-
question_answer1)
An aqueous solution of a salt X turns blood red on treatment with \[CN{{S}^{-}}\] and blue on treatment with\[{{K}_{4}}[Fe{{\left( CN \right)}_{6}}]\]. X also gives a positive chromyl chloride test. The salt X is:
A)
\[CuC{{l}_{2}}\] done
clear
B)
\[FeC{{l}_{3}}\] done
clear
C)
\[Cu{{(N{{O}_{3}})}_{2}}\] done
clear
D)
\[Fe{{(N{{O}_{3}})}_{3}}\] done
clear
View Solution play_arrow
-
question_answer2)
\[KMn{{O}_{4}}\] reacts with oxalic acid as:
| \[MnO_{4}^{-}+{{C}_{2}}O_{4}^{2-}+{{H}^{+}}\xrightarrow{{}}M{{n}^{2+}}+{{CO}_{2}}+{{H}_{2}}O\] |
| Hence, 50 ml of 0.04 M \[KMn{{O}_{4}}\] is acidic medium is chemically equivalent to |
A)
100 ml of 0.1M \[{{H}_{2}}{{C}_{2}}{{O}_{4}}\] done
clear
B)
50 ml of 0.2 M \[{{H}_{2}}{{C}_{2}}{{O}_{4}}\] done
clear
C)
50 ml of 0.1M \[{{H}_{2}}{{C}_{2}}{{O}_{4}}\] done
clear
D)
25 ml of 0.1M \[{{H}_{2}}{{C}_{2}}{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer3)
For detection of sulphur in an organic compound sodium nitroprusside is added to the sodium extract. A violet colour is obtained due to formation of
A)
\[Fe{{\left( CN \right)}_{2}}\] done
clear
B)
\[{{K}_{3}}Fe{{\left( CN \right)}_{5}}NS\] done
clear
C)
\[N{{a}_{4}}\left[ Fe{{\left( CN \right)}_{5}}NOS \right]\] done
clear
D)
\[N{{a}_{4}}Fe{{\left( CN \right)}_{6}}\] done
clear
View Solution play_arrow
-
question_answer4)
0.45 g of acid (molecular weight 90) is neutralized by 20 ml of 0.5N caustic potash. The basicity of acid is
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer5)
The cation that will not be precipitated by \[{{H}_{2}}S\] in the presence of dil. \[HCl\] is:
A)
\[P{{b}^{2+}}\] done
clear
B)
\[C{{u}^{2+}}\] done
clear
C)
\[C{{o}^{2+}}\] done
clear
D)
\[A{{s}^{3+}}\] done
clear
View Solution play_arrow
-
question_answer6)
A pink coloured salt turns blue on heating. The presence of which cation is most likely?
A)
\[C{{o}^{2+}}\] done
clear
B)
\[C{{u}^{2+}}\] done
clear
C)
\[Z{{n}^{2+}}\] done
clear
D)
\[F{{e}^{2+}}\] done
clear
View Solution play_arrow
-
question_answer7)
0.5 g mixture of \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] and \[KMn{{O}_{4}}\] was treated with excess of KI in acidic medium. \[{{I}_{2}}\]liberated required \[100c{{m}^{3}}\] of 0.15 N \[N{{a}_{2}}{{S}_{2}}{{\text{O}}_{3}}\] solution for titration. The percentage amount of \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] in the mixture is
A)
85.36% done
clear
B)
14.64% done
clear
C)
58.63% done
clear
D)
26.14% done
clear
View Solution play_arrow
-
question_answer8)
In Kjeldahl's method, \[CuS{{O}_{4}}\] acts as
A)
oxidising agent done
clear
B)
reducing agent done
clear
C)
hydrolysing agent done
clear
D)
catalytic agent done
clear
View Solution play_arrow
-
question_answer9)
In Lassaigne's test, the organic compound is fused with a piece of sodium metal in order to
A)
increase the ionisation of the compound. done
clear
