-
Write the formulas of the following coordination compounds:
(a) Tetraamminediaquacobalt (III) chloride
(b) Potassium tetracyanonickelate (II)
(c) Tris(ethane-1, 2-diamine)platinum(II) nitrate
(d) Amminebromidochloridonitrito-N-platinate (II)
(e)Dichloridobis (ethane-1, 2-diamine) platinum (IV) nitrate
(g) Iron(III) haxacyanoferrate(II).
View Answer play_arrow
-
Write IUPAC names of the following coordination compounds :
(a)[Co(NH)6] Cl3 (b) [Co(NH3)5Cl] Cl2
(c) K3 [Fe(CN)6] (d) K3 [Fe(C2O4)3]
(e) K2 [PdCl4] (f) [Pt(NH3)2 CI(NH2CH3)] Cl
View Answer play_arrow
-
Indicate the types of isomerism exhibited by the following complexes and draw the structures of these isomers :
(a)K [Cr(H2O)2 (C2O4)2]
(b) [Co(en)3] Cl3
(c) [Co(NH3)5(NO2)] (NO3)2
(d) [Pt (NH3) (H2O) Cl2]
View Answer play_arrow
-
Give evidence that [Co(NH3)5Cl] SO4 and [Co(NH3)5 SO4] Cl are ionisation isomers
View Answer play_arrow
-
Explain on the basis of valence bond theory that [Ni(CN4)]2– ion with square planar structure is damagnetic and the [NiCl4]2– ion with tetrahedral geometry is paramagnetic.
View Answer play_arrow
-
[NiCl4]2– is paramagnetic while [Ni(CO)4] is diamagnetic though both are tetrahedral. Why?
View Answer play_arrow
-
[Fe(H2O)6]3+ is strongly paramagnetic whereas [Fe(CN)6]3– is weakly paramagnetic. Explain.
View Answer play_arrow
-
Explain [Co(NH3)6]3+
is an inner orbital complex whereas 
is
an outer orbital complex.
View Answer play_arrow
-
Predict the number of unpaired electrons in the square planner [Pt(CN)4]2–?ion.
View Answer play_arrow
-
The hexaaquamanganese(II) ion contains five unpaired electrons while the hexacyano ion contains only one unpaired electron. Explain using Crystral field theory.
View Answer play_arrow
-
Explain the bonding in coordination compounds in terms of Werner’s postulates.
View Answer play_arrow
-
FeSO4 solution mixed with (NH4)2SO4 solution in the molar ratio 1 : 1 gives the test of Fe2+ ion but CuSO4 solution mixed with aqueous NH3 in the molar ratio of 1 : 4 does not give the test of Cu2+. Explain why.
View Answer play_arrow
-
Explain with two examples each of the following: Coordination entity, ligands, coordination number, coordination polyhedron, homoleptic and heteroleptic complex.
View Answer play_arrow
-
What is meant by unidentate, didentate and ambidentate ligands? Give two examples for each.
View Answer play_arrow
-
Specify the oxidation numbers of the metals in the following coordination entities :
(i) [Co(H2O)(CN) (en)2]2+ (ii) [CoBr2(en)2]+
(iii) [PtCl4]2- (iv) K3[Fe(CN)6]
(v) [Cr(NH3)3Cl3]
View Answer play_arrow
-
Using IUPAC norms write the formulae for the following :
(i) tetrahydroxozineate(II)
(ii) hexaamminecobalt(III) sulphate
(iii) potassiumtetrachloridopalladate(II)
(iv) potassium tri (oxalato) chromate (III)
(v) diamminedichloridoplatinum(II)
(vi) hexaammineplatinum (IV)
(vii)potassiumtetracyanonickelate (II)
(viii) tetrabromidocuprate(II)
(ix) pentaamminenitrito-O-cobalt(III)
(x) pentaamminenitrito-N-cobalt(III).
