer. Identify the correct choice in the given answers:
A)
1025\[\Omega \] in series
done
clear
B)
1025\[\Omega \] in parallel
done
clear
C)
975\[\Omega \]in series
done
clear
D)
975 \[\Omega \] in parallel
done
clear
View Answer play_arrow
question_answer 46) Two charges of 4\[\mu \]C each are palced at the corners of A and B of an equilateral triangle ABC of side length 0.2 m in air. The electric potential is: \[\left[ \frac{1}{4\pi {{\varepsilon }_{0}}}=9\times {{10}^{9}}\frac{N-{{m}^{2}}}{{{C}^{2}}} \right]\]
A)
\[9\times {{10}^{4}}V\]
done
clear
B)
\[18\times {{10}^{4}}V\]
done
clear
C)
\[12\times {{10}^{4}}V\]
done
clear
D)
\[72\times {{10}^{4}}V\]
done
clear
View Answer play_arrow
question_answer 47) Two identical short bar magnets each having magnetic moment M are placed at a distance of 2d with their axes perpendicular to each other in a horizontal plane. The magnetic induction at midpoint between them is:
A)
\[\frac{{{\mu }_{0}}}{4\pi }\sqrt{2}\frac{M}{{{d}^{3}}}\]
done
clear
B)
\[\frac{{{\mu }_{0}}}{4\pi }\sqrt{3}\frac{M}{{{d}^{3}}}\]
done
clear
C)
\[2\frac{{{\mu }_{0}}}{\pi }\frac{M}{{{d}^{3}}}\]
done
clear
D)
\[\frac{{{\mu }_{0}}}{4\pi }\sqrt{5}\frac{M}{{{d}^{3}}}\]
done
clear
View Answer play_arrow
question_answer 48) A bar magnet when placed at an angle of 30° to the direction of magnetic field induction of \[5\times {{10}^{2}}T\]experiences a moment of couple \[25\times {{10}^{-6}}Nm.\] If the length of the magnet is 5 cm, its pole strength is:
A)
\[2\times {{10}^{-6}}A-m\]
done
clear
B)
\[5\times {{10}^{2}}A-m\]
done
clear
C)
\[2\text{ }A-m\]
done
clear
D)
\[5\text{ }A-m\]
done
clear
View Answer play_arrow
question_answer 49) A bar magnet of moment \[3.0\text{ }A-{{m}^{2}}\]is placed in a uniform magnetic induction field of \[2\times {{10}^{-5}}T.\] If each pole of the magnet experiences a force of \[6\times {{10}^{-4}}N.\] The length of the magnet is:
A)
0.5 m
done
clear
B)
0.3 m
done
clear
C)
0.2 m
done
clear
D)
0.1 m
done
clear
View Answer play_arrow
question_answer 50) In a compound microscope the focal lengths of two lenses are 1.5 cm and 6.25 cm. If an object is placed at 2 cm from the objective and the final image is formed at 25 cm from the eye lens, the distance between the two lenses is:
A)
6.00 cm
done
clear
B)
7.75 cm
done
clear
C)
9.25 cm
done
clear
D)
11.00
done
clear
View Answer play_arrow
question_answer 51) The volume of 10 moles of an ideal gas is 1 L at \[\text{27}{{\,}^{\text{o}}}\text{C}\]and 1 atm pressure. What is the volume of 20 moles of same gas at same temperature and pressure?
A)
2
done
clear
B)
4
done
clear
C)
1
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 52) Which of the following is used in the extraction of silver?
A)
\[NaCl\]
done
clear
B)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
C)
\[\text{NaCN}\]
done
clear
D)
\[N{{a}_{2}}S{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 53) Which of the following exhibits colour in aqueous solution?
A)
\[S{{c}^{3+}}\]
done
clear
B)
\[N{{i}^{2+}}\]
done
clear
C)
\[T{{i}^{4+}}\]
done
clear
D)
\[Z{{n}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 54) What is the electrolyte used in the electrolytic method of preparation of fluorine?
A)
\[NaF+CaC{{l}_{2}}\]
done
clear
B)
\[KH{{F}_{2}}+LiF\]
done
clear
C)
\[KF+LiH{{F}_{2}}\]
done
clear
D)
\[CaC{{l}_{2}}+KF\]
done
clear
View Answer play_arrow
question_answer 55) Lead impurity is removed from silver by:
A)
poling
done
clear
B)
cupel lation
done
clear
C)
distillation
done
clear
D)
levigation
done
clear
View Answer play_arrow
question_answer 56) Aniline on heating with X in the presence of KOH gives a product with very bad smell. Which of the following is X?
A)
\[C{{H}_{3}}Cl\]
done
clear
B)
\[CC{{l}_{4}}\]
done
clear
C)
\[CHC{{l}_{3}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}Cl\]
done
clear
View Answer play_arrow
question_answer 57) Chloral hydrate is dissolved in NaOH solution and distilled. What are the compounds obtained?
A)
\[C{{H}_{3}}Cl,NaCl\]
done
clear
B)
\[C{{H}_{3}}Cl,C{{H}_{3}}COONa\]
done
clear
C)
\[CHC{{l}_{3}},HCOONa,{{H}_{2}}O~~\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}Cl,C{{H}_{3}}COONa\]
done
clear
View Answer play_arrow
question_answer 58) Bleaching action of chlorine occurs in the presence of:
A)
\[{{O}_{3}}\]
done
clear
B)
moisture
done
clear
C)
dry air
done
clear
D)
sunlight
done
clear
View Answer play_arrow
question_answer 59) Which of the following reaction conditions are used for the conversion of ethanol to ethylene?
