question_answer 1) The electromotive force of primary cell is \[2\,\,V\]. When it is short circuited it gives a current of \[4\,\,A\]. Its internal resistance in ohm is
A)
0.5
done
clear
B)
5.0
done
clear
C)
2.0
done
clear
D)
8.0
done
clear
View Answer play_arrow
question_answer 2) The value of electric permittivity of free space is
A)
\[9\times {{10}^{9}}N{{c}^{2}}/{{m}^{2}}\]
done
clear
B)
\[8.9\times {{10}^{-12}}N{{m}^{2}}/{{C}^{2}}s\]
done
clear
C)
\[8.9\times {{10}^{-12}}{{C}^{2}}/N{{m}^{2}}\]
done
clear
D)
\[9\times {{10}^{9}}{{C}^{2}}/N{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 3) Which of the following is not the unit of energy?
A)
Calorie
done
clear
B)
Joule
done
clear
C)
Electron-volt
done
clear
D)
Watt
done
clear
View Answer play_arrow
question_answer 4) A body moves with a uniform acceleration\[a\]and zero initial velocity. Another body\[B\]starts from the same point moves in the same direction with a constant velocity\[v\]. The two bodies meet after a time\[t\]. The value of\[t\]is
A)
\[\frac{2v}{a}\]
done
clear
B)
\[\frac{v}{a}\]
done
clear
C)
\[\frac{v}{2a}\]
done
clear
D)
\[\sqrt{\frac{v}{2a}}\]
done
clear
View Answer play_arrow
question_answer 5) A body starts to fall freely under gravity. The distances covered by it in first, second and third seconds are in ratio
A)
\[1:3:5\]
done
clear
B)
\[1:2:3\]
done
clear
C)
\[1:4:9\]
done
clear
D)
\[1:5:6\]
done
clear
View Answer play_arrow
question_answer 6) Galileo writes that for angles of projection of a projectile at angles\[({{45}^{o}}+\theta )\]and\[({{45}^{o}}-\theta )\], the horizontal ranges described by the projectile are in the ratio
A)
\[2:1\]
done
clear
B)
\[1:2\]
done
clear
C)
\[1:1\]
done
clear
D)
\[2:3\]
done
clear
View Answer play_arrow
question_answer 7) Two masses\[M\]and\[m\]are attached to a vertical axis by weightless thread of combined length\[l.\] They are set in rotational motion in a horizontal plane about this axis with constant angular velocity\[\omega \]. If the tensions in the thread are same during motion, the distance of M from the axis is,
A)
\[\frac{Ml}{M+m}\]
done
clear
B)
\[\frac{ml}{M+m}\]
done
clear
C)
\[\frac{M+m}{M}l\]
done
clear
D)
\[\frac{M+m}{m}l\]
done
clear
View Answer play_arrow
question_answer 8) A uniform rope of length L, resting on a frictionless horizontal surface, is pulled at one end by a force F. The tension in the rope at a distance I from this end is
A)
\[F\]
done
clear
B)
\[\frac{l}{L}F\]
done
clear
C)
\[\frac{L}{l}F\]
done
clear
D)
\[\left( 1-\frac{l}{L} \right)F\]
done
clear
View Answer play_arrow
question_answer 9) A body of mass \[5kg\] is moving with a momentum of\[10\,kg\,m/s\]. A force of\[0.2\,\,N\]acts on it in the direction of motion of the body for 10 s. The increase in its kinetic energy is
A)
\[2.8\,\,J\]
done
clear
B)
\[3.2\,\,J\]
done
clear
C)
\[3.8\,\,J\]
done
clear
D)
\[4.4\,\,J\]
done
clear
View Answer play_arrow
question_answer 10) The potential energy of a weightless spring compressed by a distance a is proportional to
A)
\[a\]
done
clear
B)
\[{{a}^{2}}\]
done
clear
C)
\[{{a}^{-2}}\]
done
clear
D)
\[{{a}^{0}}\]
done
clear
View Answer play_arrow
question_answer 11) A thin circular ring of mass\[M\]and radius\[r\]is rotating about its axis with constant angular velocity\[\omega \]. Two objects each of mass\[m\], are placed gently at the opposite ends of diameter of the ring. The ring now rotates with an angular velocity
A)
\[\omega M/(M+m)\]
done
clear
B)
\[\omega M/(M+2m)\]
done
clear
C)
\[\omega (M-2m)/(m+2M)\]
done
clear
D)
\[\omega (M+2m)/M\]
done
clear
View Answer play_arrow
question_answer 12) A wheel is rotating at\[900\,\,rpm\]about its axis. When the power is cut off it comes to rest in\[1\min \]. The angular retardation in\[rad/{{s}^{2}}\]is
A)
done
clear
B)
\[\pi /2\]
done
clear
C)
\[\pi /6\]
done
clear
D)
\[\pi /8\]
done
clear
View Answer play_arrow
question_answer 13) In a photoelectric experiment, the. Stopping potential for incident light of wavelength \[4000\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\] is \[2\,V\] . If the wavelength is changed to\[3000\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\], the stopping potential is
A)
\[2\,\,V\]
done
clear
B)
less than\[2\,\,V\]
done
clear
C)
more than\[2\,\,V\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 14) The wavelength of the most energetic\[X-\]ray emitted when a metal target is bombarded by \[100\text{ }keV\] electrons, is approximately
A)
\[12\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[4\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[0.31\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[0.124\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 15) The wavelength of the first line of Balmer series is\[6563\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]. The Rydberg constant for hydrogen is about
A)
\[1.09\times {{10}^{7}}\text{per}\,\,m\]
done
clear
B)
\[1.09\times {{10}^{8}}\text{per}\,\,m\]
done
clear
C)
\[1.09\times {{10}^{9}}\text{per}\,\,m\]
done
clear
D)
\[1.09\times {{10}^{5}}\text{per}\,\,m\]
done
clear
View Answer play_arrow
question_answer 16) \[200\,\,MeV\]of energy may be obtained per fission of\[{{U}^{235}}\]. A reactor is generating\[1000kW\]of power. The rate of nuclear fission in the reaction (in per second) is
A)
\[1000\]
done
clear
B)
\[2\times {{10}^{8}}\]
done
clear
C)
\[3.125\times {{10}^{16}}\]
done
clear
D)
\[931\]
done
clear
View Answer play_arrow
question_answer 17) \[N\]atoms of a radioactive element emit\[n-\]alpha particles per second. The half-life of the element is
A)
\[\frac{n}{N}\text{second}\]
done
clear
B)
\[\frac{N}{n}\text{second}\]
done
clear
C)
\[\frac{0.693n}{n}\text{second}\]
done
clear
D)
\[\frac{0.693n}{N}\text{second}\]
done
clear
View Answer play_arrow
question_answer 18) The depletion layer of a\[p-n\]junction
A)
is of constant width irrespective of the bias
done
clear
B)
acts like an insulating zone under reverse bias
done
clear
C)
has a width that increases with an increase in forward bias
done
clear
D)
is depleted of ions
done
clear
View Answer play_arrow
question_answer 19) An oscillator is basically an amplifier with gain
A)
less than unity
done
clear
B)
more than unity
done
clear
C)
zero
done
clear
D)
0.5
done
clear
View Answer play_arrow
question_answer 20) A charge of\[5\,\,C\] experiences a force of\[5000\,\,N\]when it is kept in a uniform electric field. What is the potential difference between two points separated by a distance of\[1\,\,cm\]?