B)
decrease the melting point of the compound. done
clear
C)
increase the reactivity of the compound. done
clear
D)
convert the covalent compound into a mixture of ionic compounds. done
clear
View Solution play_arrow
-
question_answer10)
A white sodium salt dissolves readily in water to give a solution which is neutral to litmus. When silver nitrate solution is added to the solution, a white precipitate is obtained which does not dissolve in dil. \[HN{{O}_{3}}\]. The anion could be
A)
\[CO_{3}^{2-}\] done
clear
B)
\[C{{l}^{-}}\] done
clear
C)
\[SO_{4}^{2-}\] done
clear
D)
\[{{S}^{2-}}\] done
clear
View Solution play_arrow
-
question_answer11)
An aqueous solution of a substance gives a white precipitate on treatment with dil. \[HCl\] which dissolves on heating. When \[{{H}_{2}}S\] is passed through the hot acidic solution, a black precipitate is obtained. The substance is a
A)
\[Hg_{2}^{2+}\] salt done
clear
B)
\[C{{u}^{2+}}\] salt done
clear
C)
\[A{{g}^{\text{+}}}\] alt done
clear
D)
\[P{{b}^{2+}}\] salt done
clear
View Solution play_arrow
-
question_answer12)
A is a lighter phenol and B is an aromatic carboxylic acid. Separation of mixture of A and B can be carried out easily by using a solution of
A)
sodium hydroxide done
clear
B)
sodium sulphate done
clear
C)
calcium chloride done
clear
D)
sodium bicarbonate done
clear
View Solution play_arrow
-
question_answer13)
A metal chloride dissolves appreciably in cold water. When placed on a platinum wire in Bunsen flame no distinctive colour is noticed, the cation would be
A)
\[M{{g}^{2+}}\] done
clear
B)
\[B{{a}^{2+}}\] done
clear
C)
\[P{{b}^{2+}}\] done
clear
D)
\[C{{a}^{2+}}\] done
clear
View Solution play_arrow
-
question_answer14)
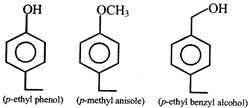
Above compounds can be differentiated by using the reagent:
A)
\[NaOH\], Tollen's reagent, \[FeC{{l}_{3}}\] done
clear
B)
\[Cr{{O}_{3}}\] Tollen's reagent, \[FeC{{l}_{3}}\] done
clear
C)
Tollen's reagent, \[Cr{{O}_{3}},FeC{{l}_{3}}\] done
clear
D)
Na, Tollen's reagent, \[FeC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer15)
Copper wire test for halogens is known as
A)
Duma's Test done
clear
B)
Beilstein's Test done
clear
C)
Liebig's Test done
clear
D)
Lassigne's Test done
clear
View Solution play_arrow
-
question_answer16)
In the separation of \[C{{u}^{2+}}\] and \[C{{d}^{2+}}\] in 2nd group qualitative analysis of cations, tetrammine copper (II) sulphate and tetrammine cadmium (II) sulphate react with KCN to form the corresponding cyano complexes. Which one of the following pairs of the complexes and their relative stability enable the separation of \[C{{u}^{2+}}\] and \[C{{d}^{2+?}}\]
A)
\[{{K}_{3}}\left[ Cu{{\left( CN \right)}_{4}} \right]\] more stable and \[{{K}_{2}}[Cd{{(CN)}_{4}}]\] less stable done
clear
B)
\[{{K}_{2}}\left[ Cu{{\left( CN \right)}_{4}} \right]\] less stable and \[{{K}_{2}}[Cd{{(CN)}_{4}}]\] more stable done
