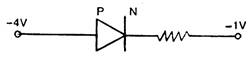
question_answer 1) In a neon discharge tube \[2.9\times {{10}^{18}}\,N{{e}^{+}}\] ions move to the right each second while\[1.2\times {{10}^{18}}\] electrons move to the left per second. Electron charge is\[1.6\times {{10}^{-19}}\]. The current in the discharge tube is:
A)
zero
done
clear
B)
0.66 A towards left
done
clear
C)
1A towards right I
done
clear
D)
0.66A towards right
done
clear
View Answer play_arrow
question_answer 2) The value of the stopping potential when the metals with work function 0.6 eV is illuminated with the light of 2 eV :
A)
2.6 V
done
clear
B)
3.6 V
done
clear
C)
0.8 V
done
clear
D)
1.4 V
done
clear
View Answer play_arrow
question_answer 3) Two trains, one coming towards and another going away from an observer both at 4 m/s produce whistle simultaneously of frequency 300 Hz. The number of beats produced, will be :
A)
5
done
clear
B)
6
done
clear
C)
7
done
clear
D)
12
done
clear
View Answer play_arrow
question_answer 4) If the metal bob of a simple pendulum is replaced by a wooden bob, then its time period will:
A)
first increase than decrease
done
clear
B)
remains the same
done
clear
C)
increase
done
clear
D)
decrease
done
clear
View Answer play_arrow
question_answer 5) The possible value of poisons ratio is :
A)
0.4
done
clear
B)
0.8
done
clear
C)
0.9
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 6) When the diameter of the capillary tube is doubled, then the height of the liquid that will rise, is:
A)
same as earlier
done
clear
B)
half
done
clear
C)
twice
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 7) An inelastic ball is dropped from a height of 100 m. Due to earth 20% of its energy is lost. The ball will rise upto the height of:
A)
80 m
done
clear
B)
40 m
done
clear
C)
75 m
done
clear
D)
20 m
done
clear
View Answer play_arrow
question_answer 8) The orbital velocity of earths satellite near surface is 7 km/s. When the radius of orbit is 4 times than that of earths radius. Then orbital velocity in that orbit will be :
A)
7 km/s
done
clear
B)
3.5 km/s
done
clear
C)
5.2 km/s
done
clear
D)
9.4 km/s
done
clear
View Answer play_arrow
question_answer 9) A body cools from \[{{60}^{o}}C\] to \[{{50}^{o}}C\] in 10 minutes. If the room temperature is 25 V and assuming Newtons law of cooling to hold good. The temperature of the body at the end of next 10 minutes will be :
A)
\[35.5{}^\circ C\]
done
clear
B)
\[50{}^\circ C\]
done
clear
C)
\[{{42.85}^{o}}C\]
done
clear
D)
\[{{45}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 10) The objective with large aperture are used in telescope for :
A)
ease of manufacture
done
clear
B)
reducing lens aberration
done
clear
C)
greater resolution
done
clear
D)
bright images
done
clear
View Answer play_arrow
question_answer 11) The frequency of a photon having energy 200 eV is \[(h=6.6\times {{10}^{-34}}Js)\]:
A)
\[4.84\times {{10}^{16}}\]
done
clear
B)
\[2.42\times {{10}^{16}}\]
done
clear
C)
\[3.84\times {{10}^{16}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 12)
The value of current in the given circuits :
A)
0.10 A
done
clear
B)
1.1 A
done
clear
C)
\[{{10}^{-2}}A\]
done
clear
D)
0 A
done
clear
View Answer play_arrow
question_answer 13) An body weighs 700 N on earth. What will be its weight on a planet having \[\frac{1}{7}\]of earths mass and half of earths radius?
A)
400 N
done
clear
B)
800 N
done
clear
C)
500 N
done
clear
D)
200 N
done
clear
View Answer play_arrow
question_answer 14) The moment of inertia of a regular disc of mass 0.4 kg and radius 100 cm about an axis perpendicular to the plane of the disc and passing through its centre, will be :
A)
0.02 kg \[{{m}^{2}}\]
done
clear
B)
1.5 kg \[{{m}^{2}}\]
done
clear
C)
0.8 kg \[{{m}^{2}}\]
done
clear
D)
0.2 kg \[{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 15) Angular velocity of seconds hand watch will be:
A)
0.105 rad/s
done
clear
B)
1.105 rad/s
done
clear
C)
0.315 rad/s
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 16) A spring 40 mm long is stretched by the application of a force, if 10 N force is required to stretch the spring through 1 mm. Then work done in stretching the spring through 40 mm is :
A)
8 J
done
clear
B)
4 J
done
clear
C)
16 J
done
clear
D)
32 J
done
clear
View Answer play_arrow
question_answer 17) If the distance between the conduction band and valence band is 1 eV, then the combination will be:
A)
semiconductor
done
clear
B)
conductor
done
clear
C)
insulator
done
clear
D)
non-metal
done
clear
View Answer play_arrow
question_answer 18) When an electron in hydrogen atom is exerted, from 4th to 5th stationary orbit the change in angular momentum of electron will be (Plancks constant = \[6.6\times {{10}^{34}}\,Js\]):
A)
\[1.05\times {{10}^{-34}}\,Js\]
done
clear
B)
\[2.05\times {{10}^{-34}}\,Js\]
done
clear
C)
\[3.10\times {{10}^{-34}}\,Js\]
done
clear
D)
\[4.05\times {{10}^{-34}}\,Js\]
done
clear
View Answer play_arrow
question_answer 19) A quality factor of LCR factor circuit having resistance R and inductance L at resonance frequency (to) is given by :
A)
\[{{\left( \frac{\omega L}{R} \right)}^{2}}\]
done
clear
B)
\[{{\left( \frac{R\omega }{L} \right)}^{2}}\]
done
clear
C)
\[\frac{R\omega }{L}\]
done
clear
D)
\[\frac{\omega L}{R}\]
done
clear
View Answer play_arrow
question_answer 20) The value of current through an inductance of 1 H and of negligible resistance when connected through an AC source of 200 V and 50 Hz will be:
A)
2.001 A
done
clear
B)
1.274 A
done
clear
C)
0.700A
done
clear
D)
0.368 A
done
clear
View Answer play_arrow
question_answer 21) A lens of refractive index p. is dipped in a liquid of refractive index p of focal length of lens in air is \[f.\] Its focal length in liquid will be :
A)
\[\frac{f\mu (\mu -1)}{(\mu -\mu )}\]
done
clear
B)
\[\frac{f(\mu -\mu )}{\mu (\mu -1)}\]
done
clear
C)
\[\frac{\mu (\mu -1)}{f(\mu -\mu )}\]
done
clear
D)
\[\frac{f\mu \mu }{\mu -\mu }\]
done
clear
View Answer play_arrow
question_answer 22) The mass of three wires of copper are in the ratio of 1 :3:5and their lengths are in the ratio of 5 :3: 1. The ratio of their electrical resistances will be :
A)
125 : 15 : 1
done
clear
B)
1 : 15 : 125
done
clear
C)
5: 3: 15
done
clear
D)
15 : 3 : 5
done
clear
View Answer play_arrow
question_answer 23) Interference was observed in interference chamber when air was present, now the chamber was evacuated and if the same light is used, a careful observer will see :
A)
interference with bright band
done
clear
B)
interference with dark band
done
clear
C)
interference in which width of the fringe will be slightly increased
done
clear
D)
interference with dark bands
done
clear
View Answer play_arrow
question_answer 24) Two whistles A and B produces notes of frequencies 660 Hz and 594 Hz respectively. There is listener at the mid point of the line joining them. Now the whistle B and the listener start moving with speed 30 m/s towards the whistle A. If speed of sound be 330 m/s, how many beats will be heard by the listener :
A)
4
done
clear
B)
5
done
clear
C)
6
done
clear
D)
7
done
clear
View Answer play_arrow
question_answer 25) In which case application of rotating body is useful?