A)
Conc. \[{{H}_{2}}S{{O}_{4}}/70{{\,}^{o}}C\]
done
clear
B)
Dil. \[{{H}_{2}}S{{O}_{4}}/140{{\,}^{o}}C\]
done
clear
C)
Dil\[{{H}_{2}}S{{O}_{4}}/100{{\,}^{o}}C\]
done
clear
D)
Conc. \[{{H}_{2}}S{{O}_{4}}/170{{\,}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 60) Which of the following has more acidic character?
A)
\[{{H}_{2}}O\]
done
clear
B)
\[{{H}_{2}}Te\]
done
clear
C)
\[{{H}_{2}}S\]
done
clear
D)
\[{{H}_{2}}Se\]
done
clear
View Answer play_arrow
question_answer 61) Which of the following compounds is formed when ethanol reacts with acetic acid in presence of conc. \[{{H}_{2}}S{{O}_{4}}\]?
A)
\[C{{H}_{3}}COO{{C}_{2}}{{H}_{5}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}OC2{{H}_{5~}}~\]
done
clear
C)
\[C{{H}_{3}}OC{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}CHO\]
done
clear
View Answer play_arrow
question_answer 62) In which of the following compounds oxygen exhibits an oxidation state of+2?
A)
\[{{H}_{2}}O\]
done
clear
B)
\[{{H}_{2}}{{O}_{2}}\]
done
clear
C)
\[O{{F}_{2}}\]
done
clear
D)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 63) What is the compound formed when acetaldehyde is reacted with \[\text{LiAl}{{\text{H}}_{\text{4}}}\]?
A)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}\]
done
clear
D)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 64) Ethyl chloride reacts with X to form \[\text{NaCl}\]and Y. One mole of Y reacts with two moles of HI to form water and ethyl iodide. Which of the following is?
A)
\[{{C}_{2}}{{H}_{5}}ONa\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}\]
done
clear
C)
\[C{{H}_{3}}COOH\]
done
clear
D)
\[C{{H}_{3}}CHO\]
done
clear
View Answer play_arrow
question_answer 65) What is the value of gas constant R in \[\text{J}\,\text{mo}{{\text{l}}^{-1}}{{\text{K}}^{-1}}\]?
A)
82.1
done
clear
B)
\[8.314\times {{10}^{7}}\]
done
clear
C)
8.314
done
clear
D)
0.0821
done
clear
View Answer play_arrow
question_answer 66) Dow metal is an alloy of
A)
Al, Mn, Zn and Mg
done
clear
B)
Sn and Mg
done
clear
C)
Cu and Zn
done
clear
D)
Fe, V, Ni and Cr
done
clear
View Answer play_arrow
question_answer 67) What is the oxidation state of carbon in carbon dioxide?
A)
+2
done
clear
B)
+4
done
clear
C)
+6
done
clear
D)
+1
done
clear
View Answer play_arrow
question_answer 68) Which of the following nuclide is unstable?
A)
\[{{\,}_{5}}{{B}^{10}}\]
done
clear
B)
\[{{\,}_{4}}{{B}^{9}}\]
done
clear
C)
\[{{\,}_{7}}{{N}^{14}}\]
done
clear
D)
\[{{\,}_{8}}{{O}^{16}}\]
done
clear
View Answer play_arrow
question_answer 69) The \[\text{p}{{\text{K}}_{\text{a}}}\]of a weak acid is 4.8. What is the ratio of\[\frac{[Salt]}{[Acid]},\] if a buffer of pH = 5.8 is to be prepared?
A)
1:1
done
clear
B)
10:1
done
clear
C)
2:1
done
clear
D)
1 :10
done
clear
View Answer play_arrow
question_answer 70) The probability of finding an electron in an orbital is approximately
A)
95%
done
clear
B)
50%
done
clear
C)
60%
done
clear
D)
25%
done
clear
View Answer play_arrow
question_answer 71) Which of the following is a Lewis acid?
A)
\[HCO{{O}^{-}}\]
done
clear
B)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
\[Si{{F}_{4}}\]
done
clear
D)
\[{{H}_{2}}S\]
done
clear
View Answer play_arrow
question_answer 72) What is the wave-length of \[{{H}_{\beta }}\]line in the Balmer series of hydrogen spectrum? (R = Rydberg constant)
A)
\[\frac{36}{5R}\]
done
clear
B)
\[\frac{5R}{36}\]
done
clear
C)
\[\frac{3R}{16}\]
done
clear
D)
\[\frac{16}{3R}\]
done
clear
View Answer play_arrow
question_answer 73) Which of the following is the correct order of atomic radii?
A)
\[N{{a}^{+}}<M{{g}^{2+}}<A{{l}^{3+}}<S{{i}^{4+}}\]
done
clear
B)
\[A{{l}^{3+}}<S{{i}^{4+}}>N{{a}^{+}}>M{{g}^{2+}}\]
done
clear
C)
\[S{{i}^{4+}}<A{{l}^{3+}}>M{{g}^{2+}}>N{{a}^{2+}}\]
done
clear
D)
\[N{{a}^{+}}>M{{g}^{2+}}>A{{l}^{3+}}>S{{i}^{4+}}\]
done
clear
View Answer play_arrow
question_answer 74) Which of the following is used to convert nitrobenzene to hydrazobenzene ?