clear
C)
\[{{K}_{2}}\left[ Cu{{\left( CN \right)}_{4}} \right]\] more stable and \[{{K}_{2}}[Cd{{(CN)}_{4}}]\] less stable done
clear
D)
\[{{K}_{3}}\left[ Cu{{\left( CN \right)}_{4}} \right]\]less stable and \[{{K}_{2}}[Cd{{(CN)}_{4}}]\] more stable done
clear
View Solution play_arrow
-
question_answer17)
Sodium carbonate cannot be used in place of \[{{(N{{H}_{4}})}_{2}}C{{O}_{3}}\] for the identification of \[C{{a}^{2+}},B{{a}^{2+}}\]and \[S{{r}^{2+}}\] ions (in group V) during mixture analysis because:
A)
\[M{{g}^{2+}}\] ions will also be precipitated. done
clear
B)
Concentration of \[CO_{3}^{2-}\] ions is very low. done
clear
C)
Sodium ions will react with acid radicals. done
clear
D)
\[N{{a}^{+}}\] ions will interfere with the detection of \[C{{a}^{2+}},B{{a}^{2+}},S{{r}^{2+}}\] ions. done
clear
View Solution play_arrow
-
question_answer18)
A solution containing \[A{{s}^{3+}},C{{d}^{2+}},N{{i}^{2+}}\] and \[Z{{n}^{2+}}\] is made alkaline with dilute \[N{{H}_{4}}OH\] and treated with \[{{H}_{2}}S\]. The precipitate obtained will consist of
A)
\[A{{s}_{2}}{{S}_{3}}\] and CdS done
clear
B)
CdS.NiS and ZnS done
clear
C)
NiS and ZnS done
clear
D)
Sulphide of all ions done
clear
View Solution play_arrow
-
question_answer19)
3.92 g of ferrous ammonium sulphate react N completely with \[50ml\frac{N}{10}KMn{{O}_{4}}\] solution. The percentage purity of the sample is
A)
50 done
clear
B)
78.4 done
clear
C)
80 done
clear
D)
39.2 done
clear
View Solution play_arrow
-
question_answer20)
1.25 g of a sample of \[N{{a}_{2}}C{{O}_{3}}\] and \[NaS{{O}_{4}}\] is dissolved in 250 ml solution. 25 ml of this solution neutralises 20 ml of 0. 1 N\[{{H}_{2}}S{{O}_{4}}\]. The % of \[N{{a}_{2}}{{O}_{2}}\] in this sample is
A)
84.8% done
clear
B)
8.48% done
clear
C)
15.2% done
clear
D)
42.4% done
clear
View Solution play_arrow
-
question_answer21)
An aqueous solution of \[FeS{{O}_{4}},A{{l}_{2}}{{(S{{O}_{4}})}_{3}}\] and chrome alum is heated with excess of \[N{{a}_{2}}{{O}_{2}}\] and filtered. The materials obtained are:-
A)
A colourless filtrate and a green residue. done
clear
B)
A yellow filtrate and a green residue. done
clear
C)
A yellow filtrate and a brown residue. done
clear
D)
A green filtrate and a brown residue. done
clear
View Solution play_arrow
-
question_answer22)
The formula of gas is \[{{[CO]}_{x}}\]. If its vapour density is 140, the value of x will be:
A)
2.5 done
clear
B)
3.0 done
clear
C)
5.0 done
clear
D)
6.0 done
clear
View Solution play_arrow
-
question_answer23)
If 0.2 gram of an organic compound containing carbon, hydrogen and oxygen on combustion, yielded 0.147 gram carbon dioxide and 0.12 gram water. What will be the content of oxygen in the substance?
A)
73.29% done
clear
B)
78.45% done
clear
C)
83.23% done
clear
D)
89.50% done
clear
View Solution play_arrow
-
question_answer24)
Which of the following statements is incorrect?
