-
Why are pentahalides more covalent than trihalides?
View Answer play_arrow
-
Why is BiH3 the strongest reducing agent amongst all the hydrides of Groups 15 elements?
View Answer play_arrow
-
Why is N2 less reactive at room temperature?
View Answer play_arrow
-
Mention the conditions to maximise the yield of ammonia.
View Answer play_arrow
-
How does ammonia react with a solution of Cu2+?
View Answer play_arrow
-
What is the
covalency of nitrogen in  ?
?
View Answer play_arrow
-
Bond angle in PH4+ is higher than in PH3. Why?
View Answer play_arrow
-
What happens when white phosphorus is heated with concentrated NaOH solution in the inert atmosphere of CO2?
View Answer play_arrow
-
What happens when PCI5 is heated?
View Answer play_arrow
-
What is the basicity of H3PO4?
View Answer play_arrow
-
What happens when H3PO3 is heated?
View Answer play_arrow
-
List the important sources of sulphur.
View Answer play_arrow
-
Write the order of thermal stability of the hydrides of group 16 elements.
View Answer play_arrow
-
Why is H2O liquid and H2S is a gas?
View Answer play_arrow
-
Which of the following does not react with oxygen directly?
Zn, Ti, Pt, Fe
View Answer play_arrow
-
Complete the
following reactions:
(i) C2H4
+ O2 (ii) 4 Al + 3O2
(ii) 4 Al + 3O2

View Answer play_arrow
-
Why does ozone act as a powerful oxidising agent?
View Answer play_arrow
-
How is O3 estimated quantitatively?
View Answer play_arrow
-
What happens when sulphur dioxide is passed into aqueous solution of Fe(III) salt?
View Answer play_arrow
-
Comment upon the nature of two S – O bonds formed in SO2 molecules? Are the two S-O bonds in this molecules equal?
View Answer play_arrow
-
How is the presence of SO2 detected?
View Answer play_arrow
-
Mention three areas in which H2SO4 plays an important role.
View Answer play_arrow
-
Write the conditions to maximize the yield of H2SO4 by contact process.
View Answer play_arrow
-
Why is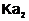 less than
less than
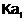 for H2SO4
in water?
for H2SO4
in water?
View Answer play_arrow
-
Considering the parameters such as bond dissociation enthalpy, electron gain enthalpy and hydration energy, compare the oxidizing power of F2 and Cl2.
View Answer play_arrow
-
Give two examples to show the anomalous behaviour of fluorine.
View Answer play_arrow
-
Sea is the greatest source of halogens. Comment.
View Answer play_arrow
-
Give the reason for the bleaching action of Cl2.
View Answer play_arrow
-
Name two poisonous gases which can be prepared from chlorine gas.
View Answer play_arrow
-
Why is 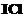 more reactive than
I2?
more reactive than
I2?
View Answer play_arrow
-
Why is helium used in diving apparatus?
View Answer play_arrow
-
Balance the
following equation
XeF6 + H2O

View Answer play_arrow
-
Why has it been difficult to study the chemistry of radon?
View Answer play_arrow
-
Discuss the general characteristics of group 15 elements with reference to their electronic configuration, oxidation state, atomic size, ionisation enthalpy and electronegativity.
View Answer play_arrow
-
question_answer35)
On
addition of cone. 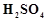 to a chloride salt, colourless fumes are evolved but in
case of iodide salt, violet fumes come out. This is because
(a)
to a chloride salt, colourless fumes are evolved but in
case of iodide salt, violet fumes come out. This is because
(a) 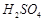 reduces
HI to
reduces
HI to 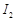 (b) HI is of violet colour,
(c) HI gets oxidised to
(b) HI is of violet colour,
(c) HI gets oxidised to 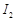 (d) HI changes to
(d) HI changes to 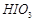
View Answer play_arrow
-
question_answer36)
In
qualitative analysis when 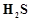 is passed through an aqueous solution of salt acidified
with dil. HCI, a black precipitate is obtained. On boiling the precipitate with
dil.
is passed through an aqueous solution of salt acidified
with dil. HCI, a black precipitate is obtained. On boiling the precipitate with
dil. 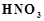 , it forms a solution of blue colour. Addition of excess of
aqueous solution of ammonia to this solution gives..........