A)
\[Zn/N{{H}_{4}}Cl\]
done
clear
B)
\[Zn/NaOH\]
done
clear
C)
\[Sn/HCl\]
done
clear
D)
\[LiAl{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 75) Which of the following has the highest electronegativity?
A)
Na
done
clear
B)
Cl
done
clear
C)
K
done
clear
D)
B
done
clear
View Answer play_arrow
question_answer 76) Which of the following has least bond energy?
A)
\[{{F}_{2}}\]
done
clear
B)
\[{{H}_{2}}\]
done
clear
C)
\[{{N}_{2}}\]
done
clear
D)
\[{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 77) Which of the following can be detected by silver mirror test?
A)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
D)
\[C{{H}_{3}}CHO\]
done
clear
View Answer play_arrow
question_answer 78) Which of the following is used to convert nitrobenzene to azobenzene?
A)
\[LiAl{{H}_{4}}\]
done
clear
B)
\[Zn/NaOH\]
done
clear
C)
\[Zn/N{{H}_{4}}Cl\]
done
clear
D)
\[Sn/HCl\]
done
clear
View Answer play_arrow
question_answer 79) Which of the following has lowest boiling point?
A)
\[C{{H}_{4}}\]
done
clear
B)
\[{{H}_{2}}O\]
done
clear
C)
HF
done
clear
D)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
View Answer play_arrow
question_answer 80) Acetic acid reacts with ethanol in presence \[{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\] to farmland water. Which of the following is\[X\]?
A)
\[C{{H}_{3}}C{{H}_{2}}COO{{C}_{2}}{{H}_{5}}\]
done
clear
B)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}CO{{C}_{2}}{{H}_{5}}\]
done
clear
D)
\[C{{H}_{3}}COO{{C}_{2}}{{H}_{5}}\]
done
clear
View Answer play_arrow
question_answer 81) Methyl benzene can be prepared by reacting benzene with bromo methane in presence of
A)
anhydrous \[AlC{{l}_{3}}\]
done
clear
B)
\[B{{r}_{2}}/CC{{l}_{4}}\]
done
clear
C)
\[Ni/{{H}_{2}}70{{\,}^{o}}C\]
done
clear
D)
dil. \[{{H}_{2}}S{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 82) Aniline is treated with \[\text{NaN}{{\text{O}}_{\text{2}}}\]and \[\text{HCl}\]at \[\text{0}{{\,}^{\text{o}}}\text{C,}\] the product formed is :
A)
Nitroaniline
done
clear
B)
Chloroaniline
done
clear
C)
Aminophenol
done
clear
D)
Benzene diazonium salt
done
clear
View Answer play_arrow
question_answer 83) What is the solution used for the preparation of hydrogen peroxide by electrolytic procedure?
A)
\[\text{0}\text{.2 N NaOH}\]
done
clear
B)
50% \[{{\text{H}}_{2}}S{{O}_{4}}\]
done
clear
C)
20% \[NaOH\]
done
clear
D)
5% \[NaCl\]
done
clear
View Answer play_arrow
question_answer 84) Heat of combustion of carbon is 96 kcal. When same quantity of carbon is burnt in oxygen, 48 kcal of heat is liberated. What is the volume of oxygen (in litre) at STP reacted with the carbon?
A)
48
done
clear
B)
22.4
done
clear
C)
11.2
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 85) What is the rate of the reaction for \[2A\to B\]?
A)
\[\frac{1}{2}\left[ \frac{-d[A]}{dt} \right]\]
done
clear
B)
\[\frac{-d[A]}{dt}\]
done
clear
C)
\[\frac{-d[B]}{dt}\]
done
clear
D)
\[\frac{+d[A]}{dt}\]
done
clear
View Answer play_arrow
question_answer 86) Benzene is used in the preparation of:
A)
polythene
done
clear
B)
gammexane
done
clear
C)
chloroform
done
clear
D)
vinegar
done
clear
View Answer play_arrow
question_answer 87) In a reaction Na is heated with Bed 3, in this reaction sodium acts as :
A)
oxidising agent
done
clear
B)
reducing agent
done
clear
C)
hydrolysing agent
done
clear
D)
precipitating agent
done
clear
View Answer play_arrow
question_answer 88) What is the molecular formula of plaster of Paris?
A)
\[2CaS{{O}_{4}}.{{H}_{2}}O\]
done
clear
B)
\[2CaS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
C)
\[CaS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
D)
\[CaS{{O}_{4}}.3{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 89) Which of the following is used in the manufacture of ethanol?
A)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
C)
\[C{{H}_{4}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 90) What are the reaction conditions used for the formation of ethylene glycol from ethylene?
A)
Bromine water
done
clear
B)
Cold alkaline \[KMn{{O}_{4}}\]
done
clear
C)
Dil. \[{{H}_{2}}S{{O}_{4}}/{{60}^{o}}\]
done
clear
D)
\[{{O}_{3}}/{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 91) Solid sodium hydroxide reacted with carbon monoxide under pressure. What is the product obtained?