A)
\[10\,\,V\]
done
clear
B)
\[250\,\,V\]
done
clear
C)
\[1000\,\,V\]
done
clear
D)
\[2500\,\,V\]
done
clear
View Answer play_arrow
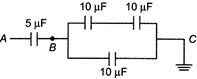
question_answer 21)
In the given circuit if point\[C\]is connected to the earth and a potential of\[+2000\,\,V\]is given to the point\[A\], the potential at\[B\]is
A)
\[1500\,\,V\]
done
clear
B)
\[1000\,\,V\]
done
clear
C)
\[500\,\,V\]
done
clear
D)
\[400\,\,V\]
done
clear
View Answer play_arrow
question_answer 22) A string vibrates according to the equation \[y=5\sin \left( \frac{2\pi x}{3} \right)\cos 20\pi t\]where\[x\]and\[y\]are in cm and t in second. The distance between two adjacent nodes is
A)
\[3cm\]
done
clear
B)
\[4.5cm\]
done
clear
C)
\[6cm\]
done
clear
D)
\[1.5cm\]
done
clear
View Answer play_arrow
question_answer 23) A single slit is located effectively at infinity in front of a lens of focal length\[1\,\,m\]and it is illuminated normally with light of wavelength\[600\,\,nm\]. The first minima on either side of central maximum are separated by\[4\,\,mm\]. Width of the slit is
A)
\[0.1mm\]
done
clear
B)
\[0.2mm\]
done
clear
C)
\[0.3mm\]
done
clear
D)
\[0.4mm\]
done
clear
View Answer play_arrow
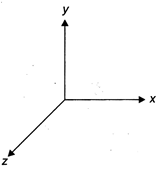
question_answer 24)
Light wave is travelling along \[y-\]direction. If the corresponding\[\mathbf{E}\]vector at any time is along the \[x-\]axis, the direction of \[\mathbf{B}\] vector at that time is along
A)
\[y-\]axis
done
clear
B)
\[x-\]axis
done
clear
C)
\[z-\]axis
done
clear
D)
\[-z-\]axis
done
clear
View Answer play_arrow
question_answer 25) An equiconvex lens of glass of focal length\[0.1m\]is cut along a plane perpendicular to principal axis into two equal parts. The ratio of focal lengths of new lenses formed is
A)
\[1:1\]
done
clear
B)
\[1:2\]
done
clear
C)
\[2:1\]
done
clear
D)
\[2:\frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 26) Time taken by light to cross a slab of thickness of\[4mm\]and refractive index 3 will be
A)
\[4\times {{10}^{-11}}s\]
done
clear
B)
\[2\times {{10}^{-11}}s\]
done
clear
C)
\[16\times {{10}^{-11}}s\]
done
clear
D)
\[8\times {{10}^{-30}}s\]
done
clear
View Answer play_arrow
question_answer 27) The value of the horizontal component of the earths magnetic field and angle of dip are\[1.8\times {{10}^{-5}}Wb/{{m}^{2}}\]and\[{{30}^{o}}\], respectively at some place. The total intensity of earths magnetic field at that place will be.
A)
\[2.08\times {{10}^{-5}}Wb/{{m}^{2}}\]
done
clear
B)
\[3.67\times {{10}^{-5}}Wb/{{m}^{2}}\]
done
clear
C)
\[3.18\times {{10}^{-5}}Wb/{{m}^{2}}\]
done
clear
D)
\[5.0\times {{10}^{-5}}Wb/{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 28) A generator produces a voltage that is given by\[V=240\sin 120t\]volt, where t is in seconds. The frequency and\[\text{rms}\]voltage are
A)
\[60Hz\]and\[240V\]
done
clear
B)
\[19Hz\]and\[120V\]
done
clear
C)
\[19Hz\]and\[170V\]
done
clear
D)
\[754Hz\]and\[170V\]
done
clear
View Answer play_arrow
question_answer 29) The pole pieces of the magnet used in a pivoted coil galvanometer are
A)
plane surfaces of a bar magnet
done
clear
B)
plane surface of a horse-shoe magnet
done
clear
C)
cylindrical surfaces of a bar magnet
done
clear
D)
cylindrical surfaces of a horse-shoe magnet
done
clear
View Answer play_arrow
question_answer 30) A\[50\,\,V\]potential difference is suddenly applied to a coil with\[L=5\times {{10}^{-2}}\]and\[R=180\Omega \]. The rate of increase of current after\[0.001s\]is
A)
\[27.3\,\,A/s\]
done
clear
B)
\[27.8\,\,A/s\]
done
clear
C)
\[2.73\,\,A/s\]
done
clear
D)
\[2.78\,\,A/s\]
done
clear
View Answer play_arrow
question_answer 31) A metal wire of specific resistance\[64\times {{10}^{-6}}\Omega cm\]and length \[198cm\] has a resistance of\[7\Omega \]. The radius of the wire will be
A)
\[2.4cm\]
done
clear
B)
\[0.24cm\]
done
clear
C)
\[0.024\text{ }cm\]
done
clear
D)
\[24cm\]
done
clear
View Answer play_arrow
question_answer 32) An electric heater, kept in vacuum, is heated continuously by passing electric current. Its temperature
A)
will go on rising with time
done
clear
B)
will stop rising after sometime as it will loose heat to the surroundings by conduction
done
clear
C)
will rise for sometime and thereafter will start falling
done
clear
D)
will become constant after sometime because of loss of heat due to radiation
done
clear
View Answer play_arrow
question_answer 33) Two straight parallel wires both carrying\[10\,\,A\]current in the same direction attracts each other with a force of\[1\times {{10}^{-3}}\,\,N\]. If both currents are doubled, the force of attraction will be
A)
\[1\times {{10}^{-3}}N\]
done
clear
B)
\[2\times {{10}^{-3}}N\]
done
clear
C)
\[4\times {{10}^{-3}}N\]
done
clear
D)
\[0.25\times {{10}^{-3}}N\]
done
clear
View Answer play_arrow
question_answer 34) A torch battery consisting of two cells of\[1.45\,\,V\]and an internal resistance\[0.15\Omega \], each cell sending current through the filament of the lamps having resistance\[1.5\Omega \]. The value of current will be
A)
\[16.11\,\,A\]
done
clear
B)
\[1.611\,\,A\]
done
clear
C)
\[0.1611\,\,A\]
done
clear
D)
\[2.6\,\,A\]
done
clear
View Answer play_arrow
question_answer 35) An external resistance\[R\]is connected with a battery of \[emf\,\,V\] and internal resistance\[r\]. Then heat produced in resistance \[R\] will be maximum, if value of \[R\] is
A)
\[r\]
done
clear
B)
\[r/2\]
done
clear
C)
\[2r\]
done
clear
D)
infinite
done
clear
View Answer play_arrow
question_answer 36) Two thin long parallel wires separated by a distance \[r\] carry equal currents\[i\]. The magnitude of the force per unit length exerted by one wire .on the other wire will be
A)
\[{{\mu }_{0}}i/2\pi r\]
done
clear
B)
\[{{\mu }_{0}}{{i}^{2}}/{{r}^{2}}\]
done
clear
C)
\[{{\mu }_{0}}{{i}^{2}}/2\pi r\]
done
clear
D)
\[{{\mu }_{0}}i/{{r}^{2}}\]
done
clear
View Answer play_arrow
question_answer 37) The expression for thermo \[emf\] in a thermocouple is given by the relation\[E=40\,\,\theta -\frac{{{\theta }^{2}}}{20}\] When \[\theta \] is the temperature difference of two junction. For this the neutral temperature will be
A)
\[{{400}^{o}}C\]
done
clear
B)
\[{{300}^{o}}C\]
done
clear
C)
\[{{200}^{o}}C\]
done
clear
D)
\[{{100}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 38) The \[X-\]ray tube is operated at\[50\,\,kV\]. The minimum wavelength produce is
A)
\[0.5\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[0.75\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[0.25\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[1.0\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 39) Bulk modulus of water is\[2\times {{10}^{9}}N/{{m}^{2}}\]. The change in pressure required to increase the density of water by 0.1 % is
A)
\[2\times {{10}^{9}}N/{{m}^{2}}\]
done
clear
B)
\[2\times {{10}^{8}}N/{{m}^{2}}\]
done
clear
C)
\[2\times {{10}^{6}}N/{{m}^{2}}\]
done
clear
D)
\[2\times {{10}^{4}}N/{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 40) A cyclotron can accelerate
A)
\[\beta -\]particle
done
clear
B)
\[\alpha -\]particle
done
clear
C)
high-velocity gamma rays
done
clear
D)
high velocity X-rays
done
clear
View Answer play_arrow
question_answer 41) The diameter of a flywheel is increased by 1%. Increase in its moment of inertia about the central axis is
A)
\[1%\]
done
clear
B)
\[0.5%\]
done
clear
C)
\[2%\]
done
clear
D)
\[4%\]
done
clear
View Answer play_arrow
question_answer 42) The ratio of the specific charge of an electron to that of an \[\text{ }\!\!\alpha\!\!\text{ -}\]particle is
A)
\[1:2\]
done
clear
B)
\[1:\frac{1}{4}\]
done
clear
C)
\[1:4\]
done
clear
D)
\[1:\frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 43) A particular star assuming it as a black body has a surface temperature of about\[5\times {{10}^{4}}K\]. The wavelength in nanometre at which its radiation becomes maximum is\[(b=0.0029\,\,m/c)\]
A)
48
done
clear
B)
58
done
clear
C)
60
done
clear
D)
70
done
clear
View Answer play_arrow
question_answer 44) Which of the following can cause the null point of a potentiometre to shift beyond the wire?
