question_answer 1) Two waves represented by: \[{{y}_{1}}=a\,\sin \left( \omega t+\frac{\pi }{3} \right)\] and \[{{y}_{2}}=a\,\sin \,\omega t\]superimpose on each other, then resultant amplitude will be:
A)
a
done
clear
B)
\[\sqrt{2}\,a\]
done
clear
C)
\[2\,a\]
done
clear
D)
\[\sqrt{3}\,a\]
done
clear
View Answer play_arrow
question_answer 2) A sound source producing waves of frequency 300 Hz and wavelength 1 m. Observer is stationary, while source is going away with the velocity 30 m/s, then apparent frequency heard by the observer is:
A)
270 Hz
done
clear
B)
273 Hz
done
clear
C)
383 Hz
done
clear
D)
300 Hz
done
clear
View Answer play_arrow
question_answer 3) The phase difference between incident wave and reflected wave is \[180{}^\circ \] when light ray:
A)
enters into glass from air
done
clear
B)
enters into air from glass
done
clear
C)
enters into glass from diamond
done
clear
D)
enters into water from glass
done
clear
View Answer play_arrow
question_answer 4) Which of the following phenomena can explain quantum nature of light?
A)
Photoelectric effect
done
clear
B)
Interference
done
clear
C)
Diffraction
done
clear
D)
Polarization
done
clear
View Answer play_arrow
question_answer 5) Image formed on retina of eye is proportional to:
A)
size of object
done
clear
B)
area of object
done
clear
C)
\[\frac{\text{size}\,\text{of}\,\text{object}}{\text{size}\,\text{of}\,\text{image}}\]
done
clear
D)
\[\frac{\text{size}\,\text{of}\,\text{image}}{\text{size}\,\text{of}\,\text{object}}\]
done
clear
View Answer play_arrow
question_answer 6) The phenomena of total internal reflection is seen when angle of incidence is :
A)
\[90{}^\circ \]
done
clear
B)
greater than critical angle
done
clear
C)
equal to critical angle
done
clear
D)
\[0{}^\circ \]
done
clear
View Answer play_arrow
question_answer 7) If applied potential is V the maximum frequency of X-rays will be :
A)
\[\frac{eV}{h}\]
done
clear
B)
\[\frac{h}{eV}\]
done
clear
C)
\[\frac{Vh}{e}\]
done
clear
D)
\[\frac{he}{V}\]
done
clear
View Answer play_arrow
question_answer 8) \[2{{n}^{2}}\] electrons are filled in any orbit of an atom, then for n = 2, the number of electrons will be:
A)
4
done
clear
B)
8
done
clear
C)
16
done
clear
D)
32
done
clear
View Answer play_arrow
question_answer 9) For a compound microscope, the focal lengths of object lens and eye lens are\[{{f}_{0}}\] and \[{{f}_{e}}\] respectively, then magnification will be done by microscope when :
A)
\[{{f}_{0}}={{f}_{e}}\]
done
clear
B)
\[{{f}_{0}}>{{f}_{e}}\]
done
clear
C)
\[{{f}_{0}}<{{f}_{e}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 10) In vacuum an electron of energy 10 keV hits tungeston target, then emitted radiation will be:
A)
cathode rays
done
clear
B)
X-rays
done
clear
C)
infrared rays
done
clear
D)
visible spectrum
done
clear
View Answer play_arrow
question_answer 11) Which of the following statement is true about de-Broglie wavelength of moving ball in cricket:
A)
de Brogliess theory does not apply on such a big body
done
clear
B)
\[\lambda =\frac{h}{\sqrt{2mE}}\]
done
clear
C)
\[\lambda =E\sqrt{\frac{h}{2m}}\]
done
clear
D)
\[\lambda =\frac{g}{2mE}\]
done
clear
View Answer play_arrow
question_answer 12) If n = 7, then angular momentum of electron will be:
A)
\[7h\]
done
clear
B)
\[42h\]
done
clear
C)
\[\sqrt{7}h\]
done
clear
D)
\[\sqrt{56}h\]
done
clear
View Answer play_arrow
question_answer 13) An electron is moving with velocity\[\upsilon \]around a nucleus in eliptically orbit, then moment of inertia of electron with respect to nucleus is:
A)
always zero
done
clear
B)
always constant
done
clear
C)
changes with tune
done
clear
D)
cant find
done
clear
View Answer play_arrow
question_answer 14) Two conducting coils are placed on the same axis, if we connect a battery with one of the coil, then :
A)
they attract each other
done
clear
B)
repel each other
done
clear
C)
attract and repel each other
done
clear
D)
both will experience no force
done
clear
View Answer play_arrow
question_answer 15) A particle moves towards east for 2 sec with velocity 15 m/s and move towards north for 8 sec with velocity 5 m/s. Then average velocity of the particle is:
A)
1 m/s
done
clear
B)
5 m/s
done
clear
C)
7 m/s
done
clear
D)
10 m/s
done
clear
View Answer play_arrow
question_answer 16) Relation between wavelength of photon and electron of same energy is :
A)
\[{{\lambda }_{ph}}>{{\lambda }_{e}}\]
done
clear
B)
\[{{\lambda }_{ph}}<{{\lambda }_{e}}\]
done
clear
C)
\[{{\lambda }_{ph}}={{\lambda }_{e}}\]
done
clear
D)
\[\frac{{{\lambda }_{e}}}{{{\lambda }_{ph}}}=\]constant
done
clear
View Answer play_arrow
question_answer 17) Work done in rotating a magnet of magnetic moment M with angle \[\] in external magnetic field H is :
A)
\[\text{MH}\,\text{cos}\,\]
done
clear
B)
\[\text{2MH}\,{{\sin }^{2}}\frac{\theta }{2}\]
done
clear
C)
\[\text{MH}\,\sin \]
done
clear
D)
\[\text{MH}\,(1-\cos \text{)}\]
done
clear
View Answer play_arrow
question_answer 18)
An electron is passing near a ring, then direction of induced current in ring is :
A)
clockwise
done
clear
B)
anticlockwise
done
clear
C)
both a and b
done
clear
D)
no current
done
clear
View Answer play_arrow
question_answer 19) Power of combination of two lens of focal lengths 20 cm and 25 cm respectively:
A)
+1D
done
clear
B)
+9D
done
clear
C)
-1D
done
clear
D)
-9D
done
clear
View Answer play_arrow
question_answer 20) A zone plate of focal length 60 cm behaves like convex lens, if wavelength of incidence light is \[6000\,\overset{\text{o}}{\mathop{\text{A}}}\,,\] then radius of first half period zone will be:
A)
\[36\times {{10}^{-8}}\,m\]
done
clear
B)
\[6\times {{10}^{-8}}\,m\]
done
clear
C)
\[\sqrt{6}\times {{10}^{-8}}\,m\]
done
clear
D)
\[6\times {{10}^{-4}}\,m\]
done
clear
View Answer play_arrow
question_answer 21) Match the following: Angular momentum (1) \[\text{ }\!\![\!\!\text{ }{{\text{M}}^{\text{-1}}}{{\text{L}}^{\text{2}}}{{\text{T}}^{\text{-2}}}\text{ }\!\!]\!\!\text{ }\] Torque (2) \[\text{ }\!\![\!\!\text{ }{{\text{M}}^{\text{-1}}}{{\text{T}}^{\text{-2}}}\text{ }\!\!]\!\!\text{ }\] Gravitational constant (3) \[\text{ }\!\![\!\!\text{ }{{\text{M}}^{\text{1}}}{{\text{L}}^{2}}{{\text{T}}^{\text{-2}}}\text{ }\!\!]\!\!\text{ }\] Tension (4) \[\text{ }\!\![\!\!\text{ }{{\text{M}}^{\text{1}}}{{\text{L}}^{2}}{{\text{T}}^{\text{-1}}}\text{ }\!\!]\!\!\text{ }\]
A)
\[c-2,d-1\]
done
clear
B)
\[a-4,b-3\]
done
clear
C)
\[a-3,c-2\]
done
clear
D)
\[b-2,a-1\]
done
clear
View Answer play_arrow
question_answer 22) If we increase kinetic energy of a body 300%, the per cent increase in its momentum is:
A)
50 %
done
clear
B)
300 %
done
clear
C)
100 %
done
clear
D)
150 %
done
clear
View Answer play_arrow
question_answer 23) Two mass of 9 g arid 6 g are tied to a string, which passes over a smooth pulley. Then common acceleration will be:
A)
\[1.96\text{ }cm/{{s}^{2}}\]
done
clear
B)
\[\text{48 }cm/{{s}^{2}}\]
done
clear
C)
\[\text{98 }cm/{{s}^{2}}\]
done
clear
D)
\[\text{196 }cm/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 24) Change in acceleration duo to gravity is same upto a height\[h\]from earth surface and below depth \[x\] then relation between \[x\] and \[h\] is \[(h\,\text{and}\,x\,<<<\,\operatorname{Re}):\]
A)
\[x=h\]
done
clear
B)
\[x=2h\]
done
clear
C)
\[x=\frac{h}{2}\]
done
clear
D)
\[x={{h}^{2}}\]
done
clear
View Answer play_arrow
question_answer 25) If a sphere is inserted in water, then it flows with \[\frac{1}{3}\text{rd}\] of it outside the water. When it is inserted in an unknown liquid then it flows with \[\frac{3}{4}\text{th}\] of it outside, then density of unknown liquid is:
A)
\[10.9\,gm\,/c.c.\]
done
clear
B)
\[\frac{9}{4}gm\,/c.c.\]
done
clear
C)
\[\frac{8}{3}gm\,/c.c.\]
done
clear
D)
\[\frac{3}{8}gm\,/c.c.\]
done
clear
View Answer play_arrow
question_answer 26) Reason for rising of kerosen oil in cloth in a lamp:
A)
viscosity of oil
done
clear
B)
less density of oil
done
clear
C)
capillary process of cloth
done
clear
D)
vaporisation of oil at low temperature
done
clear
View Answer play_arrow
question_answer 27) Escape velocity \[({{\upsilon }_{e}})\] of a body from a planet depends on its mass as:
A)
\[{{\upsilon }_{e}}\propto {{M}^{2}}\]
done
clear
B)
\[{{\upsilon }_{e}}\propto M\]
done
clear
C)
\[{{\upsilon }_{e}}\propto {{M}^{0}}\]
done
clear
D)
\[{{\upsilon }_{e}}\propto \frac{1}{M}\]
done
clear
View Answer play_arrow
question_answer 28) Which of the following quantity is conserved for a satellite revolving round the earth in a particular orbit?
A)
Angular velocity
done
clear
B)
Force
done
clear
C)
Linear momentum
done
clear
D)
Velocity
done
clear
View Answer play_arrow
question_answer 29) 200 MeV energy is obtained by fission of 1 nuclei of \[_{92}{{U}^{235}},\] to obtain 1 kW energy number of fission per second will be:
A)
\[3.125\times {{10}^{13}}\]
done
clear
B)
\[3.125\times {{10}^{14}}\]
done
clear
C)
\[3.125\times {{10}^{15}}\]
done
clear
D)
\[3.125\times {{10}^{16}}\]
done
clear
View Answer play_arrow
question_answer 30) Two bodies A and B are revolving round a planet. Radius of As orbit is 4R and that of Bs orbit is R, if speed of A is \[3\upsilon \] then speed b of B is:
A)
\[12\,\upsilon \]
done
clear
B)
\[6\,\upsilon \]
done
clear
C)
\[\frac{4}{3}\upsilon \]
done
clear
D)
\[\frac{3}{2}\upsilon \]
done
clear
View Answer play_arrow
question_answer 31) Temperature and pressure of 2g oxygen t are \[27{}^\circ C\] and 76 cm Hg, then volume of the gas is:
A)
1.53 litre
done
clear
B)
2.44 litre
done
clear
C)
3.08 litre
done
clear
D)
44.2 litre
done
clear
View Answer play_arrow
question_answer 32) In which of the following process internal energy rises?
A)
Adiabatic expansion
done
clear
B)
Adiabatic compression
done
clear
C)
Isothermal
done
clear
D)
Isothermal compression
done
clear
View Answer play_arrow
question_answer 33) True for isochoric process is:
A)
\[\Delta Q=\Delta U\]
done
clear
B)
\[\Delta W=\Delta U\]
done
clear
C)
\[\Delta Q=\Delta W\]
done
clear
D)
None
done
clear
View Answer play_arrow
question_answer 34) Significance of a and b in vander-Waals equation:
A)
\[a\] and \[b\] both show the correction in volume of gas
done
clear
B)
\[a\] and \[b\] both show cohesive force between molecules
done
clear
C)
a shows cohesive force while b shows correction in volume
done
clear
D)
\[a\] shows correction in volume while\[b\] shows cohesive force
done
clear
View Answer play_arrow
question_answer 35) If temperature of a body increases by 10%, then increase in radiated energy of the R body is:
A)
10%
done
clear
B)
40%
done
clear
C)
46%
done
clear
D)
1000%
done
clear
View Answer play_arrow
question_answer 36) In which condition a real gas behaves as an ideal gas?
