-
What are canal rays?
View Answer play_arrow
-
If an atom contains one electron and one proton. Will it carry any charge or not?
View Answer play_arrow
-
On the basis of Thomson's model of an atom, explain how the atom is neutral as a whole.
View Answer play_arrow
-
On the basis of Rutherford's model of an atom, which sub-atomic particle is present in the nucleus of an atom?
View Answer play_arrow
-
Draw a sketch of Bohr's model of an atom with three shells.
View Answer play_arrow
-
What do you think would be the observation if the a-particle scattering experiment is carried out using a foil of a metal other than gold?
View Answer play_arrow
-
Name the three sub-atomic particles of an atom.
View Answer play_arrow
-
Helium atom has an atomic mass of 4 u and two protons in its nucleus. How many neutrons does it have?
View Answer play_arrow
-
Write the distribution of electrons in carbon and sodium atoms.
View Answer play_arrow
-
If K and L shells of an atom are full, then what would be the total number of electrons in the atom?
View Answer play_arrow
-
How will you find the valency of chlorine, sulphur and magnesium?
View Answer play_arrow
-
If number of electrons in an atom is 8 and number of protons is also 8, then
(i) what is the atomic number of the atom ? and
(ii) what is the charge on the atom?
View Answer play_arrow
-
Atomic numbers of oxygen and sulphur are 8 and 16 respectively. Oxygen atom has 6 neutrons and sulphur atom has 16 neutrons. Find out the mass number of oxygen and sulphur atom.
View Answer play_arrow
-
For the symbol H, D and T tabulate three sub-atomic particles found in each of them.
View Answer play_arrow
-
Write the electronic configuration of any one pair of isotopes and isobars.
View Answer play_arrow
-
Compare the properties of electrons, protons and neutrons.
View Answer play_arrow
-
What are the limitations of J.J. Thomson's model of the atom?
View Answer play_arrow
-
What are the limitations of Rutherford's model of the atom?
View Answer play_arrow
-
Describe Bohr's model of the atom.
View Answer play_arrow
-
Compare all the proposed models of an atom given in this chapter.
View Answer play_arrow
-
Summarize the rules for writing the distribution of electrons in various shells for the first eighteen elements.
View Answer play_arrow
-
Define valency by taking examples of silicon and oxygen.
View Answer play_arrow
-
Explain with examples (i) Atomic number, (ii) Mass number, (iii) Isotopes and (iv) Isobars.
Give any two uses of isotopes.
View Answer play_arrow
-
Na+ has completely filled K and L shells. Explain.
View Answer play_arrow
-
If bromine atom is available in the form of, say two isotopes 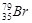 (49.70%) and
(49.70%) and 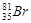 (50.30%) calculate the average atomic mass of bromine atom.
(50.30%) calculate the average atomic mass of bromine atom.
View Answer play_arrow
-
The average atomic mass of sample of an element X is 16.2 u. What are the percentages of isotopes

and

in the sample?
-
If Z = 3, what would be the valency of the element? Also, name the element.
View Answer play_arrow
-
Composition of the nuclei of two atomic species X and Y are given as under:
X Y
Protons = 6 6
Neutrons = 6 8
Give the mass numbers of X and Y. What is the relation between the two species?
View Answer play_arrow
-
For the following statements, write T for True and F for False.
(a) J.J. Thomson proposed that the nucleus of an atom contains only nucleons.
(b) A neutron is formed by an electron and a proton combining together. Therefore, it is neutral.
(c) The mass of an electron is about 1/2000 times that of proton.
(d) An isotope of iodine is used for making tincture iodine, which is used as a medicine.
View Answer play_arrow
-
Rutherford's alpha-particle scattering experiment was responsible for the discovery of
(a) atomic nucleus
(b) electron
(c) proton
(d) neutron
View Answer play_arrow
-
Isotopes of an element have
(a) the same physical properties
(b) different chemical properties
(c) different number of neutrons
(d) different atomic numbers
View Answer play_arrow
-
Number of valence electrons in Cl- ion are :
(a) 16 (b) 8
(c) 17 (d) 18
View Answer play_arrow
-
Which one of the following is a correct electronic configuration of sodium?
