A fan is moving around its axis. What will be its motion regarded as?
A)
Pure rolling
done
clear
B)
Rolling with slipping
done
clear
C)
Skidding
done
clear
D)
Pure rotation
done
clear
View Answer play_arrow
If inductance of a coil is 'L' and current passing through it is 'f. Find energy stored in it:
A)
\[L{{i}^{2}}\]
done
clear
B)
\[4L{{i}^{2}}\]
done
clear
C)
\[Li\]
done
clear
D)
\[\frac{1}{2}L{{i}^{2}}\]
done
clear
View Answer play_arrow
If \[\lambda ={{10}^{-19}}m\] changes to \[\lambda '=0.5\times {{10}^{-10}}m.\]. Find energy difference \[(\Delta E)\] give to the particle:
A)
\[\Delta E\]is equal to \[{{\left( \frac{1}{4} \right)}^{th}}\] of initial energy.
done
clear
B)
\[\Delta E\]is equal to \[{{\left( \frac{1}{2} \right)}^{th}}\] of initial energy.
done
clear
C)
\[\Delta E\]is equal to twice of initial energy.
done
clear
D)
\[\Delta E\] is equal to initial energy.
done
clear
View Answer play_arrow
If wavelength \[\lambda =5400\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]is threshold value for a certain metal, then its work function would be:
A)
\[2.3\,eV~\]
done
clear
B)
\[0.025\,eV\]
done
clear
C)
\[23\,eV\]
done
clear
D)
\[0.23\,eV\]
done
clear
View Answer play_arrow
If maximum height and range of a projectile are same. What is the angle of projection?
A)
\[{{30}^{o}}\]
done
clear
B)
\[{{76}^{o}}\]
done
clear
C)
\[{{53}^{o}}\]
done
clear
D)
\[{{90}^{o}}\]
done
clear
View Answer play_arrow
At what temperature volume of an ideal gas at \[{{0}^{o}}C\]becomes triple?
A)
\[{{546}^{o}}C\]
done
clear
B)
\[{{182}^{o}}C\]
done
clear
C)
\[{{819}^{o}}C\]
done
clear
D)
\[{{646}^{o}}C\]
done
clear
View Answer play_arrow
If kinetic energy is doubled, find fractional change in momentum:
A)
\[\sqrt{2}\]
done
clear
B)
\[3\sqrt{2}\]
done
clear
C)
\[\frac{1}{3\sqrt{2}}\]
done
clear
D)
\[\frac{1}{2\sqrt{2}}\]
done
clear
View Answer play_arrow
If velocity of a charged particle is doubled and strength of magnetic field is halved, then radius will be:
A)
8 times
done
clear
B)
6 times
done
clear
C)
4 times
done
clear
D)
3 times
done
clear
View Answer play_arrow
If phosphorus and arsenic impurities are added to a semiconductor, then it will be:
A)
transistor
done
clear
B)
p-type semiconductor
done
clear
C)
amplifier
done
clear
D)
n-type semiconductor
done
clear
View Answer play_arrow
If a sample of 16g radioactive substance disintegrate to 1g in 120 days, then what will be the half life of the sample?
A)
15 days
done
clear
B)
60days
done
clear
C)
30 days
done
clear
D)
25 days
done
clear
View Answer play_arrow
The value\[+\frac{1}{2}\]and \[-\frac{1}{2}\] of spin quantum number show:
A)
rotation of \[{{e}^{-}}\] clockwise and anticlockwise direction respectively.
done
clear
B)
rotation of \[{{e}^{-}}\] anticlockwise and clockwise direction respectively.
done
clear
C)
rotation in any direction according to convention
done
clear
D)
none of the above
done
clear
View Answer play_arrow
When resistance of wire of \[\text{50 }\!\!{}^\circ\!\!\text{ C}\] is \[5R\Omega \] and at \[{{100}^{o}}C\]is\[6R\Omega \]. Find resistance at\[{{0}^{o}}C\].
A)
\[1R\Omega \]
done
clear
B)
\[8R\Omega \]
done
clear
C)
\[3R\Omega \]
done
clear
D)
\[4R\Omega \]
done
clear
View Answer play_arrow
\[{{M}_{P}}\] and \[{{M}_{X}}\] are masses of proton and neutron, respectively at rest. When they combine to form deuterium nucleus. The mass of the nucleus will be:
A)
Less than \[{{M}_{P}}\]
done
clear
B)
Less than \[({{M}_{P}}+{{M}_{N}})\]
done
clear
C)
Less than \[({{M}_{P}}+2{{M}_{N}})\]
done
clear
D)
greater than \[({{M}_{P}}+2{{M}_{N}})\]
done
clear
View Answer play_arrow
When the door of refrigerator is opened while connected to supply, the room:
A)
gets cooled
done
clear
B)
gets heated
done
clear
C)
no effect
done
clear
D)
temperature is not given
done
clear
View Answer play_arrow
A bar magnet is dropped between a current carrying coil. what would be its acceleration:
A)
'g' downwards
done
clear
B)
greater than 'g' downwards
done
clear
C)
less than 'g' downwards
done
clear
D)
bar will be stationary
done
clear
View Answer play_arrow
When impedance is \[\sqrt{3}\] times of resistance find phase difference:
A)
\[{{90}^{o}}\]
done
clear
B)
\[{{30}^{o}}\]
done
clear
C)
\[{{60}^{o}}C\]
done
clear
D)
data is incomplete
done
clear
View Answer play_arrow
When reading of an ammeter is 10A. The peak value of current is:
A)
\[\frac{10}{\sqrt{2}}A\]
done
clear
B)
\[\frac{5}{\sqrt{2}}A\]
done
clear
C)
\[20\sqrt{2}A\]
done
clear
D)
\[10\sqrt{2}A\]
done
clear
View Answer play_arrow
Five resistance of resistance RQ are these, 3 are connectd in parallel and are joined to them in series. Find resultant resistance:
A)
\[\left( \frac{3}{7} \right)R\,\Omega \]
done
clear
B)
\[\left( \frac{7}{3} \right)R\,\Omega \]
done
clear
C)
\[\left( \frac{9}{8} \right)R\,\Omega \]
done
clear
D)
\[\left( \frac{8}{9} \right)R\,\Omega \]
done
clear
View Answer play_arrow
If a capacitor of capacitance \[10\mu f\] has potential difference of 100 volts across its ends. The energy stored in it is:
A)
\[0.05\,J\]
done
clear
B)
25
done
clear
C)
\[0.005\,J\]
done
clear
D)
\[0.001\,J\]
done
clear
View Answer play_arrow
Two conductors having current in opposite directions:
A)
attract each other
done
clear
B)
repel each other
done
clear
C)
do not affect each other
done
clear
D)
effect depends on the material of wire
done
clear
View Answer play_arrow
A cube has point changes of magnitude q at all its vertices, electric field at the centre of the cube is:
A)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{6q}{3{{a}^{2}}}\]
done
clear
B)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{8q}{{{a}^{2}}}\]
done
clear
C)
zero
done
clear
D)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{-8q}{{{a}^{2}}}\]
done
clear
View Answer play_arrow
Distance between successive compression and rare fraction is 1m and velocity of sound is \[360\text{ }m{{s}^{-1}}\] find frequency:
A)
\[180\,Hz\]
done
clear
B)
\[45\,Hz\]
done
clear
C)
\[120\,Hz\]
done
clear
D)
\[90\,Hz\]
done
clear
View Answer play_arrow
A source and observer are approaching each other with \[50\,m{{s}^{-1}}\] velocity. What will be original frequency, if the observer receives 400 cycle/sec?