A)
At high pressure
done
clear
B)
At low pressure
done
clear
C)
At low temperature
done
clear
D)
All the above
done
clear
View Answer play_arrow
question_answer 37) In a triode plate current is 2.5 mA and plate resistance 20 \[k\Omega ,\] amplification of triode is 10, the load resistance will be :
A)
5\[\text{k}\]
done
clear
B)
25\[\text{k}\]
done
clear
C)
20 \[\text{k}\]
done
clear
D)
50\[\text{k}\]
done
clear
View Answer play_arrow
question_answer 38) Volume expansion coefficient of a gas equal to:
A)
temperature
done
clear
B)
proportional to square root of temperature
done
clear
C)
inversely proportional to square root of temperature
done
clear
D)
inversely proportional to temperature
done
clear
View Answer play_arrow
question_answer 39) Minimum resonating length of a closed organ pipe is 50 cm, when it is vibrated by same frequency, then next resonating length will be:
A)
250 cm
done
clear
B)
200 cm
done
clear
C)
150 cm
done
clear
D)
100 cm
done
clear
View Answer play_arrow
question_answer 40) Velocity of a sound wave is 360 m/s and frequency is 50 Hz, then path difference will be at the phase difference of \[60{}^\circ \] is:
A)
10 cm
done
clear
B)
1.2 cm
done
clear
C)
15 cm
done
clear
D)
100 cm
done
clear
View Answer play_arrow
question_answer 41) Which of the following property is not shown by transversal wave?
A)
Reflection
done
clear
B)
Refraction
done
clear
C)
Diffraction
done
clear
D)
Polarization
done
clear
View Answer play_arrow
question_answer 42) A mass of 1 kg is suspended from a spring of force constant 400 N, executing S.H.M. total energy of the body is 2J, then maximum acceleration of the spring will be:
A)
4 m/s2
done
clear
B)
40 m/s2
done
clear
C)
200 m/s2
done
clear
D)
400 m/s2
done
clear
View Answer play_arrow
question_answer 43) A slide projector magnified a film of \[100c{{m}^{2}}\] on a screen. If linear magnification is 4, then area of the magnified image will be:
A)
\[1600c{{m}^{2}}\]
done
clear
B)
\[800c{{m}^{2}}\]
done
clear
C)
\[400c{{m}^{2}}\]
done
clear
D)
\[200c{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 44) Usually light of red colour is used to see the diffraction pattern by single slit. If green light is used in place of red light, then diffraction pattern:
A)
will become more bright
done
clear
B)
will become contract
done
clear
C)
will expand
done
clear
D)
will not be seen
done
clear
View Answer play_arrow
question_answer 45) On passing sodium light from sodium vapour, emergent light will be:
A)
line of \[5890\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
line of \[5896\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
both a and b
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 46) If in an excited state of hydrogen atom \[n=5,\]then value of \[n,\,l,\,m,\,s\] will be:
A)
\[5,\,5,\,5,\,-\frac{1}{2}\]
done
clear
B)
\[7,\,7,\,5,\,+\frac{1}{2}\]
done
clear
C)
\[6,\,6,\,5,\,-\frac{1}{2}\]
done
clear
D)
\[8,\,7,\,5,\,+\frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 47) Which of the following is true statement ?
A)
A good absorber is bad conductor
done
clear
B)
Each body emits and absorb radiation at each temperature
done
clear
C)
In a black body energy of emitted radiation is equal for all wave length
done
clear
D)
Plancks law gives the relation between maximum wavelength of black body radiation and its temperature
done
clear
View Answer play_arrow
question_answer 48) Resistance of a wire is 20 ohm, it is stretched upto, three times of its length, then its new resistance will be :
A)
\[6.67\,\Omega \]
done
clear
B)
\[60\,\Omega \]
done
clear
C)
\[120\,\Omega \]
done
clear
D)
\[180\,\Omega \]
done
clear
View Answer play_arrow
question_answer 49) Mass of hydrogen-atom is \[1.7\times {{10}^{-27}}\] kg and inter atomic distance is \[4\times {{10}^{-10}}\,m\]. Then moment of inertia of H2 molecules with respect to its centre of mass:
A)
\[7.6\,\times {{10}^{-43}}\,k{{g}^{-}}{{m}^{2}}\]
done
clear
B)
\[7.6\,\times {{10}^{-47}}\,k{{g}^{-}}{{m}^{2}}\]
done
clear
C)
\[6.7\,\times {{10}^{-47}}\,k{{g}^{-}}{{m}^{2}}\]
done
clear
D)
\[13.6\,\times {{10}^{-47}}\,k{{g}^{-}}{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 50) Two capacitors of capacity \[{{C}_{1}}\] and \[{{C}_{2}}\] are charged upto a potential \[{{V}_{1}}\] and \[{{V}_{2}}\], then condition for not flowing the charge between on connected them in parallel:
A)
\[{{C}_{1}}={{C}_{2}}\]
done
clear
B)
\[{{C}_{1}}{{V}_{1}}={{C}_{2}}{{V}_{2}}\]
done
clear
C)
\[{{V}_{1}}={{V}_{2}}\]
done
clear
D)
\[\frac{{{C}_{1}}}{{{V}_{1}}}=\frac{{{C}_{2}}}{{{V}_{2}}}\]
done
clear
View Answer play_arrow
question_answer 51) Tune constant for a series circuit R-C is:
A)
+RC
done
clear
B)
-RC
done
clear
C)
R/C
done
clear
D)
C/R
done
clear
View Answer play_arrow
question_answer 52) Unit of dipole moment is:
A)
amp-m
done
clear
B)
coulomb-m
done
clear
C)
\[amp-{{m}^{2}}\]
done
clear
D)
\[coulomb-{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 53) Three capacitors of capacity \[1\mu F\] each, are connected in such a way, that resultant capacity is \[1.5\,\mu F,\] then:
A)
all the capacitors are joined in series
done
clear
B)
all the capacitors are joined in parallel
done
clear
C)
two capacitors are in parallel, while third is in scries.
done
clear
D)
two capacitors are in series, while third is in parallel
done
clear
View Answer play_arrow
question_answer 54) According to Gauss law \[q={{\varepsilon }_{0}}o\int{\vec{E}.d\vec{S},}\]if total bound charge is zero to a Gaussian surface, then:
A)
\[E\] is 0 at the surface
done
clear
B)
number of coming and going lines of force is equal
done
clear
C)
only resultant in coming lines of force.
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 55)
Find equilvalent resistance between \[X\]and\[\text{Y}\]:
A)
R
done
clear
B)
R/2
done
clear
C)
2R
done
clear
D)
5K
done
clear
View Answer play_arrow
question_answer 56) When a cell is balanced on potentiometer wire, then balancing length is 125 cm. If resistance of 2 ohm is connected across the ends of cell, then balancing length is 100 cm, then internal resistance of cell is:
A)
\[0.5\,\Omega \]
done
clear
B)
\[0.25\,\Omega \]
done
clear
C)
\[0.05\,\Omega \]
done
clear
D)
\[5\,\Omega \]
done
clear
View Answer play_arrow
question_answer 57) In the nucleus of an atom, neutrons are in excess, then emitted particles are:
A)
neutron
done
clear
B)
electron
done
clear
C)
proton
done
clear
D)
positron
done
clear
View Answer play_arrow
question_answer 58) Water is flowing inside a tube of an uniform radius ratio of radius of entry and exit terminals of the tube is 3: 2. Then the ratio of velocities at entry and exit terminals will be:
A)
4 : 9
done
clear
B)
9 : 4
done
clear
C)
8 : 27
done
clear
D)
1 : 1
done
clear
View Answer play_arrow
question_answer 59) Total energy of a particle executing S.H.M. depends upon:
A)
amplitude and time period
done
clear
B)
amplitude, time period and displacement
done
clear
C)
amplitude and displacement
done
clear
D)
time period and displacement
done
clear
View Answer play_arrow
question_answer 60) At how many maximum points, time period of simple pendulum is same:
A)
1
done
clear
B)
2
done
clear
C)
4
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 61) Velocity at mean position of a particle executing S.H.M. is \[\upsilon ,\]then velocity of the t particle at a distance equal to half of the amplitude;
A)
\[4\upsilon \]
done
clear
B)
\[2\upsilon \]
done
clear
C)
\[\frac{\sqrt{3}}{2}\upsilon \]
done
clear
D)
\[\frac{\sqrt{3}}{4}\upsilon \]
done
clear
View Answer play_arrow
question_answer 62) A mixture of blue and red light incidence on a glass plate. After passing through the glass, rays agains come into air, then:
A)
position of both will be same, and both will propagate in same direction
done
clear
B)
position of both will be same, and both will propagate in different direction
done
clear
C)
position will be different but both will propagate in same direction.
done
clear
D)
both positions and direction of propagation will be different.
done
clear
View Answer play_arrow
question_answer 63) Ratio of average kinetic energy of \[{{H}_{2}}\] and \[{{O}_{2}}\] at a given temperature is:
A)
1 : 16
done
clear
B)
1 : 8
done
clear
C)
1 : 4
done
clear
D)
1 : 1
done
clear
View Answer play_arrow
question_answer 64) Which of following is true for diatomic gas?
A)
\[{{C}_{V}}=\frac{3}{2}R\]
done
clear
B)
\[{{C}_{P}}=\frac{7}{2}R\]
done
clear
C)
\[{{C}_{P}}=\frac{5}{2}R\]
done
clear
D)
\[{{C}_{P}}-{{C}_{V}}=R\]
done
clear
View Answer play_arrow
question_answer 65) 50 gm ice at \[0{}^\circ C\] in insulator vessel, 50 g water of \[100{}^\circ C\] is mixed in it, then final temperature of the mixture is (neglect the heat loss):
A)
\[10{}^\circ C\]
done
clear
B)
\[0{}^\circ <<{{T}_{m}}<20{}^\circ C\]
done
clear
C)
\[20{}^\circ C\]
done
clear
D)
Above \[20{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 66) Reason for potential barrier in\[p-n\] junction is:
A)
excess of positive charge at junction
done
clear
B)
deficiency of positive charge at junction
done
clear
C)
deficiency of negative charge at junction
done
clear
D)
excess of positive and negative charge at junction
done
clear
View Answer play_arrow
question_answer 67) Vibrations of rope tied by two rigid ends shown by equation \[y=\cos 2\pi t\,\sin \,2\pi t\] then minimum length of the rope will be:
A)
1 m
done
clear
B)
1/2 m
done
clear
C)
5 m
done
clear
D)
2n m
done
clear
View Answer play_arrow
question_answer 68)
If we change the value of R., then:
A)
voltage does not change on L
done
clear
B)
voltage does not change on LC combination
done
clear
C)
voltage does not change on C
done
clear
D)
voltage changes on LC combination
done
clear
View Answer play_arrow
question_answer 69) \[V=axy,\]then electric field at a point will be proportional to:
A)
r
done
clear
B)
\[{{r}^{-1}}\]
done
clear
C)
\[{{r}^{-2}}\]
done
clear
D)
\[{{r}^{2}}\]
done
clear
View Answer play_arrow
question_answer 70) Let \[{{E}_{1}}=x\hat{i}+y\hat{j}\] and \[{{E}_{2}}=x{{y}^{2}}\hat{i}+{{x}^{2}}y\,\hat{j},\] then:
A)
\[{{E}_{1}}\]represents constant electric field
done
clear
B)
\[{{E}_{2}}\]represents constant electric field
done
clear
C)
both represents constant electric field
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 71) Electric field at point 20 cm away from the centre of dielectric sphere is 100 V/m, radius of sphere is 10 cm, then value of electric field at a distance 3 cm from the centre is:
A)
100 V/m
done
clear
B)
125 V/m
done
clear
C)
120 V/m
done
clear
D)
0
done
clear
View Answer play_arrow
question_answer 72) When a X solute dissolve in Y solution then formed the precipitate. The X and Y will be:
A)
\[FeC{{l}_{3}}\] and \[SrB{{r}_{2}}\]
done
clear
B)
\[N{{a}_{2}}C{{O}_{3}}\]and \[CaC{{l}_{2}}\]
done
clear
C)
\[{{H}_{2}}S{{O}_{4}}\]and \[N{{H}_{3}}\]
done
clear
D)
\[NaN{{O}_{3}}\]and \[Pb{{(N{{O}_{3}})}_{2}}\]
done
clear
View Answer play_arrow
question_answer 73) \[Cu\] have the F.C.C. unit cell and length is 362 Pm what will be density of copper:
A)
\[6.29g/c{{m}^{3}}~\]
done
clear
B)
\[2.23\text{ }g/c{{m}^{3}}\]
done
clear
C)
\[4.45\text{ }g/c{{m}^{3}}\]
done
clear
D)
\[8.90\text{ }g/c{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 74) What does form in which of the following from the electrolysis of sodium propionate solution?