A)
when velocity of body is in straight line
done
clear
B)
when body is projected
done
clear
C)
when the body is rotated
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 26) Two bodies with kinetic energies in the ratio of 4 :1 are moving with equal linear momentum. The ratio of their masses is
A)
1 : 2
done
clear
B)
1 : 1
done
clear
C)
3:1
done
clear
D)
1 : 4
done
clear
View Answer play_arrow
question_answer 27) A body of mass 5 kg is moving in a circle of radius 1 m with an angular velocity of 2rad/sec, then the centripetal force will be:
A)
20 N
done
clear
B)
5 N
done
clear
C)
15 N
done
clear
D)
40 N
done
clear
View Answer play_arrow
question_answer 28) A truck and a car are moving with equal velocity. On applying the brakes both will stop after certain distance, then :
A)
both will cover equal distance
done
clear
B)
car will cover more distance before rest
done
clear
C)
car will cover less distance before rest
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 29) A body of mass 100 kg is rotating in a circular path of radius R with constant velocity. The work done in one revolution is:
A)
zero
done
clear
B)
\[\frac{100}{R}\,J\]
done
clear
C)
\[\frac{R}{100}\,J\]
done
clear
D)
100 R J
done
clear
View Answer play_arrow
question_answer 30) When cathode rays strike a metal target of high melting point with a very high velocity then which of the following are produced?
A)
ultrasonic rays
done
clear
B)
\[\gamma \]-waves
done
clear
C)
\[\alpha \]-ways
done
clear
D)
X-rays
done
clear
View Answer play_arrow
question_answer 31) In a circuit, the current lags behind the voltage by a phase difference of\[\frac{\pi }{2}\] The circuit contains which of the following :
A)
R and C
done
clear
B)
only C
done
clear
C)
only L
done
clear
D)
only R
done
clear
View Answer play_arrow
question_answer 32) Same length of two identical wires are first connected in series and then in parallel, the amount of heat produced in both the cases are in the ratio of :
A)
4 :1
done
clear
B)
1 : 3
done
clear
C)
1 : 2
done
clear
D)
2:1
done
clear
View Answer play_arrow
question_answer 33) Three capacitors of capacitances3\[\mu F\], 10\[\mu F\] and 15\[\mu F\]are connected in series to a voltage source of 100 V. Then the charge on 15\[\mu F\] will be :
A)
200 \[\mu C\]
done
clear
B)
150\[\mu C\]
done
clear
C)
300\[\mu C\]
done
clear
D)
400\[\mu C\]
done
clear
View Answer play_arrow
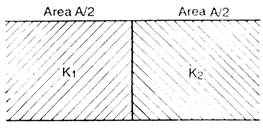
question_answer 34)
Two materials of dielectric constant \[{{k}_{1}}\] and \[{{k}_{2}}\] are filled between two parallel plates of a capacitor as shown in fig. The capacities of capacitor is :
A)
\[\frac{2A{{\varepsilon }_{o}}}{d}\left( \frac{{{k}_{1}}{{k}_{2}}}{{{k}_{1}}+{{k}_{2}}} \right)\]
done
clear
B)
\[\frac{A{{\varepsilon }_{o}}}{d}\left( \frac{{{k}_{1}}{{k}_{2}}}{{{k}_{1}}+{{k}_{2}}} \right)\]
done
clear
C)
\[\frac{2A{{\varepsilon }_{0}}({{k}_{1}}+{{k}_{2}})}{d}\]
done
clear
D)
\[\frac{A{{\varepsilon }_{0}}({{k}_{1}}+{{k}_{2}})}{d}\]
done
clear
View Answer play_arrow
question_answer 35) If induced emf produced in primary and secondary coil is 2 mV and 5 mv respectively and rate of change of current is 20 A/sec. The coefficient of mutual induction of secondary coil is :
A)
4.5 mH
done
clear
B)
1 mH
done
clear
C)
0.5 mH
done
clear
D)
0.25 mH
done
clear
View Answer play_arrow
question_answer 36) A plan convex lens is made of refractive index 1.6. The radius of curvature of the curved surface is 60 cm. The focal length of the lens is :
A)
200 cm
done
clear
B)
50 cm
done
clear
C)
150 cm
done
clear
D)
100 cm
done
clear
View Answer play_arrow
question_answer 37) Electroplating does not help in :
A)
protecting metal against corrosion .