View Answer play_arrow
-
Using IUPAC norms write the systematic names of the following :
(i)[Co(NH3)6]Cl3
(ii) [Pt(NH3)2Cl (NH2CH3)]Cl
(iii) Ti(H2O)6]3+
(iv) [Co(NH3)4Cl(NO2)] Cl
(v) [Mn(H2O)6]2+
(vi) [NiCl4]2–
(vii) [Ni(NH3)6]Cl2
(viii) [Co(en)3]3+
(ix) Ni(CO)4
View Answer play_arrow
-
List various types of isomerism possible for coordination compounds, giving an example of each.
View Answer play_arrow
-
How many geometric isomers are possible in the following coordination entity :
(i) [Cr(C2O4)3 ]3–
(ii) [Co(NH3)3Cl3]
View Answer play_arrow
-
Draw the structures of optical isomers of
(a) [Cr(C2O4)3]3– (b) [PtCl2(en)2]2+ (c) [Cr(NH3)2Cl2(en)]+
View Answer play_arrow
-
Draw the structures of optical isomers of
(a) [Cr(C2O4)3]3– (b) [PtCl2(en)2]2+ (c) [Cr(NH3)2Cl2(en)]+
View Answer play_arrow
-
Write all the geometrical isomers of [Pt(NH3)BrCI(py)] and how many of these will exhibit optical isomerism?
View Answer play_arrow
-
Write all the geometrical isomers of [Pt(NH3)BrCI(py)] and how many of these will exhibit optical isomerism ?
View Answer play_arrow
-
What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution?
View Answer play_arrow
-
Discuss the nature of bonding in the following coordination entities on the basis of valence bond theory?
(i) Fe(CN)6]4– (ii)[FeF6]3–
(iii) [Co(C2O4)3]3– (iv) [CoF6]3–
View Answer play_arrow
-
Draw figure to show splitting of degenerate d orbitals in an octahedral crystal field.
View Answer play_arrow
-
What is spectrochemical series? Explain the difference between a weak field ligand and a strong field ligand.
View Answer play_arrow
-
What is
crystal field splitting energy? How does the magnitude of 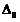 decide
the actual configuration of d-orbital in a coordination entity?
decide
the actual configuration of d-orbital in a coordination entity?
View Answer play_arrow
-
[Cr(NH3)6]3+ is paramagneticwhile [Ni(CN)4]2– is diamagnetic. Explain why?
View Answer play_arrow
-
A solution of [Ni(H2O)6]2+ is green but a solution of [Ni(CN)4]2– is colourless. Explain.
View Answer play_arrow
-
[Fe(CN)6]4– and [Fe(H2O)6]2+ are of different colours in dilute solutions. Why?
View Answer play_arrow
-
Discuss the nature of bonding in metal, carbonyls.
View Answer play_arrow
-
Give the oxidation state, d-orbital occupation and coordination number of central metal ion in the following complexes.
(a) K3[Co(C2O4)3] (b) (NH4)2[CoF4]
(c) cis – [Cr(en)2Cl2] CI (d) [Mn(H2O)6]SO4
View Answer play_arrow
-
Write down the IUPAC name for each of the following complexes and indicate the oxidation state, electronic configuration and coordination number. Also give stereochemistry and magnetic moment of the complex :
(a) K [Cr(H2)2(C2O4)2].3H2O
(b) [CrCl3(py)3]
(c) K4 [Mn(CN)6]
(d) [Co(NH3)5Cl] Cl2
(e) Cs[FeCl4]
View Answer play_arrow
-
What is meant by stability of a coordination compound in solution? State the factors which govern stability of complexes.
View Answer play_arrow
-
What is meant by Chelate effect? Given an example.
View Answer play_arrow
-
Discuss briefly the role of coordination compounds in: (i) biological systems (ii) analytical chemistry (iii) medicinal chemistry, and (iv) extraction/metallurgy of metals.