A)
\[C{{O}_{2}}\]
done
clear
B)
\[C{{H}_{3}}ONa\]
done
clear
C)
\[HCOONa\,\]
done
clear
D)
HCHO
done
clear
View Answer play_arrow
question_answer 92) What is the C-C bond length (in Angstrom) in diamond?
A)
1.54
done
clear
B)
3.35
done
clear
C)
2.0
done
clear
D)
5.2
done
clear
View Answer play_arrow
question_answer 93) In which of the following compounds the weight percentage of carbon and hydrogen are 80 and 20 respectively?
A)
\[C{{H}_{4}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
D)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
View Answer play_arrow
question_answer 94) What is the formula of diaspore?
A)
\[A{{l}_{2}}{{O}_{3}}.2{{H}_{2}}O\]
done
clear
B)
\[A{{l}_{2}}{{O}_{3}}\]
done
clear
C)
\[A{{l}_{2}}{{O}_{3}}.{{H}_{2}}O\]
done
clear
D)
\[AlP{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 95) Which of the following is calcium super phosphate?
A)
\[Ca{{(HP{{O}_{4}})}_{2}}.{{H}_{2}}O+2CaS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
B)
\[C{{a}_{3}}{{(P{{O}_{4}})}_{2}}+2CaS{{O}_{4}}\]
done
clear
C)
\[CaS{{O}_{4}}+CaO\]
done
clear
D)
\[Ca{{({{H}_{2}}P{{O}_{4}})}_{2}}.{{H}_{2}}O+CaS{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 96) What is the catalyst used during the manufacture of nitric acid by Ostwalds method?
A)
\[A{{l}_{2}}O3\]
done
clear
B)
(b ) \[Fe\]
done
clear
C)
\[{{V}_{2}}{{O}_{5}}\]
done
clear
D)
Platinum gauze
done
clear
View Answer play_arrow
question_answer 97) Which of the following is an electron deficient compound?
A)
\[NaCl\]
done
clear
B)
\[NaH\]
done
clear
C)
\[{{B}_{2}}{{H}_{6}}\]
done
clear
D)
\[KCl\]
done
clear
View Answer play_arrow
question_answer 98) Element \[{{\,}_{\text{Z}}}{{\text{M}}^{\text{A}}}\] emits one \[\alpha -\] particle followed by two \[\beta -\] particles. Which of the following is the daughter element?
A)
\[{{\,}_{z-2}}{{M}^{A-4}}\]
done
clear
B)
\[{{\,}_{Z-2}}{{M}^{A}}\]
done
clear
C)
\[{{\,}_{Z}}{{M}^{A-4}}\]
done
clear
D)
\[{{\,}_{Z+2}}{{M}^{A-4}}\]
done
clear
View Answer play_arrow
question_answer 99) Which of the following aqueous solution conduct electricity?
A)
Urea
done
clear
B)
Glucose
done
clear
C)
Sucrose
done
clear
D)
NaCI
done
clear
View Answer play_arrow
question_answer 100) 500 mL of a solution contains 12.6g of oxalic acid (mol. wt. 126). 10 mL of this solution is diluted to 100mL in a flask. What is the molarity (in mol\[{{\text{L}}^{-1}}\]) of the resultant solution?
A)
0.2
done
clear
B)
0.1
done
clear
C)
0.02
done
clear
D)
0.01
done
clear
View Answer play_arrow
question_answer 101) The insect which spreads the yellow fever is:
A)
Aedes
done
clear
B)
Prorophora
done
clear
C)
Glossina
done
clear
D)
Phlebotomns
done
clear
View Answer play_arrow
question_answer 102) The palpiger of cockroach bears:
A)
labial palp
done
clear
B)
submentum
done
clear
C)
ligula
done
clear
D)
labrum
done
clear
View Answer play_arrow
question_answer 103) The number of valves in the gut of cockroach is:
A)
4
done
clear
B)
3
done
clear
C)
2
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 104) In cockroach, the number of spiracles is:
A)
eight pairs
done
clear
B)
eight only
done
clear
C)
ten pairs
done
clear
D)
eighteen pairs
done
clear
View Answer play_arrow
question_answer 105) Which one of the following animals, can be identified as a chordate based on its larval features:
A)
Asymmetron
done
clear
B)
Ascidian
done
clear
C)
Amblystoma
done
clear
D)
Amia
done
clear
View Answer play_arrow
question_answer 106) The fossil record of placoderms is found in this era:
A)
Palaeozoic
done
clear
B)
Mesozoic
done
clear
C)
Coenozoic
done
clear
D)
Archaeozoic
done
clear
View Answer play_arrow
question_answer 107) The frog vertebra with centrum convex on its anterior side is:
A)
atlas
done
clear
B)
9th
done
clear
C)
8th
done
clear
D)
Typical
done
clear
View Answer play_arrow
question_answer 108) In Frog, Astragalus, is a part of:
A)
wrist
done
clear
B)
pectoral girdle
done
clear
C)
ankle
done
clear
D)
pelvic girdle
done
clear
View Answer play_arrow
question_answer 109) The anterior choroid plexus forms the roof of:
A)
diocoel
done
clear
B)
4th ventricle
done
clear
C)
olfactory ventricle
done
clear
D)
optocoel
done
clear
View Answer play_arrow
question_answer 110) Sodium taurocholate aids in:
A)
defaecation
done
clear
B)
absorption
done
clear
C)