A)
300cycle/sec
done
clear
B)
320 cycle/sec
done
clear
C)
340 cycle/sec
done
clear
D)
330 cycle/sec
done
clear
View Answer play_arrow
A source is approaching a stationary observer with velocity \[{{\left( \frac{1}{10} \right)}^{th}}\]that of sound. Ratio of observed and real frequencies will be:
A)
\[\frac{9}{10}\]
done
clear
B)
\[\frac{11}{10}\]
done
clear
C)
\[\frac{10}{17}\]
done
clear
D)
\[\frac{10}{9}\]
done
clear
View Answer play_arrow
Separation between slits is halved and between screen and slits is doubled. Find fringe width if original is \[\omega \]:
A)
\[\omega \]
done
clear
B)
\[9\,\omega \]
done
clear
C)
\[4\,\omega \]
done
clear
D)
\[2\,\omega \]
done
clear
View Answer play_arrow
An equilateral prism has \[\mu =\sqrt{3},\] its angle of minimum deviation is:
A)
\[{{75}^{o}}\]
done
clear
B)
\[{{60}^{o}}\]
done
clear
C)
\[{{90}^{o}}\]
done
clear
D)
\[{{45}^{o}}\]
done
clear
View Answer play_arrow
Maximum and minimum intensities obtained by two sources having intensities \[4l\] and \[l\] are:
A)
\[5I,-3I\]
done
clear
B)
\[9I,I\]
done
clear
C)
\[9I,-I\]
done
clear
D)
\[5I,3I\]
done
clear
View Answer play_arrow
Magnification of a telescope having focal length of objective lens and eye piece \[{{f}_{0}}\] and \[{{f}_{e}}\] respectively:
A)
\[\frac{{{f}_{e}}}{{{f}_{o}}}\]
done
clear
B)
\[\frac{{{f}_{o}}}{{{f}_{e}}}\]
done
clear
C)
\[\frac{1-{{f}_{o}}}{{{f}_{e}}}\]
done
clear
D)
\[\frac{{{f}_{o}}}{{{f}_{e}}-1}\]
done
clear
View Answer play_arrow
If separation between screen and source is increased by 2%,What would be the effect on the intensity?
A)
Increase by 4%
done
clear
B)
Increase by 2%
done
clear
C)
Decrease by 2%
done
clear
D)
Decrease by 4%
done
clear
View Answer play_arrow
An object is placed of a distance of 10cm from a convex lens of power 5D. Find the position of the image:
A)
\[-20cm\]
done
clear
B)
\[40cm\]
done
clear
C)
\[20cm\]
done
clear
D)
\[-40cm\]
done
clear
View Answer play_arrow
In an adiabatic process:
A)
\[{{P}^{\gamma }}V=\]constant
done
clear
B)
\[T{{V}^{\gamma -1}}=\] constant
done
clear
C)
PV= constant
done
clear
D)
all of these
done
clear
View Answer play_arrow
If there is a straight line parallel to volume axis in a P-V diagram, then it is a graph:
A)
isochoric
done
clear
B)
isobaric
done
clear
C)
isothermal
done
clear
D)
none of these
done
clear
View Answer play_arrow
Effeciency of engine working at \[{{40}^{o}}C,\text{ }{{20}^{o}}C\]is:
A)
\[0.064%\]
done
clear
B)
\[6.4\]
done
clear
C)
\[64%\]
done
clear
D)
\[6.4%\]
done
clear
View Answer play_arrow
One filament takes 10 min to heat a kattle and another takes 15 minutes. If connected in parallel they combindly take__ minute to heat the same kattle.
A)
6min
done
clear
B)
\[12.5min\]
done
clear
C)
24min
done
clear
D)
\[7.5min\]
done
clear
View Answer play_arrow
The means of energy transfer in vaccum is:
A)
irradiation
done
clear
B)
convection
done
clear
C)
radiation
done
clear
D)
conduction
done
clear
View Answer play_arrow
If electric flux varies according to \[\phi =3{{t}^{2}}+4t+2.\]Find emf at\[t=2\,sec\].
A)
32 volt
done
clear
B)
18 volt
done
clear
C)
19 volt
done
clear
D)
16 volt
done
clear
View Answer play_arrow
If relation between distance and time is \[s=a+bt+c{{t}^{2}}\]. Find initial velocity and acceleration.
A)
\[b+2ct,2c\]
done
clear
B)
\[b,2c\]
done
clear
C)
\[2c,b\]
done
clear
D)
\[b+2c,2c\]
done
clear
View Answer play_arrow
If 'n' drops of potential 'V merge. Find new potential on the big drop:
A)
\[{{n}^{2/3}}V\]
done
clear
B)
\[{{n}^{1/3}}V\]
done
clear
C)
\[nV\]
done
clear
D)
\[{{V}^{n/3}}\]
done
clear
View Answer play_arrow
If we hollow the ball of pendulum, and fill it with sand, what would be the effect of change on the centre of mass?
A)
it would distorted only
done
clear
B)
its value will decrease only
done
clear
C)
its value will increase only
done
clear
D)
its value will first increase and then decrease
done
clear
View Answer play_arrow
Amplitude of a pendulum is 60mm and angular velocity is\[2\text{ }rad\text{ }se{{c}^{-1}}\]. Find its velocity of its displacement is 20mm.
