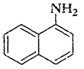
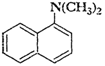
 (iii)
(iii)
question_answer27)
Which
of the following is the weakest Bronsted base?
(a) ![]() (b)
(b) ![]() (c)
(c)
![]() (d)
(d) ![]()
question_answer35)
Hofmann
Bromamide Degradation reaction is shown by..............
(a)![]() (b)
(b)![]() (c)
(c)![]() (d)
(d)![]()
question_answer37)
Methylamine
reacts with ![]() to form ?.
(a)
to form ?.
(a) ![]() (b)
(b) ![]() (c)
(c) ![]() (d)
(d) ![]()
question_answer46)
Which
of the following compounds is the weakest Bronsted base?
(a) 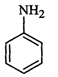 (b)
(b) ![]() (c)
(c)
![]() (d)
(d) ![]()
question_answer55)
The
product of the following reaction is...........
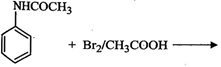 (a)
(a) 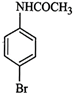 (b)
(b) 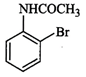 (c)
(c)
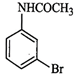 (d)
(d) 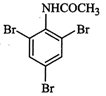
question_answer58)
Which
of the following reactions are correct?
(a) ![]() (b)
(b) ![]() (c)
(c) ![]() (d)
(d) ![]()
question_answer61)
What
is the role of ![]() in the nitrating
mixture used for nitration of benzene?
in the nitrating
mixture used for nitration of benzene?
question_answer62)
Why
is ![]() group of aniline acetylated
before carrying out nitration?
group of aniline acetylated
before carrying out nitration?
question_answer63)
What
is the product when ![]() reacts with
reacts with ![]() ?
?
question_answer64) What is the best reagent to convert nitrile to primary amine?
View Answer play_arrow question_answer65)
Give
the structure of 'A' in the following reaction.
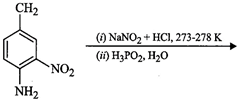
question_answer66) What is Hinsberg reagent?
View Answer play_arrow question_answer68)
Why
does acetylation of ![]() group of aniline
reduce its activating effect?
group of aniline
reduce its activating effect?
question_answer69)
Explain
why ![]() is stronger base than MeOH?
is stronger base than MeOH?
question_answer70) What is the role of pyridine in the acylation reaction of amines?
View Answer play_arrow question_answer73)
Arrange
the following compounds in increasing order of dipole moment.
![]()
question_answer74) What is the structure and IUPAC name of the compound, allyl amine?
View Answer play_arrow question_answer75)
Write
down the IUPAC name of
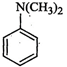
question_answer78)
Complete
the following reaction.
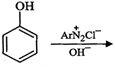
question_answer79) Why is aniline soluble in aqueous HCl?
View Answer play_arrow question_answer80)
Suggest
a route by which the following conversion can be accomplished.
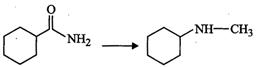
question_answer81)
Identify
A and B in the following
reaction.
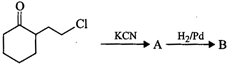
question_answer85)
How
will you bring out the following conversion?
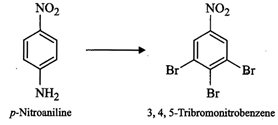
question_answer86)
How
will you carry out the following conversion?
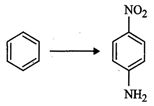
question_answer87)
How
will you carry out the following conversion?
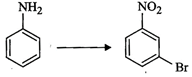
question_answer88)
How
will you carry out the following conversions?
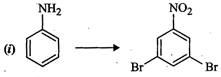
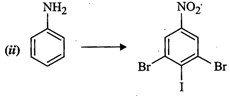
question_answer89) Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines |
| (iii) | Hermann Bromamidt reaction | (c) | Reaction of phthalimide with KOH and R?X |
| (iv) | Carbylamine reaction | (d) |
Reaction
of alkyl halides with |
question_answer90) Match the compounds given in Column I with, the items given in Column II.
| Column I | Column II | ||
| (i) | Benzenesulphonyl chloride | (a) | Zwitterion |
| (ii) | Sulphanilic acid | (b) | Hinsberg reagent |
| (iii) | Alkyldiazonium salts | (c) | Dyes |
| (iv) | Aryldiazonium salts | (d) | Conversion to alcohols |
question_answer100)
Predict
the reagent or the product in the following reaction sequence.
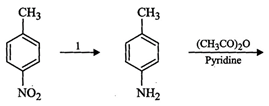
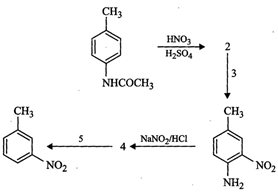
You need to login to perform this action.
You will be redirected in
3 sec