A)
Ethane
done
clear
B)
Acetylene
done
clear
C)
Ethyl alcohol
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 75) \[2A+B\xrightarrow{{}}C\] to the formation of 15 mole of C how much mole of A you will take in a jug if \[85%\] in the process?
A)
\[12.5~\]
done
clear
B)
\[17.5\]
done
clear
C)
\[35.3\]
done
clear
D)
\[30\]
done
clear
View Answer play_arrow
question_answer 76) Equal resonance length of \[C-C\]bond in benzene due to:
A)
resonance
done
clear
B)
tautomerism
done
clear
C)
\[s{{p}^{2}}\]hybridisation
done
clear
D)
isomerism
done
clear
View Answer play_arrow
question_answer 77) Match box shows:
A)
Cubic geometry
done
clear
B)
Orthorhombic geometry
done
clear
C)
Monoclinic geometry
done
clear
D)
Tetragonal geometry
done
clear
View Answer play_arrow
question_answer 78) The electro conductivity is highest in:
A)
sodium hydroxide
done
clear
B)
\[C{{H}_{3}}C{{O}_{2}}H\]
done
clear
C)
hydrogen cyanide
done
clear
D)
\[{{H}_{2}}C{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 79) Which of the following compound whose gives two combine carbon acid on oxidation is?
A)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
B)
\[C{{H}_{3}}CON{{H}_{2}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}N{{H}_{2}}\]
done
clear
D)
ethyl nitrite
done
clear
View Answer play_arrow
question_answer 80) The oxidation number of \[Ni\] and \[N{{H}_{3}}\]in complex \[{{[Ni{{(N{{H}_{3}})}_{6}}]}^{+2}}\] is:
A)
\[N{{H}_{3}}=0\,\,Ni=+2\]
done
clear
B)
\[Ni=+1,\,N{{H}_{3}}=-1/6\]
done
clear
C)
\[Ni=+1,\,N{{H}_{3}}=+1/6\]
done
clear
D)
\[Ni=+0,\,N{{H}_{3}}=+2\]
done
clear
View Answer play_arrow
question_answer 81) Which of the following order is correct c metal radius in \[IA\] group?
A)
\[K>Rb>Cs\]
done
clear
B)
\[Li>Na>K\]
done
clear
C)
\[Cs<K<Li\]
done
clear
D)
\[Na<K<Rb\]
done
clear
View Answer play_arrow
question_answer 82) The conjugated base of \[{{[{{H}_{2}}P{{O}_{4}}]}^{-}}\] is:
A)
\[{{H}_{2}}{{P}_{4}}{{O}_{7}}\]
done
clear
B)
\[{{[HP{{O}_{4}}]}^{-2}}\]
done
clear
C)
\[H\]
done
clear
D)
\[P{{O}_{4}}^{-3}\]
done
clear
View Answer play_arrow
question_answer 83) In which of the following oxidation state shows by the iodine?
A)
\[+1,-1,+3,+5\]
done
clear
B)
\[+1,-1,+3,+5,+7\]
done
clear
C)
\[+1,+3,+5\]
done
clear
D)
\[+1,+3,+7\]
done
clear
View Answer play_arrow
question_answer 84) In which of the following is not ion?
A)
\[NaN{{O}_{3}}\]
done
clear
B)
\[CaC{{O}_{3}}\]
done
clear
C)
\[CaB{{r}_{2}}\]
done
clear
D)
\[{{P}_{4}}{{S}_{3}}\]
done
clear
View Answer play_arrow
question_answer 85) This electronic configuration shows element of: \[\underset{1S}{\mathop{}}\,\,\,\,\,\,\,\,\,\,\,\underset{2S}{\mathop{}}\,\,\,\,\,\,\,\,\,\,\,\,\,\underset{2P}{\mathop{}}\,\]
A)
oxygen
done
clear
B)
neon
done
clear
C)
fluorine
done
clear
D)
nitrogen
done
clear
View Answer play_arrow
question_answer 86) In this reaction \[X+2YU+3V\] chemically known that \[10g\]of X react with \[20g\]of mixture of Y gives \[5g\]of U. Then quantity of V will be:
A)
\[30g\]
done
clear
B)
\[25g\]
done
clear
C)
\[2.5g\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 87) In which of the following will be oxidized to the formation of ketone?
A)
t-butyl alcohol
done
clear
B)
2-butanol
done
clear
C)
1-butanol
done
clear
D)
2-Propanol
done
clear
View Answer play_arrow
question_answer 88) \[C{{l}_{2}}+{{H}_{2}}SHCl+S.\]In this chemically reaction changes in oxidation number of sulphur is:
A)
\[0\] to \[2\]
done
clear
B)
\[-2\]to \[0\]
done
clear
C)
\[2\]to \[0\]
done
clear
D)
\[-2\] to \[-1\]
done
clear
View Answer play_arrow
question_answer 89) Which of the following react with \[A{{g}_{2}}S\]ore to the obtained of \[A{{g}_{2}}S\] ore in solution:
A)
\[C{{H}_{3}}CN\]
done
clear
B)
\[{{[Fe{{(CN)}_{6}}]}^{4-}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}CN\]
done
clear
D)
\[NaCN\]
done
clear
View Answer play_arrow
question_answer 90) \[X+C{{H}_{3}}COOH\xrightarrow{{}}Ester+{{H}_{2}}O,\], X is:
A)
alcohol
done
clear
B)
pyridine
done
clear
C)
alkane
done
clear
D)
acid
done
clear
View Answer play_arrow
question_answer 91) In which of the compound of urea?
A)
Ammonium thiocyanate
done
clear
B)
Ammonium cyanide
done
clear
C)
Ammonium cyanate
done
clear
D)
Glacin
done
clear
View Answer play_arrow
question_answer 92) \[C{{H}_{3}}-O-{{C}_{2}}{{H}_{5}}+HI\xrightarrow{{}}A,\] product A is:
A)
\[C{{H}_{3}}I+{{C}_{2}}{{H}_{5}}I\]
done
clear
B)
\[C{{H}_{3}}I+{{C}_{2}}{{H}_{5}}OH\]
done
clear
C)
\[C{{H}_{3}}CHI+C{{H}_{3}}OH\]
done
clear
D)
\[C{{H}_{3}}OH+C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 93) The acetone obtained from the ozonolysis of:
A)
isobutene
done
clear
B)
2-butene
done
clear
C)
butyne
done
clear
D)
1-butene
done
clear
View Answer play_arrow
question_answer 94) Which of die following example to the formation of \[C{{H}_{3}}-CH=N-N{{H}_{2}}\] from the reaction between acetaldehyde and hydrazin?
A)
Electrophilic substitution
done
clear
B)
Nucleophilic substitution
done
clear
C)
Alimesh
done
clear
D)
Condensation
done
clear
View Answer play_arrow
question_answer 95) The heat of hydrozination of \[C{{H}_{2}}=C{{H}_{2}}\] is \[30kcal/mole,\] what is the heat of hydrozination of 1, 3 butadiene?
A)
\[63\text{ }kcal/mole\]
done
clear
B)
\[57\text{ }kcal/mole\]
done
clear
C)
\[\text{60 }kcal/mole\]
done
clear
D)
\[30\text{ }kcal/mole\]
done
clear
View Answer play_arrow
question_answer 96) Which of the following is incorrect?
A)
\[{{H}_{4}}\text{ }EDTA\]tetrabasic
done
clear
B)
\[{{H}_{2}}S{{O}_{4}}\]dibasic
done
clear
C)
\[{{H}_{3}}P{{O}_{4}}\]tribasic
done
clear
D)
\[{{H}_{3}}P{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 97) In which of the following will have highest hydration?
A)
\[Rb\]
done
clear
B)
\[Cs\]
done
clear
C)
\[Na\]
done
clear
D)
\[K\]
done
clear
View Answer play_arrow
question_answer 98) Which of the following structure is false?
A)
\[C{{H}_{3}}-O=N=O\]
done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-NH\]
done
clear
C)
\[C{{H}_{3}}-CH=O-H\]
done
clear
D)
done
clear
View Answer play_arrow
question_answer 99) The \[_{p}{{K}_{a}}\] \[HN{{O}_{2}}\]is \[3.37\]. What is the pH of \[0.01mole/litre\]aqueous solution of\[HN{{O}_{2}}\]?
A)
\[2.69\]
done
clear
B)
\[8.67\]
done
clear
C)
\[1.69\]
done
clear
D)
\[3.57\]
done
clear
View Answer play_arrow
question_answer 100) \[\alpha \] particle is:
A)
\[H{{e}^{+}}\]
done
clear
B)
\[He\]
done
clear
C)
\[_{2}H{{e}^{4}}\]
done
clear
D)
\[{{[H{{e}_{2}}]}^{+2}}\]
done
clear
View Answer play_arrow
question_answer 101) Which of the following obtained from the effect of a molecule on \[Mg\]?
A)
\[Si\]
done
clear
B)
\[Pa\]
done
clear
C)
\[P\]
done
clear
D)
\[Ra\]
done
clear
View Answer play_arrow
question_answer 102) In which of the following shows resonance structure?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 103) Quick lime and slacked lime respectively is:
A)
\[CaO\]and \[CaC{{O}_{3}}\]
done
clear
B)
\[Ca{{(OH)}_{2}}\]and \[CaO\]
done
clear
C)
\[CaO\] and \[Ca{{(OH)}_{2}}\]
done
clear
D)
\[CaC{{O}_{3}}\] and \[Ca{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 104) In which of the following become reducting of metal (this reaction is not balanced equation):
A)
\[{{[C{{r}_{2}}{{O}_{7}}]}^{-2}}\xrightarrow{{}}{{[Cr{{O}_{4}}]}^{-2}}\]
done
clear
B)
\[{{[Fe{{(CN)}_{6}}]}^{-4}}\xrightarrow{{}}{{[Fe{{(CN)}_{6}}]}^{-3}}\]
done
clear
C)
\[Mn{{O}_{4}}^{-}\xrightarrow{{}}Mn{{O}_{2}}\]
done
clear
D)
\[Mn{{O}_{4}}^{-2}\xrightarrow{{}}Mn{{O}_{4}}^{-1}\]
done
clear
View Answer play_arrow
question_answer 105) The ore of copper is:
A)
Bauxite
done
clear
B)
malachite
done
clear
C)
cinnabar
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 106) What is the proper of transition element in the following:
A)
High ionization potential
done
clear
B)
Positive reduction potential
done
clear
C)
Changes oxidation state
done
clear
D)
Low melting point
done
clear
View Answer play_arrow
question_answer 107) The relation between 2C and K in equilibrium is:
A)
\[{{K}_{p}}={{K}_{c}}{{(RT)}^{\Delta n}}\]
done
clear
B)
\[{{K}_{p}}={{K}_{c}}\times RT\]
done
clear
C)
\[{{K}_{c}}={{K}_{p}}{{(RT)}^{\Delta n}}\]
done
clear
D)
\[{{K}_{c}}={{K}_{p}}\times \Delta n\]
done
clear
View Answer play_arrow
question_answer 108) In which of the following show the cis- trans geometry isomerism?
A)
\[ClC{{H}_{2}}C{{H}_{2}}Cl\]
done
clear
B)
\[C{{l}_{2}}C=C{{H}_{2}}\]
done
clear
C)
\[C{{l}_{2}}C=CC{{l}_{2}}\]
done
clear
D)
\[ClCH=CHCl\]
done
clear
View Answer play_arrow
question_answer 109) Which of the following is natural polymer?
A)
Polythene
done
clear
B)
Nitro cellulose
done
clear
C)
Backelite
done
clear
D)
Rubber
done
clear
View Answer play_arrow
question_answer 110) What is hybridisation of N in pyrole and tri-methyl amine?