done
clear
B)
fine finish to the surface
done
clear
C)
shining appearance
done
clear
D)
metal to become hard
done
clear
View Answer play_arrow
question_answer 38) Electric potential of earth is taken to be zero, because the earth is a good :
A)
semiconductor
done
clear
B)
dielectrics
done
clear
C)
insulator
done
clear
D)
conductor
done
clear
View Answer play_arrow
question_answer 39) If the vibration of a string are to be increased by a factor of 2. Then tension in the string must be made :
A)
8 times
done
clear
B)
4 times
done
clear
C)
1/3 times
done
clear
D)
half times
done
clear
View Answer play_arrow
question_answer 40) An average kinetic energy of a gas molecule at \[27{}^\circ C\] is\[6.21\times {{10}^{-21}}J.\] Its average kinetic energy at \[227{}^\circ C\] will be:
A)
\[11.35\times {{10}^{-21}}J\]
done
clear
B)
\[10.35\times {{10}^{-21}}J\]
done
clear
C)
\[15.35\times {{10}^{-21}}J\]
done
clear
D)
\[5.3\times {{10}^{-21}}J\]
done
clear
View Answer play_arrow
question_answer 41) A substance reduces to 1/16 of its original mass in 2 hours. The half-life period of the substance will be :
A)
120 min
done
clear
B)
60 min
done
clear
C)
30 min
done
clear
D)
15 min
done
clear
View Answer play_arrow
question_answer 42) A given mass of a gas is allowed to expand freely until its volume becomes double. If \[{{c}_{b}}\], and \[{{c}_{a}}\]are the velocities of sound in the gas before and after expansion respectively then \[{{c}_{a}}\] is equal to :
A)
\[{{2}_{cb}}\]
done
clear
B)
\[{{\sqrt{3}}_{cb}}\]
done
clear
C)
\[{{\sqrt{2}}_{cb}}\]
done
clear
D)
\[{{c}_{b}}\]
done
clear
View Answer play_arrow
question_answer 43) The half-life (T) and the disintegration constant \[\lambda \] of a radioactive substance are related as:
A)
\[\lambda \]T = 0.693
done
clear
B)
\[\frac{\lambda }{T}=1\]
done
clear
C)
T = 0.693\[\lambda \]
done
clear
D)
\[\frac{\lambda }{T}=0.693\]
done
clear
View Answer play_arrow
question_answer 44) One million electron volt is
A)
\[1.6\times {{10}^{-16}}J\]
done
clear
B)
\[1.6\times {{10}^{-19}}J\]
done
clear
C)
\[1.6\times {{10}^{-13}}J\]
done
clear
D)
\[1.6\times {{10}^{-11}}J\]
done
clear
View Answer play_arrow
question_answer 45) Dark lines on solar spectrum are due to :
A)
scattering
done
clear
B)
lack of certain elements
done
clear
C)
black body radiation
done
clear
D)
absorption of certain wavelengths by outer layers
done
clear
View Answer play_arrow
question_answer 46) When the amount of work done is 300 J and change in internal energy is 100 J, then heat supplied is :
A)
400 J
done
clear
B)
250 J
done
clear
C)
350 J
done
clear
D)
200 J
done
clear
View Answer play_arrow
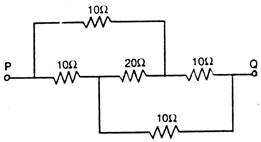
question_answer 47)
In the following circuit, the resistance between points P and Q is :
A)
10\[\Omega \]
done
clear
B)
15\[\Omega \]
done
clear
C)
5 \[\Omega \]
done
clear
D)
7.5 \[\Omega \]
done
clear
View Answer play_arrow
question_answer 48) The wavelength of first spectral line of sodium is 5896\[\overset{0}{\mathop{A}}\,\]. What is the first excitation potential of sodium atom:
A)
2.1volt
done
clear
B)
4.2 volt
done
clear
C)
1.5volt
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 49) For a common emitter circuit of a transistor, when the base current charges 80\[\mu \].i A, the collector current charges by 48 mA. The current amplification factor will be :
A)
75
done
clear
B)
50
done
clear
C)
40
done
clear
D)
60
done
clear
View Answer play_arrow
question_answer 50) In an A.C. circuit the potential difference across inductance and resistance joined in series are respectively 12 V and 16 V. The total potential difference across the circuit is:
A)
20 V
done
clear
B)
10 V
done
clear
C)
15 V
done
clear
D)
5 V
done
clear
View Answer play_arrow
question_answer 51) Which of the following electronic configuration represents the largest atomic size?
A)
\[_{20}C{{a}^{42}}\]
done
clear
B)
\[\beta \]
done
clear
C)
\[10\Omega \]
done
clear
D)
\[15\Omega \]
done
clear
View Answer play_arrow
question_answer 52) The geometry of \[5\Omega \] molecule is :
A)
trigonal pyramidal
done
clear
B)
tetrahedral
done
clear
C)
square planar
done
clear
D)
octahedral
done
clear
View Answer play_arrow
question_answer 53) \[7.5\Omega \] undergoes \[5896\overset{\text{o}}{\mathop{\text{A}}}\,\]-emission resulting nucleus will be:
A)
\[_{21}S{{c}^{41}}\]
done
clear
B)
\[_{21}S{{c}^{42}}\]
done
clear
C)
\[_{20}S{{c}^{42}}\]
done
clear
D)
\[_{21}C{{a}^{42}}\]
done
clear
View Answer play_arrow
question_answer 54) Polarising power of magnesium is closer to that of:
A)
Na
done
clear
B)
Li
done
clear
C)
K
done
clear
D)
Ca
done
clear
View Answer play_arrow
question_answer 55) Zero group elements are inert due to their:
A)
electronic configuration
done
clear
B)
monoatomic nature
done
clear
C)
rare occurrence
done
clear
D)
gaseous nature
done
clear
View Answer play_arrow
question_answer 56) CO is used in the metallurgy of :
A)
Ni
done
clear
B)
Pt
done
clear
C)
Cr
done
clear
D)
Cu
done
clear
View Answer play_arrow
question_answer 57) Which of the following is largest cation?
A)
\[_{21}C{{a}^{42}}\]
done
clear
B)
\[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\]
done
clear
C)
\[1{{s}^{2}},2{{s}^{1}}\]
done
clear
D)
\[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\]
done
clear
View Answer play_arrow
question_answer 58) In a nuclear reaction mass defect is 0.0186 amu, the energy change in the reaction will be:
A)
\[1{{s}^{2}},2{{s}^{2}}\]
done
clear
B)
\[S{{F}_{6}}\]
done
clear
C)
\[_{20}C{{a}^{42}}\]
done
clear
D)
\[\beta \]
done
clear
View Answer play_arrow
question_answer 59) The density of a gas at \[10\Omega \] and 1 atmospheric pressure is d. If the pressure remain constant, at which of the following temperature the density become 0.75 d:
A)
\[15\Omega\]
done
clear
B)
\[5\Omega \]
done
clear
C)
\[7.5\Omega \]
done
clear
D)
\[5896\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 60) \[_{21}S{{c}^{41}}\]is:
A)
paramagnetic
done
clear
B)
diamagnetic
done
clear
C)
ferromagnetic
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 61) Which of the following is linear?
A)
\[_{21}S{{c}^{42}}\]
done
clear
B)
\[_{20}S{{c}^{42}}\]
done
clear
C)
\[_{21}C{{a}^{42}}\]
done
clear
D)
\[C{{s}^{+}}\]
done
clear
View Answer play_arrow
question_answer 62) Which of the following does not conducted electricity?