View Answer play_arrow
-
How many ions are produced from the complex Co(NH3)6Cl2 in solution ?
(i) 6 (ii) 4 (iii) 3 (iv) 2
View Answer play_arrow
-
Amongst the following ions, which one has the highest magnetic moment?
(i) [Cr(H2O)6]3+
(ii)[Fe(H2O)62+
(iii) [Zn(H2O)6]2+
View Answer play_arrow
-
The oxidation number of cobalt in K[Co(CO)4] is
(i) + 1 (ii) +3 (iii) –1 (iv) –3.
View Answer play_arrow
-
Amongst the following, the most stable complex is :
(i) [Fe(H2O)6]3+ (iii) [Fe(C2O4)3]3– (iv) [FeCI6]3+
View Answer play_arrow
-
What will be the correct order for the wavelengths of absorption in the visible region of the following :
[Ni(NO2)6]4–, [Ni(NH3)6]2+, [Ni(H2O)6 ]2+
View Answer play_arrow
-
question_answer43)
Which
of the following complexes formed by 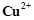 ions is most stable?
(a)
ions is most stable?
(a)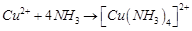 , log k =
6
(b)
, log k =
6
(b) 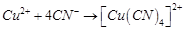 , log k = 27.3
(c)
, log k = 27.3
(c) 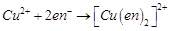 , log K=15.4
(d)
, log K=15.4
(d) 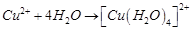 , log K = 8.9
, log K = 8.9
View Answer play_arrow
-
question_answer44)
The
colour of the coordination compounds depends on the crystal field splitting.
What will be the correct order of absorption of wavelength of light in the
visible region, for the complexes,
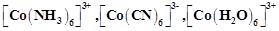 (a)
(a) 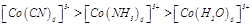 (b)
(b)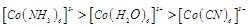 (c)
(c) 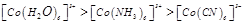 (d)
(d) 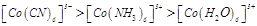
View Answer play_arrow
-
question_answer45)
When
0.1 mol 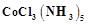 is treated with excess of
is treated with excess of 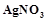 ,
0.2 mol of
,
0.2 mol of 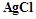 are obtained. The conductivity of solution will correspond
to
(a) 1 : 3 electrolyte (b) 1 : 2 electrolyte
(c) 1 : 1 electrolyte (d) 3 : 1 electrolyte
are obtained. The conductivity of solution will correspond
to
(a) 1 : 3 electrolyte (b) 1 : 2 electrolyte
(c) 1 : 1 electrolyte (d) 3 : 1 electrolyte
View Answer play_arrow
-
question_answer46)
When
1 mole of 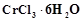 is treated with excess of
is treated with excess of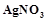 ,
3 moles of
,
3 moles of 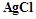 are obtained. The formula of the complex is
(a)
are obtained. The formula of the complex is
(a) 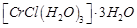 (b)
(b) 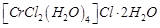 (c)
(c) 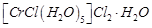 (d)
(d) 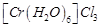
View Answer play_arrow
-
question_answer47)
The
correct IUPAC name of 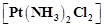 is
(a) Diamminedichloridoplatinum (II)
(b) Diamminedichloridoplatinum (IV)
(c) Diamminedichloridoplatinum (0)
(d) Dichloridodiammineplatinum (IV)
is
(a) Diamminedichloridoplatinum (II)
(b) Diamminedichloridoplatinum (IV)
(c) Diamminedichloridoplatinum (0)
(d) Dichloridodiammineplatinum (IV)
View Answer play_arrow
-
question_answer48)
The
stabilisation of coordination compounds due to chelation is called the chelate
effect. Which of the following is the most stable complex species?
(a) 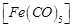 (b)
(b)
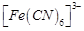 (c)
(c) 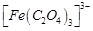 (d)
(d)
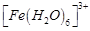
View Answer play_arrow
-
question_answer49)
Indicate
the complex ion which shows geometrical isomerism.