emulsification
done
clear
D)
assimilation
done
clear
View Answer play_arrow
question_answer 111) Epinephrine affects:
A)
rate of heart beat
done
clear
B)
regulation of minerals of body fluid
done
clear
C)
gastro intestinal co-ordinations
done
clear
D)
metamorphosis
done
clear
View Answer play_arrow
question_answer 112) In frog, innominate vein is formed by the union of the veins:
A)
lingual and mandibular
done
clear
B)
subscapular and internal jugular
done
clear
C)
branchi and musculocutaneous
done
clear
D)
external jugular and subclavian
done
clear
View Answer play_arrow
question_answer 113) In frog, the sclerotic layer is formed from:
A)
endoderm
done
clear
B)
ectoderm
done
clear
C)
mesoderm
done
clear
D)
corneous layer
done
clear
View Answer play_arrow
question_answer 114) The extinct amphibian is:
A)
Diplocaulus
done
clear
B)
Geganophis
done
clear
C)
Necturus
done
clear
D)
Palacospondylns
done
clear
View Answer play_arrow
question_answer 115) The reptile which lacks penis belongs to:
A)
Gymnophiana
done
clear
B)
Ophidia
done
clear
C)
Crocodilia
done
clear
D)
Rhynchocephalia
done
clear
View Answer play_arrow
question_answer 116) The snake having head shields and elongated, hexagonal vertebrals is:
A)
Bungarus
done
clear
B)
Eryx
done
clear
C)
Naja
done
clear
D)
Echis
done
clear
View Answer play_arrow
question_answer 117) The cervical vertebrae in birds are:
A)
heterocoelous
done
clear
B)
procoelous
done
clear
C)
amphicoelous
done
clear
D)
acoelous
done
clear
View Answer play_arrow
question_answer 118) The number of occipital condyles in mammals is:
A)
3
done
clear
B)
5
done
clear
C)
1
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 119) "Gynacomastism" is characteristic feature of:
A)
oviparous mammals
done
clear
B)
pouched mammals
done
clear
C)
oviparous reptiles
done
clear
D)
viviparous snakes
done
clear
View Answer play_arrow
question_answer 120) The phagocytic granulocyte is:
A)
neutrophil
done
clear
B)
eosinophil
done
clear
C)
monocyte
done
clear
D)
basophil
done
clear
View Answer play_arrow
question_answer 121) The term "gristle" is also used for:
A)
bone
done
clear
B)
blood
done
clear
C)
nerve
done
clear
D)
cartilage
done
clear
View Answer play_arrow
question_answer 122) The correct chronological sequence of eras is:
A)
Mesozoic \[\to \] coenozoic\[\to \] archaeozoic
done
clear
B)
Coenozoic\[\to \] proterozoic \[\to \] mesozoic
done
clear
C)
Palaeozoic\[\to \] mesozoic \[\to \] coenozoic
done
clear
D)
Palaeozoic\[\to \] archaeozoic \[\to \] coenozoic
done
clear
View Answer play_arrow
question_answer 123) The biogenetic law is based on:
A)
genetic evidences
done
clear
B)
fossil evidences
done
clear
C)
biochemical evidences
done
clear
D)
embryological evidences
done
clear
View Answer play_arrow
question_answer 124) The jinga prawn is:
A)
Penaens indicus
done
clear
B)
Metapenaeus affinis
done
clear
C)
Metapenaeus
done
clear
D)
Palaemon styliferous
done
clear
View Answer play_arrow
question_answer 125) If the red flowered F1 hybrid from the cross of red x white pea, is crossed with recessive white flowered plant, the ratio in the off springs is:
A)
2: 3
done
clear
B)
1: 1
done
clear
C)
3: 1
done
clear
D)
1: 2: 1
done
clear
View Answer play_arrow
question_answer 126) The number of genotypes in a Mendelian dihybrid cross is:
A)
3
done
clear
B)
16
done
clear
C)
12
done
clear
D)
9
done
clear
View Answer play_arrow
question_answer 127) The pH of acid rain water is:
A)
1.2
done
clear
B)
3.4
done
clear
C)
5
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 128) The pearl spot is:
A)
Wallago
done
clear
B)
Pinctada
done
clear
C)
Etroplus
done
clear
D)
Restrelliger
done
clear
View Answer play_arrow
question_answer 129) The protein efficiency ratio is:
A)
the ratio of protein consumed to increase the body weight in cultivated young animals.
done
clear
B)
the ratio of protein consumed to increase the body weight in adult and old animals
done
clear
C)
the ratio of protein to carbohydrate in feed
done
clear
D)
the ratio of protein to fat in feed
done
clear
View Answer play_arrow
question_answer 130) Which age group chicks are affected by Gumbora disease:
A)
1- 2 weeks
done
clear
B)
3 weeks
done
clear
C)
4- 8 weeks
done
clear
D)
10 weeks Gumbora is a viral disease of poultry. It affects the chick of 4 to 8 weeks.