A)
\[s{{p}^{2}}\]
done
clear
B)
\[s{{p}^{3}}\]
done
clear
C)
\[sp\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 111) Nylon is the polymer of:
A)
Acetylene
done
clear
B)
Butadyene
done
clear
C)
Adipic acid
done
clear
D)
Ethylene
done
clear
View Answer play_arrow
question_answer 112) The false name of \[NaCl\] is:
A)
Sodium chloride
done
clear
B)
GIobar salt
done
clear
C)
Rock salt
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 113) In \[C{{H}_{3}}C{{H}_{2}}OH,\] \[C{{H}_{3}}-O-C{{H}_{3}}\] isomerism is:
A)
functional isomerism
done
clear
B)
position isomerism
done
clear
C)
chain isomerism
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 114) Allotropes of carbon is:
A)
diamond
done
clear
B)
graphite
done
clear
C)
both and
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 115) \[C{{H}_{3}}CHO+G.R.\xrightarrow{{}}X\]product is:
A)
\[{{(C{{H}_{3}})}_{2}}CHOH\]
done
clear
B)
\[C{{H}_{3}}-O-C{{H}_{2}}\]
done
clear
C)
\[C{{H}_{3}}OH\]
done
clear
D)
\[C{{H}_{3}}COOH\]
done
clear
View Answer play_arrow
question_answer 116) \[10,18,36,54\]and 86 atomic number have all element is:
A)
light metal
done
clear
B)
inert gas
done
clear
C)
weak earth metal
done
clear
D)
halogen
done
clear
View Answer play_arrow
question_answer 117) The minimum configuration of an element is \[4{{s}^{1}},3{{d}^{10}}\]possible element is:
A)
element of a group 10th
done
clear
B)
a metal
done
clear
C)
a non metal
done
clear
D)
liquid at \[{{2981}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 118) Which of the following tells self going reaction:
A)
\[E>O\]
done
clear
B)
\[\Delta G>O\]
done
clear
C)
\[K>Q\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 119) \[{{C}_{6}}{{H}_{5}}COC{{H}_{3}}\]react with \[NaOH\]and bromine gives:
A)
\[CHB{{r}_{3}}\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}CONa\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}CONa+CHB{{r}_{3}}\]
done
clear
D)
\[C{{H}_{3}}COONa\]
done
clear
View Answer play_arrow
question_answer 120) The value of \[\Delta {{G}^{o}}\] of \[1\text{ }mole\] of gaseous number is \[87\text{ }kJ/mole\]at \[{{25}^{o}}C\]. At this temperature the value of 2C will be:
A)
\[1.8\times {{10}^{15}}\]
done
clear
B)
\[35\]
done
clear
C)
\[5.7\times {{10}^{-15}}\]
done
clear
D)
\[4.9\]
done
clear
View Answer play_arrow
question_answer 121) The half-life of 1 molecule is 1 hour, its rate of dissociation is 1000 per second at that time then after 3 hours. Its rate of dissociation will be:
A)
\[333\]
done
clear
B)
\[100\]
done
clear
C)
\[250\]
done
clear
D)
\[125\]
done
clear
View Answer play_arrow
question_answer 122) The magnetic moment of complex \[Kn[Mn{{F}_{6}}]\] is \[4.9\text{ }BM\]. What is the oxidation of \[Mn\] and what is the value of n:
A)
\[M(V);n=1\]
done
clear
B)
\[Mn(II);n=4\]
done
clear
C)
\[M(III);n=3\]
done
clear
D)
\[Mn(IV);n=2\]
done
clear
View Answer play_arrow
question_answer 123) Which statement is incorrect of \[AgCl\]?
A)
\[AgCl\] is the more soluble than \[KI\].
done
clear
B)
\[AgCl\] gives precipitate
done
clear
C)
\[AgCl\] is less soluble in water
done
clear
D)
\[AgI\] is less soluble than \[AgCl\]
done
clear
View Answer play_arrow
question_answer 124) In which of the following atomic weight is correct?
A)
\[B=9.016\text{ }amu~\]
done
clear
B)
\[B=10.26\text{ }amu\]
done
clear
C)
\[N=14.123\text{ }amu\]
done
clear
D)
\[0=10.02\text{ }amu\]
done
clear
View Answer play_arrow
question_answer 125) The formula of brown ring complex compound in \[[Fe{{({{H}_{2}}O)}_{5}}N{{O}^{+}}]S{{O}_{4}},\] What is the oxidation number of \[Fe\]?
A)
\[+1\]
done
clear
B)
\[+3\]
done
clear
C)
\[+2\]
done
clear
D)
\[-1\]
done
clear
View Answer play_arrow
question_answer 126) A compound which molar concentration is \[240g/mole\]and empirical formula is \[C{{H}_{2}}O\] then its molecular formula is:
A)
\[{{C}_{6}}{{H}_{16}}{{O}_{8}}\]
done
clear
B)
\[C{{l}_{2}}{{H}_{22}}{{O}_{12}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{12}}{{O}_{6}}\]
done
clear
D)
\[{{C}_{4}}{{H}_{8}}{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 127) The pH of water is 7. A substance dissolve in water then pH increase \[pH=13\] substance Y a salt is:
A)
strong acid and strong base
done
clear
B)
strong acid and weak base
done
clear
C)
weak acid and weak base
done
clear
D)
weak acid and strong base
done
clear
View Answer play_arrow
question_answer 128) In which of the following atom has minimum radii?
A)
\[Cl\]
done
clear
B)
\[K\]
done
clear
C)
\[Ar\]
done
clear
D)
\[S\]
done
clear
View Answer play_arrow
question_answer 129) In which of the following is para-uranium element?
A)
\[Po,Fm,Md\]
done
clear
B)
\[Bk,Cf,Am\]
done
clear
C)
\[TM,Nd,Pm\]
done
clear
D)
\[Th,Np,Pu\]
done
clear
View Answer play_arrow
question_answer 130) In which of the following reagent react with \[{{C}_{6}}{{H}_{6}}\]?
A)
\[Br/Fe\]
done
clear
B)
\[C{{H}_{3}}Cl/Al\]
done
clear
C)
\[HN{{O}_{3}}\]
done
clear
D)
Cold dilute \[KMn{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 131) Benzene react with \[C{{H}_{3}}Cl/AlC{{l}_{3}}\]. This reaction is called as:
A)
Friedel Craft reaction
done
clear
B)
Aldol condensation
done
clear
C)
Riemer Tiemann reaction
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 132) If the \[{{K}_{sp}}\] of \[AgCl\]is \[25\times {{10}^{-12}}\] at\[{{25}^{o}}C\]. Then solubility of \[AgCl\] is:
A)
\[5\times {{10}^{-13}}\]
done
clear
B)
\[7.4\times {{10}^{-7}}\]
done
clear
C)
\[5\times {{10}^{-6}}\]
done
clear
D)
\[2.5\times {{10}^{-6}}\]
done
clear
View Answer play_arrow
question_answer 133) The molar solubility for a solid \[[Mg{{(OH)}_{2}}]\]is 5 and solubility product is \[{{K}_{sp}}\] will be:
A)
\[s\]
done
clear
B)
\[{{s}^{3}}\]
done
clear
C)
\[4{{s}^{3}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 134) Which of the substance is used in solar cells?
A)
\[Cs\]
done
clear
B)
\[Si\]
done
clear
C)
\[Sn\]
done
clear
D)
\[Tn\]
done
clear
View Answer play_arrow
question_answer 135) Which of the following deficiency scurvy disease is obtained?
A)
follic acid
done
clear
B)
Ascorbic acid
done
clear
C)
Neocin
done
clear
D)
Thiamine
done
clear
View Answer play_arrow
question_answer 136) The special property in plastic teflon from due to which element?
A)
\[F\]
done
clear
B)
\[P\]
done
clear
C)
\[N\]
done
clear
D)
\[Cl\]
done
clear
View Answer play_arrow
question_answer 137) The addition of pH and \[pOH\]in aqueous solution will:
A)
\[7\]
done
clear
B)
\[_{p}{{K}_{\omega }}\]
done
clear
C)
\[1\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 138) Which subshell for any atom is wrong?
A)
\[2f\]
done
clear
B)
\[3s\]
done
clear
C)
\[4d\]
done
clear
D)
\[5f\]
done
clear
View Answer play_arrow
question_answer 139) Which of the following configuration is correct for the nitrogen?
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{2}}x2p{{y}^{1}}\]
done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{2}}x2p{{y}^{1}}z\]
done
clear
C)
\[1{{s}^{2}},2{{s}^{2}},2p{{x}^{1}},2p{{y}^{1}},2{{p}^{1}}z\]
done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{3}}x\]
done
clear
View Answer play_arrow
question_answer 140) Who discovered the electron on the drop of oil:
A)
Milikan
done
clear
B)
Rutherford
done
clear
C)
Chadwick
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 141) The bond order of \[He{{H}^{+}}\] is:
A)
\[3/2\]
done
clear
B)
\[1\]
done
clear
C)
\[1/2\]
done
clear
D)
\[2\]
done
clear
View Answer play_arrow
question_answer 142) In which of the following the percentage of carbon is very high?
A)
Stainless steel
done
clear
B)
Steel
done
clear
C)
Cast yon
done
clear
D)
Electric decomposed copper
done
clear
View Answer play_arrow
question_answer 143) \[{{N}_{2}}{{O}_{4}}2N{{O}_{2}}\] In this reaction the dissociation of \[{{N}_{2}}{{O}_{4}}\] is X then the how much mole will of \[{{N}_{2}}{{O}_{4}}\] and \[N{{O}_{2}}\] at equilibrium:
A)
\[{{(1-x)}^{2}}\]
done
clear
B)
\[1\]
done
clear
C)
\[(1+x)\]
done
clear
D)
\[2\]
done
clear
View Answer play_arrow
question_answer 144) The forward rate of reaction is \[2.5\times {{10}^{-4}}\]at equilibrium for a reaction and backward rate of reaction is\[7.5\times {{10}^{-4}}\]. Then velocity constant is:
A)
\[2\]
done
clear
B)
\[3\]
done
clear
C)
\[1/3\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 145) Which of the following formula is more reactive for hydrolysis?
A)
\[-COOR\]
done
clear
B)
\[-CON{{H}_{2}}\]
done
clear
C)
\[-COOH\]
done
clear
D)
\[-COCl\]
done
clear
View Answer play_arrow
question_answer 146) A element is A. Whose electronic configuration is\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{3}}\]. This element conbined with \[Ca\] and formed a compound. The formula of compound is:
A)
\[C{{a}_{3}}A\]
done
clear
B)
\[C{{a}_{3}}{{A}_{2}}\]
done
clear
C)
\[C{{a}_{2}}{{A}_{3}}\]
done
clear
D)
\[CaA\]
done
clear
View Answer play_arrow
question_answer 147) Which orbital is used in centre atom in \[N{{H}_{2}}\] bonding?
A)
\[s{{p}^{2}}\]
done
clear
B)
\[s{{p}^{3}}\]
done
clear
C)
\[sp\]
done
clear
D)
\[s\]
done
clear
View Answer play_arrow
question_answer 148) The isoelectronic of sulphide ion is:
A)
\[Kr\]
done
clear
B)
\[Ne\]
done
clear
C)
\[Ar\]
done
clear
D)
\[Cl\]
done
clear
View Answer play_arrow
question_answer 149) Which of the following is not isoelectronic species?
A)
\[Mg\] and \[N\]
done
clear
B)
\[Be\]and \[N\]
done
clear
C)
\[O\] and \[N\]
done
clear
D)
\[Mg\] and \[O\]
done
clear
View Answer play_arrow
question_answer 150) Addition of \[B{{r}_{2}}\] on \[C{{H}_{2}}=CH-C{{H}_{2}}-C{{H}_{3}}\]gives:
A)
\[Br-C{{H}_{2}}-\overset{Br}{\mathop{\overset{|}{\mathop{C}}\,}}\,H-CH-C{{H}_{3}}\]
done
clear
B)
\[Br-C{{H}_{2}}-C{{H}_{2}}\overset{Br}{\mathop{\overset{|}{\mathop{-C}}\,}}\,H-C{{H}_{3}}\]
done
clear
C)
\[B{{r}_{2}}-C{{H}_{2}}-C{{H}_{2}}-\underset{Br}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{3}}\]
done
clear
D)
\[Br-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-\underset{Br}{\mathop{\underset{|}{\mathop{C}}\,}}\,{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 151) According to Markownikoffs rule addition of water on 1-butene gives:
A)
primary alcohol
done
clear
B)
tertiary alcohol
done
clear
C)
secondary alcohol
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 152)
A)
1-amino, 1-phenyl, 2-methyl propane
done
clear
B)
2-methyl 1-amino 1-phenyl propane
done
clear
C)
1-amino 2-methyl 1-phenyl propane
done
clear
D)
1-isopropyl 1-phenyl methyl amine
done
clear
View Answer play_arrow
question_answer 153) Strong acid is:
A)
\[C{{H}_{3}}-C{{O}_{2}}H\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}-C{{O}_{2}}-H\]
done
clear
C)
\[Cl-C{{H}_{2}}-COOH\]
done
clear
D)
\[Cl-C{{H}_{2}}-C{{O}_{2}}-H\]
done
clear
View Answer play_arrow
question_answer 154) Which carbonium ion is highly stable?