(a) deep blue precipitate of
, it forms a solution of blue colour. Addition of excess of
aqueous solution of ammonia to this solution gives..........
(a) deep blue precipitate of 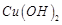 (b) deep blue solution of
(b) deep blue solution of 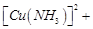 (c) deep blue solution of
(c) deep blue solution of 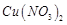 (d) deep blue solution-of
(d) deep blue solution-of
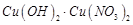
View Answer play_arrow
-
question_answer37)
In
a cydotrimetaphosphoric add molecule, how many single and double bonds are
present?
(a) 3 double bonds; 9 single bonds
(b) 6 double bonds; 6 single bonds
(c) 3 double bonds; 12 single bonds
(d) Zero double bond; 12 single bonds
View Answer play_arrow
-
question_answer38)
Which
of the following-elements can be involved in pit-die bonding?
(a) Carbon (b) Nitrogen
(c) Phosphorus (d) Boron
View Answer play_arrow
-
question_answer39)
Which
of the following pairs of ions are isoelectronic and isostructural?
(a) 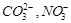 (b)
(b) 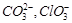 (c)
(c) 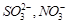 (d)
(d)
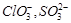
View Answer play_arrow
-
question_answer40)
Affinity
for hydrogen decreases in the group from fluorine to iodine. Which of the
halogen acids should have highest bond dissociation enthalpy?
(a) HF (b) HCl
(c) HBr (d) HI
View Answer play_arrow
-
question_answer41)
Bond
dissociation enthalpy of E?H (E= element) bonds is given below. Which of the
compounds will act as strongest reducing agent?
(a)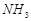 (b)
(b)
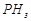 (c)
(c) 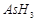 (d)
(d)
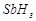 View Answer play_arrow
View Answer play_arrow
-
question_answer42)
On
heating with concentrated 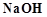 solution in an inert atmosphere of, white phosphorus
gives a gas. Which of the following statement is incorrect about the gas?
(a) It is highly poisonous and has smell like rotten fish
(b) It's solution in water decomposes in the presence of
light
(c) It is more basic than NN3
(d) It is less basic than NN3
solution in an inert atmosphere of, white phosphorus
gives a gas. Which of the following statement is incorrect about the gas?
(a) It is highly poisonous and has smell like rotten fish
(b) It's solution in water decomposes in the presence of
light
(c) It is more basic than NN3
(d) It is less basic than NN3
View Answer play_arrow
-
question_answer43)
Which
of the following acids forms three series of salts? ;
(a) 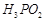 (b)
(b)
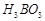 (c)
(c) 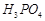 (d)
(d)
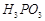
View Answer play_arrow
-
question_answer44)
Strong
reducing behaviour of 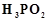 is due to
(a) low oxidation state of phosphorus
(b) presence of two ? OH groups and one P ? H bond
(c) presence of one?OH group and two P?H bonds
(d) high electron gain enthalpy of phosphorus
is due to
(a) low oxidation state of phosphorus
(b) presence of two ? OH groups and one P ? H bond
(c) presence of one?OH group and two P?H bonds
(d) high electron gain enthalpy of phosphorus
View Answer play_arrow
-
question_answer45)
On
heating lead nitrate forms oxides of nitrogen and lead. The oxides formed are..........
(a) 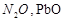 (b)
(b)
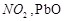 (c)
(c) 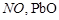 (d)
(d)
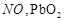
View Answer play_arrow
-
question_answer46)
Which
of the following elements does not show allotropy?
(a) Nitrogen (b) Bismuth
(c) Antimony (d) Arsenic
View Answer play_arrow
-
question_answer47)
Maximum
covalency of nitrogen is..........
(a) 3 (b) 5
(c) 4 (d) 6
View Answer play_arrow
-
question_answer48)
Which
of the following statements is wrong?
(a) Single N?N bond is stronger than the single P?P bond.
(b) PH3 can act as a ligand in the formation of
coordination compound with transition elements.
(c) NO2 is paramagnetic in nature.
(d) Covalency of nitrogen in 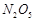 is
four.
is
four.