(a) 2, 8 (b) 8, 2, 1
(c) 2, 1, 8 (d) 2, 8, 1
View Answer play_arrow
-
Complete the following table.
|
Atomic number
|
Mass number
|
Number of neutrons
|
Number of protons
|
Number of electrons
|
Name of the atomic species
|
|
9
|
-
|
10
|
-
|
-
|
-
|
|
16
|
32
|
-
|
-
|
-
|
Sulphur
|
|
-
|
23
|
-
|
12
|
-
|
-
|
|
-
|
2
|
-
|
1
|
-
|
-
|
|
-
|
1
|
0
|
1
|
0
|
-
|
View Answer play_arrow
-
question_answer35)
Which of the following correctly represent the electronic distribution in
the Mg atom?
(a) 3, 8, 1 (b) 2, 8, 2
(c) 1, 8, 3 (d) 8, 2,
View Answer play_arrow
-
question_answer36)
Rutherford's alpha(a)
particles scattering experiment resulted into discovery of
(a) electron (b) proton (c)
nucleus in the atom (d) atomic mass
View Answer play_arrow
-
question_answer37)
The number of electrons in an element X is 15 and the
number of neutrons is 16. Which of the following is the correct representation
of the element?
(a)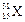 (b)
(b)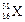 (c)
(c)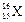 (d)
(d)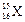
View Answer play_arrow
-
question_answer38)
Dalton's atomic theory successfully explained.
(i) law of conservation of mass (ii)
law of constant composition
(iii) law of radioactivity (iv)
law of multiple proportion
(a) (i), (ii) and (iii) (b) (i), (iii) and (iv) (c)
(ii), (iii) and (iv) (d) (i), (ii) and (iv)
View Answer play_arrow
-
question_answer39)
Which of the following statements about Rutherford's model
of atom are correct?
(i) Considered the nucleus as positively charged.
(ii) Established that the cc-particles are four times as
heavy as a hydrogen atom.
(iii) Can be compared to solar system.
(iv) Was in agreement with Thomson's model.
(a) (i) and (iii) (b) (ii) and
(iii) (c) (i) and (iv) (d) Only (i)
View Answer play_arrow
-
question_answer40)
Which of the following are true for an element?
Atomic number = number of protons + number of electrons
Mass number = number of protons + number of neutrons
Atomic number = number of protons = number of neutrons
Atomic number = number of protons = number of electrons
(a) (i) and (ii) (b) (i) and
(iii) (c) (ii) and (iii) (d) (ii) and
(iv)
View Answer play_arrow
-
question_answer41)
In the Thomson's model of atom, which of the following
statements are correct?
(i) The mass of the atom is assumed to be uniformaly
distributed over the atom.
(ii) The positive charge is assumed to be uniformaly
distributed over the atom.
(iii) The electrons are uniformaly distributed in the
positively charged sphere.
(iv) The electrons attract each other to stabilize the
atom.
(a) (i), (ii) and (iii) (b) (i) and (iii) (c)
(i) and (iv) (d) (i), (iii) and (iv)
View Answer play_arrow
-
question_answer42)
Rutherford's a-particle
scattering experiment showed that
(i) electrons have negative charge.
(ii) the mass and positive charge of the atom is
concentrated in the nucleus.
(iii) neutron exists in the nucleus.
(iv) most of the space in atom is empty.
Which of the above statements are correct?
(a) (i) and (iii) (b) (ii) and
(iv) (c) (i) and (iv) (d) (iii) and (iv)
View Answer play_arrow
-
question_answer43)
The ion of an element has 3 positive charges. Mass number
of the atom is 27 and the number of neutrons is 14. What is the number of
electrons in the ion?
(a) 13 (b)
10 (c) 14 (d)
16
View Answer play_arrow
-
question_answer44)
Identify the Mg2+ ion from the figure where, n
and p represent the number of neutrons and protons respectively.