A)
\[C{{H}_{2}}=C{{H}_{2}}C{{H}_{2}}\]
done
clear
B)
\[{{(C{{H}_{2}})}_{3}}{{C}^{+}}\]
done
clear
C)
\[P{{h}_{3}}{{C}^{+}}\]
done
clear
D)
\[{{(C{{H}_{2}})}_{2}}C{{H}^{+}}\]
done
clear
View Answer play_arrow
question_answer 155) In which of following is not classified glycin?
A)
A-zwitter ion
done
clear
B)
A base
done
clear
C)
L-amino acid
done
clear
D)
A chelating agent
done
clear
View Answer play_arrow
question_answer 156) Incorrect for the glucose
A)
combined with \[6-C\]
done
clear
B)
mutarotation
done
clear
C)
aldose
done
clear
D)
disaccharide
done
clear
View Answer play_arrow
question_answer 157) The IUPAC name of \[{{(C{{H}_{3}})}_{2}}CHCH(C{{H}_{3}})C{{H}_{2}}-CH=C{{H}_{2}}\] is:
A)
2-3 di methyl hexene
done
clear
B)
2, 3 di methyl hexane
done
clear
C)
4, 5, 5 trimethyl pentene
done
clear
D)
4, 5 di methyl hexene
done
clear
View Answer play_arrow
question_answer 158) In which gives geometry isomerism?
A)
3-flouro propene
done
clear
B)
1-chloro propene
done
clear
C)
1, 2-dibromo ethylene
done
clear
D)
Penta 2-ene
done
clear
View Answer play_arrow
question_answer 159) The electronic configuration of \[Mg\] \[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}\]. The number of proton is:
A)
\[12\]
done
clear
B)
\[10\]
done
clear
C)
\[2\]
done
clear
D)
\[6\]
done
clear
View Answer play_arrow
question_answer 160) If X obey the octate rule then the number of unpaired electron pair will on it, if \[M-X-N\]compound formed:
A)
\[1\]
done
clear
B)
\[2\]
done
clear
C)
\[0\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 161) The structure of ion of oxygen in peroxide, super oxide and ozonite respectively is:
A)
\[{{O}_{2}}^{-2},{{O}_{2}}^{-},{{O}_{3}}^{-}\]
done
clear
B)
\[{{O}_{2}}^{-2},{{O}^{-2}},{{O}^{3-}}\]
done
clear
C)
\[{{O}_{2}}^{-},{{O}_{2}}^{2},{{O}_{3}}^{-2}\]
done
clear
D)
\[{{O}_{2}}^{-},{{O}_{3}}^{-3},{{O}_{3}}^{-2}\]
done
clear
View Answer play_arrow
question_answer 162) In which ratio is correct of alkaline earth metal and family of chalcogen?
A)
\[1:1\]
done
clear
B)
\[1:3\]
done
clear
C)
\[1:2\]
done
clear
D)
\[2:1\]
done
clear
View Answer play_arrow
question_answer 163) Add \[0.01M\text{ }HCl\]in some amount in aqueous solution of acetic acid:
A)
\[E{{q}^{m\,}}cone.\,decreases\,of\,C{{H}_{3}}COOH\]
done
clear
B)
\[E{{q}^{m}}cone.\,decreases\,of\,C{{H}_{3}}CO{{O}^{-}}\]
done
clear
C)
\[E{{q}^{m}}cone.\text{ }increases\text{ }of\text{ }C{{H}_{3}}CO{{O}^{-}}\]
done
clear
D)
No any change
done
clear
View Answer play_arrow
question_answer 164) The chemically name of gammaxene:
A)
DDT
done
clear
B)
benzene hexa chloride
done
clear
C)
hexa chloro benzene
done
clear
D)
chloral
done
clear
View Answer play_arrow
question_answer 165) The adipose tissue in the skin is found in the:
A)
dermis
done
clear
B)
subcutaneous layer
done
clear
C)
Malpighian layer
done
clear
D)
epidermal layer
done
clear
View Answer play_arrow
question_answer 166) The colour of the skin is due to the presence of:
A)
keratohyalin found in the layer stratum granulosum
done
clear
B)
collagen found in the subcutaneous layer
done
clear
C)
melanin present in the dermis
done
clear
D)
both a and b
done
clear
View Answer play_arrow
question_answer 167) The mammary glands are derived from:
A)
sweat glands
done
clear
B)
adipose tissue
done
clear
C)
hair follicles
done
clear
D)
sebaceous glands
done
clear
View Answer play_arrow
question_answer 168) Which of the following is not a function of the mammalian skin?
A)
Display of emotions
done
clear
B)
Formation of vitamin D
done
clear
C)
Absorption of food scuff
done
clear
D)
Excretion of waste matter
done
clear
View Answer play_arrow
question_answer 169) Which one of the following is not the part of the epidermis ?
A)
Stratum granulosum
done
clear
B)
Stratum lucidum
done
clear
C)
Stratum cornium
done
clear
D)
Subcutaneous ayer
done
clear
View Answer play_arrow
question_answer 170) Enzymes arc essential in our body because they:
A)
supply the energy
done
clear
B)
coordinate nervous activities
done
clear
C)
catalyze biochemical reactions
done
clear
D)
are structural component of the body
done
clear
View Answer play_arrow
question_answer 171) The inorganic catalyst attached to enzymes is called:
A)
activator
done
clear
B)
lysozyme
done
clear
C)
apoenzyme
done
clear
D)
inhibitor
done
clear
View Answer play_arrow
question_answer 172) Metabolic processes in living organisms are catalyzed by:
A)
hormones
done
clear
B)
enzymes
done
clear
C)
lipids
done
clear
D)
amino acids
done
clear
View Answer play_arrow
question_answer 173) Breaking down of organic compounds in the body and liberation of energy is :
A)
a catabolic process
done
clear
B)
an anabolic process
done
clear
C)
a digestive process
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 174) The process of synthesis of glycogen formation in the liver is due to:
A)
glycolysis
done
clear
B)
glycogenolysis
done
clear
C)
glycogenesis
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 175) Vitamins are:
A)
organic compounds
done
clear
B)
hormones
done
clear
C)
minerals
done
clear
D)
salts
done
clear
View Answer play_arrow
question_answer 176) the following the function that includes all the others is :
A)
excretion
done
clear
B)
metabolism
done
clear
C)
digestion
done
clear
D)
respiration
done
clear
View Answer play_arrow
question_answer 177) A nutrient which is used to build protoplasm is;
A)
fat
done
clear
B)
protein
done
clear
C)
carbohydrate
done
clear
D)
calcium
done
clear
View Answer play_arrow
question_answer 178) Two elements needed for building healthy teeth and bones are:
A)
iron and calcium
done
clear
B)
phosphorous and iron
done
clear
C)
calcium and phosphorous
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 179) Apart of the digestive system which is not in contact with food is :
A)
large intestine
done
clear
B)
small intestine
done
clear
C)
trachea
done
clear
D)
liver
done
clear
View Answer play_arrow
question_answer 180) M. Saliva converts:
A)
starch into maltose
done
clear
B)
fats into fatty acids
done
clear
C)
glycogen into glucose
done
clear
D)
proteins into amino acids
done
clear
View Answer play_arrow
question_answer 181) A secretion that digests both carbohydrates and proteins is:
A)
ptyalin
done
clear
B)
pepsin
done
clear
C)
pancreatic juice
done
clear
D)
saliva
done
clear
View Answer play_arrow
question_answer 182) Food is moved along the alimentary canal by the contraction known as :
A)
peristalsis is
done
clear
B)
epiglottis
done
clear
C)
osmosis
done
clear
D)
cyclosis
done
clear
View Answer play_arrow
question_answer 183) Villi are present in the :
A)
large intestine
done
clear
B)
small intestine
done
clear
C)
colon
done
clear
D)
stomach
done
clear
View Answer play_arrow
question_answer 184) Fatty acids are absorbed by the :
A)
lacteals
done
clear
B)
pylorous
done
clear
C)
colon
done
clear
D)
capillaries
done
clear
View Answer play_arrow
question_answer 185) Hydrochloric acid in the stomach is secreted by some special type of cells called :
A)
peptic cells
done
clear
B)
goblet cells
done
clear
C)
oxyntic cells
done
clear
D)
gastric cells
done
clear
View Answer play_arrow
question_answer 186) Enzyme carbohydrates dominate in the digestive tract of:
A)
carnivores
done
clear
B)
omnivores
done
clear
C)
parasites
done
clear
D)
herbivores
done
clear
View Answer play_arrow
question_answer 187) In Horses, Rabbits, Hares, the cellulose gets digested in the :
A)
caecum
done
clear
B)
stomach
done
clear
C)
appendix
done
clear
D)
rumen
done
clear
View Answer play_arrow
question_answer 188) The pylorous is the constricted part of the alimentary canal which is situated between:
A)
stomach and duodenum
done
clear
B)
oesophagus and stomach
done
clear
C)
duodenum and ileum
done
clear
D)
ileum and rectum
done
clear
View Answer play_arrow
question_answer 189) The intestinal juice, succus entericus is secreted by:
A)
Brunners gland
done
clear
B)
Kupffer cells
done
clear
C)
crypts of Leiberkuhn
done
clear
D)
goblet cells
done
clear
View Answer play_arrow
question_answer 190) Below freezing point, the pepsin :
A)
becomes over activated
done
clear
B)
gets destroyed
done
clear
C)
remains unaffected
done
clear
D)
gets inactivated
done
clear
View Answer play_arrow
question_answer 191) The correct order of structures through which the air passes in the body is :
A)
nasal cavity, larynx, trachea, bronchi, air sacs
done
clear
B)
bronchi, nasal cavity, larynx, trachea, air sacs
done
clear
C)
larynx bronchi, nasal cavity, trachea, air sacs
done
clear
D)
nasal cavity, trachea, larynx, bronchi, air sacs
done
clear
View Answer play_arrow
question_answer 192) The trachea is prevented from collapsing by the presence by rings of:
A)
striated muscles
done
clear
B)
cartilage
done
clear
C)
adenoids
done
clear
D)
sinuses
done
clear
View Answer play_arrow
question_answer 193) The process of breathing is controlled by respiratory centre located in the:
A)
spinal cord
done
clear
B)
cerebellum
done
clear
C)
medulla oblongata
done
clear
D)
hypothalamus
done
clear
View Answer play_arrow
question_answer 194) When the carbon dioxide concentration in blood increases, the rate of breathing will:
A)
remain unaffected
done
clear
B)
decrease
done
clear
C)
stop
done
clear
D)
increase
done
clear
View Answer play_arrow
question_answer 195) During inspiration :
A)
the diaphragm gets raised and ribs get lowered
done
clear
B)
both diaphragm and ribs raised
done
clear
C)
both diaphragm and ribs get lowered
done
clear
D)
the diaphragm get flattened and ribs get raised
done
clear
View Answer play_arrow
question_answer 196) Which one of the following can respire in the absence of oxygen?
A)
Amoeba
done
clear
B)
Tapeworm
done
clear
C)
House fly
done
clear
D)
Hydra
done
clear
View Answer play_arrow
question_answer 197) The breathing rate gets increased by the increase in the content of :
A)
nitrogen
done
clear
B)
oxygen
done
clear
C)
carbon dioxide
done
clear
D)
carbon monoxide
done
clear
View Answer play_arrow
question_answer 198) The normal rate of respiration in man per minute is about:
A)
10 to 15 times
done
clear
B)
16 to 20 times
done
clear
C)
19 to 21 times
done
clear
D)
23 to 25 times
done
clear
View Answer play_arrow
question_answer 199) A normal man at rest inspires and expires about 500 millilitres of air, this amount is known as:
A)
complimental volume of air
done
clear
B)
tidal volume of air
done
clear
C)
reserve volume of air
done
clear
D)
residual volume of air
done
clear
View Answer play_arrow
question_answer 200) One of the following is not a respiratory pigment?