View Answer play_arrow
-
question_answer49)
A
brown ring is formed in the ring test for 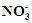 ion. It is due to the formation of
(a)
ion. It is due to the formation of
(a) 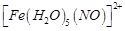 (b)
(b) 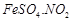 (c)
(c) 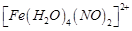 (d)
(d) 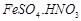
View Answer play_arrow
-
question_answer50)
Elements
of group- 15 form compounds in +5 oxidation state. However, bismuth forms only
one well characterised compound in +5 oxidation state. The compound is
(a) 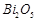 (b)
(b)
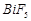 (c)
(c) 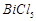 (d)
(d)
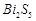
View Answer play_arrow
-
question_answer51)
On
heating ammonium dichromate and barium azide separately we get
(a) 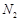 in both cases
(b)
in both cases
(b) 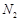 with ammonium
dichromate and NO with barium azide
(c)
with ammonium
dichromate and NO with barium azide
(c) 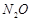 with ammonium
dichromate and N^ with barium azide
(d)
with ammonium
dichromate and N^ with barium azide
(d) 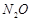 with ammonium
dichromate and NO; with barium azide
with ammonium
dichromate and NO; with barium azide
View Answer play_arrow
-
question_answer52)
In
the preparation of NN03, we get NO gas by catalytic oxidation of ammonia. The
moles of NO produced by the oxidation of two moles of 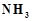 will be..........
(a) 2 (b) 3
(c) 4 (d) 6
will be..........
(a) 2 (b) 3
(c) 4 (d) 6
View Answer play_arrow
-
question_answer53)
The
oxidation state of central atom in the anion of compound 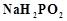 will be...........
(a) + 3 (b) +5
(c) + 1 (d)-3
will be...........
(a) + 3 (b) +5
(c) + 1 (d)-3
View Answer play_arrow
-
question_answer54)
Which
of the following is not tetrahedral in shape?
(a) 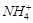 (b)
(b)
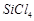 (c)
(c) 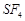 (d)
(d)
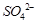 It has trigonal bipyramidal geometry having
It has trigonal bipyramidal geometry having 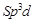 hybridisation.
hybridisation.
View Answer play_arrow
-
question_answer55)
Which
of the following are peroxoacids of sulphur?
(a) 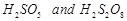 (b)
(b) 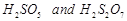 (c)
(c) 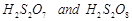 (d)
(d) 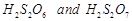
View Answer play_arrow
-
question_answer56)
Hot
cone. 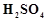 acts as moderately strong oxidising agent. It oxidises both
metals and non-metals. Which of the following element is oxidised by cone.
acts as moderately strong oxidising agent. It oxidises both
metals and non-metals. Which of the following element is oxidised by cone. 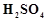 into
two gaseous products?
(a) Cu (b) S
(c) C (d) Zn
into
two gaseous products?
(a) Cu (b) S
(c) C (d) Zn
View Answer play_arrow
-
question_answer57)
A
black compound of manganese reacts with a halogen acid to give greenish yellow
gas. When excess of this gas reacts with 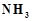 an unstable trihalide is formed. In this process the
oxidation state of nitrogen changes from..........
(a) -3 to + 3 (b) -3 to 0
(c) - 3 to + 5 (d)
0 to ? 3
an unstable trihalide is formed. In this process the
oxidation state of nitrogen changes from..........
(a) -3 to + 3 (b) -3 to 0
(c) - 3 to + 5 (d)
0 to ? 3
View Answer play_arrow
-
question_answer58)
In
the preparation of compounds of Xe, Bartlett had taken 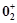 Pt
Pt 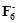 as a base
compound. This is because
(a) both
as a base
compound. This is because
(a) both 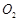 and Xe have
same size.
(b) both
and Xe have
same size.
(b) both 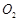 and Xe have
same electron gain enthalpy.
(c) both
and Xe have
same electron gain enthalpy.
(c) both 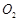 and Xe have
almost same ionisation enthalpy.
(d) both Xe and
and Xe have
almost same ionisation enthalpy.
(d) both Xe and 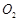 are gases.
are gases.
View Answer play_arrow
-
question_answer59)
In
solid state PCl5 is a..........