(a) 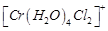 (b)
(b) 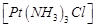 (c)
(c) 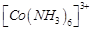 (d)
(d) 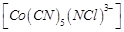
View Answer play_arrow
-
question_answer50)
The
CFSE for octahedral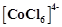 . is 18,000
. is 18,000  . The CFSE for tetrahedral
. The CFSE for tetrahedral  -willbe
(a) 18,000 cm-1 (b) 16,000 cm-1
(c) 8,000 cm-1 (d) 20,000 cm-1
-willbe
(a) 18,000 cm-1 (b) 16,000 cm-1
(c) 8,000 cm-1 (d) 20,000 cm-1
View Answer play_arrow
-
question_answer51)
Due
to the presence of ambidentate ligands coordination compounds show isomerism.
Palladium complexes of the type 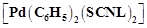 and
and
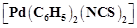 are
(a) linkage isomers
(b) coordination isomers
(c) ionisation isomers
(d) geometrical isomers
are
(a) linkage isomers
(b) coordination isomers
(c) ionisation isomers
(d) geometrical isomers
View Answer play_arrow
-
question_answer52)
The
compound 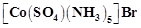 and
and
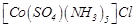
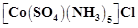 represent
(a) linkage isomerism
(b) ionisation isomerism
(c) coordination isomerism
(d) no isomerism
represent
(a) linkage isomerism
(b) ionisation isomerism
(c) coordination isomerism
(d) no isomerism
View Answer play_arrow
-
question_answer53)
A
chelating agent has two or more than two donor atoms to bind to a single metal
ion. Which of the following is not a chelating agent?
(a) Thiosulphato (b) Qxalato
(c) Glycinato (d) Ethane-1, 2-diamine
View Answer play_arrow
-
question_answer54)
Which
of the following species is not expected to be a ligand?
(a) NO (b) 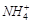 (c)
(c) 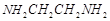 (d)
CO
(d)
CO
View Answer play_arrow
-
question_answer55)
What
kind of isomerism exists between 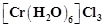 (violet) and
(violet) and
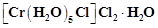 (Greyish-green)?
(a) Linkage isomerism
(b) Solvate isomerism
(c) Ionisation isomerism
(d) Coordination isomerism
(Greyish-green)?
(a) Linkage isomerism
(b) Solvate isomerism
(c) Ionisation isomerism
(d) Coordination isomerism
View Answer play_arrow
-
question_answer56)
IUPAC
name of is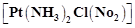 (a) Platinum diaminechloronitrite
(b) Chloronitrito-N-ammineplatinum (II)
(c) Diamminechloridonitrito-N-platinum (II)
(d) Diamminechloronitrito-N-platinate (II)
(a) Platinum diaminechloronitrite
(b) Chloronitrito-N-ammineplatinum (II)
(c) Diamminechloridonitrito-N-platinum (II)
(d) Diamminechloronitrito-N-platinate (II)
View Answer play_arrow
-
question_answer57)
Atomic
number of Mn. Fe and Co are 25, 26 and 27 respectively. Which of the following
inner orbital octahedral complex ions are diamagnetic?
(a) 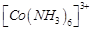 (b)
(b)
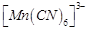 (c)
(c) 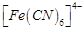 ( d)
( d)
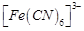
View Answer play_arrow
-
question_answer58)
Atomic
number of Mn, Fe, Co and Ni are 25, 26, 27 and 28 respectively. Which of the
following outer orbital octahedral complexes have same number of unpaired
electrons?
(a) 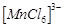 (b)
(b)
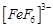 (c)
(c) 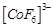 (d)
(d)
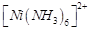
View Answer play_arrow
-
question_answer59)
Which
of the following options are correct for 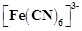 complex?
(a)
complex?