A)
Anthocyanin
done
clear
B)
Haemoglobin
done
clear
C)
Haemoerythrin
done
clear
D)
Haemocyanin
done
clear
View Answer play_arrow
question_answer 201) The disease that occurs when the haemoglobin content of the blood goes down is :
A)
plurisy
done
clear
B)
emphysema
done
clear
C)
anaemia
done
clear
D)
pneumonia
done
clear
View Answer play_arrow
question_answer 202) The percentage of carbon dioxide, oxygen and nitrogen in exhaled air of man are about:
A)
\[0.04-16{}^\circ 79\]
done
clear
B)
\[0.4-20{}^\circ 70\]
done
clear
C)
\[4.0-20{}^\circ 79\]
done
clear
D)
\[4.0-16{}^\circ 79\]
done
clear
View Answer play_arrow
question_answer 203) The transport of oxygen by the blood takes place by :
A)
leucocytes
done
clear
B)
thrombocytes
done
clear
C)
plasma
done
clear
D)
erythrocytes
done
clear
View Answer play_arrow
question_answer 204) Theory throcytes of mammals are, without nucleus except in one of the following?
A)
Man
done
clear
B)
Monkey
done
clear
C)
Elephant
done
clear
D)
Camel
done
clear
View Answer play_arrow
question_answer 205) The blood in the vessels does not coagulate under normal conditions because of the presence of:
A)
calcium
done
clear
B)
heparin
done
clear
C)
prothrombin
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 206) Erythrocytes are formed in the:
A)
thymus
done
clear
B)
liver
done
clear
C)
bone marrow
done
clear
D)
spleen
done
clear
View Answer play_arrow
question_answer 207) All the following statements in respect of arteries are true except:
A)
they contain valves
done
clear
B)
they are thick walled and contractile
done
clear
C)
they carry blood away from the heart
done
clear
D)
they contain elastic tissue
done
clear
View Answer play_arrow
question_answer 208) All of the following veins carry deoxygenated blood except:
A)
renal vein
done
clear
B)
superior vena cava
done
clear
C)
pulmonary vein
done
clear
D)
inferior vena cava
done
clear
View Answer play_arrow
question_answer 209) All of the following statements are true in respect of white blood corpuscles except:
A)
they are formed in lymph glands
done
clear
B)
they eat bacteria and-foreign bodies
done
clear
C)
they move like Amoeba
done
clear
D)
they move like Paramecium
done
clear
View Answer play_arrow
question_answer 210) The impulse of the heart beat originates from:
A)
Hensens node
done
clear
B)
Sinu auricular node
done
clear
C)
Ranviers node
done
clear
D)
Auriculo ventricular node
done
clear
View Answer play_arrow
question_answer 211) A person with type A blood group may safely receive a transfusion of:
A)
type A B
done
clear
B)
type A and type O
done
clear
C)
type A and type AB
done
clear
D)
type AB and type O
done
clear
View Answer play_arrow
question_answer 212) Serum is like plasma except that it has no:
A)
haemoglobin
done
clear
B)
fibrin
done
clear
C)
fibrinogen
done
clear
D)
red blood corpuscles
done
clear
View Answer play_arrow
question_answer 213) Circulation of blood is called open in :
A)
Man
done
clear
B)
Frog
done
clear
C)
Cockroach
done
clear
D)
Fish
done
clear
View Answer play_arrow
question_answer 214) The correct sequence of steps in clotting of blood is:
A)
platelet, fibrin, fibrinogen, thrombin, prothrombin
done
clear
B)
platelet, thrombin, fibrin, fibrinogen, prothrombin
done
clear
C)
platelet, fibrin, fibrinogen, prothrombin, thrombin
done
clear
D)
platelet, prothrombin, thrombin, fibrinogen, fibrin
done
clear
View Answer play_arrow
question_answer 215) Persons having no antigen in RBC and both a and b antibodies are known as universal donors. Their blood group is :
A)
A
done
clear
B)
AB
done
clear
C)
O
done
clear
D)
B
done
clear
View Answer play_arrow
question_answer 216) The elimination of urea is termed as :
A)
ureotelism
done
clear
B)
ammonotelism
done
clear
C)
urecotelism
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 217) The filterate from the glomerulus contains :
A)
urea, uric acid, ammonia and water
done
clear
B)
urea and uric acid only
done
clear
C)
urea, uric acid and water
done
clear
D)
urea, uric acid, glucose and water
done
clear
View Answer play_arrow
question_answer 218) Urea in the body of animals is formed as a result of:
A)
deamination of amino acids
done
clear
B)
oxidation of glucose
done
clear
C)
hydrolysis of fats
done
clear
D)
emunification of fats
done
clear
View Answer play_arrow
question_answer 219) The part of the brain which is most developed in higher primates is :
A)
cerebellum
done
clear
B)
cerebrum
done
clear
C)
medulla oblongata
done
clear
D)
olfactory lobes
done
clear
View Answer play_arrow
question_answer 220) The cerebellum is concerned with :
A)
memory
done
clear
B)
perception
done
clear
C)
intelligence
done
clear
D)
coordination of muscular movement
done
clear
View Answer play_arrow
question_answer 221) A mass of cytons is called as :
A)
synapse
done
clear
B)
ganglion
done
clear
C)
motor nerve
done
clear
D)
nerve fibre
done
clear
View Answer play_arrow
question_answer 222) The pathway of reflex arc as:
A)
sense organ, spinal cord, motor neuron, sensory nerve, muscle
done
clear
B)
sense organ, sensory neuron, motor neuron, spinal cord, muscle
done
clear
C)
sense organ, motor neuron, spinal cord, sensory neuron, muscles
done
clear
D)
sense organ, motor neuron, spinal cord, sensory neuron, muscle
done
clear
View Answer play_arrow
question_answer 223) The sense of balance maintained with the help of:
A)
hammer
done
clear
B)
strirrup
done
clear
C)
semicircular canals
done
clear
D)
cochlea
done
clear
View Answer play_arrow
question_answer 224) The small canal leading from pinna into the head in mammals is :
A)
Haversian canal
done
clear
B)
auditory meatus
done
clear
C)
inguinal canal
done
clear
D)
eustachian tube
done
clear
View Answer play_arrow
question_answer 225) The fluid present between the lens and the cornea in the eye is :
A)
aqueous humor
done
clear
B)
endolymphatic fluid
done
clear
C)
vitreous humor
done
clear
D)
perilymphatic fluid
done
clear
View Answer play_arrow
question_answer 226) The eye disease hypermetropia can becured by using:
A)
concave lens
done
clear
B)
convex lens
done
clear
C)
concavo-convex lens
done
clear
D)
plane lens
done
clear
View Answer play_arrow
question_answer 227) The unstriped muscles are found in :
A)
neck region
done
clear
B)
heart region
done
clear
C)
urinary bladder
done
clear
D)
thigh region
done
clear
View Answer play_arrow
question_answer 228) Hormones are distributed to the different body parts by:
A)
ducts
done
clear
B)
blood
done
clear
C)
nerves
done
clear
D)
lymph
done
clear
View Answer play_arrow
question_answer 229) Metamorphosis of Frog from tadpole to adult is controlled by :
A)
adrenalin
done
clear
B)
thyroxin
done
clear
C)
secretin
done
clear
D)
insulin
done
clear
View Answer play_arrow
question_answer 230) The basal metabolism rate of the body is regulated by:
A)
parathyroid
done
clear
B)
thymus
done
clear
C)
pituitary
done
clear
D)
thyroid
done
clear
View Answer play_arrow
question_answer 231) An endocrine gland known as gland of emergency is :
A)
pituitary
done
clear
B)
parathyroid
done
clear
C)
adrenal
done
clear
D)
pancreas
done
clear
View Answer play_arrow
question_answer 232) One of the hormone, which when secreted increase the blood pressure, heart beat output of glucose in the blood :
A)
thyroxin
done
clear
B)
aldosterone
done
clear
C)
adrenalin
done
clear
D)
testosterone
done
clear
View Answer play_arrow
question_answer 233) Water vascular system is a pecularity of:
A)
Porifera
done
clear
B)
Coelenterata
done
clear
C)
Mollusca
done
clear
D)
Echinodermata
done
clear
View Answer play_arrow
question_answer 234) A definite number of segments is found in :
A)
Slug
done
clear
B)
Earthworm
done
clear
C)
Leech
done
clear
D)
Tapeworm
done
clear
View Answer play_arrow
question_answer 235) Which of the following is a Coelenterate?
A)
Sea urchin
done
clear
B)
Sea horse
done
clear
C)
Sea pen
done
clear
D)
Sea cucumber
done
clear
View Answer play_arrow
question_answer 236) What is common amongst Tapeworm Liver fluke and Planaria?
A)
They are all segmented
done
clear
B)
They are all found in gut
done
clear
C)
They all have a coelom
done
clear
D)
They are all Hat
done
clear
View Answer play_arrow
question_answer 237) You are examining a water sample from a pond which is full of water plants. You find an animal which is cylindrical and shows a long filamentous structure at one end. It is not stationary and shows variety of movements. It could:
A)
Paramecium
done
clear
B)
Tadpole
done
clear
C)
Hydra
done
clear
D)
Amoeba
done
clear
View Answer play_arrow
question_answer 238) Kala-azar is caused by :
A)
Trypanosoma
done
clear
B)
Leishmania
done
clear
C)
Amoeba
done
clear
D)
tse-tse fly
done
clear
View Answer play_arrow
question_answer 239) Which is the secretion of body of an insect?
A)
Lac
done
clear
B)
Wax
done
clear
C)
Sulphur
done
clear
D)
Phosphorus
done
clear
View Answer play_arrow
question_answer 240) Basic unit of compound eyes of Cockroach is:
A)
ocselli
done
clear
B)
ommatidia
done
clear
C)
ratinule
done
clear
D)
crystalline core
done
clear
View Answer play_arrow
question_answer 241) The larva of Housefly breathes by means of:
A)
gills
done
clear
B)
trachea tubes
done
clear
C)
body surface
done
clear
D)
does not breathe
done
clear
View Answer play_arrow
question_answer 242) A normal human being will have:
A)
22 pairs of chromosome
done
clear
B)
23 pairs of chromosome
done
clear
C)
44 pairs of chromosome
done
clear
D)
46 pairs of chromosome
done
clear
View Answer play_arrow
question_answer 243) The human sperm cell contains:
A)
46 chromosomes
done
clear
B)
44 chromosomes and 1X and 1Y chromosome
done
clear
C)
23 chromosomes only
done
clear
D)
23 chromosomes and 1X and 1Y chromosome
done
clear
View Answer play_arrow
question_answer 244) The haemoglobin of people suffering from sickle cell anaemia differs from normal haemoglobin by one:
A)
lipid
done
clear
B)
monosaccharide
done
clear
C)
fatty acid
done
clear
D)
amino acid
done
clear
View Answer play_arrow
question_answer 245) If a man who is colour blind marries a woman who is pure normal for colour vision, the chances of their sons having colour blindness is:
A)
100%
done
clear
B)
50 : 50
done
clear
C)
(zero) 0%
done
clear
D)
75 : 25
done
clear
View Answer play_arrow
question_answer 246) Absence of a digit or limb in a man is due to:
A)
germinal variation
done
clear
B)
somatic variation
done
clear
C)
continuous variation
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 247) Brachydactyly is due to :
A)
dominant gene on the autosome
done
clear
B)
recessive gene on the autosome
done
clear
C)
dominant gene on the sex chromosome
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 248) Uncontrolled twitching of voluntary muscles is a symptom of:
A)
phenylketonuria
done
clear
B)
Hutingtons chorea
done
clear
C)
alkaptonuria
done
clear
D)
haemophilia
done
clear
View Answer play_arrow
question_answer 249) The gene for haemophilia is located on:
A)
Y-chromosome
done
clear
B)
X-chromosome
done
clear
C)
autosome
done
clear
D)
both a and b
done
clear
View Answer play_arrow
question_answer 250) Cause of Downs syndrome is due to :
A)
trisomy of 21st chromosome
done
clear
B)
trisomy of X chromosome
done
clear
C)
monosomy of 21s1 chromosome
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 251) A lady becomes haemophilic when the recessive gene responsible for it is located on:
A)
both autosome and sex chromosome
done
clear
B)
on X chromosome
done
clear
C)
both the X chromosome
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 252) The cause of Cat-cry syndrome is due to :
A)
loss of a segment of X chromosome
done
clear
B)
loss of a segment of 5th chromosome
done
clear
C)
loss of segment of \[\text{Y}\] chromosome
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 253) Deletition of a part of 22nd chromosome and its subsequent attachments to the 9th chromosome results in:
A)
bleeders disease
done
clear
B)
phenylketonuria
done
clear
C)
granulocytic leukemia
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 254) Webbed neck, low set ears and broad chest are the symptoms associated with :
A)
females having XXX chromosomes
done
clear
B)
females having XO chromosomes
done
clear
C)
females having XX chromosomes
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 255) Abnormal sterile male, with underdeveloped genetilia, feeble breasts and sponsely hairy body are symptoms associates with:
A)
males having XXY chromosomes
done
clear
B)
males having XY chromosomes
done
clear
C)
males having XYY chromosomes
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 256) XYY males show the following symptoms :
A)
underdeveloped genitilia, sterile and feeble breasts
done
clear
B)
over developed genitilia, unusual increase in height, criminal bent of mind
done
clear
C)
normal height and normal development of genetilia
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 257) PKU is caused by the lack of gene for:
A)
a hormone
done
clear
B)
an enzyme
done
clear
C)
liver cells
done
clear
D)
brain cells
done
clear
View Answer play_arrow
question_answer 258) The genotype for blood group B will be:
A)
AB or AB
done
clear
B)
BB or BO
done
clear
C)
AB or OO
done
clear
D)
BB or AA
done
clear
View Answer play_arrow
question_answer 259) The blood-group AB is considered universal recipient because it has :
A)
antibodies for A and B
done
clear
B)
antibodies neither for A nor for B
done
clear
C)
antibodies for B only
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 260) Chromosome other than sex chromoson is known as
A)
sex chromosome
done
clear
B)
autosome
done
clear
C)
autophage
done
clear
D)
chromomere
done
clear
View Answer play_arrow
question_answer 261) The gene responsible for causing re green colour blindness is located on:
A)
autosome
done
clear
B)
X-chromosome
done
clear
C)
Y-chromosome
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 262) Gene in a test-tube was synthesized by:
A)
Watson and Crick
done
clear
B)
H.G. Khorana
done
clear
C)
Arthur Komberg
done
clear
D)
Stanley Miller
done
clear
View Answer play_arrow
question_answer 263) Surigical removal of testes is termed as:
A)
castration
done
clear
B)
IUD
done
clear
C)
vasectomy
done
clear
D)
gastrula
done
clear
View Answer play_arrow
question_answer 264) The sex ratio in India in 2000 stands
A)
1000 males per 1200 females
done
clear
B)
1000 males per 965 females
done
clear
C)
1000 males per 1071 females
done
clear
D)
1000 males per 960 females
done
clear
View Answer play_arrow
question_answer 265) Angiospermic companion cell is associated with:
A)
sieve tube
done
clear
B)
vessels
done
clear
C)
tracheids
done
clear
D)
gymnospermic tracheids
done
clear
View Answer play_arrow
question_answer 266) Lateral roots are originated from:
A)
cortex
done
clear
B)
pericycle
done
clear
C)
endodermis
done
clear
D)
pith
done
clear
View Answer play_arrow
question_answer 267) Cricket bats are formed by the wood of plant:
A)
Pinus roxburghii
done
clear
B)
Morns alba
done
clear
C)
Cedrus deodara
done
clear
D)
Salix purpurea
done
clear
View Answer play_arrow
question_answer 268) Oil is obtained from which plant of Compositae family?