done
clear
View Answer play_arrow
question_answer 131) Budding in protozoa is seen in
A)
hydrozoa
done
clear
B)
mastigophora
done
clear
C)
sporozoa
done
clear
D)
ciliata
done
clear
View Answer play_arrow
question_answer 132) The number of merozoites at the end of an erythrocytic cycle in Plasmodium malariae is:
A)
6- 12
done
clear
B)
14- 24
done
clear
C)
26- 30
done
clear
D)
32- 38
done
clear
View Answer play_arrow
question_answer 133) The description of amoeboid movement like spreading of a water drop on an uneven surface of a glass plate is:
A)
Contraction theory
done
clear
B)
Adhesian theory
done
clear
C)
Surface tension theory
done
clear
D)
Change of viscosity theory
done
clear
View Answer play_arrow
question_answer 134) The parasite of endothelial system of man is:
A)
Enthamoeba
done
clear
B)
Schistosoma
done
clear
C)
Taenia
done
clear
D)
Leishmania
done
clear
View Answer play_arrow
question_answer 135) In benign tertian fever, the erythrocytic schizogony begins with the entry of:
A)
trophozoite
done
clear
B)
sporozoite
done
clear
C)
micro-meta cryptomerozoites
done
clear
D)
macro-meta cryptomerozoites
done
clear
View Answer play_arrow
question_answer 136) The narrow middle part of chromatophore in Euglena is:
A)
pyrenoid
done
clear
B)
pyrenophore
done
clear
C)
endosome
done
clear
D)
paramylum
done
clear
View Answer play_arrow
question_answer 137) An example for the group which is considered as blind ally in animal kingdom is:
A)
Chalina
done
clear
B)
Chelone
done
clear
C)
Chamaeleon
done
clear
D)
Chiroptera
done
clear
View Answer play_arrow
question_answer 138) The cnidoblasts used during looping movement of Hydra are:
A)
stenoteles
done
clear
B)
volvents
done
clear
C)
desmonemes
done
clear
D)
atrichous isorrhiza
done
clear
View Answer play_arrow
question_answer 139) An example of Scyphozoa is:
A)
jelly fish
done
clear
B)
cuttle fish
done
clear
C)
silver fish
done
clear
D)
cat fish
done
clear
View Answer play_arrow
question_answer 140) An acoelomate with Protandrous condition is:
A)
Taenia
done
clear
B)
Schistosoma
done
clear
C)
Megascolex
done
clear
D)
Ascaris
done
clear
View Answer play_arrow
question_answer 141) The total number of moults an Ascaris larva undergoes to become an adult is:
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 142) A metazoan adult whose body is covered by cilia is:
A)
Dugesia
done
clear
B)
Paramoecium
done
clear
C)
Amphiblastula
done
clear
D)
Fasciola
done
clear
View Answer play_arrow
question_answer 143) An annelid in which internal fertilization occurs is:
A)
Neanthes
done
clear
B)
Polygordius
done
clear
C)
Hirudmaria
done
clear
D)
Eunice
done
clear
View Answer play_arrow
question_answer 144) Spermatophores in earthworm are formed by the secretion of the glands:
A)
clitellar
done
clear
B)
prostrate
done
clear
C)
conglobate
done
clear
D)
accessory
done
clear
View Answer play_arrow
question_answer 145) Which one of the following is a molluscan larva:
A)
lovens larva
done
clear
B)
veligei
done
clear
C)
parenchymula
done
clear
D)
bipinnaria
done
clear
View Answer play_arrow
question_answer 146) The pentamerous symmetry is found in :
A)
sea mouse
done
clear
B)
sea urchin
done
clear
C)
sea pew
done
clear
D)
sea horse
done
clear
View Answer play_arrow
question_answer 147) The earthworms, insects and snails are
A)
denterostomes
done
clear
B)
protostomes
done
clear
C)
acoelomates
done
clear
D)
pseudo coelomates
done
clear
View Answer play_arrow
question_answer 148) Identify a Deuterostome:
A)
Leucosolenia
done
clear
B)
Dentalium
done
clear
C)
Dugesia
done
clear
D)
Ophiothrix
done
clear
View Answer play_arrow
question_answer 149) Biramous appendages are present in:
A)
Insecta
done
clear
B)
Crustacea
done
clear
C)
Cephalopoda
done
clear
D)
Onycophora
done
clear
View Answer play_arrow
question_answer 150) Mandibles are absent in:
A)
Periplaneta
done
clear
B)
Anopheles
done
clear
C)
Musca
done
clear
D)
Culex
done
clear
View Answer play_arrow
question_answer 151) To which of the following the sepals, petals, stamens and carpels are in the ratio of 2:2:3:1?