(a) covalent
solid
(b) octahedral structure
(c) ionic solid with 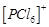 octahedral
and
octahedral
and 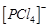 tetrahedral
(d) ionic solid with
tetrahedral
(d) ionic solid with 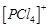 tetrahedral
and
tetrahedral
and 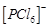 octahedral
octahedral
View Answer play_arrow
-
question_answer60)
Reduction
potentials of some ions are given below. Arrange them in decreasing order of
oxidising power.
(a) 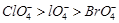 (b)
(b) 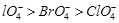 (c)
(c)  (d)
(d) 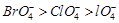 View Answer play_arrow
View Answer play_arrow
-
question_answer61)
Which
of the following is isoelectronic pair?
(a) 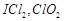 (b)
(b)
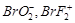 (c)
(c) 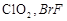 (d)
(d)
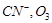
View Answer play_arrow
-
question_answer62)
If
chlorine gas is passed through hot 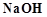 solution, two changes are observed in the oxidation number
of chlorine during the reaction. These are ......... and.........
(a) 0 to +5 (b) 0 to +3
(c) 0 to -1 (d) 0 to +1
solution, two changes are observed in the oxidation number
of chlorine during the reaction. These are ......... and.........
(a) 0 to +5 (b) 0 to +3
(c) 0 to -1 (d) 0 to +1
View Answer play_arrow
-
question_answer63)
Which
of the following options are not in accordance with the property mentioned
against them?
(a) 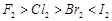 Oxidising
power
(b)
Oxidising
power
(b) 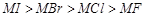 Ionic
character of metal halide
(c)
Ionic
character of metal halide
(c) 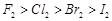 Bond dissociation enthalpy
(d) HI< HBr < HCI < HF Hydrogen-halogen bond
strength
Bond dissociation enthalpy
(d) HI< HBr < HCI < HF Hydrogen-halogen bond
strength
View Answer play_arrow
-
question_answer64)
Which
of the following is correct for P4 molecule of white phosphorus?
(a) It has 6 lone pairs of electrons
(b) It has six P?P single bonds
(c) It has three P ? P single bonds
(d) It has four lone pairs of electrons
View Answer play_arrow
-
question_answer65)
Which
of the following statements are correct?
(a) Among halogens, radius ratio between iodine and
fluorine is maximum.
(b) Leaving F ? F bond, all halogens have weaker X ? X bond
than X ? X bond in interhalogens.
(c) Among interhalogen compounds maximum number of atoms
are present in iodine fluoride.
(d) Interhalogen compounds are more reactive than halogen
compounds.
View Answer play_arrow
-
question_answer66)
Which
of the following statements are correct for SO2 gas?
(a) It acts as bleaching agent in moist conditions.
(b) Its molecule has linear geometry.
(c) Its dilute solution is used as disinfectant.
(d) It can be prepared by the reaction of dilute 1-12804
with metal sulphide.
View Answer play_arrow
-
question_answer67)
Which
of the following statements are correct?
(a) All the three N ? 0 bond lengths in 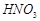 are equal.
(b) All P? Cl bond lengths in PCI5 molecule in
gaseous state are equal
(C) P4 molecule
in white phosphorus have angular strain therefore white phosphorus is very
reactive
(d) PCI5 is ionic in solid state in which cation
is tetrahedral and anion is octahedral.
are equal.
(b) All P? Cl bond lengths in PCI5 molecule in
gaseous state are equal
(C) P4 molecule
in white phosphorus have angular strain therefore white phosphorus is very
reactive
(d) PCI5 is ionic in solid state in which cation
is tetrahedral and anion is octahedral.
View Answer play_arrow
-
question_answer68)
Which
of the following orders are correct as per the properties mentioned against
each?
(a) 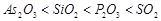 Acid strength.
(b)
Acid strength.
(b) 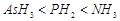 Enthalpy of vaporisation.
(c)
Enthalpy of vaporisation.
(c) 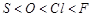 More negative electron gain enthalpy.
(d)
More negative electron gain enthalpy.
(d) 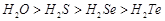 Thermal stability.
Thermal stability.
View Answer play_arrow
-
question_answer69)
Which
of the following statements are correct?