View Answer play_arrow
-
question_answer45)
(a) 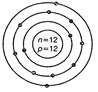 (b)
(b) 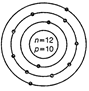 (c)
(c) 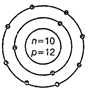 (d)
(d) 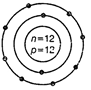 In a sample of ethyl ethanoate
In a sample of ethyl ethanoate 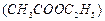 the
two oxygen atoms have the same number of electrons but different, number of
neutrons, which of the following is the correct reason for it?
(a) One of the oxygen atoms have gained electrons
(b) One of the oxygen atoms has gained two neutrons
(c) The two oxygen atoms are isotopes
(d) The two oxygen atoms are isobars
the
two oxygen atoms have the same number of electrons but different, number of
neutrons, which of the following is the correct reason for it?
(a) One of the oxygen atoms have gained electrons
(b) One of the oxygen atoms has gained two neutrons
(c) The two oxygen atoms are isotopes
(d) The two oxygen atoms are isobars
View Answer play_arrow
-
question_answer46)
Elements with valency 1 are
(a) always metals (b)
always metalloids
(c) either metals or non-metals (d)
always non-metals
View Answer play_arrow
-
question_answer47)
The first model of an atom was given by
(a) N Bohr (b) E Goldstein (c)
Rutherford (d) JJ Thomson
View Answer play_arrow
-
question_answer48)
An atom with 3 protons and 4 neutrons will have a valency
of
(a) 3 (b)
7 (c) 1 (d)
4
View Answer play_arrow
-
question_answer49)
The electron distribution in an aluminium atom is
(a) 1, 8, 3 (b) 2, 8,
2 (c) 8, 2, 3 (d) 2,
3, 8
View Answer play_arrow
-
question_answer50)
Which of the following in figure do not represent Bohr's
model of an atom correctly?
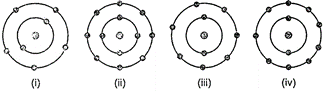 (a) (i) and (ii) (b) (ii) and
(iii) (c) (ii) and (iv) (d) (i) and (iv)
(a) (i) and (ii) (b) (ii) and
(iii) (c) (ii) and (iv) (d) (i) and (iv)
View Answer play_arrow
-
question_answer51)
Which of the following statement is always correct?
(a) An atom has equal number of electrons and protons
(b) An atom has equal number of electrons and neutrons
(c) An atom has equal number of protons and neutrons
(d) An atom has equal number of electrons, protons and
neutrons
View Answer play_arrow
-
question_answer52)
Atomic models have been improved over the years. Arrange
the following atomic models in the order of their chronological order.
(i) Rutherford's atomic model
(ii) Thomson's atomic model
(iii) Bohr's atomic model
(a) (i), (ii) and (iii) (b) (ii), (iii) and
(i) (c) (ii), (i) and (iii) (d) (iii), (ii) and (i)
View Answer play_arrow
-
question_answer53)
Is it possible for the atom of an element to have one electron,
one proton and no neutron. If so, name the element.
View Answer play_arrow
-
question_answer54)
Write any two observations which support the fact that
atoms are divisible.
View Answer play_arrow
-
question_answer55)
Will 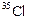 and
and
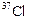 have
different valencies? Justify your answer.
have
different valencies? Justify your answer.
View Answer play_arrow
-
question_answer56)
Why did Rutherford select a gold foil in his 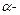 ray
scattering experiment?
ray
scattering experiment?
View Answer play_arrow
-
question_answer57)
Find out the valency of the atoms represented by the
Figure (a) and (b).
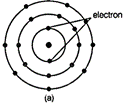

View Answer play_arrow
-
question_answer58)
One electron is present in the outermost shell of the atom
of an element X. What would be the nature and value of charge on the ion formed
if this electron is removed from the outermost shell?
View Answer play_arrow
-
question_answer59)
Write down the electron distribution of chlorine atom. How
many electrons are there in the L-shell? Atomic number of chlorine is = 17.
View Answer play_arrow
-
question_answer60)
In the atom of an element X, 6 electrons are present in
the outermost shell. If it acquires noble gas configuration by accepting requisite
number of electrons, then what would be the charge on the
ion so formed?
View Answer play_arrow
-
question_answer61)
What information do you get from the figure about the
atomic number, mass number and valency of atoms X, Y and Z? Give your answer in
a tabular form.