A)
Molten \[C{{s}^{+}}\]
done
clear
B)
Molten \[{{K}^{+}}\]
done
clear
C)
Solid \[{{K}^{+}}\]
done
clear
D)
Aqueous \[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\]
done
clear
View Answer play_arrow
question_answer 63) Oxidation number of sulphur in \[1{{s}^{2}},2{{s}^{1}}\] will be:
A)
\[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\]
done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}\]
done
clear
C)
\[S{{F}_{6}}\]
done
clear
D)
\[_{20}C{{a}^{42}}\]
done
clear
View Answer play_arrow
question_answer 64) Which of the following act as a reducing agent?
A)
\[\beta \]
done
clear
B)
\[10\Omega \]
done
clear
C)
\[15\Omega \]
done
clear
D)
\[5\Omega \]
done
clear
View Answer play_arrow
question_answer 65) Which of the following is Bronsted acid?
A)
\[7.5\Omega \]
done
clear
B)
\[5896\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[_{21}S{{c}^{41}}\]
done
clear
D)
\[_{21}S{{c}^{42}}\]
done
clear
View Answer play_arrow
question_answer 66) Which of the following electronic configuration represents a transition element?
A)
\[_{20}S{{c}^{42}}\]
done
clear
B)
\[_{21}C{{a}^{42}}\]
done
clear
C)
\[C{{s}^{+}}\]
done
clear
D)
\[{{K}^{+}}\]
done
clear
View Answer play_arrow
question_answer 67) When \[N{{a}^{+}}\] reacts with \[N{{a}^{+}}\] a blood red colour is obtained due to the formation of
A)
\[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\]
done
clear
B)
\[1{{s}^{2}},2{{s}^{1}}\]
done
clear
C)
\[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\]
done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 68) Which of the following is not an ore of copper?
A)
Limonite
done
clear
B)
Malachite
done
clear
C)
Chalcopyrite
done
clear
D)
Cuprite
done
clear
View Answer play_arrow
question_answer 69) The hvdrochlorides of amines form double salt with:
A)
\[S{{F}_{6}}\]
done
clear
B)
\[_{20}C{{a}^{42}}\]
done
clear
C)
both a and b
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 70) Which of the following does not act as Lewis acid?
A)
\[\beta \]
done
clear
B)
\[10\Omega \]
done
clear
C)
\[15\Omega \]
done
clear
D)
\[5\Omega \]
done
clear
View Answer play_arrow
question_answer 71) Gun cotton is obtained when cone. \[7.5\Omega \] reacts with:
A)
starch
done
clear
B)
glycol
done
clear
C)
glycerine
done
clear
D)
cellulose
done
clear
View Answer play_arrow
question_answer 72) The number of electrons present m the valence shell of phosphorus in \[5896\overset{\text{o}}{\mathop{\text{A}}}\,\] is:
A)
8
done
clear
B)
10
done
clear
C)
12
done
clear
D)
18
done
clear
View Answer play_arrow
question_answer 73) The greatest strain is involved in cycloalkane, when the bond angle is:
A)
\[_{21}S{{c}^{41}}\]
done
clear
B)
\[_{21}S{{c}^{42}}\]
done
clear
C)
\[_{20}S{{c}^{42}}\]
done
clear
D)
\[_{21}C{{a}^{42}}\]
done
clear
View Answer play_arrow
question_answer 74) The next higher homologue of \[C{{s}^{+}}\] is:
A)
\[{{K}^{+}}\]
done
clear
B)
\[N{{a}^{+}}\]
done
clear
C)
\[L{{i}^{+}}\]
done
clear
D)
\[L{{i}^{+}}\]
done
clear
View Answer play_arrow
question_answer 75) The acid which does not contain \[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\]group is :
A)
ethanoic acid
done
clear
B)
picric acid
done
clear
C)
lactic acid
done
clear
D)
palmitic acid
done
clear
View Answer play_arrow
question_answer 76) An alcohol on oxidation produces \[1{{s}^{2}},2{{s}^{1}}\] and \[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\]. The alcohol is:
A)
\[1{{s}^{2}},2{{s}^{2}}\]
done
clear
B)
\[S{{F}_{6}}\]
done
clear
C)
\[_{20}C{{a}^{42}}\]
done
clear
D)
\[\beta \]
done
clear
View Answer play_arrow
question_answer 77) Cyclohexanol is a:
A)
primary alcohol
done
clear
B)
secondary alcohol
done
clear
C)
tertiary alcohol
done
clear
D)
phenol
done
clear
View Answer play_arrow
question_answer 78) Which of the following is the strongest acid?
A)
\[10\Omega \]
done
clear
B)
\[15\Omega \]
done
clear
C)
\[5\Omega \]
done
clear
D)
\[7.5\Omega \]
done
clear
View Answer play_arrow
question_answer 79) When n-pentane is isomerised to \[5896\overset{\text{o}}{\mathop{\text{A}}}\,\] using \[_{21}S{{c}^{41}}\] catalyst at 575 K, traces of olefine formed is?
A)
\[_{21}S{{c}^{42}}\]
done
clear
B)
\[_{20}S{{c}^{42}}\]
done
clear
C)
\[_{21}C{{a}^{42}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 80) Acylation of benzene to produce aliphatic aromatic ketones is called :
A)
Benzoin condensation
done
clear
B)
Friedal Crafts reaction
done
clear
C)
Hydroformylation
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 81) Sodium salt of formic acid on strong heating followed by acid hydrolysis produces:
A)
\[C{{s}^{+}}\]
done
clear
B)
\[{{K}^{+}}\]
done
clear
C)
\[N{{a}^{+}}\]
done
clear
D)
\[L{{i}^{+}}\]
done
clear
View Answer play_arrow
question_answer 82) R COOH after treatment with PCIs and KCN is subjected to hydrolysis followed by Clemmensons reduction, product obtained is:
A)
\[18.600\text{ }MeV\]
done
clear
B)
\[18.600\text{ }MeV\]
done
clear
C)
\[16.84\text{ }MeV\]
done
clear
D)
\[16.84\text{ }MeV\]
done
clear
View Answer play_arrow
question_answer 83) Formaldehyde polymerises from 6 to 50 molecules to form:
A)
meta formaldehyde
done
clear
B)
para formaldehyde
done
clear
C)
formalin
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 84) \[17.34\text{ }MeV\] on treatment with \[17.34\text{ }MeV\] produces:
A)
\[11.24\text{ }MeV\]
done
clear
B)
\[11.24\text{ }MeV\]
done
clear
C)
\[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\]
done
clear
D)
\[1{{s}^{2}},2{{s}^{1}}\]
done
clear
View Answer play_arrow
question_answer 85) Photochemical chlorination of alkane is initiated by the process of:
A)
pyrolysis
done
clear
B)
substitution
done
clear
C)
homolysis
done
clear
D)
peroxidation
done
clear
View Answer play_arrow
question_answer 86) Which of the following is the strongest nucleophile ?
A)
\[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\]
done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}\]
done
clear
C)
\[S{{F}_{6}}\]
done
clear
D)
\[_{20}C{{a}^{42}}\]
done
clear
View Answer play_arrow
question_answer 87) Atomic weight of radium is 226 and its half life period is 1600 years. The number of disintegration produced per second from 1 g are:
A)
\[\beta \]
done
clear
B)
\[10\Omega \]
done
clear
C)
\[15\Omega \]
done
clear
D)
\[5\Omega \]
done
clear
View Answer play_arrow
question_answer 88) If in the reaction \[7.5\Omega \]the degree of dissociation of N204 is a, then total number of molecules present at equilibrium will be:
A)
1
done
clear
B)
\[5896\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[_{21}S{{c}^{41}}\]
done
clear
D)
\[_{21}S{{c}^{42}}\]
done
clear
View Answer play_arrow
question_answer 89) If 342 g of cane sugar \[_{20}S{{c}^{42}}\]are dissolved in 1000 g of water, the solution will freeze at (\[_{21}C{{a}^{42}}\] for water \[C{{s}^{+}}\]):
A)
\[{{K}^{+}}\]
done
clear
B)
\[N{{a}^{+}}\]
done
clear
C)
\[L{{i}^{+}}\]
done
clear
D)
\[18.600\text{ }MeV\]
done
clear
View Answer play_arrow
question_answer 90) The electrode potential of \[16.84\text{ }MeV\] electrode in which the concentration of \[17.34\text{ }MeV\] is 0.01 M will be, given that \[11.24\text{ }MeV\]:
A)
\[{{27}^{o}}C\]
done
clear
B)
\[{{27}^{o}}C\]
done
clear
C)
\[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\]
done
clear
D)
\[1{{s}^{2}},2{{s}^{1}}\]
done
clear
View Answer play_arrow
question_answer 91) For which electrolyte \[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\] does not hold good:
A)
\[1{{s}^{2}},2{{s}^{2}}\]
done
clear
B)
\[S{{F}_{6}}\]
done
clear
C)
\[_{20}C{{a}^{42}}\]
done
clear
D)
\[\beta \]
done
clear
View Answer play_arrow
question_answer 92) During electrolysis of an aqueous solution of \[10\Omega \] sulphate 0.635 g of Cu was deposited at cathode. The amount of electricity consumed is:
A)
9650 coulomb
done
clear
B)
3860 coulomb
done
clear
C)
4825 coulomb
done
clear
D)
1930 coulomb
done
clear
View Answer play_arrow
question_answer 93) If the \[15\Omega \] ion concentration of a given solution is \[5\Omega \], the pH of the solution is:
A)
\[7.5\Omega \]
done
clear
B)
\[5896\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[_{21}S{{c}^{41}}\]
done
clear
D)
\[_{21}S{{c}^{42}}\]
done
clear
View Answer play_arrow
question_answer 94) Given that, \[_{20}S{{c}^{42}}\] ...(i) and \[_{21}C{{a}^{42}}\]...(ii) which of the following relation is correct?
A)
\[C{{s}^{+}}\]
done
clear
B)
\[{{K}^{+}}\]
done
clear
C)
\[N{{a}^{+}}\]
done
clear
D)
\[L{{i}^{+}}\]
done
clear
View Answer play_arrow
question_answer 95) Given that \[18.600\text{ }MeV\] ??.(1) \[16.84\text{ }MeV\] ??(2) the heat of sublimation of A will be
A)
x or y
done
clear
B)
\[17.34\text{ }MeV\]
done
clear
C)
\[11.24\text{ }MeV\]
done
clear
D)
\[{{27}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 96) The ratio of the value of any colligative property for \[342\text{ }K\] solution to that for sugar solution is nearly:
A)
1
done
clear
B)
0.5
done
clear
C)
2.0
done
clear
D)
2.5
done
clear
View Answer play_arrow
question_answer 97) For the reaction, \[342\text{ }K\] \[400\text{ }K\] An increase in pressure, favours
A)
backward reaction
done
clear
B)
forward reaction
done
clear
C)
no effect
done
clear
D)
all
done
clear
View Answer play_arrow
question_answer 98) The vapour density of a gas A is two times that of the vapour density of gas B If the molecular weight of gas B is M, then the molecular weight of gas A will be:
A)
M
done
clear
B)
\[400\text{ }K\]
done
clear
C)
\[440K\]
done
clear
D)
\[440K\]
done
clear
View Answer play_arrow
question_answer 99) 10 g of a gas at NTP occupies a volume of 2 litre. If the pressure and the amount of gas remain the same, then the temperature at which the volume doubles will be:
A)
\[520\text{ }K\]
done
clear
B)
\[520\text{ }K\]
done
clear
C)
\[{{H}_{2}}{{O}_{2}}\]
done
clear
D)
\[{{H}_{2}}{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 100) Given that, \[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\] ?.(i) \[1{{s}^{2}},2{{s}^{1}}\] ?..(ii) Heat of evaporation AH\[1{{s}^{2}},2{{s}^{2}},2{{p}^{3}}\] ° of \[1{{s}^{2}},2{{s}^{2}}\] will be:
A)
\[S{{F}_{6}}\]
done
clear
B)
\[_{20}C{{a}^{42}}\]
done
clear
C)
\[\beta \]
done
clear
D)
\[10\Omega \]
done
clear
View Answer play_arrow
question_answer 101) Green plants are :
A)
autotrophs
done
clear
B)
heterotrophs
done
clear
C)
chemotrophs
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 102) The protein part of enzyme is called :
A)
holoenzyme
done
clear
B)
cofactor
done
clear
C)
apoenzyme
done
clear
D)
isozyme
done
clear
View Answer play_arrow
question_answer 103) Lomasomes are found in:
A)
algal cell
done
clear
B)
fungal cell
done
clear
C)
yeast
done
clear
D)
E. coli
done
clear
View Answer play_arrow
question_answer 104) Genetically active area of chromosome is called:
A)
euchromatin
done
clear
B)
heterochromatin
done
clear
C)
heptan
done
clear
D)
cistron
done
clear
View Answer play_arrow
question_answer 105) Hurlers disease is caused by:
A)
presence of lysosome
done
clear
B)
absence of lysosome
done
clear
C)
presence of tryosinogeh
done
clear
D)
absence tryosinogen
done
clear
View Answer play_arrow
question_answer 106) Vacuolar membrane is called :
A)
plasmalemma
done
clear
B)
cell membrane
done
clear
C)
tonoplast
done
clear
D)
symplast
done
clear
View Answer play_arrow
question_answer 107) DNA is a ..... chains :
A)
nucleotide
done
clear
B)
nucleoside
done
clear
C)
polynucleoside
done
clear
D)
polynucleotide
done
clear
View Answer play_arrow
question_answer 108) Protoplasm is a physical basis of life. This statement belongs to :
A)
Robert Hooke
done
clear
B)
Robert Brown
done
clear
C)
Huxley
done
clear
D)
Leeuwenhoek
done
clear
View Answer play_arrow
question_answer 109) \[\alpha \]-helical model of protein was discovered by:
A)
Pauling and Corey
done
clear
B)
Watson
done
clear
C)
Morgan
done
clear
D)
Berzelus
done
clear
View Answer play_arrow
question_answer 110) Cell wall is absent in :
A)
gametes
done
clear
B)
Amoeba
done
clear
C)
Mycoplasma
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 111) KNA contains:
A)
hexose sugar
done
clear
B)
deoxyribose sugar
done
clear
C)
dextrose sugar
done
clear
D)
ribose sugar
done
clear
View Answer play_arrow
question_answer 112) Which of the following can photosynthesize at low temperature?