(a) 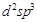 hybridisation
(b)
hybridisation
(b) 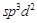 hybridisation
(c) Paramagnetic
(d) Diamagnetic
hybridisation
(c) Paramagnetic
(d) Diamagnetic
View Answer play_arrow
-
question_answer60)
An
aqueous pink solution of cobalt (II) chloride changes to deep blue on addition
of excess of HCl. This is because..........
(a) 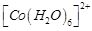 is
transformed into
is
transformed into
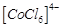 (b)
(b) 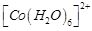 is
transformed into
is
transformed into
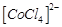 (c) tetrahedral complexes have smaller crystal field
splitting than octahedral complexes
(d) tetrahedral complexes have larger crystal field
splitting than octahedral complex
(c) tetrahedral complexes have smaller crystal field
splitting than octahedral complexes
(d) tetrahedral complexes have larger crystal field
splitting than octahedral complex
View Answer play_arrow
-
question_answer61)
Which
of the following complexes are homoleptic?
(a) 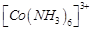 (b)
(b)
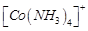 (c)
(c)  (d)
(d)
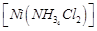
View Answer play_arrow
-
question_answer62)
Which
of the following complexes are heteroleptic?
(a) 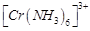 (b)
(b)
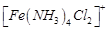 (c)
(c) 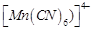 (d)
(d)
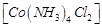
View Answer play_arrow
-
question_answer63)
Identify
the optically active compounds from the following
(a) 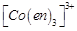 (b)
trans -
(b)
trans - 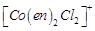 (c)
(c) 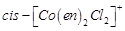 (d)
(d) 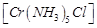
View Answer play_arrow
-
question_answer64)
Identify
the correct statements for the behaviour of ethane-1, 2- diamine as a ligand.
(a) It is a neutral ligand
(b) It is a didentate ligand
(c) It is a chelating ligand
(d) It is a unidentate ligand
View Answer play_arrow
-
question_answer65)
Which
of the following complexes show linkage isomerism?
(a) 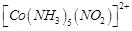 (b)
(b) 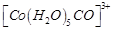 (c)
(c) 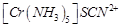 (d)
(d) 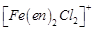
View Answer play_arrow
-
question_answer66)
Arrange
the following complexes in the increasing order of conductivity of their
solution
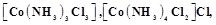
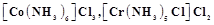
View Answer play_arrow
-
question_answer67)
A
coordination compound 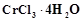 precipitates silver chloride when treated with silver
nitrate. The molar conductance of its solution corresponds to a total of two
ions. Write structural formula of the compound and name it.
precipitates silver chloride when treated with silver
nitrate. The molar conductance of its solution corresponds to a total of two
ions. Write structural formula of the compound and name it.
View Answer play_arrow
-
question_answer68)
A
complex of the type 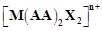 is known to be optically active.
What does this indicate about the structure of the complex?
Give one example of such complex.
is known to be optically active.
What does this indicate about the structure of the complex?
Give one example of such complex.
View Answer play_arrow
-
question_answer69)
Magnetic
moment of 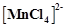 is 5.92 BM. Explain giving reason present.
is 5.92 BM. Explain giving reason present.
View Answer play_arrow
-
question_answer70)
On
the basis of crystal field theory explain why Co (III) forms paramagnetic octahedral
complex with weak field ligands whereas it forms diamagnetic octahedral complex
with strong field ligands.
View Answer play_arrow
-
question_answer71)
Why
are low spin tetrahedral complexes not formed?
View Answer play_arrow
-
question_answer72)
Give
the electronic configuration of the following complexes on the basis of crystal
field splitting theory.
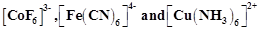 .
.
View Answer play_arrow
-
question_answer73)
Explain
why 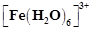 has magnetic moment value of 5.92 BM whereas
has magnetic moment value of 5.92 BM whereas 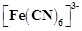 has
a value of only 1.74 BM?
has
a value of only 1.74 BM?