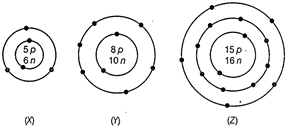
View Answer play_arrow
-
question_answer62)
In response to a question, a student stated that in an
atom, the number of protons is greater than the number of neutrons, which in
turn is greater than the number of electrons. Do you agree with the statement?
Justify your answer.
View Answer play_arrow
-
question_answer63)
Calculate the number of neutrons present in the nucleus of
an element X which is represented as 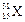
View Answer play_arrow
-
question_answer64)
Match the names of the Scientists given in Column A with their contributions
towards the understanding of the atomic structure as given in Column B.
|
|
Column A
|
Column B
|
|
(a)
|
Ernest Rutherford
|
(i) Indivisibility of atoms
|
|
(b)
|
JJ Thomson
|
(ii) Stationary orbits
|
|
(c)
|
Dalton
|
(iii) Concept or nucleus
|
|
(d)
|
Neils Bohr
|
(iv) Discovery of electrons
|
|
(e)
|
James Chadwick
|
(v) Atomic number
|
|
(f)
|
E Goldstein
|
(vi) Neutron
|
|
(g)
|
Mosely
|
(vii)Canal rays
|
View Answer play_arrow
-
question_answer65)
The atomic number of calcium and argon are 20 and 18 respectively, but the
mass number of both these elements is 40. What is the name given to such a pair
of elements?
A pair of elements having same mass number but different
atomic number is called isobars.
View Answer play_arrow
-
question_answer66)
Complete the table on the basis of information available
in the symbols given below.
View Answer play_arrow
-
question_answer67)
Helium atom has 2 electrons in its valence shell but its
valency is not 2. Explain.
View Answer play_arrow
-
question_answer68)
Fill in the blanks in the following statements.
(a) Rutherford's a-particle scattering experiment lead to
the discovery of the..........
(b) Isotopes have same ......... but different...........
(c) Neon and chlorine have atomic numbers 10 and 17
respectively. Their valencies will be ......... and ......... respectively,
(d) The electronic configuration of silicon is .........
and that of sulphur is
View Answer play_arrow
-
question_answer69)
An element X has a mass number 4 and atomic number 2.
Write the valency of this element.
View Answer play_arrow
-
question_answer70)
Why do helium, neon and argon have a zero valency?
View Answer play_arrow
-
question_answer71)
The ratio of the radii of hydrogen atom and its nucleus is
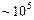 .
Assuming the atom and the nucleus to be spherical.
(a) What will be the ratio of their sizes ?
(b) If atom is represented by planet earth
.
Assuming the atom and the nucleus to be spherical.
(a) What will be the ratio of their sizes ?
(b) If atom is represented by planet earth 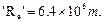 estimate
the size of the nucleus.
estimate
the size of the nucleus.
View Answer play_arrow
-
question_answer72)
Enlist the conclusions drawn by Rutherford from his 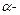 ray
scattering experiment.
ray
scattering experiment.
View Answer play_arrow
-
question_answer73)
In what way is the Rutherford's atomic model different
from that of Thomson's atomic model?
View Answer play_arrow
-
question_answer74)
What were the drawbacks of Rutherford's model of an atom?
View Answer play_arrow
-
question_answer75)
What are the postulates of Bohr's model of an atom?
View Answer play_arrow
-
question_answer76)
Show diagrammatically the electron distributions in a
sodium atom and a sodium ion and also give their atomic number.
View Answer play_arrow
-
question_answer77)
In the gold foil experiment of Geiger and Marsden, that paved the way for
Rutherford's model of an atom, ~ 1.00% of the α - particles were found to deflect at
angles > 50°. If one mole of 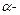 particles
were bombarded on the gold foil, compute the number of a-particles that would
deflect at angles less than 50°.
particles
were bombarded on the gold foil, compute the number of a-particles that would
deflect at angles less than 50°.
View Answer play_arrow
![]() and
and
![]() have
different valencies? Justify your answer.
have
different valencies? Justify your answer.
![]() ray
scattering experiment?
ray
scattering experiment?


![]() ray
scattering experiment.
ray
scattering experiment.