A)
Bacteria
done
clear
B)
Lichen
done
clear
C)
Yeast
done
clear
D)
Batrachospemium
done
clear
View Answer play_arrow
question_answer 113) Ctilvin cycle takes place in :
A)
cytoplasm
done
clear
B)
chloroplast
done
clear
C)
mitochondria
done
clear
D)
peroxisomes
done
clear
View Answer play_arrow
question_answer 114) Krebs cycle is also called as :
A)
Tricarboxylic acid cycle
done
clear
B)
Oxidative phosphorylation
done
clear
C)
EMP pathway
done
clear
D)
Glycolysis
done
clear
View Answer play_arrow
question_answer 115) Lichens are very sensitive to:
A)
\[S{{O}_{2}}\] pollution
done
clear
B)
noise pollution
done
clear
C)
soil pollution
done
clear
D)
water pollution
done
clear
View Answer play_arrow
question_answer 116) Organism which can respire in absence of\[{{O}_{2}}\] is :
A)
Chlorella
done
clear
B)
Solanum
done
clear
C)
Sacdiamm
done
clear
D)
Saccharomyces
done
clear
View Answer play_arrow
question_answer 117) Citrus canker is a:
A)
fungal disease
done
clear
B)
algal disease
done
clear
C)
viral disease
done
clear
D)
bacterial disease
done
clear
View Answer play_arrow
question_answer 118) In bryophytes, which part of archegonium encloses egg:
A)
neck
done
clear
B)
cover cell
done
clear
C)
center
done
clear
D)
neck canal cells
done
clear
View Answer play_arrow
question_answer 119) How many hydrogen bonds are present between guanine and cytosine?
A)
Four
done
clear
B)
Three
done
clear
C)
Two
done
clear
D)
One
done
clear
View Answer play_arrow
question_answer 120) Ratio of the cross between f] hybrid and recessive parent will be :
A)
3:1
done
clear
B)
1:1
done
clear
C)
2:1
done
clear
D)
4:1
done
clear
View Answer play_arrow
question_answer 121) Interferon suppresses the pathogenic activity of:
A)
virus
done
clear
B)
bacteria
done
clear
C)
Mycoplasma
done
clear
D)
Candidas
done
clear
View Answer play_arrow
question_answer 122) Protonema is :
A)
juvenile phase of moss gametophyte
done
clear
B)
a part of Fumaria in sporophyte
done
clear
C)
fossil plant
done
clear
D)
young alga
done
clear
View Answer play_arrow
question_answer 123) The first stable compound of dark reaction of photosynthesis is : .
A)
phosphoglyceraldehyde
done
clear
B)
phosphoglyceric acid
done
clear
C)
fructose
done
clear
D)
starch
done
clear
View Answer play_arrow
question_answer 124) Anthocyanins are pigments which are :
A)
water soluble and located in cell sap
done
clear
B)
water insoluble and located in cell sap
done
clear
C)
water soluble and located in chromoplast
done
clear
D)
water soluble and located in leucoplast
done
clear
View Answer play_arrow
question_answer 125) RR-21 is a variety of :
A)
Rice
done
clear
B)
Sugarcane
done
clear
C)
Potato
done
clear
D)
Wheat
done
clear
View Answer play_arrow
question_answer 126) Genetically engineered human insulin is prepared by using:
A)
E. coli
done
clear
B)
Rhizopus
done
clear
C)
Pseudomonas
done
clear
D)
Yeast
done
clear
View Answer play_arrow
question_answer 127) Which RNA carries information from DNA in protein synthesis ?
A)
s-RNA
done
clear
B)
f-RNA
done
clear
C)
r-RNA
done
clear
D)
m-RNA
done
clear
View Answer play_arrow
question_answer 128) Peanut oil is obtained from :
A)
Glycine max
done
clear
B)
Arachis hypogeal
done
clear
C)
Pisum sativum
done
clear
D)
Lathyrus odoratus
done
clear
View Answer play_arrow
question_answer 129) Acacia nrnbica is a :
A)
hydrophyte
done
clear
B)
mesophyte
done
clear
C)
halophyte
done
clear
D)
xerophytes
done
clear
View Answer play_arrow
question_answer 130) Fruitless flowering plants are :
A)
angiosperms
done
clear
B)
gymnosperms
done
clear
C)
sterile plants
done
clear
D)
primitive plants
done
clear
View Answer play_arrow
question_answer 131) Best soil for healthy and vigorous growth of a plant is
A)
sandy soil
done
clear
B)
loam
done
clear
C)
clay
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 132) When plant cells are kept in a concentrated salt solution, they are :
A)
deplasmolysed
done
clear
B)
plasmolysed
done
clear
C)
Remains as such
done
clear
D)
becomes turgid
done
clear
View Answer play_arrow
question_answer 133) Heterospory and seed habit are exhibited by a plant possessing :
A)
petiole
done
clear
B)
ligule
done
clear
C)
bract
done
clear
D)
spathe
done
clear
View Answer play_arrow
question_answer 134) Aswagandha is called as :
A)
Raiiwolfin serpentine
done
clear
B)
Withania somnifera
done
clear
C)
Datura stramonium
done
clear
D)
Cannabis sativa
done
clear
View Answer play_arrow
question_answer 135) Bordered pits are found in :
A)
gymnosperms
done
clear
B)
bryophytes
done
clear
C)
monocots
done
clear
D)
Hydrilla
done
clear
View Answer play_arrow
question_answer 136) Late blight of Potato is caused by :
A)
Ustilago
done
clear
B)
Altermaria solani
done
clear
C)
Phytophthora infestans
done
clear
D)
Helminthosporium
done
clear
View Answer play_arrow
question_answer 137) Cuscuta is a :
A)
mangrove plant
done
clear
B)
saprophytic plant
done
clear
C)
epiphytic plant
done
clear
D)
parasitic plant
done
clear
View Answer play_arrow
question_answer 138) Penicillium belongs to
A)
Phycomycetes
done
clear
B)
Ascomycetes
done
clear
C)
Basidiomycetes
done
clear
D)
none of the these
done
clear
View Answer play_arrow
question_answer 139) Bacteriophage transfers DNA from donor to recipient cell, this is called :
A)
translation
done
clear
B)
transduction
done
clear
C)
conjugation
done
clear
D)
transcription
done
clear
View Answer play_arrow
question_answer 140) Velamen tissue is found in :
A)
stem of orchids
done
clear
B)
aerial roots of orchids
done
clear
C)
clinging roots of orchd
done
clear
D)
pneumatophorcs
done
clear
View Answer play_arrow
question_answer 141) In angiosperms, latcml roots arise from -
A)
endodermis
done
clear
B)
cortex
done
clear
C)
pericycle
done
clear
D)
epiblcmma
done
clear
View Answer play_arrow
question_answer 142) Bulliform celts are found in upper epidermis of:
A)
cladodes
done
clear
B)
phyllocttide
done
clear
C)
monocot leaf
done
clear
D)
dicot leaf
done
clear
View Answer play_arrow
question_answer 143) Laminarm and Fucus arc examples of:
A)
red algae
done
clear
B)
green algae
done
clear
C)
blue-green algae
done
clear
D)
brown algae
done
clear
View Answer play_arrow
question_answer 144) Hexoses and pentoses are :
A)
mono saccharides
done
clear
B)
disaccharides
done
clear
C)
polysaccharides
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 145) Tectona grandis belongs to family :
A)
Verbenaceae
done
clear
B)
Anacardiaceae
done
clear
C)
Rubiaceae
done
clear
D)
Labiatae
done
clear
View Answer play_arrow
question_answer 146) Radial vascular bundles are found in :
A)
monocot stem
done
clear
B)
dicot stem
done
clear
C)
root
done
clear
D)
leaf
done
clear
View Answer play_arrow
question_answer 147) The term vascular cryptogames is used for:
A)
pteridophyta
done
clear
B)
bryophyte
done
clear
C)
angiosperm
done
clear
D)
gymnosperm
done
clear
View Answer play_arrow
question_answer 148) National flower of India is:
A)
Lotus
done
clear
B)
Lily
done
clear
C)
Rose
done
clear
D)
Sunflower
done
clear
View Answer play_arrow
question_answer 149) Companion cells are absent is phloem of:
A)
angiosperm
done
clear
B)
gymnosperm
done
clear
C)
algae
done
clear
D)
lichen
done
clear
View Answer play_arrow
question_answer 150) Fungi can be stained with :
A)
fast green
done
clear
B)
cotton blue
done
clear
C)
Grams stain
done
clear
D)
PAS
done
clear
View Answer play_arrow
question_answer 151) The joint, radius-ulna with arm is :
A)
hinge
done
clear
B)
ball and socket
done
clear
C)
pivot
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 152) The casein is a :
A)
milk protein
done
clear
B)
blood protein
done
clear
C)
wheat protein
done
clear
D)
fish protein
done
clear
View Answer play_arrow
question_answer 153) When \[{{O}_{2}}\]is inadequate during respiration. The condition is called:
A)
anoxia
done
clear
B)
piaunsy
done
clear
C)
asphyxia
done
clear
D)
hypoxia
done
clear
View Answer play_arrow
question_answer 154) Heamocyanin is found in :
A)
Nereis
done
clear
B)
Leech
done
clear
C)
Earthworm
done
clear
D)
Cockroach
done
clear
View Answer play_arrow
question_answer 155) Which hormone promotes cell division, bone growth and protein synthesis?
A)
GH
done
clear
B)
ADH
done
clear
C)
DTH
done
clear
D)
ACTH
done
clear
View Answer play_arrow
question_answer 156) Territorially occurs as result of:
A)
co-operation
done
clear
B)
competition
done
clear
C)
parasitism
done
clear
D)
predation
done
clear
View Answer play_arrow
question_answer 157) Sympathetic nerves in mammal arise from:
A)
3rd, 7th, 9th, 11th cranial nerves
done
clear
B)
cervical region
done
clear
C)
thoraco-lumber region
done
clear
D)
sacral region
done
clear
View Answer play_arrow
question_answer 158) Reserpine is an alkaloid obtained from:
A)
Aswagandha
done
clear
B)
Serpgandha
done
clear
C)
Opium
done
clear
D)
Cannabis
done
clear
View Answer play_arrow
question_answer 159) Trypsin acts on :
A)
lipid
done
clear
B)
protein
done
clear
C)
carbohydrates
done
clear
D)
bile
done
clear
View Answer play_arrow
question_answer 160) Gauchers disease is related with abnormal:
A)
glucose metabolism
done
clear
B)
fat metabolism
done
clear
C)
protein metabolism
done
clear
D)
carbohydrate metabolism
done
clear
View Answer play_arrow
question_answer 161) Split genes are made up of exons and , introns. The coding sequences arc called as:
A)
exons
done
clear
B)
introns
done
clear
C)
cistrons
done
clear
D)
mutans
done
clear
View Answer play_arrow
question_answer 162) Wobble hypothesis was given by :
A)
Holley
done
clear
B)
Khorana
done
clear
C)
Crick
done
clear
D)
Nirenberg
done
clear
View Answer play_arrow
question_answer 163) Drosophila melanogastor has 8 chromosomes in somatic cell. How many linkage groups will be there ?
A)
4
done
clear
B)
8
done
clear
C)
2
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 164) Binary fission is found in :
A)
Amoeba
done
clear
B)
Paramedum
done
clear
C)
Planaria
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 165) Thyroxine is:
A)
an enzyme
done
clear
B)
a hormone
done
clear
C)
n vitamin
done
clear
D)
a by product
done
clear
View Answer play_arrow
question_answer 166) Which is absent in plants?