A)
Brassicaceae
done
clear
B)
Asteraceae
done
clear
C)
Solanaceae
done
clear
D)
Liliaceae
done
clear
View Answer play_arrow
question_answer 152) The female gametophyte in angiosperms is called:
A)
prothallus
done
clear
B)
embryo sac
done
clear
C)
fruiting body
done
clear
D)
ovule
done
clear
View Answer play_arrow
question_answer 153) Hardwickia yields:
A)
stain
done
clear
B)
alkaloids
done
clear
C)
timber
done
clear
D)
aromatic oil
done
clear
View Answer play_arrow
question_answer 154) Chicory is obtained from :
A)
roots
done
clear
B)
flowers
done
clear
C)
fruits
done
clear
D)
seeds
done
clear
View Answer play_arrow
question_answer 155) The variety varalakshmi belongs to the crop:
A)
rice
done
clear
B)
cotton
done
clear
C)
ground nut
done
clear
D)
cocount
done
clear
View Answer play_arrow
question_answer 156) In the life cycle of Rhizopus, the unicellular structure with diploid nuclei is :
A)
sporangiophore
done
clear
B)
sporangium
done
clear
C)
zygospore
done
clear
D)
gametangium
done
clear
View Answer play_arrow
question_answer 157) The number of neck canal cells in the archegonium of Selaginella is:
A)
8
done
clear
B)
4
done
clear
C)
3
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 158) The unique anatomical character of Cycas leaflet is the presence of:
A)
girdle leaf traces
done
clear
B)
transfusion tissue
done
clear
C)
algal layer
done
clear
D)
trabeculae
done
clear
View Answer play_arrow
question_answer 159) Which of the following virus has spherical shape?
A)
polio virus
done
clear
B)
tobacco mosaic virus
done
clear
C)
bacteriophage
done
clear
D)
vaccinia virus
done
clear
View Answer play_arrow
question_answer 160) Bacillus mycoides is:
A)
saprophyte
done
clear
B)
autotroph
done
clear
C)
parasite
done
clear
D)
symbiont
done
clear
View Answer play_arrow
question_answer 161) Root pockets can be seen in :
A)
Nelumbo
done
clear
B)
Nymphaea
done
clear
C)
Hydrilla
done
clear
D)
Pistia
done
clear
View Answer play_arrow
question_answer 162) Muchlenbeckia belongs to this category :
A)
xerophyte
done
clear
B)
epiphyte
done
clear
C)
mesophyte
done
clear
D)
hydrophyte
done
clear
View Answer play_arrow
question_answer 163) One of the following is an amphibious plant:
A)
Nelumbo
done
clear
B)
Azolla
done
clear
C)
Typha
done
clear
D)
Hydrilla
done
clear
View Answer play_arrow
question_answer 164) Emasculation means:
A)
removal of sepals
done
clear
B)
removal of petals
done
clear
C)
removal of stamens
done
clear
D)
removal of carpels
done
clear
View Answer play_arrow
question_answer 165) The maize varieties Dixie 11, 22 were introduced into India from this country :
A)
Australia
done
clear
B)
China
done
clear
C)
Taiwan
done
clear
D)
America
done
clear
View Answer play_arrow
question_answer 166) Which fungus causes black coal like spots of Anthracnose in plants:
A)
Gloeosporium
done
clear
B)
Tolyposporium
done
clear
C)
Ustilago
done
clear
D)
Alternaria
done
clear
View Answer play_arrow
question_answer 167) Species belonging to this genus cause "downy mildew disease":
A)
Uromyces
done
clear
B)
Fusarium
done
clear
C)
Ustilago
done
clear
D)
Peronospora
done
clear
View Answer play_arrow
question_answer 168) Vascular wilts are caused by this pathogen :
A)
Pyricularia
done
clear
B)
Puccinia
done
clear
C)
Erwinia
done
clear
D)
Helminthosporium
done
clear
View Answer play_arrow
question_answer 169) The purple colour formed in the petioles and veins of tomato plants during nitrogen deficiency is due to :
A)
xanthophylls
done
clear
B)
carotenes
done
clear
C)
anthocyanins
done
clear
D)
leghaemoglobin
done
clear
View Answer play_arrow
question_answer 170) When fats are the respiratory substrate, the RQ is:
A)
zero
done
clear
B)
one
done
clear
C)
less than one
done
clear
D)
more than one
done
clear
View Answer play_arrow
question_answer 171) Oxidative phosphorylation occurs in
A)
chloroplast
done
clear
B)
mitochondria
done
clear
C)
peroxisomes
done
clear
D)
ribosome
done
clear
View Answer play_arrow
question_answer 172) Synchronous flowering in pine apple can be achieved by the application of :
A)
kinetin
done
clear
B)
2,4, D
done
clear
C)
abscisic acid
done
clear
D)
NAA
done
clear
View Answer play_arrow
question_answer 173) Which of the following type of phytohormones, resemble the nucleic acids in some structural aspects :
A)
cytokinin
done
clear
B)
auxins
done
clear
C)
gibberellins
done
clear
D)
abscisic acid
done
clear
View Answer play_arrow
question_answer 174) The terminal acceptor of electrons in non-cyclic photophosphorylation is
A)
plastoquinone
done
clear
B)
plastocyanin
done
clear
C)
NAD phosphate
done
clear
D)
ferredoxin
done
clear
View Answer play_arrow
question_answer 175) Wareing and his co-workers were associated with the discovery of :
A)
abscisic acid
done
clear
B)
auxins
done
clear
C)
cytokinins
done
clear
D)
gibberellins
done
clear
View Answer play_arrow
question_answer 176) The scientist associated with discovery of photsystems :