A)
Delbergia sissoo
done
clear
B)
Glycine max
done
clear
C)
Carthamus tinctorius
done
clear
D)
Brassica
done
clear
View Answer play_arrow
question_answer 269) Study of flower is called:
A)
Anthology
done
clear
B)
Spermology
done
clear
C)
Malicology
done
clear
D)
Agrostology
done
clear
View Answer play_arrow
question_answer 270) Which of the following inhibits the enzymatic reaction by changing in the structure of enzyme?
A)
Competitive inhibition
done
clear
B)
CoA
done
clear
C)
Irreversible inhibition
done
clear
D)
Allosteric inhibition
done
clear
View Answer play_arrow
question_answer 271) The light reaction of photosynthesis does not show:
A)
photolysis of water
done
clear
B)
release of \[{{H}_{2}}\]
done
clear
C)
release of \[{{O}_{2}}\]
done
clear
D)
electron transport
done
clear
View Answer play_arrow
question_answer 272) Epipetalous stamens, oblique septum and berry fruit is the characteristics of family:
A)
Solanaceae
done
clear
B)
Malvaceae
done
clear
C)
Cruciferae
done
clear
D)
Labiatae
done
clear
View Answer play_arrow
question_answer 273) Legume or pod is developed from:
A)
monocarpellary ovary
done
clear
B)
multicarpellary ovary
done
clear
C)
bicarpellary ovary
done
clear
D)
epigynous ovary
done
clear
View Answer play_arrow
question_answer 274) What will be the sequence of nucleotides in RNA if DNA have ATTGGC sequence?
A)
TAAGGG
done
clear
B)
UGATCA
done
clear
C)
ATAGCG
done
clear
D)
UAACCG
done
clear
View Answer play_arrow
question_answer 275) Which structure of enzyme help in active site formation?
A)
Quartemary
done
clear
B)
Tertiary
done
clear
C)
Secondary
done
clear
D)
Primary
done
clear
View Answer play_arrow
question_answer 276) Bulliform cells are found in:
A)
leaf of Wheat
done
clear
B)
pod of Pea
done
clear
C)
tuber of Potato
done
clear
D)
sped of Sunflower
done
clear
View Answer play_arrow
question_answer 277) If a pin is hammered in Mango tree above 2 metre of base, what will be the height of pin after 5 year:
A)
5 metre
done
clear
B)
3 metre
done
clear
C)
10 metre
done
clear
D)
2 metre
done
clear
View Answer play_arrow
question_answer 278) Endosperm of angiosperm is differ from gymnosperm because :
A)
endosperm forms before fertilization
done
clear
B)
it remains for some time only
done
clear
C)
endosperm does not form before fertilization
done
clear
D)
fat is present in angiosperm
done
clear
View Answer play_arrow
question_answer 279) Parasexuality is involved with:
A)
fusion of gamete and protoplast
done
clear
B)
fusion of male gamete with secondary nucleus
done
clear
C)
fusion of protoplast
done
clear
D)
fusion of male and female gamete
done
clear
View Answer play_arrow
question_answer 280) The flower divisible into two equal halves by a single vertical plane is called:
A)
zygomorphic
done
clear
B)
symmetrical
done
clear
C)
bilateral
done
clear
D)
actinomorphic
done
clear
View Answer play_arrow
question_answer 281) When pollen of a flower is transferred to stigma of other flower of same plant the phenomenon is called ?
A)
Allogamy
done
clear
B)
Autogamy
done
clear
C)
Dichogamy
done
clear
D)
Cleistogamy
done
clear
View Answer play_arrow
question_answer 282) The smallest flowering angiosperm is:
A)
Wolffia microsperma
done
clear
B)
Moms alba
done
clear
C)
Ricinus communis
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 283) How many types of gametes may be produced by genotype D/d: E/e: F/f?
A)
27
done
clear
B)
8
done
clear
C)
3
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 284) Jumping genes were discovered by:
A)
H.G. Khorana
done
clear
B)
Jacob and Monad
done
clear
C)
B. McClintock
done
clear
D)
Beadle and Tatum
done
clear
View Answer play_arrow
question_answer 285) Chromosome is consist of:
A)
DNA and ATP
done
clear
B)
DNA and histone
done
clear
C)
RNA and histone
done
clear
D)
DNA and RNA
done
clear
View Answer play_arrow
question_answer 286) Ions involved in photosystem-\[\text{II}\] are
A)
\[M{{n}^{++}}\] and \[C{{l}^{-}}\]
done
clear
B)
\[M{{g}^{+2}}\] and \[NO_{3}^{-}\]
done
clear
C)
\[F{{e}^{+2}}\] and \[C{{l}^{-}}\]
done
clear
D)
\[{{K}^{+}}\] and \[N{{a}^{+}}\]
done
clear
View Answer play_arrow
question_answer 287) Formation of \[NADP{{H}_{2}}\] in chloroplast takes place in:
A)
non-cyclic photophosphorylation
done
clear
B)
cyclic photophosphorylation
done
clear
C)
oxidative phosphorylation
done
clear
D)
substrate level phosphorylation
done
clear
View Answer play_arrow
question_answer 288) Xerophytic plant is:
A)
Brassica
done
clear
B)
Cuscuta
done
clear
C)
Hydrilla
done
clear
D)
Capparis
done
clear
View Answer play_arrow
question_answer 289) In fungus:
A)
absence of chlorophyII
done
clear
B)
presence of chlorophyll
done
clear
C)
presence of leucophyll
done
clear
D)
absence of leucophyu
done
clear
View Answer play_arrow
question_answer 290) Chitgoza obtains from:
A)
Cycos revolute
done
clear
B)
Cedrus
done
clear
C)
Zamia
done
clear
D)
Pinus gerardiana
done
clear
View Answer play_arrow
question_answer 291) Fucoxanthin is found in:
A)
Chlorophyceae
done
clear
B)
Phaeophyceae
done
clear
C)
Rhodophyceae
done
clear
D)
Bacillariophyceae
done
clear
View Answer play_arrow
question_answer 292) Cycas contains
A)
resin canal
done
clear
B)
mucilagenous canal
done
clear
C)
lactiferous canal
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 293) Mature sieve tube lacks:
A)
nucleus
done
clear
B)
nucleus and chloroplast
done
clear
C)
nucleolus and ER
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 294) The name cytokinin was proposed by:
A)
Letham (1963)
done
clear
B)
Miller (1964)
done
clear
C)
Skoog (1955)
done
clear
D)
both (a) and (c)
done
clear
View Answer play_arrow
question_answer 295) Which of the following transfer continuously from nucleus to cytoplasm?
A)
Cellulose
done
clear
B)
Glucose
done
clear
C)
RNA
done
clear
D)
DNA
done
clear
View Answer play_arrow
question_answer 296) The cell organdies involved in energy transformation are:
A)
chloroplast and mitochondria
done
clear
B)
chloroplast and leucoplast
done
clear
C)
golgi body and mitochondria
done
clear
D)
mitochondria and leucoplast
done
clear
View Answer play_arrow
question_answer 297) The process by which water enters into plant cell is called :
A)
diffusion
done
clear
B)
osmosis
done
clear
C)
endosmosis
done
clear
D)
both (b) and (c)
done
clear
View Answer play_arrow
question_answer 298) The life cycle of yeast is:
A)
diplobiontic
done
clear
B)
haplobiontic
done
clear
C)
haplodiplobiontic
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 299) Morphologically similar but physiologically dissimilar gametes are called :
A)
heterogamous
done
clear
B)
isogamous
done
clear
C)
anisogamous
done
clear
D)
syngamous
done
clear
View Answer play_arrow
question_answer 300) In which of the following pyrenoids are present?
A)
Mnrchantia
done
clear
B)
Riccia
done
clear
C)
Anthoceros
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 301) Autecology is related with:
A)
animal ecology
done
clear
B)
plant ecology
done
clear
C)
ecological study of an individual species
done
clear
D)
study of growing community
done
clear
View Answer play_arrow
question_answer 302) Liquid part of Coconut is called:
A)
male gametophyte
done
clear
B)
nucellus
done
clear
C)
endosperm
done
clear
D)
embryo
done
clear
View Answer play_arrow
question_answer 303) Which of the following is biofertilizor?
A)
Polyporus
done
clear
B)
Azotobactor
done
clear
C)
Slaginella
done
clear
D)
Bacillus
done
clear
View Answer play_arrow
question_answer 304) Drug ephedrine is obtained from:
A)
Ephedra
done
clear
B)
Cycas
done
clear
C)
Pinus
done
clear
D)
Taxus
done
clear
View Answer play_arrow
question_answer 305) Reindeer moss is:
A)
Cladonia rangifera
done
clear
B)
Funaria
done
clear
C)
Sphnyinm
done
clear
D)
Usnea
done
clear
View Answer play_arrow
question_answer 306) Red rot of Sugarcane is caused by:
A)
Helminthosporium oryzae
done
clear
B)
Colletorichum falcatum
done
clear
C)
Cercospora personata
done
clear
D)
Alternaria solani
done
clear
View Answer play_arrow
question_answer 307) If a tube is filled after one hour when rate of cell division is one division per minute. Then half of tube will filled in:
A)
30 minute
done
clear
B)
61 minute
done
clear
C)
59 minute
done
clear
D)
45 minute
done
clear
View Answer play_arrow
question_answer 308) Which of the following reaction takes place in Krebs cycle?