A)
Shorter length of wire
done
clear
B)
Longer length of wire
done
clear
C)
Low\[emf\]of auxiliary battery
done
clear
D)
High\[emf\]of auxiliary battery
done
clear
View Answer play_arrow
question_answer 45) The kinetic energy of an electron is\[5eV\]. Calculate the de-Broglie wavelength associated with it\[(h=6.6\times {{10}^{-34}}J\,\,s,\]\[{{m}_{e}}=9.1\times {{10}^{-31}}kg)\]
A)
\[5.47\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[10.9\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[2.7\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 46) A ray of light is incident at polarising angle such that its deviation is \[{{24}^{o}}\], then angle of incidence is
A)
\[{{24}^{o}}\]
done
clear
B)
\[{{57}^{o}}\]
done
clear
C)
\[{{66}^{o}}\]
done
clear
D)
\[{{90}^{o}}\]
done
clear
View Answer play_arrow
question_answer 47) A particle of mass m is projected with velocity\[v\]making an angle of \[{{45}^{o}}\] with the horizontal. When the particle lands on the level ground the magnitude of the change in its momentum will be
A)
\[2\,\,mv\]
done
clear
B)
\[\frac{mv}{\sqrt{2}}\]
done
clear
C)
\[mv\sqrt{2}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 48) A Carnot engine takes heat from a reservoir at\[{{627}^{o}}C\]and rejects heat to a sink at\[{{27}^{o}}C\]. Its efficiency will be
A)
\[\frac{3}{5}\]
done
clear
B)
\[\frac{1}{3}\]
done
clear
C)
\[\frac{2}{3}\]
done
clear
D)
\[\frac{200}{209}\]
done
clear
View Answer play_arrow
question_answer 49) If\[{{r}_{1}}\]and\[{{r}_{2}}\]are the radii of the atomic nuclei of mass number 64 and 125 respectively, then the ratio\[\left( \frac{{{r}_{1}}}{{{r}_{2}}} \right)\]is
A)
\[\frac{64}{125}\]
done
clear
B)
\[\sqrt{\frac{64}{125}}\]
done
clear
C)
\[\frac{5}{4}\]
done
clear
D)
\[\frac{4}{5}\]
done
clear
View Answer play_arrow
question_answer 50) The working of which of the following is similar to that of a slide projector?
A)
Electron microscope
done
clear
B)
Scanning electron microscope
done
clear
C)
Transmission electron microscope
done
clear
D)
Atomic force microscope
done
clear
View Answer play_arrow
question_answer 51) Which of the following compounds are tranquilizer on the basis of their medicinal properties?
A)
Chloramphenicol
done
clear
B)
Seconal
done
clear
C)
Luminal
done
clear
D)
Both (b) and (c)
done
clear
View Answer play_arrow
question_answer 52) In alkaline earth metal sulphates, the value of hydration energy decreases down the group because of the
A)
decrease in size
done
clear
B)
increase in size
done
clear
C)
greater lattice energy
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 53) Which of the following is used in the preparation of chlorine?
A)
Only\[Mn{{O}_{2}}\]
done
clear
B)
Only\[KMn{{O}_{4}}\]
done
clear
C)
Both\[Mn{{O}_{2}}\]and\[KMn{{O}_{4}}\]
done
clear
D)
Either\[Mn{{O}_{2}}\]or\[KMn{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 54) Which of the following configuration is associated with biggest jump between 2nd and 3rd\[IE\]?
A)
\[1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{2}}\]
done
clear
B)
\[1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}},\,\,3{{s}^{1}}\]
done
clear
C)
\[1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}},\,\,3{{s}^{2}}\]
done
clear
D)
\[1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{1}}\]
done
clear
View Answer play_arrow
question_answer 55) Calculate\[\Delta {{G}^{o}}\]for conversion of oxygen to ozone,\[\frac{3}{2}{{O}_{2}}(g)\xrightarrow{{}}{{O}_{3}}(g)\]at\[298\,\,K\], if\[{{K}_{p}}\]for this conversion is\[2.47\times {{10}^{-29}}\].
A)
\[163\,\,kJ\,\,mo{{l}^{-1}}\]
done
clear
B)
\[2.4\times {{10}^{2}}\,\,kJ\,\,mo{{l}^{-1}}\]
done
clear
C)
\[1.63\,\,kJ\,\,mo{{l}^{-1}}\]
done
clear
D)
\[2.38\times {{10}^{6}}\,\,kJ\,\,mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 56) Which gives only mono substituted product?
A)
\[o-\]dinitrobenzene
done
clear
B)
\[m-\]dinitrobenzene
done
clear
C)
\[p-\]dinitrobenzene
done
clear
D)
Nitrobenzene
done
clear
View Answer play_arrow
question_answer 57) The\[{{K}_{sp}}\]of\[Mg{{(OH)}_{2}}\]is\[1\times {{10}^{-12}}\]. \[0.01M\]\[Mg{{(OH)}_{2}}\]will precipitate at the limited\[pH\]of
A)
3
done
clear
B)
9
done
clear
C)
5
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 58) Which of the following transition have minimum wavelength?
A)
\[{{n}_{4}}\to {{n}_{1}}\]
done
clear
B)
\[{{n}_{2}}\to {{n}_{1}}\]
done
clear
C)
\[{{n}_{4}}\to {{n}_{2}}\]
done
clear
D)
\[{{n}_{3}}\to {{n}_{1}}\]
done
clear
View Answer play_arrow
question_answer 59) \[C{{H}_{3}}COOH\]is reacted with\[CH\equiv CH\]in presence of\[H{{g}^{2+}}\]the product is
A)
\[\underset{\begin{smallmatrix} | \\ C{{H}_{2}} \end{smallmatrix}}{\mathop{C{{H}_{2}}}}\,(OOC{{H}_{3}})\]
done
clear
B)
\[\underset{\begin{smallmatrix} | \\ C{{H}_{2}}-(OOC-C{{H}_{3}}) \end{smallmatrix}}{\mathop{C{{H}_{3}}}}\,\]
done
clear
C)
\[{{\underset{\begin{smallmatrix} | \\ CH-(OOC-C{{H}_{3}}) \end{smallmatrix}}{\mathop{C{{H}_{3}}}}\,}_{2}}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 60) Which of the following metal does not react with the solution of copper sulphate?
A)
\[Fe\]
done
clear
B)
\[Zn\]
done
clear
C)
\[Mg\]
done
clear
D)
\[Ag\]
done
clear
View Answer play_arrow
question_answer 61)
The reaction,\[A+B+C\xrightarrow{{}}Z\]occurs by the following mechanism (i)\[A+B\to Y\](fast) (ii)\[X+C\to Y\](slow) (iii) \[Y+B\to Z\](very fast)
Rate law for this reaction is
A)
\[k[C]\]
done
clear
B)
\[k[A]{{[B]}^{2}}[C]\]
done
clear
C)
\[k[Z]\]
done
clear
D)
\[k[X][Y][Z]\]
done
clear
View Answer play_arrow
question_answer 62) \[\underset{\begin{smallmatrix} -10.7 \\ {{S}^{o}}(298\,\,K) \\ JK\,\,mo{{l}^{-1}} \end{smallmatrix}}{\mathop{{{H}^{+}}(aq)}}\,+O{{H}^{-}}(aq)\xrightarrow{{}}\underset{+70}{\mathop{{{H}_{2}}O}}\,(l)\]Standard entropy change for the above reaction is
A)
\[60.3\,\,J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
B)
\[80.7\,\,J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
C)
\[-70\,\,J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
D)
\[+10.7\,\,J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 63) Which of the following is an example of coordination isomerism?
A)
\[[Co{{(N{{H}_{3}})}_{3}}{{({{H}_{2}}O)}_{2}}Cl]B{{r}_{2}}\]
done
clear
B)
\[[Co{{(N{{H}_{3}})}_{6}}][Cr{{({{C}_{2}}{{O}_{4}})}_{3}}]\]
done
clear
C)
\[[Co{{(N{{H}_{3}})}_{5}}N{{O}_{3}}]S{{O}_{4}}\]
done
clear
D)
\[[Pt{{(N{{H}_{3}})}_{4}}C{{l}_{2}}]B{{r}_{2}}\]
done
clear
View Answer play_arrow
question_answer 64) Which of the following is threo isomer? \[\underset{(I)}{\mathop{\underset{A}{\mathop{\underset{X-\underset{|}{\overset{|}{\mathop{C-}}}\,Z}{\overset{\begin{smallmatrix} Y \\ | \end{smallmatrix}}{\mathop{X-C-Z}}}\,}}\,}}\,\,\,\,\,\,\,\,\underset{(II)}{\mathop{\underset{A}{\mathop{\underset{Z-\underset{|}{\overset{|}{\mathop{C-}}}\,X}{\overset{\begin{smallmatrix} Y \\ | \end{smallmatrix}}{\mathop{X-C-Z}}}\,}}\,}}\,\,\,\,\,\,\,\,\underset{(III)}{\mathop{\underset{A}{\mathop{\underset{Z-\underset{|}{\overset{|}{\mathop{C-}}}\,X}{\overset{\begin{smallmatrix} Y \\ | \end{smallmatrix}}{\mathop{X-C-Y}}}\,}}\,}}\,\]
A)
Only I
done
clear
B)
Only II
done
clear
C)
Only III
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 65) The\[IUPAC\]name of\[C{{H}_{3}}CH=CHCOO{{C}_{2}}{{H}_{5}}\]is
A)
ethyl but -1-enoate
done
clear
B)
ethyl but-2-enoate
done
clear
C)
ethyl prop-2-enoate
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 66) 1-3-butadiene when treated with\[B{{r}_{2}}\]gives
A)
1, 4-dibromo-2-butene
done
clear
B)
1, 3-dibromo-1-butene
done
clear
C)
3, 4-dibropio-2-butene
done
clear
D)
2, 3-dibromo-2-butene
done
clear
View Answer play_arrow
question_answer 67) In\[NO_{3}^{-}\]ion, the number of bond pair and lone pair of electrons on nitrogen atom are
A)
2, 2
done
clear
B)
3, 1
done
clear
C)
1, 3
done
clear
D)
3, 0
done
clear
View Answer play_arrow
question_answer 68) Neutral refractories are
A)
\[Si{{O}_{2}}\]
done
clear
B)
\[CaO\]
done
clear
C)
graphite
done
clear
D)
\[MgO\]
done
clear
View Answer play_arrow
question_answer 69) The method usually employed for the destruction of a colloidal solution is
A)
dialysis
done
clear
B)
addition of electrolytes
done
clear
C)
diffusion through animal membrane
done
clear
D)
condensation
done
clear
View Answer play_arrow
question_answer 70) Which among the following conditions, increase the yield of the product in the equilibrium? \[3A+B\underset{Endothermic}{\mathop{}}\,4C\]
A)
Increase in pressure
done
clear
B)
Increase in volume
done
clear
C)
Increase in temperature
done
clear
D)
Decrease in temperature
done
clear
View Answer play_arrow
question_answer 71) A sugar syrup of weight \[214.2g\] contains \[34.2g\] of sugar. The molar concentration is
A)
\[0.55\]
done
clear
B)
\[5.5\]
done
clear
C)
\[55\]
done
clear
D)
\[0.1\]
done
clear
View Answer play_arrow
question_answer 72) The relation which describes the variation of vapour pressure with temperature is called
A)
Hess law
done
clear
B)
Arrhenius equation
done
clear
C)
Kirchoffs law
done
clear
D)
Clausius-Clapeyron equation
done
clear
View Answer play_arrow
question_answer 73) \[59g\]of an amide obtained from a carboxylic acid,\[RCOOH\], upon heating with alkali liberated \[17g\] of ammonia. Atomic masses of \[C,\,\,H,\,\,N\]and\[O\]are 12, 1, 14 and 16 respectively. The acid is
A)
formic acid
done
clear
B)
acetic acid
done
clear
C)
propionic acid
done
clear
D)
benzoic acid
done
clear
View Answer play_arrow
question_answer 74) The gas evolved in tailing of mercury is
A)
\[C{{O}_{2}}\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
\[{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 75) In which of the following ores calcination process is not applicable?
A)
\[CaC{{O}_{3}}\]
done
clear
B)
\[A{{l}_{2}}{{O}_{3}}\cdot 2{{H}_{2}}O\]
done
clear
C)
\[CuC{{O}_{3}}\cdot Cu{{(OH)}_{2}}\]
done
clear
D)
\[ZnS\]
done
clear
View Answer play_arrow
question_answer 76) The reaction,\[{{({{C}_{6}}{{H}_{5}})}_{2}}C=O+C{{H}_{2}}=P{{({{C}_{6}}{{H}_{5}})}_{3}}\xrightarrow[ether]{Dry}\]\[{{({{C}_{6}}{{H}_{5}})}_{2}}C=C{{H}_{2}}+{{({{C}_{6}}{{H}_{5}})}_{3}}PO\], is called
A)
Wittig reaction
done
clear
B)
Claisen reaction
done
clear
C)
Benzoin condensation
done
clear
D)
Perkins reaction
done
clear
View Answer play_arrow
question_answer 77) Which of the following is a plasticizer?
A)
Cresyl phosphate
done
clear
B)
Ethyl acetate
done
clear
C)
Phthalic anhydride
done
clear
D)
Phthalic acid
done
clear
View Answer play_arrow
question_answer 78) The order of decreasing polarity in the compounds\[CaO,\,\,CsF,\,\,KCl,\,\,MgO\]is
A)
\[CaO,\,\,CsF,\,\,KCl,\,\,MgO\]
done
clear
B)
\[MgO,\,\,KCl,\,\,CaO,\,\,CsF\]
done
clear
C)
\[KCl,\,\,CaO,\,\,CsF,\,\,MgO\]
done
clear
D)
\[CsF,\,\,KCl,\,\,CaO,\,\,MgO\]
done
clear
View Answer play_arrow
question_answer 79) Which of the following is a solid compound?
A)
\[{{N}_{2}}O\]
done
clear
B)
\[{{N}_{2}}{{O}_{4}}\]
done
clear
C)
\[NO\]
done
clear
D)
\[{{N}_{2}}{{O}_{5}}\]
done
clear
View Answer play_arrow
question_answer 80) Which of the following is incorrect about vulcanisation of rubber?
A)
Vulcanised rubber has excellent elasticity
done
clear
B)
Vulcanised rubber increases resistance to oxidation and organic solvent
done
clear
C)
Vulcanisation usually establishes cross links at allylic position type reactive sites
done
clear
D)
Vulcanised rubber has high-water absorption tendency
done
clear
View Answer play_arrow
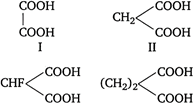
question_answer 81)
Mark out the correct set of decreasing\[p{{K}_{a}}\]for the following molecules
A)
\[IV>II>III>I\]
done
clear
B)
\[I>III>II>IV\]
done
clear
C)
\[IV>III>II>I\]
done
clear
D)
\[IV>II>I>III\]
done
clear
View Answer play_arrow
question_answer 82) Which one is the most stable carbocation?
A)
\[C{{H}_{2}}FCH_{2}^{+}\]
done
clear
B)
\[C{{F}_{3}}CH_{2}^{+}\]
done
clear
C)
\[CC{{l}_{3}}-CH_{2}^{+}\]
done
clear
D)
\[^{+}C{{F}_{3}}\]
done
clear
View Answer play_arrow
question_answer 83) The correct order of dipole mpmwt is
A)
\[C{{H}_{4}}<N{{H}_{3}}<N{{H}_{3}}<{{H}_{2}}O\]
done
clear
B)
\[N{{F}_{3}}<C{{H}_{4}}<N{{H}_{3}}<{{H}_{2}}O\]
done
clear
C)
\[N{{H}_{3}}<N{{F}_{3}}<C{{H}_{4}}<{{H}_{2}}O\]
done
clear
D)
\[{{H}_{2}}O<N{{H}_{3}}<N{{F}_{3}}<C{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 84) The vapour density of a chloride of a metal is \[81.5\,\,(H=1)\] and the chloride contains \[34.46%\] metal. The specific heat of the metal is\[0.115\]. The molecular formula of chloride is
A)
\[MC{{l}_{2}}\]
done
clear
B)
\[{{M}_{2}}C{{l}_{3}}\]
done
clear
C)
\[MC{{l}_{3}}\]
done
clear
D)
\[MC{{l}_{4}}\]
done
clear
View Answer play_arrow
question_answer 85) What is the freezing point of a\[10%\](by weight) solution of\[C{{H}_{3}}OH\]in water?
A)
\[-{{6.45}^{o}}C\]
done
clear
B)
\[-{{5.1}^{o}}C\]
done
clear
C)
\[{{1.02}^{o}}C\]
done
clear
D)
\[-{{3.5}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 86) Bohrs radius of 2nd orbit of Be34 is equal to that of
A)
4th orbit of hydrogen
done
clear
B)
2nd orbit of\[H{{e}^{+}}\]
done
clear
C)
3rd orbit of\[L{{i}^{2+}}\]
done
clear
D)
first orbit of hydrogen
done
clear
View Answer play_arrow
question_answer 87) Half-life period of a radioactive element is\[100yr\]. How long will it take for its \[93.75%\] decay?
A)
\[400\,\,yr\]
done
clear
B)
\[300\,\,yr\]
done
clear
C)
\[200\,\,yr\]
done
clear
D)
\[193\,\,yr\]
done
clear
View Answer play_arrow
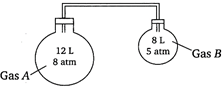
question_answer 88)
Two flasks containing gases\[A\]and\[B\]are interconnected as shown in the figure. The stopper is opened, the gases are allowed to mix homogeneously. The partial pressures of\[A\]and\[B\]in the mixture will be, respectively
A)
8 and 5 atm
done
clear
B)
9.6 and 4 atm
done
clear
C)
4.8 and 2 atm
done
clear
D)
6.4 and 4 atm
done
clear
View Answer play_arrow
question_answer 89) Aniline is not the major product in one of the following reactions. Identify that reaction
A)
\[{{C}_{6}}{{H}_{5}}OH+N{{H}_{3}}\xrightarrow[{{300}^{o}}C]{ZnC{{l}_{2}}}\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}N{{O}_{2}}+Z{{n}_{powder}}\xrightarrow{Alcoholic\,\,KOH}\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}Cl+N{{H}_{3}}\xrightarrow[C{{u}_{2}}O,\,high\,\,pressure]{{{200}^{o}}C}\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}N{{O}_{2}}+6(H)\xrightarrow[HCl]{Fe+{{H}_{2}}O}\]
done
clear
View Answer play_arrow
question_answer 90) Acetaldehyde forms a white crystalline precipitate on mixing with a ...... solution of
A)
acidic,\[Zn-Hg\]
done
clear
B)
alcoholic,\[N{{a}_{2}}S{{O}_{3}}\]
done
clear
C)
saturated aqueous,\[NaHS{{O}_{3}}\]
done
clear
D)
aqueous,\[NaCl\]
done
clear
View Answer play_arrow
question_answer 91) Which one of the following equations represents Freundlich adsorption isotherm?
A)
\[\frac{x}{m}=Kp\]
done
clear
B)
\[\frac{x}{m}=K{{p}^{n}}\]
done
clear
C)
\[\log \frac{x}{m}=K{{p}^{n}}\]
done
clear
D)
\[\log \frac{x}{m}=Kn\log p\]
done
clear
View Answer play_arrow
question_answer 92) The carbohydrate that will yield glucose and fructose on homogeneous catalytic hydrolysis in presence of dilute sulphuric acid is
A)
cellulose
done
clear
B)
maltose
done
clear
C)
starch
done
clear
D)
sucrose
done
clear
View Answer play_arrow
question_answer 93) Which pair of elements has similar properties?
A)
13, 31
done
clear
B)
11, 20
done
clear
C)
12, 10
done
clear
D)
21, 33
done
clear
View Answer play_arrow
question_answer 94) Ethylmine can be prepared by the action of bromine and caustic potash on
A)
acetamide
done
clear
B)
propionamide
done
clear
C)
form amide
done
clear
D)
methylcyanide
done
clear
View Answer play_arrow
question_answer 95) Which of the following alkyl halide will react according to\[{{S}_{N}}1\]mechanism?
A)
1-chloropentane
done
clear
B)
2-methyl-2-chloropentane
done
clear
C)
neo-pentyl chloride
done
clear
D)
2-chloropentane
done
clear
View Answer play_arrow
question_answer 96) In a balanced equation,\[{{H}_{2}}S{{O}_{4}}+xHI\xrightarrow{{}}{{H}_{2}}S+y{{I}_{2}}+z{{H}_{2}}O\]the value of\[x,\,\,y\]and\[z\]are respectively
A)
3, 5, 2
done
clear
B)
4, 8, 5
done
clear
C)
8, 4, 4
done
clear
D)
5, 3, 4
done
clear
View Answer play_arrow
question_answer 97) Which of the following statement is not correct?
A)
\[La{{(OH)}_{3}}\]is less basic than\[Lu{{(OH)}_{3}}\]
done
clear
B)
In lanthanide series, ionic radius of Lu^ ion decreases
done
clear
C)
\[La\]is actually an element of transition series rather lanthanide
done
clear
D)
Atomic radius of\[Zr\]and\[Hf\]are same because of lanthanide contraction
done
clear
View Answer play_arrow
question_answer 98) Diethyl ether on heating with cone. \[HI\]gives two moles of
A)
ethyl iodide
done
clear
B)
ethanol
done
clear
C)
iodoform
done
clear
D)
methyl iodide
done
clear
View Answer play_arrow
question_answer 99) Which of the following is reducing agent?
A)
\[LiAl{{H}_{4}}\]
done
clear
B)
\[Zn+HCl\]
done
clear
C)
\[Sn+HCl\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 100) The brass is an alloy of
A)
gold and copper
done
clear
B)
silver and zinc
done
clear
C)
copper and zinc
done
clear
D)
copper and aluminium
done
clear
View Answer play_arrow
question_answer 101) The Devonian period is considered to be as age of
A)
fishes
done
clear
B)
amphibians
done
clear
C)
reptiles
done
clear
D)
mammals
done
clear
View Answer play_arrow
question_answer 102) Which of the following show paleontological evidence for evolution?
A)
Darwin finches
done
clear
B)
Duck-billed platypus
done
clear
C)
Archaeopteryx
done
clear
D)
Biston betularia
done
clear
View Answer play_arrow
question_answer 103) Kingdom -Plantae was divided into ......... divisions by Engler and Pranti
A)
10
done
clear
B)
11
done
clear
C)
13
done
clear
D)
14
done
clear
View Answer play_arrow
question_answer 104) Which one of the following is a disease causing bacterium in human beings?
A)
E. coli
done
clear
B)
Xanthomonas citri
done
clear
C)
TMV
done
clear
D)
Pilobolus
done
clear
View Answer play_arrow
question_answer 105) Schizogony of Plasmodium is also called as
A)
sporogony
done
clear
B)
gamogony
done
clear
C)
agamogony
done
clear
D)
binary fission
done
clear
View Answer play_arrow
question_answer 106) Dum-dum fever is caused by
A)
Leishmania tropica
done
clear
B)
Leishmania donovani
done
clear
C)
Trypanosoma evansi
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 107) The retort cells are found in the stem of
A)
Sphagnum
done
clear
B)
Poly trichum
done
clear
C)
Funaria
done
clear
D)
Pogonatum
done
clear
View Answer play_arrow
question_answer 108) Nervous system is first formed in
A)
platyhelminthes
done
clear
B)
cnidarian
done
clear
C)
annelids
done
clear
D)
sponges
done
clear
View Answer play_arrow
question_answer 109) The resin duct of a gymnosperm stem is an example of
A)
schizogenous cavity
done
clear
B)
lysigenous cavity
done
clear
C)
big vacuole
done
clear
D)
intercellular space
done
clear
View Answer play_arrow
question_answer 110) Which among dicotyledons are considered to have primitive genera?
A)
Papilionaceae
done
clear
B)
Cucurbitaceae
done
clear
C)
Magnoliaceae
done
clear
D)
Rosaceae
done
clear
View Answer play_arrow
question_answer 111) Enterocoelom appeared first in the course of evolution in
A)
Echinodermata
done
clear
B)
Annelida
done
clear
C)
Chordata
done
clear
D)
Aschelminthes
done
clear
View Answer play_arrow
question_answer 112) The generic name of brain coral is
A)
Fungia
done
clear
B)
Meandrina
done
clear
C)
Madrepora
done
clear
D)
Astraea
done
clear
View Answer play_arrow
question_answer 113) The secondary host in the case of blood fluke is
A)
snail
done
clear
B)
pig
done
clear
C)
man
done
clear
D)
sand fly
done
clear
View Answer play_arrow
question_answer 114) The secretory as well as excretory cells of plants are
A)
cells of oil glands
done
clear
B)
hydathode
done
clear
C)
latex cells
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 115) The plasma membrane of mycoplasma is rich in
A)
cellulose
done
clear
B)
myosin
done
clear
C)
myosin
done
clear
D)
glycogen
done
clear
View Answer play_arrow
question_answer 116) Purgative to expel Ascaris is
A)
oil of Chenopodium
done
clear
B)
tetrachlorethylene
done
clear
C)
hexylresorcinol
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 117) The repeating unit in glycogen is
A)
glucose
done
clear
B)
fructose
done
clear
C)
galactose
done
clear
D)
cellulose
done
clear
View Answer play_arrow
question_answer 118) Which one is a marine leech?
A)
Spiunculus
done
clear
B)
Pontobdella
done
clear
C)
Hirudo
done
clear
D)
Polygordius
done
clear
View Answer play_arrow
question_answer 119) The general body plan and body symmetry are similar in
A)
frog and lofcust
done
clear
B)
sea horse and star fish
done
clear
C)
annelids and sponge
done
clear
D)
pigeon and liver fluke
done
clear
View Answer play_arrow
question_answer 120) Which one is not a carp?
A)
Labeo
done
clear
B)
Catia catia
done
clear
C)
Mystus
done
clear
D)
Cyprinus
done
clear
View Answer play_arrow
question_answer 121) Endo peptidase of plant kingdom is
A)
trysin
done
clear
B)
pepsin
done
clear
C)
papain
done
clear
D)
urease
done
clear
View Answer play_arrow
question_answer 122) One of the following cells can divide by meiosis
A)
sporocyte
done
clear
B)
meiocyte
done
clear
C)
primary spermatocyte and primary oocyte
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 123) Mutations brings about......... change.
A)
small
done
clear
B)
large
done
clear
C)
negligible
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 124) Chromosome complement with\[2n-1\]is called
A)
monosomy
done
clear
B)
trisomy
done
clear
C)
nullisomy
done
clear
D)
tetrasomy
done
clear
View Answer play_arrow
question_answer 125) Interferon is the bodys first line of defence against......... attacks.
A)
viral
done
clear
B)
bacterial
done
clear
C)
protozoan
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 126) One of them is recently domestic carnivore
A)
cat
done
clear
B)
dog
done
clear
C)
polar hear
done
clear
D)
camel
done
clear
View Answer play_arrow
question_answer 127) Dolichos lablab is a
A)
Straggler
done
clear
B)
climber
done
clear
C)
twiner
done
clear
D)
runner
done
clear
View Answer play_arrow
question_answer 128) Sweat glands are scanty in
A)
man
done
clear
B)
rabbit
done
clear
C)
polar bear
done
clear
D)
elephant
done
clear
View Answer play_arrow
question_answer 129) In sugarcane, the adventitious roots are called
A)
prop roots
done
clear
B)
prop and tuberous roots
done
clear
C)
pneumatophores
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 130) In tamarind, the pinnate leaf is
A)
bipinnate
done
clear
B)
tripinnate
done
clear
C)
peripinnate
done
clear
D)
imperipinnate
done
clear
View Answer play_arrow
question_answer 131) Camouflage of Chameleon is associated with
A)
chromosome
done
clear
B)
chromomere
done
clear
C)
chromoplast
done
clear
D)
chromatophore
done
clear
View Answer play_arrow
question_answer 132) Which of these is the place of cellulose digestion in numinants?