A)
\[120mm/se{{c}^{-1}}\]
done
clear
B)
\[133mm/se{{c}^{-1}}\]
done
clear
C)
\[155mm/se{{c}^{-1}}\]
done
clear
D)
\[12mm/se{{c}^{-1}}\]
done
clear
View Answer play_arrow
A body of mass \[0.1\,kg\]attains a velocity of \[10m{{s}^{-1}}\]in\[0.1\text{ }sec\]. The force acting on the body is:
A)
\[10\,N\]
done
clear
B)
\[10.01\,N\]
done
clear
C)
\[5.\text{1}N\]
done
clear
D)
\[100\text{ }N\]
done
clear
View Answer play_arrow
A man is height of\[100\,m\]. He sees a car which makes an angle of \[\frac{\pi }{6}\]with man's position. If the car moves at a point-where angle is\[\frac{\pi }{3},\] what is the distance moved by it?
A)
\[\left( \frac{400}{\sqrt{3}} \right)\,m\]
done
clear
B)
\[\left( 200\sqrt{3} \right)m\]
done
clear
C)
\[\left( \frac{200}{\sqrt{3}} \right)\,m\]
done
clear
D)
\[\left( \frac{150}{\sqrt{3}} \right)\,m\]
done
clear
View Answer play_arrow
A ball is thrown upward, it takes 4sec. to reach back to the ground. Find its initial velocity:
A)
\[60\,m{{s}^{-1}}\]
done
clear
B)
\[10\,m{{s}^{-1}}\]
done
clear
C)
\[40\,m{{s}^{-1}}\]
done
clear
D)
\[20\,m{{s}^{-1}}\]
done
clear
View Answer play_arrow
A ball is thrown from height 'h' and another from '2h' The ratio of time taken by the two ball to reach ground is:
A)
\[1:\sqrt{2}\]
done
clear
B)
\[\sqrt{2}:1\]
done
clear
C)
\[4:1\]
done
clear
D)
\[1:4\]
done
clear
View Answer play_arrow
Dimension of relative density is:
A)
\[Kg\,{{m}^{-3}}\]
done
clear
B)
\[M{{L}^{-3}}\]
done
clear
C)
dimensionless
done
clear
D)
\[{{M}^{2}}{{L}^{-6}}\]
done
clear
View Answer play_arrow
Dissolving \[120\text{ }g\]of urea (mol. wt. 60) in \[1000\text{ }g\]of water gave a solution of density\[1.15\text{ }g/ml\]. The molarity of the solution is
A)
\[1.78\text{ }M\]
done
clear
B)
\[2.00\text{ }M\]
done
clear
C)
\[2.05\text{ }M\]
done
clear
D)
\[2.22M\]
done
clear
View Answer play_arrow
The reaction, \[3ClO_{(aq)}^{-}\xrightarrow{{}}ClO_{3(aq)}^{-}+2Cl_{(aq)}^{-}\] is an example of
A)
oxidation reaction
done
clear
B)
reduction reaction
done
clear
C)
disproportionate reaction
done
clear
D)
decomposition reaction
done
clear
View Answer play_arrow
The density of neon will be highest at
A)
STP
done
clear
B)
\[{{0}^{o}}C,\]2 atm
done
clear
C)
\[{{273}^{o}}C,\]1 atm
done
clear
D)
\[{{273}^{o}}C,\]2 atm
done
clear
View Answer play_arrow
For a monoatomic gas kinetic energy = E. The relation with rms velocity is
A)
\[u={{\left( \frac{2E}{m} \right)}^{{\scriptstyle{}^{1}/{}_{2}}}}\]
done
clear
B)
\[u={{\left( \frac{3E}{2m} \right)}^{{\scriptstyle{}^{1}/{}_{2}}}}\]
done
clear
C)
\[u={{\left( \frac{E}{2m} \right)}^{{\scriptstyle{}^{1}/{}_{2}}}}\]
done
clear
D)
\[u={{\left( \frac{E}{3m} \right)}^{{\scriptstyle{}^{1}/{}_{2}}}}\]
done
clear
View Answer play_arrow
The wavelength associated with a golf ball weighing 200 g and moving at a speed of 5m/h is of the order
A)
\[{{10}^{-10}}m\]
done
clear
B)
\[{{10}^{-20}}m\]
done
clear
C)
\[{{10}^{-30}}m\]
done
clear
D)
\[{{10}^{-40}}m\]
done
clear
View Answer play_arrow
At the critical temperature
A)
liquid and vapour exist in equilibrium
done
clear
B)
vapour state does not exist at all
done
clear
C)
the vapour condense into solid
done
clear
D)
the meniscus between liquid and vapour disappears.
done
clear
View Answer play_arrow
Potassium has a bcc structure with nearest neighbour distance \[4.52\text{ }A{}^\circ \]. Its atomic weight is 39. Its density will be
A)
\[454\text{ }kg\text{ }{{m}^{-3}}\]
done
clear
B)
\[804\text{ }kg\text{ }{{m}^{-3}}\]
done
clear
C)
\[852\text{ }kg\text{ }{{m}^{-3}}\]
done
clear
D)
\[908k\,g{{m}^{-3}}\]
done
clear
View Answer play_arrow
Which of the following is the least stable ion?