(a) S?S bond is present-in 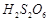 (b) In peroxosulphuric acid
(b) In peroxosulphuric acid 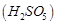 sulphur
is in + 6 oxidation state
(c) Iron powder along with
sulphur
is in + 6 oxidation state
(c) Iron powder along with 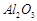 and
and
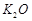 is used as a catalyst in the
preparation of
is used as a catalyst in the
preparation of 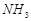 by Haber's process
(d) Change in enthalpy is positive for the preparation of
803 by catalytic oxidation of
by Haber's process
(d) Change in enthalpy is positive for the preparation of
803 by catalytic oxidation of 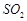
View Answer play_arrow
-
question_answer70)
In
which of the following reactions cone. 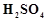 is used as an oxidizing reagent?
(a)
is used as an oxidizing reagent?
(a) 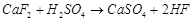 (b)
(b) 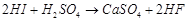 (c)
(c) 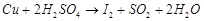 (d)
(d) 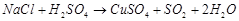
View Answer play_arrow
-
question_answer71)
Which
of the following statements are true?
(a) Only type of interactions between particles of noble
gases are due to weak dispersion forces.
(b) lonisation enthalpy of molecular oxygen is very close
to that of xenon.
(c) Hydrolysis of 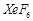 is
a redox reaction.
(d) Xenon fluorides are not. reactive.
is
a redox reaction.
(d) Xenon fluorides are not. reactive.
View Answer play_arrow
-
question_answer72)
In
the preparation of 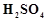 by Contact process, why is
by Contact process, why is  not
absorbed directly in water to form
not
absorbed directly in water to form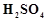 ?
?
View Answer play_arrow
-
question_answer73)
Write
a balanced chemical equation for the reaction showing catalytic oxidation of 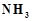 by
atmospheric oxygen.
by
atmospheric oxygen.
View Answer play_arrow
-
question_answer74)
Write
the structure of pyrophosphoric acid.
View Answer play_arrow
-
question_answer75)
PH3
forms bubbles, when passed slowly in water but NN3 dissolves. Explain why?
View Answer play_arrow
-
question_answer76)
In
PCl5, phosphorus is in 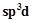 hybridised state but all its five bonds are not
equivalent. Justify your answer with .reason.
hybridised state but all its five bonds are not
equivalent. Justify your answer with .reason.
View Answer play_arrow
-
question_answer77)
Why
is nitric oxide paramagnetic in gaseous state but the solid obtained on cooling
it is diamagnetic?
View Answer play_arrow
-
question_answer78)
Give
one reason to explain why CIF3 exists but FCl3 does not
exist?
View Answer play_arrow
-
question_answer79)
Out
of 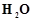 and
and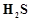 , which one has higher bond angle and why?
, which one has higher bond angle and why?
View Answer play_arrow
-
question_answer80)
SF6
is known but SCl6 is not. Why?
View Answer play_arrow
-
question_answer81)
On
reaction with Cl2, phosphorus forms two types of halides 'A' and 'S.
Halide 'A' is yellowish-white powder but halide 'S is colourless oily liquid. Identify
A and B and write the formulae of their hydrolysis products.
View Answer play_arrow
-
question_answer82)
In
the ring test of 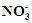 ion,
ion, 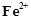 ion reduces nitrate ion to nitric oxide, which combines
with
ion reduces nitrate ion to nitric oxide, which combines
with 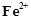 (aq) ion to form
brown complex. Write the reactions involved in the formation of brown ring.
(aq) ion to form
brown complex. Write the reactions involved in the formation of brown ring.
View Answer play_arrow
-
question_answer83)
Explain
why the stability of oxoacids of chlorine increases in the order given below.
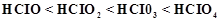
View Answer play_arrow
-
question_answer84)
Explain
why ozone is thermodynamically less stable than oxygen?
View Answer play_arrow
-
question_answer85)
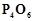 reacts with water according to equation
reacts with water according to equation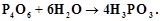 Calculate
the volume of 0.1 M
Calculate
the volume of 0.1 M 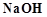 solution required to neutralise the acid formed by
dissolving 1.1 g of
solution required to neutralise the acid formed by
dissolving 1.1 g of 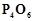 in
in 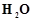 .