A)
Rumen
done
clear
B)
Reticulum
done
clear
C)
Omassum
done
clear
D)
Abomassum
done
clear
View Answer play_arrow
question_answer 133) Cyathjum and hypanthodium resemble each other in possessing
A)
involucre
done
clear
B)
dioecious nature
done
clear
C)
receptacle
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 134) Vital capacity of lungs is
A)
Inspiratory Reserve Volume (IRV) + Expiratory Reserve Volume (ERV) + Tidal Volume (TV) + Residual Volume (RV)
done
clear
B)
IRV + ERV + TV
done
clear
C)
IRV + ERV
done
clear
D)
IRV + ERV + TV - RV
done
clear
View Answer play_arrow
question_answer 135) Spiral valve is present in
A)
right auricle
done
clear
B)
sinus venosus
done
clear
C)
truncus arteriosus
done
clear
D)
right ventricle
done
clear
View Answer play_arrow
question_answer 136) Edible part of mango is
A)
epicarp
done
clear
B)
mesocarp
done
clear
C)
thalamus
done
clear
D)
endocarp
done
clear
View Answer play_arrow
question_answer 137) Smallest parasitic plant with two cotyledons in the seed is
A)
Lemna
done
clear
B)
Wolffia
done
clear
C)
Arceuthobium
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 138) Inflorescence of Liliaceae is
A)
Zygomorphic
done
clear
B)
trimerous
done
clear
C)
pentamerous
done
clear
D)
imperfect
done
clear
View Answer play_arrow
question_answer 139) The presence of cuticle in arthropods indicate
A)
metamorphosis
done
clear
B)
moulting
done
clear
C)
aquatic environment
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 140) Autonomy, a phenomenon is well marked in
A)
annelids
done
clear
B)
arthropods
done
clear
C)
mollusc
done
clear
D)
Echinodermata
done
clear
View Answer play_arrow
question_answer 141) Cotton fibre is part of
A)
epidermal tissue system
done
clear
B)
vascular tissue system
done
clear
C)
meristematic tissue system
done
clear
D)
ground tissue system
done
clear
View Answer play_arrow
question_answer 142) Hyperparasite is
A)
which kills host
done
clear
B)
which completes life cycle in ones host
done
clear
C)
which uses body of host to reproduce
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 143) Actively moving animal organisms in any aquatic systems are
A)
benthic
done
clear
B)
phytoplanktons
done
clear
C)
nectons
done
clear
D)
zooplanktons
done
clear
View Answer play_arrow
question_answer 144) How deep is photic or euphotic zone from the upper surface of sea?
A)
10 m
done
clear
B)
100 m
done
clear
C)
200 m
done
clear
D)
500 m
done
clear
View Answer play_arrow
question_answer 145) Heavy metals are detoxified in plants by
A)
ABA
done
clear
B)
all elopathins
done
clear
C)
phytochelatins
done
clear
D)
phytoalexins
done
clear
View Answer play_arrow
question_answer 146) If the number of a species increase in any national park it can be due to
A)
intraspecific competition
done
clear
B)
interspecific competition
done
clear
C)
emigration
done
clear
D)
mutualism
done
clear
View Answer play_arrow
question_answer 147) The concept of genetic drift was introduced by
A)
Hardy Weinberg
done
clear
B)
Sewall Wright
done
clear
C)
GG Simpson
done
clear
D)
Julian Huxley
done
clear
View Answer play_arrow
question_answer 148) Which one among the following chemicals is used for preventing defoliation of forest trees?
A)
AMO - 1618
done
clear
B)
Phosphon D
done
clear
C)
Malic hydrazide
done
clear
D)
2, 4-D
done
clear
View Answer play_arrow
question_answer 149) The angiosperm showing symbiotic association with blue green algae is
A)
Lens
done
clear
B)
Cyamopsis
done
clear
C)
Trifolium
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 150) Sago which we use as food comes from this plant
A)
Calamus ritung
done
clear
B)
Metroxylon rumphii
done
clear
C)
Areca catechu
done
clear
D)
Phoenix dactylifera
done
clear
View Answer play_arrow
question_answer 151) Which one is the hardest wood?
A)
Shorea robusta
done
clear
B)
Tectona grandis
done
clear
C)
Cedrus deodara
done
clear
D)
Dalbergia sissoo
done
clear
View Answer play_arrow
question_answer 152) Medium used in culturing hybridoma cells is
A)
HAT
done
clear
B)
HAP
done
clear
C)
TAP
done
clear
D)
TAC
done
clear
View Answer play_arrow
question_answer 153) Agar is obtained from
A)
bacteria
done
clear
B)
blue-green algae
done
clear
C)
red algae
done
clear
D)
brown algae
done
clear
View Answer play_arrow
question_answer 154) Against which foreign organism (antigen) antibiotic is most effective?
A)
Virus
done
clear
B)
Bacteria
done
clear
C)
Fungal infection
done
clear
D)
Protozoan infection
done
clear
View Answer play_arrow
question_answer 155) Toxin produced by plants are
A)
phytoalexins
done
clear
B)
phytochrome
done
clear
C)
phytoferritin
done
clear
D)
phytotoxins
done
clear
View Answer play_arrow
question_answer 156) Which disease is not caused by triple antigens?
A)
Tetanus
done
clear
B)
Diphtheric
done
clear
C)
Whooping cough
done
clear
D)
Typhoid
done
clear
View Answer play_arrow
question_answer 157) Glaucoma is caused due to
A)
opacity of lens
done
clear
B)
increase in size of eye ball
done
clear
C)
canal of Schlemn is blocked
done
clear
D)
vitreous humour becomes dry
done
clear
View Answer play_arrow
question_answer 158) Which cranial nerve controls the heart muscles?
A)
Facial
done
clear
B)
Vagus
done
clear
C)
Auditory
done
clear
D)
Trochlear
done
clear
View Answer play_arrow
question_answer 159) Corolloid roots of Cycas has
A)
Anabaena
done
clear
B)
My corrhiza
done
clear
C)
Rhizobium
done
clear
D)
Azotobacter
done
clear
View Answer play_arrow
question_answer 160) The sun loving plants are referred to as
A)
halophytes
done
clear
B)
heliophytes
done
clear
C)
heterotrophs
done
clear
D)
sciophytes
done
clear
View Answer play_arrow
question_answer 161) Die back of citrus and reclamation of legumes and cereals is due to deficiency of
A)
copper
done
clear
B)
zinc
done
clear
C)
sodium
done
clear
D)
molybdenum
done
clear
View Answer play_arrow
question_answer 162) Which enzyme fixes\[C{{O}_{2}}\]in Calvin cycle?
A)
RuBP carboxylase
done
clear
B)
PEP carboxylase
done
clear
C)
carbonic anhydrase
done
clear
D)
hexokinase
done
clear
View Answer play_arrow
question_answer 163) In which of the following are orchreate stipules found?
A)
Cruciferae
done
clear
B)
Papilionaceae
done
clear
C)
Compositae
done
clear
D)
Polygonaceae
done
clear
View Answer play_arrow
question_answer 164) Surfactant in alveoli is secreted by
A)
dust cells
done
clear
B)
type I epithelial cells
done
clear
C)
type II epithelial cells
done
clear
D)
Kupffer cells
done
clear
View Answer play_arrow
question_answer 165) Which one is not fully formed in birds?