View Answer play_arrow
-
question_answer74)
Arrange
following complex ions in increasing order of crystal field splitting energy (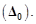
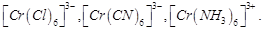
View Answer play_arrow
-
question_answer75)
Why
do compounds having similar geometry have different magnetic moment?
View Answer play_arrow
-
question_answer76)
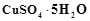 is blue in colour while
is blue in colour while 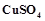 is
colourless. Why?
is
colourless. Why?
View Answer play_arrow
-
question_answer77)
Name
the type of isomerism when ambidentate ligands are attached to central metal
ion. Give two examples of ambidentate ligands.
View Answer play_arrow
-
question_answer78)
Match
the complex ions given in Column I with the colours given in Column II and
assign the correct code.
|
|
Column I (Complex ion)
|
Column II (Colour)
|
|
A.
|
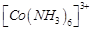
|
1. Violet
|
|
B.
|
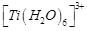
|
2. Green
|
|
C.
|
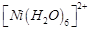
|
3. Pale blue
|
|
D.
|
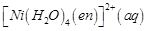
|
4. Yellowish orange
|
Codes
A B C D A B C D
(a) 1 2 4 5 (b) 4 3 2 1
(c) 3 2 4 1 (d) 4 1 2 3
View Answer play_arrow
-
question_answer79)
Match
the coordination compounds given in Column I with the central metal atoms given
in Column II and assign the correct code.
|
|
Column I (Coordination compound)
|
Column II (Central metal atom)
|
|
A.
|
Chlorophyll
|
1. Rhodium
|
|
B.
|
Blood pigment
|
2. Cobalt
|
|
C.
|
Wilkinson catalyst
|
3. Magnesium
|
|
D.
|
Vitamin 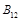
|
4. Iron
|
Codes
A B C D A B C D
(a) 3 4 1 2 (b) 3 4 5 1
(c) 4 3 2 1 (d) 3 4 1 2
View Answer play_arrow
-
question_answer80)
Match
the complex ions given in Column I with the hybridisation and number of
unpaired electrons given in Column II and assign the correct code.
|
Column I
(Complex ion)
|
Column II (Hybridisation, number of unpaired electrons)
|
A. 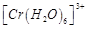 B.
B. 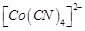 C.
C. 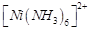 D.
D. 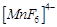
|
1. 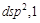 2.
2. 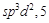 3.
3. 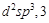 4.
4. 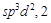
|
Codes
A B C D A B C D
(a) 3 1 4 2 (b) 4 3 2 1
(c) 3 2 4 1 (d) 4 1 2 3
View Answer play_arrow
-
question_answer81)
Match
the complex species given in Column I with the possible isomerism given in
Column II and assign the correct code.
|
|
Column 1 (Complex species)
|
Column 11 (Isomerism)
|
|
A.
B.
C.
D.
|
  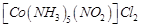 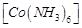 
|
1. Optical
2. lonisation
3. Coordination
4. Geometrical
|
Codes
A B C D A B C D
(a) 1 2 4 3 (b) 4 3 2 1
(c) 4 2 1 3 (d) 4 1 2 3
View Answer play_arrow
-
question_answer82)
Match
the compounds given in Column I with the oxidation state of cobalt present in
it (given in column II) and assign the correct code.
|
Column I
(Compound)
|
Column II
(Oxidation state of Co)
|
A. 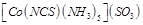
|
1. +4
|
B. 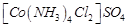
|
2. 0
|
C. 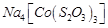
|
3. +2
|
D. 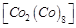
|
4. +3
|
Code
.
A B C D A B C D
(a) 1 2 4 3 (b) 4 3 2 1
(C) 3 1 4 2 (d) 4 1 3 2
View Answer play_arrow
-
question_answer83)
Assertion (A) Toxic metal ions are removed by the chelating ligands.
Reason (R) Chelate
complexes tend to be more stable.
View Answer play_arrow
-
question_answer84)
Assertion (A) 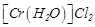 and
and 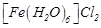 are reducing in nature.
Reason (R) Unpaired
electrons are present in their d-orbitals.
are reducing in nature.
Reason (R) Unpaired
electrons are present in their d-orbitals.
View Answer play_arrow
-
question_answer85)
Assertion (A) Linkage isomerism arises in coordination compounds containing
ambidentate ligand.
Reason (R) Ambidentate
ligand has two different donor atoms.
View Answer play_arrow
-
question_answer86)
Assertion (A) Complexes of 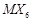 and
and 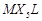 type (X and L are unidentate) do. not show geometrical
isomerism.
Reason (R) Geometrical
isomerism is not shown by complexes of coordination number 6.
type (X and L are unidentate) do. not show geometrical
isomerism.
Reason (R) Geometrical
isomerism is not shown by complexes of coordination number 6.
View Answer play_arrow
-
question_answer87)
Assertion (A) 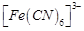 ion shows magnetic moment corresponding to two unpaired
electrons.
Reason (R) Because it
has
ion shows magnetic moment corresponding to two unpaired
electrons.
Reason (R) Because it
has 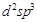 type hybridisation.
type hybridisation.
View Answer play_arrow
-
question_answer88)
Using
crystal field theory, draw energy level diagram, write electronic configuration
of the central metal atom/ion and determine the magnetic moment value in the
following
(a) 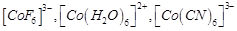 (b)
(b) 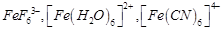
View Answer play_arrow
-
question_answer89)
Using
valence bond theory, explain the following in relation to the complexes given
below
 (a) Type of hybridisation
(b) Inner or outer orbital complex
(c) Magnetic behaviour
(d) Spin only magnetic moment value.
(a) Type of hybridisation
(b) Inner or outer orbital complex
(c) Magnetic behaviour
(d) Spin only magnetic moment value.
View Answer play_arrow
-
question_answer90)
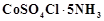 exists in two isomeric forms 'A' and 'S. Isomer 'A' reacts
with
exists in two isomeric forms 'A' and 'S. Isomer 'A' reacts
with 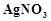 to give white precipitate, but does not react with
to give white precipitate, but does not react with 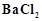 .
Isomer 'B' gives white precipitate with
.
Isomer 'B' gives white precipitate with 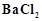 but does not react with
but does not react with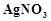 .
Answer the following questions.
(a) Identify 'A' and 'B' and write their structural
formulae.
(b) Name the type of isomerism involved.
(c) Give the IUPAC name of 'A' and 'B'.
.
Answer the following questions.
(a) Identify 'A' and 'B' and write their structural
formulae.
(b) Name the type of isomerism involved.
(c) Give the IUPAC name of 'A' and 'B'.
View Answer play_arrow
-
question_answer91)
What
is the relationship between observed colour of the complex and the wavelength
of light absorbed by the complex?
View Answer play_arrow
-
question_answer92)
Why
are different colours observed in octahedral and tetrahedral complexes for the
same metal and same ligands?
View Answer play_arrow
![]() (b)
(b) ![]() (c)
(c) ![]() (d)
(d) ![]()
![]() (b)
(b)
![]() (c)
(c) ![]() (d)
(d)
![]()
![]() (b)
(b)
![]() (c)
(c) ![]() (d)
(d)
![]()
![]() (b)
(b) ![]() (c)
(c) ![]() (d)
(d) ![]()
![]() is 5.92 BM. Explain giving reason present.
is 5.92 BM. Explain giving reason present.
![]() is blue in colour while
is blue in colour while ![]() is
colourless. Why?
is
colourless. Why?