.
View Answer play_arrow
-
question_answer86)
White
phosphorus reacts with chlorine and the product hydrolyses in the presence of
water. Calculate the mass of HCl obtained by
the hydrolysis of the product formed by the reaction of 62 g of white
phosphorus with chlorine in the presence of water.
View Answer play_arrow
-
question_answer87)
Name
three oxoacids of nitrogen. Write the disproportionation reaction of that
oxoacid of nitrogen in which nitrogen is in + 3 oxidation state.
View Answer play_arrow
-
question_answer88)
Nitric
acid forms an oxide of nitrogen on reaction with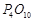 .
Write the reaction involved. Also write the resonating structures of the oxide
of nitrogen formed.
.
Write the reaction involved. Also write the resonating structures of the oxide
of nitrogen formed.
View Answer play_arrow
-
question_answer89)
Give
an example to show the effect of concentration of nitric acid on the formation
of oxidation product.
View Answer play_arrow
-
question_answer90)
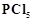 reacts with finely divided silver on heating and a white
silver salt is obtained, which dissolves on adding excess aqueous NN3 solution.
reacts with finely divided silver on heating and a white
silver salt is obtained, which dissolves on adding excess aqueous NN3 solution.
View Answer play_arrow
-
question_answer91)
Write
the reactions involved to explain what happens.
View Answer play_arrow
-
question_answer92)
Phosphorus
forms a number of oxoacids. Out of these oxoacids, phosphinic acid has strong
reducing property. Write its structure and also write a reaction showing its
reducing behaviour.
View Answer play_arrow
-
question_answer93)
Match
the compounds given in Column I with the hybridisation and shape given in
Column II and mark the correct option.
|
Column I
|
Column II
|
A. 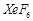
|
1. 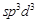 - distorted
octahedral - distorted
octahedral
|
B. 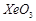
|
2. 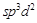 - square
planar - square
planar
|
C. 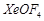
|
3. 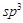 pyramidal pyramidal
|
D. 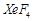
|
4.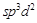 square
pyramidal square
pyramidal
|
Codes
A B C D A B C
D
(a) 1 3 4 2 (b) 1 2 4
3
(c) 4 3 1 2 (d) 4 1 2
3
View Answer play_arrow
-
question_answer94)
Match
the formulas of oxides given in Column I with the type of oxide given in Column
II and mark the correct option.
|
|
Column I
|
|
Column II
|
|
A.
|
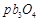
|
1,
|
Neutral oxide
|
|
B.
|
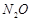
|
2.
|
Acidic oxide
|
|
C.
|

|
3.
|
Basic oxide
|
|
D.
|
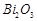
|
4.
|
Mixed oxide
|
Codes
A B C D A B C D
(a) 1 2 3 4 (b) 4 1 2 3
(c) 3 2 4 1 (d) 4 3 1 2
View Answer play_arrow
-
question_answer95)
Match
the items of Columns I and II and mark the correct option.
|
|
Column I
|
Column II
|
|
A. B. C. D.
|
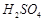
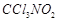
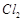 Sulphur
Sulphur
|
1. Highest electron gain enthalpy
2. Chalcogen
3. Tear gas
4. Storage batteries
|
Codes
A B C D A B C D
(a) 4 3 1 2 (b) 3 4 1 2
(c) 4 1 2 3 (d) 2 1 3 4
View Answer play_arrow
-
question_answer96)
Match
the species given in Column I with the shape given in Column I and mark the
correct option.
|
|
Column 1
|
Column 11
|
|
A.
|
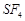
|
1. Tetrahedral
|
|
B.
|
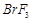
|
2. Pyramidal
|
|
C.
|

|
3. Sea-saw shaped
|
|
D.
|
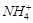
|
4. Bent T-shaped
|
Codes
A B C D A B C D
(a) 3 2 1 4 (b) 3 4 2 1
(c) 1 2 3 4 (d) 1 4 3 2
View Answer play_arrow
-
question_answer97)
Match
the items of Columns I and II and mark the correct option.
|
|
Column 1
|
Column 11
|
|
A.