A)
\[L{{i}^{+}}\]
done
clear
B)
\[B{{e}^{-}}\]
done
clear
C)
\[{{B}^{-}}\]
done
clear
D)
\[{{C}^{-}}\]
done
clear
View Answer play_arrow
The correct order of hybridisation of the central atom in the following species \[N{{H}_{3}},\,{{[PtC{{l}_{4}}]}^{2}},\,PC{{l}_{5}}\]and \[BC{{l}_{3}}\] is
A)
\[ds{{p}^{2}},ds{{p}^{3}},s{{p}^{2}}\]and \[s{{p}^{3}}\]
done
clear
B)
\[s{{p}^{3}},ds{{p}^{2}},s{{p}^{3}}d\]and \[s{{p}^{2}}\]
done
clear
C)
\[ds{{p}^{2}},\text{ }s{{p}^{3}},\text{ }s{{p}^{3}}\]and \[ds{{p}^{3}}\]
done
clear
D)
\[ds{{p}^{2}},s{{p}^{3}},s{{p}^{2}}\] and \[ds{{p}^{3}}\]
done
clear
View Answer play_arrow
The species having bond order different from that in \[CO\]is
A)
\[N{{O}^{-}}\]
done
clear
B)
\[N{{O}^{+}}\]
done
clear
C)
\[C{{N}^{-}}\]
done
clear
D)
\[{{N}_{2}}\]
done
clear
View Answer play_arrow
Identify the correct order of solubility of \[N{{a}_{2}}S,\]\[CuS\] and \[ZnS\]in aqueous medium
A)
\[CuS>ZnS>N{{a}_{2}}S\]
done
clear
B)
\[ZnS>N{{a}_{2}}S>CuS\]
done
clear
C)
\[N{{a}_{2}}S>CuS>ZnS\]
done
clear
D)
\[N{{a}_{2}}S>ZnS>CuS\]
done
clear
View Answer play_arrow
\[C{{H}_{3}}N{{H}_{2}}\](\[0.1\] mole,\[{{K}_{b}}=5\times {{10}^{-4}}\]) is added to \[0.08\] moles of \[HCl\]and the solution is diluted to one litre, resulting hydrogen ion concentration is
A)
\[1.6\times {{10}^{-11}}\]
done
clear
B)
\[8\times {{10}^{-11}}\]
done
clear
C)
\[5\times {{10}^{-5}}\]
done
clear
D)
\[8\times {{10}^{-2}}\]
done
clear
View Answer play_arrow
The species which by defamation has zero standard molar enthalpy of formation at 298 K is
A)
\[B{{r}_{2(g)}}\]
done
clear
B)
\[C{{l}_{2(g)}}\]
done
clear
C)
\[{{H}_{2}}{{O}_{(g)}}\]
done
clear
D)
\[C{{H}_{4}}_{(g)}\]
done
clear
View Answer play_arrow
The molar ionic conductance?s at infinite dilution of \[M{{g}^{2+}}\] and \[C{{l}^{-}}\] are \[106.1\]and \[76.3\text{ }oh{{m}^{-1}}\text{ }c{{m}^{2}}\text{ }mo{{l}^{-1}}\]respectively. The molar conductance of solution of at infinite dilution will be
A)
\[29.8\text{ }oh{{m}^{-1}}\text{ }c{{m}^{2}}\text{ }mo{{l}^{-1}}\]
done
clear
B)
\[183.4\text{ }oh{{m}^{-1}}\text{ }c{{m}^{2}}\text{ }mo{{l}^{-1}}\]
done
clear
C)
\[285.7\text{ }oh{{m}^{-1}}\text{ }c{{m}^{2}}\text{ }mo{{l}^{-1}}\]
done
clear
D)
\[258.7\text{ }oh{{m}^{-1}}\text{ }c{{m}^{2}}\text{ }mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
When a lead storage battery is discharged
A)
\[S{{O}_{2}}\]is evolved
done
clear
B)
lead is formed
done
clear
C)
lead sulphate is consumed
done
clear
D)
sulphuric acid is consumed
done
clear
View Answer play_arrow
What is the equilibrium constant for the reaction?
Given, \[F{{e}^{2+}}+C{{e}^{4+}}F{{e}^{3+}}+C{{e}^{3+}}\] \[{{E}^{o}}(C{{e}^{4+}}/C{{e}^{3+}})=1.44\,V\] \[{{E}^{o}}(F{{e}^{3+}}/F{{e}^{2+}})=0.68\,V\]
A)
\[4.52\times {{10}^{12}}\]
done
clear
B)
\[6.88\times {{10}^{12}}\]
done
clear
C)
\[7.13\times {{10}^{10}}\]
done
clear
D)
\[5.02\times {{10}^{10}}\]
done
clear
View Answer play_arrow
DDT on exposure to water decomposes. Half ife is equal to 10 years. How much time will take for its decomposition to 99%?
A)
50 years
done
clear
B)
67 years
done
clear
C)
500 years
done
clear
D)
600 years
done
clear
View Answer play_arrow
Cow milk is an example of natural emulsion stabilized by
A)
Fat
done
clear
B)
Water
done
clear
C)
Casein
done
clear
D)
ions
done
clear
View Answer play_arrow
The heat capacity of a bomb calorimeter is\[500\text{ }J{{/}^{o}}C\]. When \[0.1\text{ }g\]of methane was burnt in this calorimeter, the temperature rose by\[{{2}^{o}}C\]. The value of \[\Delta E\]per mole will be
A)
\[+1\,kJ\]
done
clear
B)
\[-1\,kJ\]
done
clear
C)
\[+160\,kJ\]
done
clear
D)
\[-160\,kJ\]
done
clear
View Answer play_arrow
A compound gives a positive test with \[{{I}_{2}}/NaOH\]and is extracted from benzene by saturated \[NaHS{{O}_{3}}\]. The compound is:
A)
\[C{{H}_{3}}{{(C{{H}_{2}})}_{3}}COC{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}{{(C{{H}_{2}})}_{4}}CHO\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}COC{{H}_{2}}C{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}{{(C{{H}_{2}})}_{4}}C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
Which species represents the electrophile in aromatic nitration?
A)
\[N{{O}_{2}}\]
done
clear
B)
\[NO_{2}^{-}\]
done
clear
C)
\[NO_{2}^{+}\]
done
clear
D)
\[NO_{3}^{-}\]
done
clear
View Answer play_arrow
The IUPAC name of the compound \[C{{H}_{3}}C{{H}_{2}}-O-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,}}\,}}\,H\]
A)
ethyl propyloxide
done
clear
B)
ethoxy propane
done
clear
C)
2-ethoxy propane
done
clear
D)
2-propoxyethane
done
clear
View Answer play_arrow
Which of the following statement is correct?
A)
Tin stone is magnetic in nature
done
clear
B)
Wolframite is non-magnetic in nature
done
clear
C)
Wolframite is \[FeW{{O}_{4}}\]
done
clear
D)
Cassiterite and rutile are sulphides of the metals.
done
clear
View Answer play_arrow
Hydrogen burns in air or oxygen with
A)
Invisible pale blue flame
done
clear
B)
blue flame
done
clear
C)
red flame
done
clear
D)
golden flame
done
clear
View Answer play_arrow
Select the incorrect statement
A)
All oxides of alkali metals except \[{{C}_{S2}}O\]possess anti-fluorite structure
done
clear
B)
The crystal structures of \[NaHC{{O}_{3}}\]and \[KHC{{O}_{3}}\]are similar
done
clear
C)
\[L{{i}_{2}}C{{O}_{3}},\]\[L{{i}_{3}}P{{O}_{4}}\] and LiF are insoluble in water
done
clear
D)
None of these
done
clear
View Answer play_arrow
The indicator used in titrating oxalic acid with caustic soda solution is
A)
methyl orange
done
clear
B)
methyl red
done
clear
C)
fluorescein
done
clear
D)
phenolphthalein
done
clear
View Answer play_arrow
The most stable conformation of chlorohydrin at room temperature is
A)
fully eclipsed
done
clear
B)
partially eclipsed
done
clear
C)
gauche,
done
clear
D)
staggered
done
clear
View Answer play_arrow
Which of the following will be aromatic?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
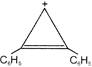
Identify X in the following reaction
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
When propene is allowed to react with diazomethane in presence of UV light the product formated is
A)
methyl cyclopropane
done
clear
B)
cyclopropane
done
clear
C)
butene
done
clear
D)
cyclobutane
done
clear
View Answer play_arrow
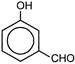
Identify the product Y
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following does not give a white precipitate when boiled with alcoholic silver nitrate solution?