A)
Calvin
done
clear
B)
Hill
done
clear
C)
Emerson
done
clear
D)
Ruben
done
clear
View Answer play_arrow
question_answer 177) The wing like portion on the stipe of Agaricus basidiocarp is the remnant of:
A)
velum
done
clear
B)
hymenium
done
clear
C)
paraphyses
done
clear
D)
gill
done
clear
View Answer play_arrow
question_answer 178) The 4-carbon compound formed during RuBP regeneration reactions of calvin cycle is :
A)
sedaheptulose phosphate
done
clear
B)
xylulose phosphate
done
clear
C)
erythrose phosphate
done
clear
D)
glyceraldehyde phosphate
done
clear
View Answer play_arrow
question_answer 179) Embryo culture in angiosperms was first tried by:
A)
Manning
done
clear
B)
Guha and Maheshwari
done
clear
C)
Nitsch
done
clear
D)
White
done
clear
View Answer play_arrow
question_answer 180) One of the following is involved denitrification reactions:
A)
Nitrosomonas
done
clear
B)
Nitrobacter
done
clear
C)
Thiobacillus
done
clear
D)
Rhodospirillwn
done
clear
View Answer play_arrow
question_answer 181) The book "Historia Plantarum" was written by:
A)
Bauhin
done
clear
B)
Theophrastus
done
clear
C)
Malpighi
done
clear
D)
Bentham
done
clear
View Answer play_arrow
question_answer 182) Chlorophyll containing roots are found in:
A)
Avicennia
done
clear
B)
Vanda
done
clear
C)
Taemophyllum
done
clear
D)
Lorwthus
done
clear
View Answer play_arrow
question_answer 183) Dahlia has:
A)
tendrillar roots
done
clear
B)
respiratory roots
done
clear
C)
nodular roots
done
clear
D)
storage roots
done
clear
View Answer play_arrow
question_answer 184) The stem modification in Hydrocotyle vulgaris is:
A)
stolen
done
clear
B)
sucker
done
clear
C)
offset
done
clear
D)
runner
done
clear
View Answer play_arrow
question_answer 185) Unifacial leaves are found in :
A)
Cestrum
done
clear
B)
Allium
done
clear
C)
Hibiscus
done
clear
D)
Eucalyptus
done
clear
View Answer play_arrow
question_answer 186) Thorns which help in climbing are found in
A)
Carissa
done
clear
B)
Ulex
done
clear
C)
Bougainvillea
done
clear
D)
Dawsonia
done
clear
View Answer play_arrow
question_answer 187) Amentum is found in :
A)
A chyranthes
done
clear
B)
Betula
done
clear
C)
Dolichos
done
clear
D)
Oryza
done
clear
View Answer play_arrow
question_answer 188) In Benincase the condition of androecium described as:
A)
diadelphous
done
clear
B)
oolyadelphous
done
clear
C)
synandrous
done
clear
D)
syngenesious
done
clear
View Answer play_arrow
question_answer 189) Malacophily is observed in the species one of the following:
A)
Zostera
done
clear
B)
Lemna
done
clear
C)
Ruppia
done
clear
D)
Bignonia
done
clear
View Answer play_arrow
question_answer 190) A true fruit along with a false fruit is found
A)
Mangifera
done
clear
B)
Terminalia
done
clear
C)
Anacardium
done
clear
D)
Cocas
done
clear
View Answer play_arrow
question_answer 191) The remnant of nucellus in the seed is:
A)
endosperm
done
clear
B)
aril
done
clear
C)
cotyledon
done
clear
D)
perisperm
done
clear
View Answer play_arrow
question_answer 192) The nucleolus is concerned with the synthesis of:
A)
carbohydrates
done
clear
B)
proteins
done
clear
C)
fats
done
clear
D)
resins
done
clear
View Answer play_arrow
question_answer 193) The organdies which play an important role in the formation of cell plate are :
A)
microtubules
done
clear
B)
endopiasmic reticulum
done
clear
C)
ribosomes
done
clear
D)
Golgi complexes
done
clear
View Answer play_arrow
question_answer 194) In one of the following plants chromosomes were first observed:
A)
Tradescantia
done
clear
B)
Allium
done
clear
C)
Colchicum
done
clear
D)
Vallisneria
done
clear
View Answer play_arrow
question_answer 195) Pairing of homologous chromosomes takes place during :
A)
leptotene
done
clear
B)
zygotene
done
clear
C)
pachytene
done
clear
D)
diplotene
done
clear
View Answer play_arrow
question_answer 196) The extension of pith between the vascular bundies as seen in the transverse section of a young dicot stem is called :
A)
Phloem
done
clear
B)
Medullary ray
done
clear
C)
Cambium
done
clear
D)
Xylem
done
clear
View Answer play_arrow
question_answer 197) The ground tissue is undifferentiated in :
A)
dicot stem
done
clear
B)
dicot root
done
clear
C)
monocot stem
done
clear
D)
monocot root
done
clear
View Answer play_arrow
question_answer 198) Periblem is a part of:
A)
cortex
done
clear
B)
stele
done
clear
C)
apical meristem
done
clear
D)
vascular bundle
done
clear
View Answer play_arrow
question_answer 199) Reticulate and scleriform thickenings are found in one of the following:
A)
sieve tubes
done
clear
B)
metaxylem vessels
done
clear
C)
protoxylem vessels
done
clear
D)
companion cells
done
clear
View Answer play_arrow
question_answer 200) The sclerenchymatous part of pericycle which gives mechanical support is called:
A)
hard bast
done
clear
B)
bast fibres
done
clear
C)
sclereids
done
clear
D)
wood fibres
done
clear
View Answer play_arrow