|
Its partial hydrolysis does not change oxidation
state of central atom.
|
1. He
|
|
B.
|
It is used in modern diving apparatus.
|
2. 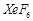
|
|
C.
|
It is used to provide inert atmosphere for
filling electrical bulbs.
|
3. 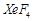
|
|
D.
|
Its central atom is in sp^d2
hybridisation.
|
4. Ar
|
Codes
A B C D A B C D
(a) 1 4 2 3 (b) 1 2 3 4
(c) 2 1 4 3 (d) 1 3 2 4
View Answer play_arrow
-
question_answer98)
Assertion (A) N2 is less reactive than P4.
Reason (R) Nitrogen
has more electron gain enthalpy than phosphorus.
View Answer play_arrow
-
question_answer99)
Assertion (A) 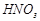 makes iron passive.
Reason (R)
makes iron passive.
Reason (R) 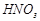 forms a protective layer of ferric
nitrate on the surface of iron.
forms a protective layer of ferric
nitrate on the surface of iron.
View Answer play_arrow
-
question_answer100)
Assertion (A) 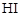 cannot be prepared by the reaction of
cannot be prepared by the reaction of 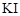 with
concentrated
with
concentrated 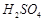 Reason (R)
Reason (R) 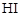 has Lowest H?X bond strength
among halogen acids.
has Lowest H?X bond strength
among halogen acids.
View Answer play_arrow
-
question_answer101)
Assertion
(A) Both rhombic and monocline sulphur
exist as 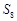 but
oxygen exists as
but
oxygen exists as 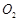 .
Reason (R) Oxygen
forms
.
Reason (R) Oxygen
forms 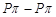 multiple bond due to small size
and small bond length but
multiple bond due to small size
and small bond length but 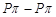 bonding is not
possible in sulphur.
bonding is not
possible in sulphur.
View Answer play_arrow
-
question_answer102)
Assertion (A) 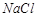 reacts with concentrated
reacts with concentrated 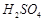 to give colourless fumes with pungent smell. But on adding
to give colourless fumes with pungent smell. But on adding 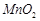 the
fumes become
greenish yellow.
Reason (R)
the
fumes become
greenish yellow.
Reason (R) 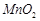 oxidises HCl to chlorine gas which
is greenish yellow.
oxidises HCl to chlorine gas which
is greenish yellow.
View Answer play_arrow
-
question_answer103)
Assertion (A) SF6 cannot be hydrolysed butSF4 can be.
Reason (R) Six F-atoms
in SF6 prevent the attack of H2O on sulphur atom Of SF6.
View Answer play_arrow
-
question_answer104)
An
amorphous solid "A" burns in air to form a gas "B" which
turns lime water milky. The gas is also produced as a by-product during
roasting of sulphide ore. This gas decolourises acidified aqueous 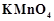 solution
and reduces
solution
and reduces 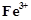 to
to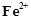 . Identify the solid "A" and the gas "B"
and write the reactions involved.
. Identify the solid "A" and the gas "B"
and write the reactions involved.
View Answer play_arrow
-
question_answer105)
On
heating lead (II) nitrate gives a brown gas "A". The gas "A"
on cooling changes to colourless solid "B". Solid "B" on
heating with NO changes to a blue solid 'C. Identify 'A', 'B' and 'C and also
write reactions involved and draw the structures of 'B' and 'C'.
View Answer play_arrow
-
question_answer106)
On
heating compound (A) gives a gas (B) which is a constituent of air. This gas
when treated with 3 moles of hydrogen (H2) in the presence of a
catalyst gives another gas (C) which is basic in nature. Gas C on further Oxidation
in moist condition gives a compound (D) which is a part of acid rain. Identify
compounds (A) to (D) and also give necessary equations of all the steps involved.
View Answer play_arrow
![]() (b)
(b)
![]() (c)
(c) ![]() (d)
(d)
![]()
![]() (b)
(b) ![]() (c)
(c) ![]() (d)
(d) ![]()
![]() (b)
(b)
![]() (c)
(c) ![]() (d)
(d)
![]()
![]() and
and![]() , which one has higher bond angle and why?
, which one has higher bond angle and why?