A)
Pectoral girdle
done
clear
B)
Pelvic girdle
done
clear
C)
Hind limb
done
clear
D)
Forelimb
done
clear
View Answer play_arrow
question_answer 166) Cells of Deiter occur in
A)
utriculus
done
clear
B)
retina of eyes
done
clear
C)
organ of Corti
done
clear
D)
sebaceous glands
done
clear
View Answer play_arrow
question_answer 167) Epinephrine is
A)
clusters of glomeruli in mammalian kidney
done
clear
B)
nephrostomal part of mesoderm
done
clear
C)
hormone of the adrenal gland
done
clear
D)
frontal lobe of nephridia
done
clear
View Answer play_arrow
question_answer 168) Plumose stigma is adaptation for
A)
hydrophily
done
clear
B)
ornithophily
done
clear
C)
zoophily
done
clear
D)
anemophily
done
clear
View Answer play_arrow
question_answer 169) Embryo sac is
A)
megaspore
done
clear
B)
micro gametophyte
done
clear
C)
female gametophyte
done
clear
D)
megasporangium
done
clear
View Answer play_arrow
question_answer 170) Middle piece of a mammalian sperm contains
A)
nucleus
done
clear
B)
centriole
done
clear
C)
mitochondria
done
clear
D)
vacuole
done
clear
View Answer play_arrow
question_answer 171) The type of immunoglobulin present in the foetus are
A)
\[IgD\]
done
clear
B)
\[IgE\]
done
clear
C)
\[IgG\]
done
clear
D)
\[IgM\]
done
clear
View Answer play_arrow
question_answer 172) One of the following controls the population from going beyond its limits
A)
carrying capacity
done
clear
B)
environmental resistance
done
clear
C)
family planning programme
done
clear
D)
shortage of food supply
done
clear
View Answer play_arrow
question_answer 173) Viroids differ from viruses in being
A)
naked RNA molecules only
done
clear
B)
naked DNA molecules
done
clear
C)
naked DNA packaged with viral genome
done
clear
D)
satellite RNA packaged with viral genome
done
clear
View Answer play_arrow
question_answer 174) The codon for anticodon 3UUAS is
A)
5AAUS
done
clear
B)
3AAU57
done
clear
C)
5AAT3
done
clear
D)
3AAGS
done
clear
View Answer play_arrow
question_answer 175) Pureline breed refers to
A)
homozygosity
done
clear
B)
heterozygosity
done
clear
C)
homozygosity with only dominant genes
done
clear
D)
heterozygosity and linkage
done
clear
View Answer play_arrow
question_answer 176) Major function of mitochondria in a cell is
A)
secretion
done
clear
B)
excretion
done
clear
C)
osmoregulation
done
clear
D)
fat synthesis
done
clear
View Answer play_arrow
question_answer 177) Feet in King fisher are modified for
A)
walking
done
clear
B)
perching
done
clear
C)
running
done
clear
D)
catching
done
clear
View Answer play_arrow
question_answer 178) Echinoderms are exclusively
A)
marine
done
clear
B)
riverine
done
clear
C)
esturine
done
clear
D)
fresh water
done
clear
View Answer play_arrow
question_answer 179) Instead of seta in capsule of Sphagnum the structure present in
A)
columella
done
clear
B)
pseudopodium
done
clear
C)
haustoria
done
clear
D)
root
done
clear
View Answer play_arrow
question_answer 180) Kappa particles in Paramecium aurelia making killer traits were discovered by
A)
Hill
done
clear
B)
Sonneborn
done
clear
C)
Rosenh of
done
clear
D)
Morgan
done
clear
View Answer play_arrow
question_answer 181) Dodder can transmit
A)
mycoplasmal disease
done
clear
B)
viral diseases
done
clear
C)
Both (a) and (b)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 182) Acquired characters are
A)
inherited
done
clear
B)
artificial
done
clear
C)
superficial
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 183) Sexual selection in Darwins theory was the
A)
weakest part
done
clear
B)
strong part
done
clear
C)
strongest part
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 184) The statement All biological catalysts are proteins is no more valid because of the discovery of
A)
ribonuclease
done
clear
B)
ribozymes
done
clear
C)
lysozymes
done
clear
D)
enzymes
done
clear
View Answer play_arrow
question_answer 185) From the given pair of proteins where both members of the pair are either structural or functional. Mark the odd pair.
A)
Haemoglobin and myoglobin
done
clear
B)
Fibrinogen and trypsin
done
clear
C)
Insulin and glucagon
done
clear
D)
Keratin and fibrin
done
clear
View Answer play_arrow
question_answer 186) Ethylene is involved in
A)
aerobic respiration
done
clear
B)
anaerobic respiration
done
clear
C)
climacteric respiration
done
clear
D)
fermentation
done
clear
View Answer play_arrow
question_answer 187) Suspensor formation in Sagittaria occurs at ......... celled stage.
A)
6-10
done
clear
B)
4-5
done
clear
C)
2-3
done
clear
D)
single
done
clear
View Answer play_arrow
question_answer 188) Insulin and glucagon are
A)
polypeptides
done
clear
B)
polysaccharides
done
clear
C)
polynucleotides
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 189) Preganglionic sympathetic fibres are
A)
adrenergic
done
clear
B)
cholinergic
done
clear
C)
synergic
done
clear
D)
hypergonic
done
clear
View Answer play_arrow
question_answer 190) Kranz anatomy is found in
A)
most of monocots
done
clear
B)
few dicots
done
clear
C)
Both (a) and (b)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 191) Osphradium of Pila globosa is
A)
chemoreceptor
done
clear
B)
photoreceptor
done
clear
C)
thermo receptor
done
clear
D)
tangoreceptor
done
clear
View Answer play_arrow
question_answer 192) Caudal sensory organs (phasmids) are present in
A)
rotifers
done
clear
B)
nematomorphs
done
clear
C)
nematodes
done
clear
D)
cestodes
done
clear
View Answer play_arrow
question_answer 193) The total number of species belonging to present day gymnosperm is
A)
1000
done
clear
B)
2000
done
clear
C)
1500
done
clear
D)
722
done
clear
View Answer play_arrow
question_answer 194) It is a copper moss
A)
Bryum
done
clear
B)
Tortella
done
clear
C)
Merceya
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 195) A phage that invades but does not destroy the host is
A)
temperate phage
done
clear
B)
sexduction
done
clear
C)
phycophages
done
clear
D)
virulent phage
done
clear
View Answer play_arrow
question_answer 196) The drug calchicine has inhibitory effect on cell cycle in which stage?
A)
Prophase
done
clear
B)
Anaphase
done
clear
C)
Metaphase
done
clear
D)
Telophase
done
clear
View Answer play_arrow
question_answer 197) Endoparasite worms belong to class
A)
Turbellaria
done
clear
B)
Trematoda
done
clear
C)
Cestoda
done
clear
D)
Encestoda
done
clear
View Answer play_arrow
question_answer 198) Roots are the source of cammercial rubber in the species of
A)
Hevea
done
clear
B)
Ficus
done
clear
C)
Taraxacum
done
clear
D)
Parthenium
done
clear
View Answer play_arrow
question_answer 199) The inherent capacity of a cell to regenerate a new whole organism is called
A)
ontogeny
done
clear
B)
totipotency
done
clear
C)
phylogeny
done
clear
D)
proliferation
done
clear
View Answer play_arrow
question_answer 200) Trypanosoma brucei causes
A)
dysentery
done
clear
B)
Kala-azar
done
clear
C)
sleeping sickness in man
done
clear
D)
a disease of animals
done
clear
View Answer play_arrow