A)
\[F{{e}^{2+}}\] ion also gives blood red colour with \[SC{{N}^{-}}\] ion. done
clear
B)
\[F{{e}^{3+}}\] ion also gives blood red colour with \[SC{{N}^{-}}\] ion. done
clear
C)
On passing \[{{H}_{2}}S\] into \[N{{a}_{2}}Zn{{O}_{2}}\] solution a white ppt of ZnS is formed. done
clear
D)
Cupric ion reacts with excess of ammonia solution to give deep blue colour of \[{{[Cu{{(N{{H}_{3}})}_{4}}]}^{2+}}\]ion. done
clear
View Solution play_arrow
-
question_answer25)
An aqueous solution of colourless metal sulphate M gives a white precipitate with\[N{{H}_{4}}OH\]. This was soluble in excess of\[N{{H}_{4}}OH\]. On passing \[{{H}_{2}}S\] through this solution a white ppt. is formed. The metal M in the salt is
A)
Ca done
clear
B)
Ba done
clear
C)
Al done
clear
D)
Zn done
clear
View Solution play_arrow
-
question_answer26)
Volume of 3% solution of sodium carbonate necessary to neutralise a litre of 0.1N sulphuric acid
A)
176.66 ml done
clear
B)
156.6 ml done
clear
C)
116.0 ml done
clear
D)
196.1 ml done
clear
View Solution play_arrow
-
question_answer27)
The Lassaigne 's extract is boiled with dil. \[HN{{O}_{3}}\] before testing for halogens because
A)
silver halides are soluble in \[HN{{O}_{3}}\] done
clear
B)
\[N{{a}_{2}}S\] and NaCN are decomposed by \[HN{{O}_{3}}\] done
clear
C)
\[A{{g}_{2}}S\] is soluble in \[HN{{O}_{3}}\] done
clear
D)
\[AgCN\] is soluble in \[HN{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer28)
For preparing 250 mL of N/20 solution of Mohr 's salt, the amount of Mohr's salt needed is
A)
9.8 g done
clear
B)
4.9 g done
clear
C)
19.6 g done
clear
D)
3.2 g done
clear
View Solution play_arrow
-
question_answer29)
Potassium chromate solution is added to an aqueous solution of a metal chloride. The precipitate thus obtained is insoluble in acetic acid. When precipitate is subjected to flame test the colour of the flame is
A)
lilac done
clear
B)
apple green done
clear
C)
crimson red done
clear
D)
golden yellow done
clear
View Solution play_arrow
-
question_answer30)
Three separate samples of a solution of a single salt gave these results. One formed a white precipitate with excess ammonia solution, one formed a white precipitate with dil. \[NaCl\] solution and one formed a black precipitate with \[{{H}_{2}}S\]. The salt could be
A)
\[AgN{{O}_{3}}\] done
clear
B)
\[Pb{{\left( N{{O}_{3}} \right)}_{2}}\] done
clear
C)
\[Hg{{\left( N{{O}_{3}} \right)}_{2}}\] done
clear
D)
\[MnS{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer31)
Compound
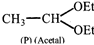
and \[\underset{\left( Q \right)}{\mathop{C{{H}_{3}}-C{{H}_{2}}-O-C{{H}_{2}}-C{{H}_{2}}}}\,\] can be differentiated by:
A)
\[{{N}_{3}}{{O}^{\oplus }},Na\] done
clear
B)
\[{{H}_{3}}{{O}^{\oplus }}\], Tollens' test done
clear
C)
\[{{H}_{3}}{{O}^{\oplus }}\] Fehlingtest done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer32)
The best method for the separation of naphthalene and benzoic acid from their mixture is:
A)
Distillation done
clear
B)
Sublimation done
clear
C)
Chromatography done
clear
D)
Crystallisation done
clear
View Solution play_arrow
-
question_answer33)
A salt on treatment with dil. \[HCl\] gives a pungent smelling gas and a yellow precipitate. The salt gives green flame when tested. The solution gives a yellow precipitate with potassium chromate. The salt is
A)
\[NiS{{O}_{4}}\] done
clear
B)
\[Ba{{S}_{2}}{{O}_{3}}\] done
clear
C)
\[Pb{{S}_{2}}{{O}_{3}}\] done
clear
D)
\[CuS{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer34)
0.24 g of a volatile liquid on vaporization gives 45 ml of vapours at NTP. What will be the vapour density of the substance? (Density of \[{{H}_{2}}=0.089g\text{ }{{L}^{-1}}\])
A)
95.39 done
clear
B)
39.95 done
clear
C)
99.53 done
clear
D)
59.93 done
clear
View Solution play_arrow
-
question_answer35)
The salt used for performing 'bead' test in qualitative inorganic analysis is
A)
\[{{K}_{2}}S{{O}_{4}}.A{{l}_{2}}{{\left( S{{O}_{4}} \right)}_{3}}.24{{H}_{2}}O\] done
clear
B)
\[FeS{{O}_{4}}.{{\left( N{{H}_{4}} \right)}_{2}}S{{O}_{4}}4{{H}_{2}}O\] done
clear
C)
\[Na\left( N{{H}_{4}} \right)HP{{O}_{4}}.4{{H}_{2}}O\] done
clear
D)
\[CaS{{O}_{4}}.2{{H}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer36)
A gas "X" is passed through water to form a saturated solution. The aqueous solutions on treatment with the \[AgN{{O}_{3}}\] gives a white preciptate. The saturated aqueous solution also dissolves magnesium ribbon with evolution of a colourless gas "Y" Identify 'X' and 'Y?.
A)
\[X=C{{O}_{2}},Y=C{{l}_{2}}\] done
clear
B)
\[X=C{{l}_{2}},Y=C{{O}_{2}}\] done
clear
C)
\[X=C{{l}_{2}},Y={{H}_{2}}\] done
clear
D)
\[X={{H}_{2}},Y=C{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer37)
In Kjeldahl's method for the estimation of\[{{\operatorname{N}}_{2}}\], potassium sulphate and copper sulphate are used. On the basis of their functions which of the following statement(s) is/are correct?
| I. Potassium sulphate raises the bpt. And ensures complete reaction. |
| II. Copper sulphate acts as catalyst. |
| III. Potassium sulphate acts as catalyst and copper sulphate raises the bpt. |
A)
Only III is correct done
clear
B)
I and II are correct done
clear
C)
Only II is correct done
clear
D)
None is correct done
clear
View Solution play_arrow
-
question_answer38)
Aniline is usually purified by
A)
chromatographic technique done
clear
B)
steam distillation done
clear
C)
by addition of oxalic acid done
clear
D)
fractional crystallization done
clear
View Solution play_arrow
-
question_answer39)
Prussian blue is formed when
A)
ferrous sulphate reacts with \[FeC{{l}_{3}}\] done
clear
B)
ferric sulphate reacts with \[{{K}_{4}}Fe{{\left( CN \right)}_{6}}\] done
clear
C)
ferrous ammonium sulphate reacts with \[FeC{{l}_{3}}\] done
clear
D)
ammonium sulphate reacts with \[N{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer40)
0.59 g of an organic substance when treated with caustic soda evolved ammonia which required 20 cc of N/2 sulphuric acid for neutralisation. The percentage of nitrogen is
A)
26.32% done
clear
B)
40% done
clear
C)
53.6% done
clear
D)
63.6% done
clear
View Solution play_arrow
-
question_answer41)
An organic compound has C and H percentage in the ratio 6 : 1 and C and O percentage in the ratio 3:4 the compound is
A)
HCHO done
clear
B)
\[C{{H}_{3}}OH\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
D)
\[{{(COOH)}_{2}}\] done
clear
View Solution play_arrow
-
question_answer42)
The presence of magnesium is confirmed in the qualitative analysis by the formation of a white crystalline precipitate of
A)
\[Mg{{\left( HC{{O}_{3}} \right)}_{2}}\] done
clear
B)
\[MgN{{H}_{4}}P{{O}_{4}}\] done
clear
C)
\[MgN{{H}_{4}}{{\left( HC{{O}_{3}} \right)}_{3}}\] done
clear
D)
\[MgC{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer43)
A salt which gives \[C{{O}_{2}}\] with hot conc. \[{{H}_{2}}S{{O}_{4}}\] and also decolourizes acidified\[KMn{{O}_{4}}\]. On warming is:
A)
\[HCO_{3}^{-}\] done
clear
B)
\[CO_{3}^{2-}\] done
clear
C)
Qxalate done
clear
D)
Acetate done
clear
View Solution play_arrow
-
question_answer44)
2.79 g of an organic compound when heated in Carius tube with cone. \[HN{{O}_{3}}\] and \[{{H}_{2}}P{{O}_{4}}\] formed is converted into \[MgN{{H}_{4}}.P{{O}_{4}}\]ppt. The ppt. on heating gave 1.332 g of \[M{{g}_{2}}{{P}_{2}}{{O}_{7}}\]. The percentage of P in the compound is
A)
23.33% done
clear
B)
13.33% done
clear
C)
33.33% done
clear
D)
26.66% done
clear
View Solution play_arrow
-
question_answer45)
| \[[X]+{{H}_{2}}S{{O}_{4}}\xrightarrow{{}}[Y]\] a colourless gas with irritating smell, |
| \[[Y]+{{K}_{2}}C{{r}_{2}}O+{{H}_{2}}S{{O}_{4}}\xrightarrow{{}}\] green solution. |
| [X] and [Y] is: |
A)
\[S{{O}_{3}}^{2-},S{{O}_{2}}\] done
clear
B)
\[C{{l}^{-}},HCl\] done
clear
C)
\[{{S}^{2-}},{{H}_{2}}S\] done
clear
D)
\[C{{O}_{3}}^{2-},C{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer46)
0.75 g platinic chloride of a mono- acidic base on ignition gives 0.245 g platinum. The molecular weight of the base is
A)
75.0 done
clear
B)
93.5 done
clear
C)
100 done
clear
D)
80.0 done
clear
View Solution play_arrow
-
question_answer47)
The sodium extract prepared from sulphanilic acid, contains \[SC{{N}^{-}}\]. It gives blood red colouration with
A)
a mixture of \[N{{a}_{2}}S\] and \[C{{S}_{2}}\] done
clear
B)
\[FeC{{l}_{3}}\] done
clear
C)
\[FeS{{O}_{4}}\] done
clear
D)
\[N{{a}_{2}}S{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer48)
An orange precipitate of II group is dissolved in cone. \[HCl\]; the solution when treated with excess of water turns milky due to formation of
A)
\[Sn\left( OH \right)Cl\] done
clear
B)
\[Sb\left( OH \right)C{{l}_{2}}\] done
clear
C)
\[SbOCl\] done
clear
D)
\[Sb{{\left( OH \right)}_{2}}Cl\] done
clear
View Solution play_arrow
-
question_answer49)
A substance on treatment with dil. \[{{H}_{2}}S{{O}_{4}}\] liberates a colourless gas which produces
| (I) turbidity with baryta water and |
| (II) turns acidified dichromate solution green. |
| The reaction indicates the presence of |
A)
\[CO_{3}^{2-}\] done
clear
B)
\[{{s}^{2-}}\] done
clear
C)
\[SO_{3}^{2-}\] done
clear
D)
\[NO_{3}^{-}\] done
clear
View Solution play_arrow
-
question_answer50)
Which of the following gives a precipitate with \[Pb{{(N{{O}_{3}})}_{2}}\] but not with \[Ba{{(N{{O}_{3}})}_{2}}\]?
A)
Sodium chloride done
clear
B)
Sodium acetate done
clear
C)
Sodium nitrate done
clear
D)
Sodium hyrogen phosphate done
clear
View Solution play_arrow
 Above compounds can be differentiated by using the reagent:
Above compounds can be differentiated by using the reagent: 