A)
Cell wall
done
clear
B)
Vascular system
done
clear
C)
Nervous system
done
clear
D)
Epidermal tissue system
done
clear
View Answer play_arrow
question_answer 167) The metal constituent of haemoglobin is :
A)
iron
done
clear
B)
copper
done
clear
C)
cobalt
done
clear
D)
magnesium
done
clear
View Answer play_arrow
question_answer 168) Book lungs are respiratory organ of:
A)
Scorpion
done
clear
B)
Birds
done
clear
C)
Fishes
done
clear
D)
Sea bird
done
clear
View Answer play_arrow
question_answer 169) The presence of feathers and power of flight are characteristic feature of:
A)
Aves
done
clear
B)
Reptilia
done
clear
C)
Mammals
done
clear
D)
Amphibians
done
clear
View Answer play_arrow
question_answer 170) The important character of Cobra is:
A)
presence of hood
done
clear
B)
small scales on head
done
clear
C)
rounded tail
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 171) Ptyalin acts at pH :
A)
3.4
done
clear
B)
6.8
done
clear
C)
7.8
done
clear
D)
9.0
done
clear
View Answer play_arrow
question_answer 172) The vital capacity of lung is equal to :
A)
4000 ml
done
clear
B)
2200 ml
done
clear
C)
1100 ml
done
clear
D)
500 ml
done
clear
View Answer play_arrow
question_answer 173) The golden age of Dinosaurs was in which of the following era?
A)
Mesozoic
done
clear
B)
Coenozoic
done
clear
C)
Palaeozoic
done
clear
D)
Archaeozoic
done
clear
View Answer play_arrow
question_answer 174) Balbiani rings are present in:
A)
Polysomes
done
clear
B)
Autosomes
done
clear
C)
polytene chromosomes
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 175) Which is a gaseoues hormone ?
A)
GA
done
clear
B)
ABA
done
clear
C)
Ethylene
done
clear
D)
Kn
done
clear
View Answer play_arrow
question_answer 176) Diabetes is due to :
A)
enzyme deficiency
done
clear
B)
hormonal deficiency
done
clear
C)
iodine deficiency
done
clear
D)
fat deficiency
done
clear
View Answer play_arrow
question_answer 177) In Earthworms, self-fertilization does not occur due to:
A)
hypogyny
done
clear
B)
protogyny
done
clear
C)
protandry
done
clear
D)
epigyny
done
clear
View Answer play_arrow
question_answer 178) Phagocytosis was seen by:
A)
Matchnikoff
done
clear
B)
Strassburger
done
clear
C)
Huxley
done
clear
D)
Haekel
done
clear
View Answer play_arrow
question_answer 179) Pace-maker is:
A)
SA-node
done
clear
B)
AV-node
done
clear
C)
SV-node
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 180) The great Indian bustard is an endangered species due to:
A)
hunting
done
clear
B)
climate change
done
clear
C)
spreading desert
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 181) Which of the following are multicellular grade organisms?
A)
Sponges
done
clear
B)
Coelanterates
done
clear
C)
Prokaryotes
done
clear
D)
Monerans
done
clear
View Answer play_arrow
question_answer 182) Two sister chromatids are attached with:
A)
spindle fibre
done
clear
B)
centromere
done
clear
C)
chromocentre
done
clear
D)
chromatid
done
clear
View Answer play_arrow
question_answer 183) Which enzyme helps in digestion of milk?
A)
Rennin
done
clear
B)
In vertase
done
clear
C)
Amylase
done
clear
D)
Maltase
done
clear
View Answer play_arrow
question_answer 184) Birds lack:
A)
fore limb
done
clear
B)
hind limb
done
clear
C)
pelvic girdle
done
clear
D)
pectoral girdle
done
clear
View Answer play_arrow
question_answer 185) Pyramid of energy is :
A)
upright
done
clear
B)
inverted
done
clear
C)
oblique
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 186) Which of the following organelle is rich in hydrolysing enzymes ?
A)
Lysosomes
done
clear
B)
Mitochondria
done
clear
C)
Golgi bodies
done
clear
D)
ER
done
clear
View Answer play_arrow
question_answer 187) Regeneration of lost tail of Lizard in an example of:
A)
epimorphosis
done
clear
B)
morpholaxis
done
clear
C)
autolysis
done
clear
D)
restructury
done
clear
View Answer play_arrow
question_answer 188) Sporogony of malarial parasite occurs in:
A)
liver of human being
done
clear
B)
RBCs of human beings
done
clear
C)
stomach wall of mosquite
done
clear
D)
salivary gland of mosquite
done
clear
View Answer play_arrow
question_answer 189) Amoebiasis is caused by :
A)
Entaniovba liisioSytica
done
clear
B)
Taenia solium
done
clear
C)
Ascariy
done
clear
D)
Phsmodium
done
clear
View Answer play_arrow
question_answer 190) The branch of science which deals with the process of improvement of human race by selective breeding is called :
A)
eugenics
done
clear
B)
euphenics
done
clear
C)
euthenics
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 191) Cobalt is a constituent of vitamin :
A)
\[{{B}_{6}}\]
done
clear
B)
\[{{B}_{12}}\]
done
clear
C)
\[{{B}_{1}}\]
done
clear
D)
\[{{B}_{2}}\]
done
clear
View Answer play_arrow
question_answer 192) Blood cells which show phagocytosis is :
A)
platelet
done
clear
B)
eosinophil
done
clear
C)
basophil
done
clear
D)
monocyte
done
clear
View Answer play_arrow
question_answer 193) Protein which shows structural and enzymatic activity :
A)
trypsin
done
clear
B)
actin
done
clear
C)
myosin
done
clear
D)
troponin
done
clear
View Answer play_arrow
question_answer 194) The largest muscle in human body is :
A)
masseter
done
clear
B)
Sartorius
done
clear
C)
stapeduis
done
clear
D)
gluteus
done
clear
View Answer play_arrow
question_answer 195) Surrogate mother is used for :
A)
induction of lactation
done
clear
B)
artificially inseminated female
done
clear
C)
future mother with transplanted embryo
done
clear
D)
artificial in semination
done
clear
View Answer play_arrow
question_answer 196) Incipient nucleus is present in :
A)
Chlorophyceae
done
clear
B)
Rhodophyceae
done
clear
C)
Myxophyceae
done
clear
D)
Phaeophyceae
done
clear
View Answer play_arrow
question_answer 197) Reason of death of Cobra bite person is :
A)
inactivation of nerves
done
clear
B)
contraction of muscle
done
clear
C)
destruction of RBCs
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 198) Spongin fibres are secreted by :
A)
Choanocytes
done
clear
B)
Pinacocytes
done
clear
C)
Amoebocytes
done
clear
D)
Spongioblasts
done
clear
View Answer play_arrow
question_answer 199) Animals which live at the bottom of sea are:
A)
nekton
done
clear
B)
diatom
done
clear
C)
banthos
done
clear
D)
phnkton
done
clear
View Answer play_arrow
question_answer 200) Who reported that Downs syndrome is due to extra 21st chromosome?
A)
J.L Down (1866)
done
clear
B)
Lejeune (1959)
done
clear
C)
Klinefeltcr (1942)
done
clear
D)
Huntington (1872)
done
clear
View Answer play_arrow