A)
Methyl chloride
done
clear
B)
Allyl chloride
done
clear
C)
Vinyl chloride
done
clear
D)
Benzyl chloride
done
clear
View Answer play_arrow
In which of the following compounds \[-OH\]group is least reactive?
A)
done
clear
B)
done
clear
C)
done
clear
D)
All are equally reactive
done
clear
View Answer play_arrow
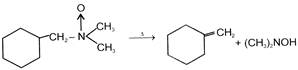
The products of the following reaction are
A)
done
clear
B)
done
clear
C)
done
clear
D)
\[C{{H}_{3}}CON{{H}_{2}}+C{{H}_{2}}=CH-CH=C{{H}_{2}}\]
done
clear
View Answer play_arrow
Which of the following acids on heating readily undergoes decarboxylation?
A)
2-Oxocyclohexane carboxylic acid
done
clear
B)
3-Oxocyclohexane carboxylic acid
done
clear
C)
4-Oxocyclohexane carboxylic acid
done
clear
D)
All undergo decarboxylation
done
clear
View Answer play_arrow
This reaction is called
A)
Cope reaction
done
clear
B)
Ritter reaction
done
clear
C)
Schmidt reaction
done
clear
D)
Gabriel reaction
done
clear
View Answer play_arrow
Which of the following acts as an inhibitor in vinyl polymerization
A)
Benzophenone
done
clear
B)
P-benzoquinone
done
clear
C)
benzoic anhydride
done
clear
D)
benzoic peroxide
done
clear
View Answer play_arrow
Arsenic drugs are mainly used in the treatment of
A)
Jaundice
done
clear
B)
Typhoid
done
clear
C)
Syphilis
done
clear
D)
Cholera
done
clear
View Answer play_arrow
A white crystalline solid A on boiling with caustic soda solution gave a gas B which when passed through an alkaline solution of potassium mercuric iodide gave a brown ppt. The substance A on heating gave a gas C which rekindled a glowing splinter but did not give brown fumes with nitric oxide. The gas B and C are respectively.
A)
\[{{H}_{2}}S,\,{{O}_{2}}\]
done
clear
B)
\[N{{H}_{3}},{{N}_{2}}O\]
done
clear
C)
\[HCl,NO\]
done
clear
D)
\[C{{O}_{2}},{{O}_{2}}\]
done
clear
View Answer play_arrow
Which will give borax bead test with blue bead?
A)
\[C{{o}^{3+}}\]
done
clear
B)
\[C{{d}^{2+}}\]
done
clear
C)
\[C{{r}^{3+}}\]
done
clear
D)
\[N{{i}^{2+}}\]
done
clear
View Answer play_arrow
The hybridization of central metal ion and shape of wilkinson's catalyst is
A)
\[ds{{p}^{2}},\]square planar
done
clear
B)
\[~s{{p}^{3}},\] tetrahedral ,
done
clear
C)
\[{{d}^{2}}~s{{p}^{3}},\] octahedral
done
clear
D)
\[~s{{p}^{3}}d,\]trigonal bipyramidal
done
clear
View Answer play_arrow
In alkaline condition \[KMn{{O}_{4}}\] reacts as \[2KMn{{O}_{4}}+2KOH\xrightarrow{{}}2{{K}_{2}}Mn{{O}_{4}}+{{H}_{2}}O+O\] The equivalent weight of \[KMn{{O}_{4}}\] would be
A)
\[158.0~\]
done
clear
B)
\[79.0\]
done
clear
C)
\[52.7\]
done
clear
D)
\[31.6\]
done
clear
View Answer play_arrow
The noble gas which forms .interstitial compounds is
A)
\[Ar\]
done
clear
B)
\[Xe\]
done
clear
C)
\[Kr\]
done
clear
D)
\[He\]
done
clear
View Answer play_arrow
Which of the following has stronger bond?
A)
\[F-C1\]
done
clear
B)
\[F-Br\]
done
clear
C)
\[F-B\]
done
clear
D)
\[Cl-Br\]
done
clear
View Answer play_arrow
Which of the following trichlorides of nitrogen family hydrolyses through nucleophilic attack by\[{{H}_{2}}O\] molecule?
A)
\[NC{{l}_{3}}\]
done
clear
B)
\[P{{F}_{3}}\]
done
clear
C)
\[N{{F}_{3}}\]
done
clear
D)
\[AsC{{l}_{3}}\]
done
clear
View Answer play_arrow
Choose incorrect statement with respect to taxa
A)
Term given by H. J. Lam for plants
done
clear
B)
It is abstract term and represents rank in hierarchy
done
clear
C)
Can indicate categories at very different levels
done
clear
D)
Mammals and dogs represent taxa at different levels
done
clear
View Answer play_arrow
Descendent of a common ancestor which have monophyletic origin is
A)
Clade
done
clear
B)
Cline
done
clear
C)
Deme
done
clear
D)
Grade
done
clear
View Answer play_arrow
Find odd one out for taxonomic keys
A)
Statement in key is called lead
done
clear
B)
Generally analytical in nature
done
clear
C)
Separate taxonomic keys are not needed for each taxonomic category
done
clear
D)
Concept of key was developed by John Ray
done
clear
View Answer play_arrow
Archae bacteria are often referred as living fossils and they can thrive well even in extremes of conditions because
A)
They have introns in DNA
done
clear
B)
They have branched chain lipids in cell membrane
done
clear
C)
Sequence of 16 s rRNA is different from bacteria
done
clear
D)
Can reproduce through elementary bodies like mycoplasma
done
clear
View Answer play_arrow
Jokers of plant Kingdom are not killed by the antibiotic penicillin because
A)
They have replicating disc in DNA
done
clear
B)
Lack mesosome and membrane bound organelles
done
clear
C)
They do not have cell wall
done
clear
D)
They are the smallest organism
done
clear
View Answer play_arrow
Fuligo and Physarum are characterized by all except
A)
Capillitia in sporangium
done
clear
B)
Absence of cell wall in vegetative phase
done
clear
C)
Non-motile swarm cells
done
clear
D)
Poly centric plasmodium
done
clear
View Answer play_arrow
Cellular slime moulds
A)
Produce naked sporangia
done
clear
B)
Have dominant diploid phase
done
clear
C)
From haploid macrocyst by mitosis
done
clear
D)
Posses saprobic nutrition only
done
clear
View Answer play_arrow
Which is known as squirting fungi?
A)
Saprolegnia
done
clear
B)
PiIobolus
done
clear
C)
Rhizopus
done
clear
D)
Arthrobotrys
done
clear
View Answer play_arrow
Find odd one out with respect to spirogyra
A)
Primordial utricle
done
clear
B)
Sinistral chloroplast
done
clear
C)
Morphological anisogamy
done
clear
D)
Haplontic life cycle
done
clear
View Answer play_arrow
Which is not a feature of spike moss?
A)
Precocious gametophyte
done
clear
B)
Cespitose habit
done
clear
C)
Meroblastic embryo
done
clear
D)
Gemetic meiosis
done
clear
View Answer play_arrow
Find out correct match
A)
Pagoda tree - non-motile sperm
done
clear
B)
Sago palm - living fossil
done
clear
C)
Gnetum - homoxylous wood
done
clear
D)
Ephedra - chilgoza
done
clear
View Answer play_arrow
Largest angiospermic family ofdicot has
A)
Axile placentation
done
clear
B)
Synandrous stamen
done
clear
C)
Syngenesious and epipetalous condition
done
clear
D)
Ligulate corolla in disc florets
done
clear
View Answer play_arrow
Which is not a feature of monocot stem?
A)
Schizoly sigenous water cavity
done
clear
B)
Endarchxylem
done
clear
C)
Undifferentied ground tissue
done
clear
D)
Bulliform cells
done
clear
View Answer play_arrow
Water potential of a cell is affected by
A)
Solute potential
done
clear
B)
Pressure potential
done
clear
C)
Osmotic potential
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which of these micro-elements is an activator for both rubisco and PEP case?
A)
\[Zn\]
done
clear
B)
\[Mg\]
done
clear
C)
\[Mn\]
done
clear
D)
\[Cu\]
done
clear
View Answer play_arrow
During symbiotic nitrogen fixation in leguminous plants, ATP needed to reduce one mole of dinitrogen into ammonia with the release of hydrogen is
A)
8
done
clear
B)
16
done
clear
C)
12
done
clear
D)
6
done
clear
View Answer play_arrow
How may additional pairs of protons are pumped towards outer mitochondrial chamber when three \[NAD{{H}_{2}}\]molecules are oxidized through ETS?
A)
3
done
clear
B)
6
done
clear
C)
9
done
clear
D)
4
done
clear
View Answer play_arrow
Which of the following is odd with respect to segmentation?
A)
Earthworm
done
clear
B)
Cockroach
done
clear
C)
Star Fish
done
clear
D)
Prawn
done
clear
View Answer play_arrow
Which of the following cnidarian belongs to class hydrozoa?
A)
Madrepora
done
clear
B)
Heliopora
done
clear
C)
Millepora
done
clear
D)
Tubipora
done
clear
View Answer play_arrow
Larva of liver fluke which infact secondary host is
A)
Cercaria
done
clear
B)
Metacercaria
done
clear
C)
Miracidium
done
clear
D)
Redia
done
clear
View Answer play_arrow
In which of the following arthropod, chelicerae are present?
A)
Crab
done
clear
B)
Prawn
done
clear
C)
Centipede
done
clear
D)
Scorpion
done
clear
View Answer play_arrow
Which of the following poikilotherm vertebrate has dicondylic skull?
A)
Proteus
done
clear
B)
Testudo
done
clear
C)
Anobas
done
clear
D)
Draco
done
clear
View Answer play_arrow
Eustachian tube, epiglottis and pinna of ear consist of
A)
Elastic cartilage
done
clear
B)
Hyaline cartilage
done
clear
C)
Calcified cartilage
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which of the following bone is/are formed by the replacement of cartilage?
A)
Humerus
done
clear
B)
Scapula
done
clear
C)
Femur
done
clear
D)
All of these
done
clear
View Answer play_arrow
The most abundant blood plasma protein is
A)
Albumin
done
clear
B)
Globulin
done
clear
C)
Fibrinogen
done
clear
D)
Both b and c
done
clear
View Answer play_arrow
Which of the following statement is incorrect with respect to earthworm?
A)
Blood gland - 4th, 5th, 6th segment
done
clear
B)
Lateral hearts - 7th, 9th segment
done
clear
C)
Pharyngeal nephridia - 15th segment onwards
done
clear
D)
Ovaries - 13th segment
done
clear
View Answer play_arrow
Which of the following monosaccharide is pentose sugar?
A)
Mannose
done
clear
B)
Ribose
done
clear
C)
Galactose
done
clear
D)
Glucose
done
clear
View Answer play_arrow
Which of the following is not an essential amino acid?
A)
Tryptophan
done
clear
B)
Valine
done
clear
C)
Serine
done
clear
D)
Leucine
done
clear
View Answer play_arrow
Puccinia graminis tritici which causes black rust of wheat produces
A)
Dikaryotic spores only on primary host
done
clear
B)
Four basidiospores per basidium endogenously
done
clear
C)
Monokaryotic pycniospores on upper side of leaf in secondary host
done
clear
D)
Dikaryotic, non-stalked, thin walled bicelled teleutospores on wheat plant
done
clear
View Answer play_arrow
Cord moss
A)
lacks multicellular rhizoids
done
clear
B)
possesses transverse bands of cellulose in exostome of capsule
done
clear
C)
is antoicous and protogynous
done
clear
D)
is always non-vascular and non-embryophyte
done
clear
View Answer play_arrow
Choose correct statement with respect to selaginella
A)
Main plant body is always isophyllous
done
clear
B)
Absence of vascular tissue in gametophyte
done
clear
C)
Leaves have furcate venation and liyule
done
clear
D)
Single binucleate neck canal cell in archegonium
done
clear
View Answer play_arrow
External water is not required for fertilization in smallest group of plant Kingdom because
A)
Pollination is indirect as they lack stigma
done
clear
B)
Fertilization is siphonogamous
done
clear
C)
All are perennial woody plants either shrubs or trees
done
clear
D)
Development of male gametophyte is precocious
done
clear
View Answer play_arrow
Which is not related to endoplasmic reticulum?
A)
Endomembrane system
done
clear
B)
Intracellular digestion
done
clear
C)
Formation of sphaerosomes
done
clear
D)
GERL
done
clear
View Answer play_arrow
Select the incorrect statement with respect to water potential
A)
Pure water will have the greatest \[{{\Psi }_{w}}\]
done
clear
B)
For a solution at atmospheric pressure its value is equal to sum of solute potential and pressure potential
done
clear
C)
Addition of solute decreases water potential
done
clear
D)
\[{{\Psi }_{w}}=-{{\Psi }_{s}}-{{\Psi }_{p}}\]for a fully plasmolysed cell
done
clear
View Answer play_arrow
Which of the following essential element is an activator of nitrogenase enzyme during nitrogen fixation?
A)
\[Zn\]
done
clear
B)
\[Mn\]
done
clear
C)
\[Mo\]
done
clear
D)
\[Mg\]
done
clear
View Answer play_arrow
Find out the number of additional protons pumped to inter membranal space from matrix of mitochondria when two moles of NADH + IP are oxidized through ETS
A)
2 pairs
done
clear
B)
4 pairs
done
clear
C)
3 pairs
done
clear
D)
1 pairs
done
clear
View Answer play_arrow
Match the following plant and their chromosome number in gametes
Column-I Column-II A. Onion 1. 24 B. Maize 2. 17 C. Apple 3, 10 D. Potato 4. 8
A)
A-1 B-2 C-3 D-4
done
clear
B)
A-4 B-3 C-2 D-1
done
clear
C)
A-4 B-1 C-2 D-3
done
clear
D)
A-3 B-1 C-2 D-4
done
clear
View Answer play_arrow
Maximum and minimum average weight of a fruit is 50 and 3g respectively. Find out the approximate weight of fruit with genotype AaBBCc
A)
44g
done
clear
B)
37g
done
clear
C)
31g
done
clear
D)
13g
done
clear
View Answer play_arrow
Number of Phosphodiester linkages in DNA of \[\phi \]X174 is
A)
5385
done
clear
B)
48501
done
clear
C)
2691
done
clear
D)
24250
done
clear
View Answer play_arrow
Find the correct match
A)
Barley : NP 836
done
clear
B)
HVW - 468 : Mass selection
done
clear
C)
Lectin : French bean
done
clear
D)
Pruteen : Energy cropping
done
clear
View Answer play_arrow
Which is not a role of predation?
A)
Energy transfer from low to higher tropic levels
done
clear
B)
Resource partitioning
done
clear
C)
Predators keep population of prey under control
done
clear
D)
Help in maintain species diversity
done
clear
View Answer play_arrow
Which of the following condition does not promote decomposition of detritus?
A)
Detritus rich in proteins and sugars
done
clear
B)
Less aerated soil
done
clear
C)
Neutral or slightly alkaline pH in soil
done
clear
D)
\[25-{{30}^{o}}C\]temperature of soil
done
clear
View Answer play_arrow
Tropics account for their greater biological diversity because
A)
Speciation is function of time
done
clear
B)
It has remained relatively undisturbed
done
clear
C)
Constant and predictable environment
done
clear
D)
More than one option is correct
done
clear
View Answer play_arrow
Presence of large amount of nutrients in water that causes excessive growth of planktonic algae is
A)
Bio-magnification
done
clear
B)
Accelerated eutrophication
done
clear
C)
Algal bloom
done
clear
D)
Ecological boomrang
done
clear
View Answer play_arrow
Which of the following is not mesodermal in origin?
A)
Lens
done
clear
B)
Adrenal cortex
done
clear
C)
Dentine of teeth
done
clear
D)
Choroid layer
done
clear
View Answer play_arrow
Which of the following cannot be taken as a character of non-striated muscle fibres?
A)
Do not get fatigue
done
clear
B)
Involuntary
done
clear
C)
Multinucleate
done
clear
D)
Spindle shaped
done
clear
View Answer play_arrow
Which of the following chemical substance of saliva act as antimicrobial agent?. A. Lysozyme B. Ptyalin C. Thiocyanate ions D. Bicarbonate ions
A)
A only
done
clear
B)
A and B
done
clear
C)
A and C
done
clear
D)
A,B and C
done
clear
View Answer play_arrow
Which of the following statement is true?
A)
The chief cells of the gastric glands secrete Intrinsic factor, essential for absorption of vitamin \[{{B}_{12}}\]
done
clear
B)
Water absorption occurs in large intestine mainly maximum amount of the total water is absorbed here
done
clear
C)
High protein diet increase the salivary secretion
done
clear
D)
Success entericus has brush bordered enzymes
done
clear
View Answer play_arrow
Pineal gland functions as a A. Biological clock B. Neurosecretory transducer C. Master gland D. Tropic gland
A)
A Only
done
clear
B)
A and B
done
clear
C)
B and C
done
clear
D)
C and D
done
clear
View Answer play_arrow
After which period of foetal growth, it becomes less susceptible to teratogens?
A)
After 6th week
done
clear
B)
After 12th week
done
clear
C)
After gastrulation
done
clear
D)
After 20th week
done
clear
View Answer play_arrow
Which of the following stage of early embryonic development passes through the phase of compaction?
A)
Morula
done
clear
B)
Blastocyst
done
clear
C)
Gastrula
done
clear
D)
Secondary yolk sec formation
done
clear
View Answer play_arrow
The levels of which of the following hormone will decrease after menopause?
A)
FSH
done
clear
B)
Estrogen
done
clear
C)
LH
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which of the following may cause toxic shock syndrome in females?
A)
Tampon
done
clear
B)
Diaphragm
done
clear
C)
Toxins released by staphylococcus aureous
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which of the following antibody is the earliest immunoglobin to be synthesized by the foetus?
A)
IgM
done
clear
B)
IgG
done
clear
C)
IgA
done
clear
D)
IgD
done
clear
View Answer play_arrow
Which of the following statement is incorrect about opium and its derivatives?
A)
Opioids bind to specific opioid receptors present in our central nervous system and gatrointestinal tract
done
clear
B)
Heroin is diacetylmorphine
done
clear
C)
Codeine is used in cough syrup
done
clear
D)
Generally taken by smoking and injection, heroin is a stimulant and increases the body functions
done
clear
View Answer play_arrow
During separation and isolation of DNA fragments, that can be seen after staning with a compound called as
A)
Safranin
done
clear
B)
Acetocarmine
done
clear
C)
Fast green
done
clear
D)
Ethidium bromide
done
clear
View Answer play_arrow
Which of the mouth parts of cockroach acts like a tongue?
A)
Labrum
done
clear
B)
Labium
done
clear
C)
Hypopharynx
done
clear
D)
Maxillae
done
clear
View Answer play_arrow
In opuntia the spines represent the modification of
A)
Stem
done
clear
B)
Bud
done
clear
C)
Leaves
done
clear
D)
Roots
done
clear
View Answer play_arrow
Rizome is found in
A)
Ginger and Turmeric
done
clear
B)
Colocasia and Ginger
done
clear
C)
Helianthus and Turmeric
done
clear
D)
Dryopteris and Gladiolus
done
clear
View Answer play_arrow
In cockroach the forewings are articulated with
A)
Prothorax
done
clear
B)
Mesothorax
done
clear
C)
Metathorax
done
clear
D)
First abdominal segent
done
clear
View Answer play_arrow
In Bougainvillea the thorns are modification of
A)
lateral bud
done
clear
B)
stipule
done
clear
C)
axillary bud
done
clear
D)
leaf
done
clear
View Answer play_arrow
In Periplaneta SP heptic caeca open at the junction of
A)
Fore gut and mid gut
done
clear
B)
Mid gut and ileum
done
clear
C)
Ileum and Colon
done
clear
D)
Colon and rectum
done
clear
View Answer play_arrow
When stem is changed into fleshy green long structure then it is termed as
A)
Phylloclade
done
clear
B)
Cladode
done
clear
C)
Phyllode
done
clear
D)
Corm
done
clear
View Answer play_arrow
Phallic gland in cockroach is related to
A)
Male excretory system
done
clear
B)
Male reproductive system
done
clear
C)
Female excretory system
done
clear
D)
Female reproductive system
done
clear
View Answer play_arrow
Tomato, Chilli, Ashwagandha, Petunia belong to which family?
A)
Solanaceae
done
clear
B)
Poaceae
done
clear
C)
Fabaceae
done
clear
D)
Liliaceae
done
clear
View Answer play_arrow
Stilt roots are
A)
adventitious root
done
clear
B)
tap roots
done
clear
C)
secondary roots
done
clear
D)
present in Bombax
done
clear
View Answer play_arrow
Male cockroach can be identified by the presence of
A)
Anal style
done
clear
B)
Anal cercus
done
clear
C)
Broad abdomen
done
clear
D)
Antenna
done
clear
View Answer play_arrow
Haemocoel is found in
A)
Cockroach and Ascaris
done
clear
B)
Periplaneta and Pila
done
clear
C)
Blatta and Asterias
done
clear
D)
Asterias and Balanoglossus
done
clear
View Answer play_arrow
How many pairs of spiracles are present in cockroach?
A)
4
done
clear
B)
6
done
clear
C)
8
done
clear
D)
10
done
clear
View Answer play_arrow
\[{{C}_{1+2+(2)}}\] condition is related to
A)
Pea
done
clear
B)
Mustard
done
clear
C)
Rose
done
clear
D)
More than one are correct
done
clear
View Answer play_arrow
Inferior ovary is found in
A)
Sunflower
done
clear
B)
Mustard
done
clear
C)
China Rose
done
clear
D)
Rose
done
clear
View Answer play_arrow
Perisperm is
A)
Remnants of nucleus
done
clear
B)
Remnants of endosperm
done
clear
C)
Remnants of secondary nucleus
done
clear
D)
Persistent embryo
done
clear
View Answer play_arrow
Coleorhiza is a cup like covering present on
A)
Radicle of dicots
done
clear
B)
Plumule of dicots
done
clear
C)
Plumule of monocots
done
clear
D)
Radicle of monocots
done
clear
View Answer play_arrow
Syconus fruit develop from
A)
Catkin
done
clear
B)
Verticillaster
done
clear
C)
Hypanthodium
done
clear
D)
Cyathium
done
clear
View Answer play_arrow
Operation flood is related to
A)
Curvier
done
clear
B)
Haeckel
done
clear
C)
Kurein
done
clear
D)
Frisch
done
clear
View Answer play_arrow
Dicetyl morphine is
A)
Smack
done
clear
B)
Charas
done
clear
C)
Ganja
done
clear
D)
Hashish
done
clear
View Answer play_arrow
Select exotic breed of cattle
A)
Gir
done
clear
B)
Deoni
done
clear
C)
Sahiwal
done
clear
D)
Jersey
done
clear
View Answer play_arrow
Lichens are sensitive to
A)
\[N{{O}_{2}}\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
\[{{N}_{2}}O\]
done
clear
View Answer play_arrow
India covers the world's land area of
A)
\[2.4%\]
done
clear
B)
\[1.8%\]
done
clear
C)
\[8.1%\]
done
clear
D)
\[4.2%\]
done
clear
View Answer play_arrow
How many biosphere reserve are in India
A)
25
done
clear
B)
90
done
clear
C)
14
done
clear
D)
448
done
clear
View Answer play_arrow
Humulin is related to
A)
E. coli
done
clear
B)
S. Typhi
done
clear
C)
Agro bacterium
done
clear
D)
Rhizobium
done
clear
View Answer play_arrow
Select the wrong match with respect to relative contribution of various green house gases to total global warming
A)
Methane - 30%
done
clear
B)
CFCS - 14%
done
clear
C)
\[C{{o}_{2}},-60%~\]
done
clear
D)
\[{{N}_{2}}o-6%\]
done
clear
View Answer play_arrow
Government of India has passed the Water Act in
A)
1974
done
clear
B)
1980
done
clear
C)
2010
done
clear
D)
2013
done
clear
View Answer play_arrow
According to one estimate _____ gm of carbon is fixed in the biosphere through photosynthesis annually.
A)
\[4\times {{10}^{6}}\]
done
clear
B)
\[~4\times {{10}^{13}}\]
done
clear
C)
\[5\times {{10}^{12}}\]
done
clear
D)
\[170\times {{10}^{6}}\]
done
clear
View Answer play_arrow
Connell's elegant field experiment showed that on the rocky sea coasts of Scotland, the larger and competitively superior barnacle _____dominates the intertidal area.
A)
Chathamalus
done
clear
B)
Balanus
done
clear
C)
Shark
done
clear
D)
Euglena
done
clear
View Answer play_arrow
It is now routinely used to detect HIV in suspected patient.
A)
PCR
done
clear
B)
Southern blotting
done
clear
C)
Gel electrophoresis
done
clear
D)
Hybridization
done
clear
View Answer play_arrow
A procedure through which a piece of DNA is introduced in a host bacterium is
A)
Transfection
done
clear
B)
Transformation
done
clear
C)
Transduction
done
clear
D)
Transgenesis
done
clear
View Answer play_arrow
Homo erectus had a large brain around
A)
900 CC
done
clear
B)
1400CC
done
clear
C)
1300 CC
done
clear
D)
1450CC
done
clear
View Answer play_arrow
UTRs are related to
A)
t-RNA
done
clear
B)
m-RNA
done
clear
C)
r-RNA
done
clear
D)
All above
done
clear
View Answer play_arrow
Apomixis is found in
A)
Asteraceal
done
clear
B)
Grasses
done
clear
C)
Cotton
done
clear
D)
More than one are correct
done
clear
View Answer play_arrow
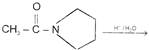
 This reaction is called
This reaction is called 