A)
Reduction of acetyle CoA into \[C{{O}_{2}}\] and \[{{H}_{2}}O\]
done
clear
B)
Oxidation of acetyle CoA into \[C{{O}_{2}}\] and \[{{H}_{2}}O\]
done
clear
C)
Oxidation of acetyle CoA with electron transport system into \[C{{O}_{2}}\] and \[{{H}_{2}}O\]
done
clear
D)
Oxidation of citric acid into \[{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 309) The correct statement is:
A)
there is no diploid structure in sporogonium of Riccia
done
clear
B)
sporogonium of Riccia is divided into foot, seta and capsule
done
clear
C)
chloroplast is present in sporogonium and spore mother cell of Riccia
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 310) Gossypium is an:
A)
bast fibre
done
clear
B)
pappus
done
clear
C)
outgrowth of testa
done
clear
D)
outgrowth of pericarp
done
clear
View Answer play_arrow
question_answer 311) Electron microscope was discovered by:
A)
Knoll and Ruska
done
clear
B)
Ivanowski
done
clear
C)
Lederberg and Tatum
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 312) The leaves of Mimosa pudica droops down when touch because:
A)
leaves has nervous system
done
clear
B)
leaves are very tender
done
clear
C)
the turgor pressure of leaf base changes
done
clear
D)
vascular bundle become dead
done
clear
View Answer play_arrow
question_answer 313) Angiopperm and gymnosperm both are similar in respect of:
A)
fertilization method
done
clear
B)
presence of ovule
done
clear
C)
presence of vessels
done
clear
D)
nature of endosperm
done
clear
View Answer play_arrow
question_answer 314) Root pressure is measured by:
A)
barometer
done
clear
B)
auxanometer
done
clear
C)
manometer
done
clear
D)
photometer
done
clear
View Answer play_arrow
question_answer 315) The living fossil is:
A)
Finns
done
clear
B)
Cedrus
done
clear
C)
Ginkgo
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 316) In snapdragon when red and white flowered plants are crossed \[{{F}_{1}}\] generation (is pink in colour of flower. Now \[{{F}_{1}}\] offspring is crossed with white flowered plant than off springs will be:
A)
ratio of red and white flower is 3 : 1
done
clear
B)
10% red flower
done
clear
C)
ratio of red, pink and white is 1 : 2 : 1
done
clear
D)
ratio of pink and white flowered plants is 1 : 1
done
clear
View Answer play_arrow
question_answer 317) Kaziranga is associated with:
A)
Rhinoceros
done
clear
B)
Tigers
done
clear
C)
Birds
done
clear
D)
Lions
done
clear
View Answer play_arrow
question_answer 318) Blue-green algae is found in:
A)
Cycas
done
clear
B)
Pinus
done
clear
C)
Scales of Marchantia
done
clear
D)
Rice in
done
clear
View Answer play_arrow
question_answer 319) Turpentine oil is obtained from:
A)
Pinus
done
clear
B)
Cycas
done
clear
C)
Cedrus
done
clear
D)
Petroleum
done
clear
View Answer play_arrow
question_answer 320) In gametophyte of fern:
A)
prothallus is independent from sporophyte
done
clear
B)
prothallus is attached with sporophyte
done
clear
C)
it is main plant body
done
clear
D)
a structure attached with rhizome
done
clear
View Answer play_arrow
question_answer 321) The lack of fruit formation in gymnosperm is due to:
A)
absence of fertilization
done
clear
B)
lack of seeds
done
clear
C)
naked ovule
done
clear
D)
sheding of immature seed
done
clear
View Answer play_arrow
question_answer 322) In Pteridium meiosis takes place at the time of:
A)
spore formation
done
clear
B)
organ formation in prothallus
done
clear
C)
spore germination
done
clear
D)
gamete formation
done
clear
View Answer play_arrow
question_answer 323) Club moss is the common name of:
A)
Selaginella
done
clear
B)
Funaria
done
clear
C)
Polaniogeton
done
clear
D)
Lycopodium
done
clear
View Answer play_arrow
question_answer 324) Pneumatophores (respiratory roots) are found in:
A)
Pistia
done
clear
B)
Cahbranchia
done
clear
C)
Rhizophora
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 325) All tissues consist of cells sentence was said by:
A)
Wallace
done
clear
B)
Virchow
done
clear
C)
Schleiden
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 326) The part of mushroom comes out from soil is called:
A)
basidiocarp
done
clear
B)
ascogonium
done
clear
C)
ascocarp
done
clear
D)
pericarp
done
clear
View Answer play_arrow
question_answer 327) The red colour of red sea is due to which of the following blue-green algae:
A)
Chlamydomonas nivalis
done
clear
B)
Anabaena
done
clear
C)
Microcystis
done
clear
D)
Trichodesmhim
done
clear
View Answer play_arrow
question_answer 328) The vascular bundle which have phloem in both side of xylem is called:
A)
Radial
done
clear
B)
Bicollateral
done
clear
C)
Collateral
done
clear
D)
Amphicribral
done
clear
View Answer play_arrow
question_answer 329) Bean is non-endospermic seed because :
A)
at the germination time, cotyledons come out from soil
done
clear
B)
endosperm is absent
done
clear
C)
embryo have two cotyledons with starch
done
clear
D)
endosperm is filled with albumin
done
clear
View Answer play_arrow
question_answer 330) Parthenogenesis takes place when:
A)
unfertilized egg develops into an embryo
done
clear
B)
embryo forms with the fusion of egg and sperm
done
clear
C)
embryo formation occurs from other cell
done
clear
D)
sperm forms the embryo
done
clear
View Answer play_arrow
question_answer 331) Which of the following is prokaryotes?
A)
Vaucheria
done
clear
B)
Ficus
done
clear
C)
Sargassum
done
clear
D)
Oscillatoria
done
clear
View Answer play_arrow
question_answer 332) Rauwolfiu serpentina is used for the treatment of:
A)
blood pressure
done
clear
B)
diabetes
done
clear
C)
lack of sleeping
done
clear
D)
paralysis
done
clear
View Answer play_arrow
question_answer 333) Caryopsis is the characteristic of family:
A)
Poaceae
done
clear
B)
Fabaceae
done
clear
C)
Asteraceae
done
clear
D)
Malvaceae
done
clear
View Answer play_arrow
question_answer 334) Extra chromosomal DNA is found in:
A)
golgi body and lysosome
done
clear
B)
chloroplast and mitochondria
done
clear
C)
plastid and ribosome
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 335) Enzymes used in glycolysis are found in:
A)
inner membrane of mitochondria
done
clear
B)
outer membrane of mitochondria
done
clear
C)
cytoplasm
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 336) In the glucose synthesis \[{{H}_{2}}\] obtains from:
A)
\[FAD{{H}_{2}}\]
done
clear
B)
\[NADP{{H}_{2}}\]
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
\[n(C{{H}_{2}}O)\]
done
clear
View Answer play_arrow
question_answer 337) In T.S. of root:
A)
metaxylem present inner side and protoxylem in in outer side
done
clear
B)
protoxylem present in inner side and metaxylem in outer side
done
clear
C)
protoxylem remains absent
done
clear
D)
protoxylem and metaxylem are not present on same radius
done
clear
View Answer play_arrow
question_answer 338) The reason of dropsy is:
A)
Ricinus communis
done
clear
B)
Argemon maxicana
done
clear
C)
Sunflower
done
clear
D)
Datura
done
clear
View Answer play_arrow
question_answer 339) The function of restriction enzyme is:
A)
cleavages of protein
done
clear
B)
cleavages of DNA
done
clear
C)
cleavages of fat
done
clear
D)
cleavages of starch
done
clear
View Answer play_arrow
question_answer 340) When a solute is dissolved in pure solvent, the change in diffusion pressure called:
A)
diffusion
done
clear
B)
DFD
done
clear
C)
osmosis
done
clear
D)
endosmosis
done
clear
View Answer play_arrow
question_answer 341) The correct statement is:
A)
perithecium is the fruiting body of Aspergillus
done
clear
B)
sexual reproduction of Albugo is oogamous
done
clear
C)
Ascomycetes are known as club fungi
done
clear
D)
sexual reproduction in Helminthosporium is oogamous
done
clear
View Answer play_arrow
question_answer 342) In pond ecosystem which of the following is not affected by temperature?
A)
Metalimnion
done
clear
B)
Epilimnion
done
clear
C)
Hypolimnion
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 343) Phylloclade is the modification of:
A)
root
done
clear
B)
petiole
done
clear
C)
stem
done
clear
D)
leaf
done
clear
View Answer play_arrow
question_answer 344) Rennin used in cheese industry is an:
A)
antibiotic
done
clear
B)
enzyme
done
clear
C)
inhibitor
done
clear
D)
alkaloid
done
clear
View Answer play_arrow
question_answer 345) Nucellus is found in:
A)
leaf
done
clear
B)
ovule
done
clear
C)
pollen
done
clear
D)
cell
done
clear
View Answer play_arrow
question_answer 346) Hormone used for ripening fruits is:
A)
ethylene
done
clear
B)
auxin
done
clear
C)
cytokinin
done
clear
D)
ABA
done
clear
View Answer play_arrow
question_answer 347) Cystolith is made up of:
A)
calcium oxide
done
clear
B)
calcium oxilate
done
clear
C)
calcium carbonate
done
clear
D)
calcium chloride
done
clear
View Answer play_arrow
question_answer 348) Maximum number of chromosomes are present in:
A)
Lycopodium
done
clear
B)
Ophioglossum
done
clear
C)
Triticum
done
clear
D)
Cycas
done
clear
View Answer play_arrow
question_answer 349) The fruit of orange is:
A)
pome
done
clear
B)
pepo
done
clear
C)
drupe
done
clear
D)
hesperidium
done
clear
View Answer play_arrow
question_answer 350) Rubber is obtained from:
A)
latex
done
clear
B)
wood
done
clear
C)
wool
done
clear
D)
Coconut epicarp
done
clear
View Answer play_arrow
question_answer 351) Tunica-corpus theory was proposed by:
A)
Hanstem
done
clear
B)
C. Nagelli
done
clear
C)
Schmidt
done
clear
D)
Skoog
done
clear
View Answer play_arrow
question_answer 352) Synapsis takes place in:
A)
pachytene
done
clear
B)
zygotene
done
clear
C)
diplotene
done
clear
D)
leptotene
done
clear
View Answer play_arrow
question_answer 353) The edible part of Cabbage is:
A)
stem
done
clear
B)
bud (leafy)
done
clear
C)
inflorescence
done
clear
D)
flower
done
clear
View Answer play_arrow
question_answer 354) In monocot grafting is impossible due to the presence of:
A)
open vascular bundles
done
clear
B)
closed vascular bundles
done
clear
C)
scattered vascular bundles
done
clear
D)
sclerenchymatus hypodermis
done
clear
View Answer play_arrow
question_answer 355) The algae used in space research is:
A)
Cephalcuros
done
clear
B)
Gelidium
done
clear
C)
Chlorella
done
clear
D)
Gracilaria
done
clear
View Answer play_arrow
question_answer 356) Water blooms are caused by:
A)
Chlamydomonas
done
clear
B)
Microcystis
done
clear
C)
Hydrodictyon
done
clear
D)
Oedogonium
done
clear
View Answer play_arrow
question_answer 357) The movement of pollen tube towards ovule is called:
A)
chemotropism
done
clear
B)
hydrotropism
done
clear
C)
thigmotropism
done
clear
D)
geotropism
done
clear
View Answer play_arrow
question_answer 358) Diakinesis is found in:
A)
metaphase-I of meiosis
done
clear
B)
prophase-I of meiosis
done
clear
C)
prophase-II of meiosis
done
clear
D)
metaphase of mitosis
done
clear
View Answer play_arrow
question_answer 359) The site of lipid synthesis is:
A)
smooth endoplasmic reticulum
done
clear
B)
rough endoplasmic reticulum
done
clear
C)
golgi body
done
clear
D)
free ribosomes
done
clear
View Answer play_arrow
question_answer 360) The father of Botany is:
A)
Theophrastus
done
clear
B)
Robert Hook
done
clear
C)
Louis Pasteur
done
clear
D)
Mendel
done
clear
View Answer play_arrow
question_answer 361) Botanical herbarium of India are classified according to classification of:
A)
Bentham and Hooker
done
clear
B)
Hutchinson
done
clear
C)
Engler and Pranti
done
clear
D)
Bauhin
done
clear
View Answer play_arrow
question_answer 362) Taxa is a:
A)
order
done
clear
B)
genera
done
clear
C)
taxonomic group of any rank
done
clear
D)
taxonomic groups below than species
done
clear
View Answer play_arrow
question_answer 363) Indian rubber is obtained from:
A)
Picas elastic
done
clear
B)
Mangifera indica
done
clear
C)
Citrus
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 364) If a plant species does not occur naturally. That will be classified in Red Data Book as a/an:
A)
extinct
done
clear
B)
rare
done
clear
C)
endangered
done
clear
D)
commonly found
done
clear
View Answer play_arrow
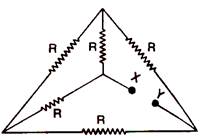
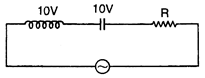
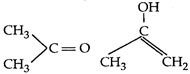
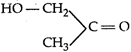
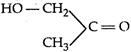
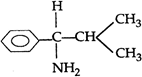 IUPAC name is:
IUPAC name is:
