question_answer 1) A cylinder of mass 10 kg is rolling on a rough plane with a velocity of 10 m/s. If the coefficient of friction between the surface and cylinder is 0.5, then before stopping, it will cover a distance of \[(Take\text{ }g=10\text{ }m/{{s}^{2}})\]
A)
10m
done
clear
B)
7.5 m
done
clear
C)
5 m
done
clear
D)
2.5 m
done
clear
View Answer play_arrow
question_answer 2) If a ladder weighing 250 N is placed against a smooth vertical wall having coefficient of friction between it and floor is 0.3, then what is the maximum force of friction available at the point of contact between the ladder and the floor?
A)
75 N
done
clear
B)
50 N
done
clear
C)
35N
done
clear
D)
25 N
done
clear
View Answer play_arrow
question_answer 3) A ball of mass 150 g moving with an acceleration 20 \[{{10}^{-10}}s\]is hit by a force, which acts on it for 0.1 s. The impulsive force is
A)
0.5 N-s
done
clear
B)
0.1 N-s
done
clear
C)
0.3 N-s
done
clear
D)
1.2 N-s
done
clear
View Answer play_arrow
question_answer 4) A body, thrown upwards with some velocity reaches the maximum height of 50 m. Another body with double the mass thrown up with double the initial velocity will reach a maximum height of
A)
100 m
done
clear
B)
200 m
done
clear
C)
300 m
done
clear
D)
400 m
done
clear
View Answer play_arrow
question_answer 5) A 100 m long train is moving with a uniform velocity of 45 km/h. The time taken by the train to cross a bridge of length 1 km is
A)
58 s
done
clear
B)
68s
done
clear
C)
78 s
done
clear
D)
88 s
done
clear
View Answer play_arrow
question_answer 6) Velocity-time curvefora body, projected vertically upwards, is
A)
parabola
done
clear
B)
ellipse
done
clear
C)
hyperbola
done
clear
D)
straight line
done
clear
View Answer play_arrow
question_answer 7) The numerical ratio of displacement to the distance covered is always
A)
less than one
done
clear
B)
equal to one
done
clear
C)
equal to or less than one
done
clear
D)
equal to or greater than one
done
clear
View Answer play_arrow
question_answer 8) In the relation\[{{10}^{-16}}s\],the dimensional formula for k is
A)
\[{{10}^{-1}}s\]
done
clear
B)
\[y=5\sin \frac{\pi x}{3}\cos 40\pi t\]
done
clear
C)
\[\mu F\]
done
clear
D)
\[\mu F\]
done
clear
View Answer play_arrow
question_answer 9) While measuring the diameter of a wire by screw gauge, three readings were taken are 1.002 cm, 1.004 cm and 1.006 cm. The absolute error in the third reading is
A)
0.002 cm
done
clear
B)
0.004 cm
done
clear
C)
zero
done
clear
D)
1.002 cm \[\,\omega \] \[I=\frac{1}{2}M{{R}^{2}}\] \[\therefore \]
done
clear
View Answer play_arrow
question_answer 10) Masses of two substances are 1 g and 9 g respectively. If their kinetic energies are same, then the ratio of their momentum will be
A)
1 : 9
done
clear
B)
9 : 1
done
clear
C)
3:1
done
clear
D)
1 : 3
done
clear
View Answer play_arrow
question_answer 11) A particle of mass 100 g is thrown vertically upwards with a speed of 5 m/s. The work done by the force of gravity during the time when particle goes up is
A)
-0.5 J
done
clear
B)
-1.25 J
done
clear
C)
1.25 J
done
clear
D)
0.5 J
done
clear
View Answer play_arrow
question_answer 12) A circular disc is rotating with angular velocity to. If a man standing at the edge of the disc walks towards its centre, then the angular velocity of the disc will
A)
decrease
done
clear
B)
increase
done
clear
C)
be halved
done
clear
D)
not change
done
clear
View Answer play_arrow
question_answer 13) The moment of inertia of a disc of mass m and radius R about an axis, which is tangential to the circumference of the disc and parallel to its diameter is
A)
\[{{m}^{3}}\]
done
clear
B)
\[{{m}^{3}}\]
done
clear
C)
\[{{m}^{3}}\]
done
clear
D)
\[{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 14) A body weighs 500 N on the surface of the earth. How much would it weigh half way below the surface of the earth?
A)
1000 N
done
clear
B)
500 N
done
clear
C)
250 N
done
clear
D)
125 N
done
clear
View Answer play_arrow
question_answer 15) Escape velocity at surface of earth is 11.2 km/s. Escape velocity from a planet whose mass is the same as that of earth and radius 1/4 that of earth, is
A)
2.8 km/s
done
clear
B)
15.6 km/s
done
clear
C)
22.4 km/s
done
clear
D)
44.8 km/s
done
clear
View Answer play_arrow
question_answer 16) The rms value of the electric field of the light coming from the sun is 720 N/C. The average total energy density of the electromagnetic wave is
A)
\[4.58\times {{10}^{-6}}J/{{m}^{3}}\]
done
clear
B)
\[6.37\times {{10}^{-9}}J/{{m}^{3}}\]
done
clear
C)
\[81.35\times {{10}^{-12}}J/{{m}^{3}}\]
done
clear
D)
\[3.3\times {{10}^{-3}}J/{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 17) The time taken by a photoelectron to come out after the photon strikes is approximately
A)
\[2\times {{10}^{-2}}\]
done
clear
B)
\[\sqrt{3}\]
done
clear
C)
\[\frac{W}{3}\]
done
clear
D)
\[\frac{W}{\sqrt{3}}\]
done
clear
View Answer play_arrow
question_answer 18) A man is watching two trains, one leaving and the other coming with equal speed of 4 m/s. If they sound their whistles each of frequency 240 Hz, the number of beats heard by man (velocity of sound in air = 320 m/s) will be equal to
A)
12
done
clear
B)
zero
done
clear
C)
3
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 19) A tuning fork of frequency 392 Hz, resonates with 50 cm length of a string under tension (T). If length of the string is decreased by 2%, keeping the tension constant, the number of beats heard when the string and the tuning fork made to vibrate simultaneously is
A)
4
done
clear
B)
6
done
clear
C)
8
done
clear
D)
12
done
clear
View Answer play_arrow
question_answer 20) The equation of stationary wave along a stretched string is given by\[8\times {{10}^{-4}}\]where x and y are in centimetre and t in second. The separation between two adjacent nodes is
A)
6 cm
done
clear
B)
4 cm
done
clear
C)
3 cm
done
clear
D)
1.5 cm
done
clear
View Answer play_arrow
question_answer 21) A and B are two hills at a distance 510 m apart. A person standing between the hills, claps his hands and hears two echoes at the end of 1 s and 2 s respectively. The velocity of sound in air is
A)
225 m/s
done
clear
B)
340 m/s
done
clear
C)
510 m/s
done
clear
D)
1020 m/s
done
clear
View Answer play_arrow
question_answer 22)
The coefficient of thermal conductivity of copper is nine times that of steel. In the composite cylindrical bar shown in the figure, what will be the temperature at the junction of copper and steel?
A)
\[25{}^\circ C\]
done
clear
B)
\[33{}^\circ C\]
done
clear
C)
\[67{}^\circ C\]
done
clear
D)
\[75{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 23) The amount of work, which can be obtained by supplying 200 cal of heat, is
A)
840 J
done
clear
B)
840 erg
done
clear
C)
840 W
done
clear
D)
840 dyne
done
clear
View Answer play_arrow
question_answer 24) A Carnot engine operates between 227 °C and 27 °C. Efficiency of the engine will be
A)
1/3
done
clear
B)
2/5
done
clear
C)
3/4
done
clear
D)
3/5
done
clear
View Answer play_arrow
question_answer 25) A capacitor of 20\[8\times {{10}^{-3}}V\] capacity charged up to 500 V is connected in parallel with another capacitor of 10\[6\times {{10}^{-3}}V\] which is charged up to 200 V. Their common potential is
A)
500 V
done
clear
B)
400 V
done
clear
C)
300 V
done
clear
D)
200 V
done
clear
View Answer play_arrow
question_answer 26) What will happen when a 40 W, 220 V lamp and 100 W, 220 V lamp are connected in series across 40 V supply?
A)
100 W lamp will fuse
done
clear
B)
40 W lamp will fuse
done
clear
C)
Both lamps will fuse
done
clear
D)
Neither lamp will fuse
done
clear
View Answer play_arrow
question_answer 27) A parallel plate capacitor having a plate separation of 2 mm is charged by connecting it to a 300 V supply. The energy density is
A)
0.01 J/\[4\times {{10}^{-3}}V\]
done
clear
B)
0.1 J/\[2\times {{10}^{-3}}V\]
done
clear
C)
1.0 J/\[{{f}_{R}}<{{f}_{G}}<{{f}_{V}}\]
done
clear
D)
10 J/\[{{f}_{V}}<{{f}_{G}}<{{f}_{R}}\]
done
clear
View Answer play_arrow
question_answer 28) A \[5{}^\circ C\] rise in the temperature is observed in a conductor by passing some current. When the current is doubled, then rise in temperature will be equal to
A)
\[5{}^\circ C\]
done
clear
B)
\[10{}^\circ C\]
done
clear
C)
\[20{}^\circ C\]
done
clear
D)
\[40{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 29) If a 2 kW boiler is used every day for 1 h, then electrical energy consumed by boiler in thirty days is
A)
15 unit
done
clear
B)
60unit
done
clear
C)
120 unit
done
clear
D)
240 unit
done
clear
View Answer play_arrow
question_answer 30) Two identical wires are first connected in series and then in parallel. The amount of heat produced in both the conditions are in the ratio of
A)
1 : 2
done
clear
B)
2 :1
done
clear
C)
1 : 4
done
clear
D)
4 :1
done
clear
View Answer play_arrow
question_answer 31) A galvanometer can be changed into an ammeter by using
A)
low resistance shunt in series
done
clear
B)
low resistance shunt in parallel
done
clear
C)
high resistance shunt in series
done
clear
D)
high resistance shunt in parallel
done
clear
View Answer play_arrow
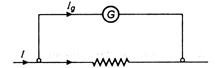
question_answer 32)
A galvanometer gives full scale deflection with 0.2 A in a coil as shown in the figure. The resistance of its coil is 10 How much shunt resistance is required to convert it into an ammeter to read current upto 1.8 A ?
A)
1.25 \[{{f}_{G}}<{{f}_{R}}<{{f}_{V}}\]
done
clear
B)
2.5 \[{{f}_{G}}<{{f}_{V}}<{{f}_{R}}\]
done
clear
C)
50 \[\mu \]
done
clear
D)
10.0\[6.6\times {{10}^{-34}}\]
done
clear
View Answer play_arrow
question_answer 33) The coefficient of mutual inductance, when magnetic flux is \[\frac{16}{R}\] Wb and change in current is 0.01 A, is
A)
2 H
done
clear
B)
3H
done
clear
C)
4 H
done
clear
D)
8H
done
clear
View Answer play_arrow
question_answer 34) The magnetic needle lying parallel to the magnetic field requires W units of work to rotate it through \[60{}^\circ \]. The torque needed to maintain the needle in this position is
A)
3W
done
clear
B)
\[\frac{16}{3R}\]W
done
clear
C)
\[\frac{16}{5R}\]
done
clear
D)
\[\frac{16}{7R}\]
done
clear
View Answer play_arrow
question_answer 35) If two electric bulbs have 40 W and 60 W rating at 220 V, then the ratio of their resistances will be
A)
3 : 2
done
clear
B)
3 : 8
done
clear
C)
4 : 3
done
clear
D)
9 : 4
done
clear
View Answer play_arrow
question_answer 36) A small piece of metal wire is dragged across the gap between the poles of a magnet is 0.4 s. If the change in magnetic flux in the wire is \[{{E}_{n}}\] Wb, then emf induced in the wire is
A)
\[4{{E}_{n}}\]
done
clear
B)
\[{{E}_{n}}/4\]
done
clear
C)
\[2{{E}_{n}}\]
done
clear
D)
\[{{E}_{n}}/2\]
done
clear
View Answer play_arrow
question_answer 37) In 0.1 s, the current in a coil increases from 1 A to 1.5 A. If inductance of this coil is 60 mH, then induced current in an external resistance of 3 Q will be
A)
1 A
done
clear
B)
0.5A
done
clear
C)
0.2A
done
clear
D)
0.1 A
done
clear
View Answer play_arrow
question_answer 38) The focal lengths for violet, green and red light rays are \[{{f}_{V}},{{f}_{G}}\]are \[{{f}_{r}}\]respectively. Which of the following is the true relationship?
A)
\[\overset{0}{\mathop{A}}\,\]
done
clear
B)
\[\overset{0}{\mathop{A}}\,\]
done
clear
C)
\[\overset{0}{\mathop{A}}\,\]
done
clear
D)
\[\overset{0}{\mathop{A}}\,\]
done
clear
View Answer play_arrow
question_answer 39) The power of combination of two lenses of powers + 1.5 D and - 2.5 D is
A)
+1.5D
done
clear
B)
- 2.5 D
done
clear
C)
- 1 D
done
clear
D)
+ 1 D
done
clear
View Answer play_arrow
question_answer 40) A ray of monochromatic light suffers minimum deviation of 38°, while passing through a prism of refracting angle 60°. Refractive index of the prism material is
A)
2.4
done
clear
B)
0.8
done
clear
C)
1.3
done
clear
D)
1.5
done
clear
View Answer play_arrow
question_answer 41) In a plano-convex lens the radius of curvature of the convex lens is 10 cm. If the plane side is polished, then the focal length will be (Refractive index \[K=\frac{1}{2}m{{v}^{2}}\]= 1.5)
A)
15 cm
done
clear
B)
10 cm
done
clear
C)
2.5 cm
done
clear
D)
5 cm
done
clear
View Answer play_arrow
question_answer 42) The blue colour of sky is due to
A)
scattering
done
clear
B)
absorption
done
clear
C)
reflection
done
clear
D)
diffraction
done
clear
View Answer play_arrow
question_answer 43) The photoelectric threshold wavelength for potassium (work function being 2 eV) is (Take h \[W=\mu ngs\]J-s)
A)
310 nm
done
clear
B)
620 nm
done
clear
C)
1200 nm
done
clear
D)
2100 nm
done
clear
View Answer play_arrow
question_answer 44) The half-life period of a radioactive substance is 140 days. After how much time, 15 g will decay from a 16 g sample of the substance?
A)
140 days
done
clear
B)
280 days
done
clear
C)
420 days
done
clear
D)
560 days
done
clear
View Answer play_arrow
question_answer 45) An electron changes its position from orbitn = 2 to the orbit n = 4 of an atom. The wavelength of the emitted radiations is (R = R vdbergs constant)
A)
\[\mu \]
done
clear
B)
\[\frac{1}{2}m{{v}^{2}}=\mu mgs\]
done
clear
C)
\[\frac{1}{2}m{{v}^{2}}=\frac{1}{2}\times 10\times {{(10)}^{2}}=500kg.m/s.\]
done
clear
D)
\[v=10m/s,\mu =0.5,\,m=10kg,\,g=10m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 46) If the energy of a hydrogen atom in nth orbit is \[\Rightarrow \] then energy in the nth orbit of a singly ionized helium atom will be
A)
\[s=\frac{\frac{1}{2}\times m\times {{(10)}^{2}}}{\mu mg}\]
done
clear
B)
\[=\frac{50}{0.5\times 10}=10m.\]
done
clear
C)
\[F=\mu R\]
done
clear
D)
\[R=w=250N.\]
done
clear
View Answer play_arrow
question_answer 47) If the work function of a photometal is 6.875eV, its threshold wavelength will be \[(Take\text{ }c=3\times {{10}^{8}}m/s)\]
A)
3600\[\mu =0.3\]
done
clear
B)
2400 \[\therefore \]
done
clear
C)
1800\[F=0.3\times 250=75N.\]
done
clear
D)
1200 \[\overrightarrow{\mathbf{F}}\]
done
clear
View Answer play_arrow
question_answer 48) Which of the following is unipolar transistor ?
A)
p-n-p transistor
done
clear
B)
n-p-n transistor
done
clear
C)
Field effect transistor
done
clear
D)
Point contact transistor
done
clear
View Answer play_arrow
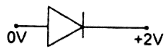
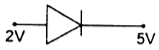
question_answer 49) In which of the following figures, junction diode is forward biased?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 50) The Boolean function given is for which gate ? Y = AB + AB
A)
NOR gate
done
clear
B)
AND gate
done
clear
C)
XOR gate
done
clear
D)
NAND gate
done
clear
View Answer play_arrow
question_answer 51) The de-Broglie wavelength associated with a particle of mass \[{{10}^{-6}}\] kg moving with a velocity of \[10m{{s}^{-1}}\] is
A)
\[6.63\times {{10}^{-7}}\]
done
clear
B)
\[6.63\times {{10}^{-16}}\]
done
clear
C)
\[6.63\times {{10}^{-21}}\]
done
clear
D)
\[6.63\times {{10}^{-29}}\]
done
clear
View Answer play_arrow
question_answer 52) The state of hybridisation of B in \[BC{{l}_{3}}\]is
A)
\[sp\]
done
clear
B)
\[s{{p}^{2}}\]
done
clear
C)
\[s{{p}^{3}}\]
done
clear
D)
\[s{{p}^{2}}{{d}^{2}}\]
done
clear
View Answer play_arrow
question_answer 53) The significant figures in 3400 are
A)
2
done
clear
B)
5
done
clear
C)
6
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 54) One mole of an ideal gas for which \[{{C}_{V}}=\frac{3}{2}R\] is heated reversibly at a constant pressure of 1 atm from \[{{25}^{o}}C\] to \[{{100}^{o}}C\], the \[\Delta H\] is
A)
\[3.775cal\]
done
clear
B)
\[37.256cal\]
done
clear
C)
\[372.56cal\]
done
clear
D)
\[3725.6cal\]
done
clear
View Answer play_arrow
question_answer 55) If 20 g of a solute was dissolved in 500 mL of water and osmotic pressure of the solution was found to be 600 mm of Hg at \[{{15}^{o}}C\], then molecular weight of the solute is
A)
1000
done
clear
B)
1200
done
clear
C)
1400
done
clear
D)
1800
done
clear
View Answer play_arrow
question_answer 56) Work done during isothermal expansion of one mole of an ideal gas from 10 atm to 1 atm at 300 K is
A)
\[4938.8\text{ }J\]
done
clear
B)
\[4138.8\text{ }J\]
done
clear
C)
\[5744.1\text{ }J\]
done
clear
D)
\[6257.2\text{ }J\]
done
clear
View Answer play_arrow
question_answer 57) The number of sodium atoms in 2 moles of sodium ferrocyanide is
A)
\[12\times {{10}^{23}}\]
done
clear
B)
\[26\times {{10}^{23}}\]
done
clear
C)
\[34\times {{10}^{23}}\]
done
clear
D)
\[48\times {{10}^{23}}\]
done
clear
View Answer play_arrow
question_answer 58) According to Bohrs principle, the relation between principle quantum number (n) and radius of orbit is
A)
\[r\propto n\]
done
clear
B)
\[r\propto {{n}^{2}}\]
done
clear
C)
\[r\propto \frac{1}{n}\]
done
clear
D)
\[r\propto \frac{1}{{{n}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 59) A solution having hydrogen ion concentration is 0.0005 g eqvti/L, its pOH is
A)
\[8.2798\]
done
clear
B)
\[10.6990\]
done
clear
C)
\[12.7854\]
done
clear
D)
\[13.3344\]
done
clear
View Answer play_arrow
question_answer 60) The number of lattice arrangements in different crystal systems are
A)
8
done
clear
B)
7
done
clear
C)
9
done
clear
D)
14
done
clear
View Answer play_arrow
question_answer 61) If equilibrium constants of reaction,\[{{N}_{2}}+{{O}_{2}}2NO\] is \[{{K}_{1}}\] and\[\frac{1}{2}{{N}_{2}}+\frac{1}{2}{{O}_{2}}NO\] is \[{{K}_{2}}\] then
A)
\[{{K}_{1}}={{K}_{2}}\]
done
clear
B)
\[{{K}_{2}}=\sqrt{{{K}_{1}}}\]
done
clear
C)
\[{{K}_{1}}=2{{K}_{2}}\]
done
clear
D)
\[{{K}_{1}}=\frac{1}{2}{{K}_{2}}\]
done
clear
View Answer play_arrow
question_answer 62) Wolframite ore is separated from tin stone ore by the process of
A)
roasting
done
clear
B)
electromagnetic
done
clear
C)
smelting
done
clear
D)
calcination
done
clear
View Answer play_arrow
question_answer 63) If solubility product of \[HgS{{O}_{4}}\] is \[6.4\times {{10}^{-5}}\], then its solubility is
A)
\[8\times {{10}^{-3}}mol/L\]
done
clear
B)
\[6.4\times {{10}^{-5}}mol/L\]
done
clear
C)
\[6.4\times {{10}^{-3}}mol/L\]
done
clear
D)
\[2.8\times {{10}^{-6}}mol/L\]
done
clear
View Answer play_arrow
question_answer 64) Disintegration constant for a radioactive substance is 0.58 per hour. Its half-life period is
A)
\[8.2\text{ }h\]
done
clear
B)
\[5.2h\]
done
clear
C)
\[1.2h\]
done
clear
D)
\[2.4\text{ }h\]
done
clear
View Answer play_arrow
question_answer 65) The rate constant of a reaction depends upon
A)
extent of reaction
done
clear
B)
time of reaction
done
clear
C)
temperature of the system
done
clear
D)
concentration of the system
done
clear
View Answer play_arrow
question_answer 66) A cylinder of 5 L capacity, filled with air at NTP, is connected with another evacuated cylinder of 30 L capacity. The resultant air pressure in both the cylinders will be
A)
10.8 cm of Hg
done
clear
B)
14.9 cm of Hg
done
clear
C)
21.8 cm of Hg
done
clear
D)
38.8 cm of Hg
done
clear
View Answer play_arrow
question_answer 67) The maximum number of hydrogen bonds formed by a water molecule in ice is
A)
4
done
clear
B)
5
done
clear
C)
7
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 68) The volume strength of \[1.5N\] \[{{H}_{2}}{{O}_{2}}\] solution is
A)
\[8.4L\]
done
clear
B)
\[4.2\text{ }L\]
done
clear
C)
\[16.8\text{ }L\]
done
clear
D)
\[5.2\text{ }L\]
done
clear
View Answer play_arrow
question_answer 69) When a substance is dissolved in a solvent, the vapour pressure of solvent decreases. It brings
A)
a decrease in boiling point of the solution
done
clear
B)
an increase in boiling point of the solution
done
clear
C)
a decrease in freezing point of the solution
done
clear
D)
an increase in freezing point of the solution
done
clear
View Answer play_arrow
question_answer 70) Acetone reacts with iodine to form iodoform in the presence of
A)
\[CaC{{O}_{3}}\]
done
clear
B)
\[NaOH\]
done
clear
C)
\[KOH\]
done
clear
D)
\[MgC{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 71) The chemical formula of diammine silver (I) chloride is
A)
\[[Ag(N{{H}_{3}})]Cl\]
done
clear
B)
\[[Ag{{(N{{H}_{3}})}_{2}}]Cl\]
done
clear
C)
\[{{[Ag{{(N{{H}_{3}})}_{2}}]}_{2}}Cl\]
done
clear
D)
\[{{[Ag{{(N{{H}_{3}})}_{2}}]}_{2}}Cl\]
done
clear
View Answer play_arrow
question_answer 72) When alkyi halide is heated with dry \[A{{g}_{2}}O\], it produces
A)
ester
done
clear
B)
ether
done
clear
C)
ketone
done
clear
D)
alcohol
done
clear
View Answer play_arrow
question_answer 73) The IUPAC name of the compound\[\overset{OC{{H}_{3}}}{\mathop{\overset{|}{\mathop{C{{H}_{3}}-C{{H}_{2}}-CH-C{{H}_{2}}-OH}}\,}}\,\] is
A)
2-methoxy-l-butanol
done
clear
B)
3-methoxy-l-butanol
done
clear
C)
2-methoxy-l-butanal
done
clear
D)
1, 2-methoxy-butanol
done
clear
View Answer play_arrow
question_answer 74) The strongest oxidising agent is
A)
\[{{F}_{2}}\]
done
clear
B)
\[C{{l}_{2}}\]
done
clear
C)
\[{{I}_{2}}\]
done
clear
D)
\[B{{r}_{2}}\]
done
clear
View Answer play_arrow
question_answer 75) Which of the following is used widely in the manufacture of lead storage battery?
A)
Arsenic
done
clear
B)
Lithium
done
clear
C)
Bismuth
done
clear
D)
Antimony
done
clear
View Answer play_arrow
question_answer 76) By annealing, steel
A)
becomes soft
done
clear
B)
becomes liquid
done
clear
C)
becomes hard and brittle
done
clear
D)
is covered with a thin film of \[F{{e}_{3}}{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 77) Iron pipes, lying in acidic soil, are often attached to the blocks of magnesium for their protection from rusting, because magnesium
A)
is lighter than iron
done
clear
B)
is readily converted into positive ion
done
clear
C)
forms a corrosion-resistant alloy with iron
done
clear
D)
prevents air from reaching the surface of iron
done
clear
View Answer play_arrow
question_answer 78) How many atoms of calcium will be deposited from a solution of \[CaC{{l}_{2}}\] by a current of 5 mA flowing for 60s?
A)
\[4.68\times {{10}^{18}}\]
done
clear
B)
\[4.68\times {{10}^{15}}\]
done
clear
C)
\[4.68\times {{10}^{12}}\]
done
clear
D)
\[4.68\times {{10}^{9}}\]
done
clear
View Answer play_arrow
question_answer 79) In the reaction\[A{{g}_{2}}O+{{H}_{2}}{{O}_{2}}\xrightarrow{{}}2Ag+{{H}_{2}}O+{{O}_{2}},\]\[{{H}_{2}}{{O}_{2}}\] acts as
A)
reducing agent
done
clear
B)
oxidising agent
done
clear
C)
bleaching agent
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 80) Which of the following acid is formed when\[Si{{F}_{4}}\] reacts with water?
A)
\[Si{{F}_{4}}\]
done
clear
B)
\[{{H}_{2}}Si{{F}_{4}}\]
done
clear
C)
\[{{H}_{2}}S{{O}_{4}}\],
done
clear
D)
\[{{H}_{2}}Si{{F}_{6}}\]
done
clear
View Answer play_arrow
question_answer 81) Which of the following statement is correct with respect to the property of elements with an increase in atomic number in the carbon family (group 14)?
A)
Atomic size decreases
done
clear
B)
lonisation energy increases
done
clear
C)
Metallic character decreases
done
clear
D)
Stability of \[+2\] oxidation state increases
done
clear
View Answer play_arrow
question_answer 82) Boron halides behave as Lewis base, because of their
A)
ionic nature
done
clear
B)
acidic nature
done
clear
C)
covalent nature
done
clear
D)
electron deficient nature
done
clear
View Answer play_arrow
question_answer 83) The most efficient agent for the absorption of \[S{{O}_{3}}\] is
A)
\[80%{{H}_{2}}S{{O}_{4}}~~\]
done
clear
B)
\[98%{{H}_{2}}S{{O}_{4}}~~\]
done
clear
C)
\[50%{{H}_{2}}S{{O}_{4}}~~\]
done
clear
D)
\[20%{{H}_{2}}{{S}_{2}}{{O}_{7}}~~\]
done
clear
View Answer play_arrow
question_answer 84) Alkaline earth metals are denser than alkali metals, because metallic bonding in alkaline earth metals, is
A)
stronger
done
clear
B)
weaker
done
clear
C)
volatile
done
clear
D)
not present
done
clear
View Answer play_arrow
question_answer 85) Toluene is oxidised to benzoic acid by
A)
\[KMn{{O}_{4}}\]
done
clear
B)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]
done
clear
C)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 86) Methane and ethane both can be prepared in one step by which of the following compound?
A)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
B)
\[C{{H}_{3}}OH\]
done
clear
C)
\[C{{H}_{3}}Br\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 87) Phenol can be converted into salicylaldehyde by
A)
Etard reaction
done
clear
B)
Perkin reaction
done
clear
C)
Cannizaro reaction
done
clear
D)
Reimer-Tiemann reaction
done
clear
View Answer play_arrow
question_answer 88) Which of the following oil is obtained from benzene after fractional distillation of coal tar?
A)
Light oil
done
clear
B)
Heavy oil
done
clear
C)
Middle oil
done
clear
D)
Anthracene oil
done
clear
View Answer play_arrow
question_answer 89) Azo dyes are prepared from
A)
phenol
done
clear
B)
ethyl bromide
done
clear
C)
benzaldehyde
done
clear
D)
benzoic acid
done
clear
View Answer play_arrow
question_answer 90) Which of the following is strongest base?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 91) Which of the following is the most stable compound?
A)
\[P{{h}_{3}}\overset{+}{\mathop{C}}\,\]
done
clear
B)
\[P{{h}_{2}}\overset{+}{\mathop{C}}\,\,H\]
done
clear
C)
\[P{{h}_{2}}\overset{+}{\mathop{C}}\,\,{{H}_{2}}\]
done
clear
D)
\[Ph\overset{+}{\mathop{C}}\,\,{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 92) A biological catalyst is essentially
A)
an enzyme
done
clear
B)
a carbohydrate
done
clear
C)
an amino acid
done
clear
D)
a nitrogen compound
done
clear
View Answer play_arrow
question_answer 93) Haemoglobin is an example of
A)
blood
done
clear
B)
glycoprotein
done
clear
C)
fibrous protein
done
clear
D)
globular protein
done
clear
View Answer play_arrow
question_answer 94) In elastomer, intermolecular forces are
A)
nil
done
clear
B)
weaker
done
clear
C)
strong
done
clear
D)
very strong
done
clear
View Answer play_arrow
question_answer 95) When glucose reacts with bromine water, the main product is
A)
acetic acid
done
clear
B)
saccharic acid
done
clear
C)
glyceraldehyde
done
clear
D)
gluconic acid
done
clear
View Answer play_arrow
question_answer 96) In the reaction\[C{{H}_{3}}CN+2H\xrightarrow[ether]{HCl}X\xrightarrow[{{H}_{2}}O]{Boiling}Y;\]the term Y is
A)
acetone .
done
clear
B)
ethyl amine
done
clear
C)
acetaldehyde
done
clear
D)
dimethyl amine
done
clear
View Answer play_arrow
question_answer 97)
A)
Streckers reaction
done
clear
B)
Sandmeyefs reaction
done
clear
C)
Wohl-Ziegler reaction
done
clear
D)
Stephens reaction
done
clear
View Answer play_arrow
question_answer 98) A reagent used to test for unsaturation of alkene is
A)
cone. \[{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
ammoniacaI \[C{{u}_{2}}C{{l}_{2}}\]
done
clear
C)
ammoniacal \[AgN{{O}_{3}}\]
done
clear
D)
solution of \[B{{r}_{2}}\] in \[CC{{l}_{4}}\]
done
clear
View Answer play_arrow
question_answer 99) A primary amine is formed from an amide by the treatment of bromine and alkali. The primary amine has
A)
1 carbon atom less than amide
done
clear
B)
1 carbon atom more than amide
done
clear
C)
1 hydrogen atom less than amide
done
clear
D)
1 hydrogen atom more than amide
done
clear
View Answer play_arrow
question_answer 100) Which of the following is industrially prepared by passing ethylene into hypochlorous acid?
A)
Ethylene glycol
done
clear
B)
Ethylene oxide
done
clear
C)
Ethylene dinitrate
done
clear
D)
Ethane
done
clear
View Answer play_arrow
question_answer 101) The branch of biology, which deals with the study of processes and functions of an organism, is called
A)
histology
done
clear
B)
anatomy
done
clear
C)
physiology
done
clear
D)
entomology
done
clear
View Answer play_arrow
question_answer 102) Virus multiplies in
A)
soil
done
clear
B)
dead tissue
done
clear
C)
living tissue
done
clear
D)
culture medium
done
clear
View Answer play_arrow
question_answer 103) Which of the following technique, other than microscopy is used for study of cell?
A)
Maceration
done
clear
B)
Piasmolys is
done
clear
C)
Chromatography
done
clear
D)
Autoradiography
done
clear
View Answer play_arrow
question_answer 104) Robert Hooke used the term cell in the year
A)
1650
done
clear
B)
1665
done
clear
C)
1865
done
clear
D)
1960
done
clear
View Answer play_arrow
question_answer 105) Protein synthesis takes place in
A)
ribosomes
done
clear
B)
chloroplasts
done
clear
C)
mitochondria
done
clear
D)
Golgi bodies
done
clear
View Answer play_arrow
question_answer 106) Replication of centriole occurs during
A)
imerphase
done
clear
B)
prophase
done
clear
C)
late prophase
done
clear
D)
late telophase
done
clear
View Answer play_arrow
question_answer 107) L-shaped chromosomes are also called
A)
acrocentric
done
clear
B)
telocentric
done
clear
C)
sub-metacentric
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 108) Which of the following is/are grouped under phanerogams ?
A)
Angiosperms
done
clear
B)
Gymnosperms
done
clear
C)
Pteridophyles
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 109) A bacterium divides after every 35 minutes. If a culture containing \[{{10}^{s}}\]cells per mL is grown, then cell concentration per mL after 175 minutes wilt be
A)
\[175\times {{10}^{5}}\]
done
clear
B)
\[125\times {{10}^{s}}\]
done
clear
C)
\[48\times {{10}^{5}}\]
done
clear
D)
\[32\times {{10}^{5}}\]
done
clear
View Answer play_arrow
question_answer 110) Cladonia rangiferina is a/an
A)
algae
done
clear
B)
lichen
done
clear
C)
fungus
done
clear
D)
angiosperm
done
clear
View Answer play_arrow
question_answer 111) Which of the following is an algal parasite ?
A)
Volvax
done
clear
B)
Ulothrix
done
clear
C)
Porphyra
done
clear
D)
Cephaleuros
done
clear
View Answer play_arrow
question_answer 112) Which of the following is true about bryophytes ?
A)
They are thalloid
done
clear
B)
They contain chloroplast
done
clear
C)
They possess archegonia
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 113) The kidney-shaped covering of sorus in Dryopteris, is called
A)
placenta
done
clear
B)
ramentum
done
clear
C)
sporophyll
done
clear
D)
indusium
done
clear
View Answer play_arrow
question_answer 114) Which of the following is a wheat fruit ?
A)
Achene
done
clear
B)
Cypsella
done
clear
C)
Caryopsis
done
clear
D)
Endosperm
done
clear
View Answer play_arrow
question_answer 115) A gymnospennic leaf carries 16 chromosomes. The number of chromosomes in its endosperm is
A)
24
done
clear
B)
16
done
clear
C)
12
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 116) Tyioses thickenings are seen in
A)
collencnyma
done
clear
B)
phloem cells
done
clear
C)
ray parenchyma only
done
clear
D)
ray parenchyma and xylem cells
done
clear
View Answer play_arrow
question_answer 117) Which of the following plant shows multiple epidermis?
A)
Croton
done
clear
B)
Atlium
done
clear
C)
Nerium
done
clear
D)
Cucurbits
done
clear
View Answer play_arrow
question_answer 118) Double fertilization occurs among
A)
algae
done
clear
B)
bryophytes
done
clear
C)
angiospenns
done
clear
D)
gymnosperms
done
clear
View Answer play_arrow
question_answer 119) The endosperm in angiosperm develops from
A)
zygote
done
clear
B)
secondary nucleus
done
clear
C)
chalazal polar nucleus
done
clear
D)
micropylar polar nucleus
done
clear
View Answer play_arrow
question_answer 120) The fertilization in which male gametes are carried through pollen tube is known as
A)
syngamy
done
clear
B)
porogamy
done
clear
C)
siphonogamy
done
clear
D)
chalazogamy
done
clear
View Answer play_arrow
question_answer 121) Which of the following theory gives the latest explanation for the closure of stomata?
A)
ABA theory
done
clear
B)
Munch theory
done
clear
C)
Starch glucose theory
done
clear
D)
Active \[{{K}^{+}}\] transport theory
done
clear
View Answer play_arrow
question_answer 122) Loss of liquid water by guttation occurs through
A)
hydathodes
done
clear
B)
stomata
done
clear
C)
cuticle
done
clear
D)
bark
done
clear
View Answer play_arrow
question_answer 123) Which of the following is the first compound that accepts carbon dioxide during dark phase of photosynthesis?
A)
NADP
done
clear
B)
RuBP
done
clear
C)
Ferredox in
done
clear
D)
Cytochrome
done
clear
View Answer play_arrow
question_answer 124) Oxygen which is liberated during photosynthesis, comes from
A)
\[C{{O}_{\mathbf{2}}}\]
done
clear
B)
\[{{H}_{2}}p\]
done
clear
C)
chlorophyll
done
clear
D)
phosphoglyceric acid
done
clear
View Answer play_arrow
question_answer 125) In \[{{C}_{4}}\] plants, the carbon dioxide fixation occurs in
A)
guard cells
done
clear
B)
spongy cells
done
clear
C)
palisade cells
done
clear
D)
bundle sheath cells
done
clear
View Answer play_arrow
question_answer 126) Ethyl alcohol is commercially manufactured from
A)
bajra
done
clear
B)
grapes
done
clear
C)
maize
done
clear
D)
sugarcane
done
clear
View Answer play_arrow
question_answer 127) Last electron acceptor in respiration is
A)
\[{{O}_{2}}\]
done
clear
B)
\[{{H}_{2}}\]
done
clear
C)
\[C{{O}_{2}}\]
done
clear
D)
NADH
done
clear
View Answer play_arrow
question_answer 128) A hormone delaying senescence is
A)
auxin
done
clear
B)
cytokinins
done
clear
C)
ethylene
done
clear
D)
gibberellin
done
clear
View Answer play_arrow
question_answer 129) Which of the following induces flowering in long day plants ?
A)
Gibberellins
done
clear
B)
Cyrokinin
done
clear
C)
Auxins
done
clear
D)
Lthylene
done
clear
View Answer play_arrow
question_answer 130) What name has been assigned to the genus produced by a cross between cabbage and radish ?
A)
Secale
done
clear
B)
Bursa pastoris
done
clear
C)
Lysogenicophyll
done
clear
D)
Raphanobrassica
done
clear
View Answer play_arrow
question_answer 131) Genetic counsellors can identify heterozygous individuals by
A)
height of individuals
done
clear
B)
colour of individuals
done
clear
C)
screening procedures
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 132) The term heterosis was first coined by
A)
McClintock
done
clear
B)
Power
done
clear
C)
Swaminathan
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 133) Somatic hybridization is a technique of
A)
natural breeding
done
clear
B)
natural pollination
done
clear
C)
artificial pollination
done
clear
D)
somatic cells hybridization
done
clear
View Answer play_arrow
question_answer 134) Which of the following is initiation codon is eukaryotes?
A)
UAG
done
clear
B)
UGA
done
clear
C)
UAA
done
clear
D)
AUG
done
clear
View Answer play_arrow
question_answer 135) Which of the following species are restricted to an area?
A)
Sibling species
done
clear
B)
Endemic species
done
clear
C)
Allopatric species
done
clear
D)
Sympatric species
done
clear
View Answer play_arrow
question_answer 136) Biological concept of species is mainly based on
A)
reproductive isolation
done
clear
B)
morphological features only
done
clear
C)
methods of reproduction only
done
clear
D)
morphology and methods of reproduction
done
clear
View Answer play_arrow
question_answer 137) Which of the following supports a dena population of plankton and littoral vegetation
A)
Oligoirophic
done
clear
B)
Eutrophic
done
clear
C)
Lithotrophic
done
clear
D)
Agroecoirophic
done
clear
View Answer play_arrow
question_answer 138) A man-made ecosystem is
A)
less in diversity
done
clear
B)
more in diversity
done
clear
C)
man does not make ecosystem
done
clear
D)
more stable than natural ecosystem
done
clear
View Answer play_arrow
question_answer 139) Crop rotation is used by fanners to increase
A)
soil fertility
done
clear
B)
community area
done
clear
C)
organic content of soil
done
clear
D)
nitrogenous content in the soil
done
clear
View Answer play_arrow
question_answer 140) Which of the following organism form the decomposers ?
A)
Pteris
done
clear
B)
Bacteria
done
clear
C)
Saprophytic fungi
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 141) Fly-ash is a/an
A)
insectivorous plant
done
clear
B)
light airbom paniculate matter
done
clear
C)
new name of orchid plant
done
clear
D)
causal organism of various diseases
done
clear
View Answer play_arrow
question_answer 142) Some effects of sulphur dioxide and its transformation products on plant include
A)
plasmolysis
done
clear
B)
Golgi body destruction
done
clear
C)
protein dis-imegration
done
clear
D)
chlorophyll destruction
done
clear
View Answer play_arrow
question_answer 143) Which of the following plant is used for the purification of water ?
A)
Beggiata
done
clear
B)
Chlorella
done
clear
C)
Spirogyra
done
clear
D)
Eichhornia
done
clear
View Answer play_arrow
question_answer 144) In the treatment of waste water discharge, which treatment stage involves biological treatment ?
A)
Primary treatment
done
clear
B)
Secondary treatment
done
clear
C)
Tertiary treatment
done
clear
D)
Reverse osmosis stage
done
clear
View Answer play_arrow
question_answer 145) Which of the following is considered to be the best chemical method of fixing atmospheric nitrogen ?
A)
Fisher method
done
clear
B)
De can method
done
clear
C)
Haber-Bosch method
done
clear
D)
Paranas-Meyerhoff method
done
clear
View Answer play_arrow
question_answer 146) Desired improved varieties of economically useful crops are raised by
A)
migration
done
clear
B)
biofertitizer
done
clear
C)
hybridization
done
clear
D)
natural selection
done
clear
View Answer play_arrow
question_answer 147) Indian rose wood tree is a common name of
A)
Acacia
done
clear
B)
Shorea
done
clear
C)
Dalbergia
done
clear
D)
Eucalyptus
done
clear
View Answer play_arrow
question_answer 148) Emasculation is concerned with
A)
hybridization
done
clear
B)
clonal selection
done
clear
C)
mass selection
done
clear
D)
pure line selection
done
clear
View Answer play_arrow
question_answer 149) Which of the following insecticide is obtained from the roots of Derris elleptica?
A)
Cinerin
done
clear
B)
Nicotine
done
clear
C)
Roienone
done
clear
D)
Pyrethrum
done
clear
View Answer play_arrow
question_answer 150) In maize, hybrid vigour is exploited by
A)
bombarding the protoplast with DNA
done
clear
B)
crossing of two inbreed parental lines
done
clear
C)
harvesting seeds from the most productive plants
done
clear
D)
inducing mutations
done
clear
View Answer play_arrow
question_answer 151) Which of the following is not vestigial in man?
A)
Tail vertebrae
done
clear
B)
Nails
done
clear
C)
Nictitating membrane
done
clear
D)
Vermiform appendix
done
clear
View Answer play_arrow
question_answer 152) The chemical used in National Malaria Eradication Programme is
A)
2.4-D
done
clear
B)
BHC
done
clear
C)
DOT
done
clear
D)
Pyrethroid
done
clear
View Answer play_arrow
question_answer 153) A eukaryoric gene contains two kinds of base sequences. Which of these plays an important role in protein synthesis ?
A)
Imrons
done
clear
B)
Exons
done
clear
C)
Both (a) and (b)
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 154) The number of hydrogen bonds between adenine and thymine in a DNA molecule is
A)
two
done
clear
B)
three
done
clear
C)
four
done
clear
D)
eight
done
clear
View Answer play_arrow
question_answer 155) The enzyme, which combines with non-protein pan to form a functional enzyme known as
A)
Co-enzyme
done
clear
B)
holoenzyme
done
clear
C)
apoenzyme
done
clear
D)
prosthetic group
done
clear
View Answer play_arrow
question_answer 156) Which of the following enzyme digest protein in stomach?
A)
Trypsin
done
clear
B)
Pepsin
done
clear
C)
Crepsin
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 157) Passive food ingestion in Amoeba is known as
A)
impon
done
clear
B)
invagination
done
clear
C)
circumfluence
done
clear
D)
circumvallation
done
clear
View Answer play_arrow
question_answer 158) The slime moulds are characterized by the presence of
A)
elaiers
done
clear
B)
pseudo elaters
done
clear
C)
capiilirium
done
clear
D)
capitulum
done
clear
View Answer play_arrow
question_answer 159) Ecdysone is secreted from
A)
Insecta
done
clear
B)
Trematoda
done
clear
C)
Nemaroda
done
clear
D)
Polychaeta
done
clear
View Answer play_arrow
question_answer 160) In the life cycle of mosquito, comma-shaped stage is
A)
larval stage
done
clear
B)
pupal stage
done
clear
C)
imago stage
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 161) Haemocoel is found in
A)
Hydra and Aurelia
done
clear
B)
Taenia and Ascaris
done
clear
C)
Cockroach and Pi/a
done
clear
D)
Balanoglossus and Herdmania
done
clear
View Answer play_arrow
question_answer 162) The group of Anamniota includes
A)
reptiles and birds
done
clear
B)
birds and mammals
done
clear
C)
fishes and amphibians
done
clear
D)
reptiles and mammals
done
clear
View Answer play_arrow
question_answer 163) The excretory material of bony Fish is
A)
urea
done
clear
B)
protein
done
clear
C)
ammonia
done
clear
D)
amino acid
done
clear
View Answer play_arrow
question_answer 164) Different colour of frog skin are controlled by:
A)
hormones
done
clear
B)
melanocytes
done
clear
C)
nervous system
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 165) Blastula of frog has
A)
blastopore
done
clear
B)
blastocoel
done
clear
C)
archenteron
done
clear
D)
gastro pore
done
clear
View Answer play_arrow
question_answer 166) Carotene pigment is found in the cells of
A)
dermis
done
clear
B)
epidermis
done
clear
C)
adipose cell
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 167) Deboses membrane is a layer of
A)
muscular tissue
done
clear
B)
epithelial tissue
done
clear
C)
connective tissue
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 168) Achilles tendon is associated with
A)
gluteus muscle
done
clear
B)
hamstring muscle
done
clear
C)
quadriceps muscle
done
clear
D)
gastrocnemius muscle
done
clear
View Answer play_arrow
question_answer 169) The leucocytes contain, which of the following in large quantity ?
A)
Basophils
done
clear
B)
Neutrophils
done
clear
C)
Eosinophils
done
clear
D)
Monocytes
done
clear
View Answer play_arrow
question_answer 170) Which pan of our body secreted the hormone secrer in?
A)
Ileum
done
clear
B)
Stomach
done
clear
C)
Duodenum
done
clear
D)
Oesophagus
done
clear
View Answer play_arrow
question_answer 171) During inspiration, the diaphragm
A)
expands
done
clear
B)
shows no change
done
clear
C)
contracts and flattens
done
clear
D)
relaxes to become dome-shaped
done
clear
View Answer play_arrow
question_answer 172) The oxygen toxicity is related with
A)
blood poisoning
done
clear
B)
collapse of alveolar walls
done
clear
C)
failure of ventilation of lungs
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 173) Cardiac output is determined by
A)
heart rate
done
clear
B)
stroke volume
done
clear
C)
blood now
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 174) The important function of lymph is to
A)
transport oxygen to the brain
done
clear
B)
transport carbon dioxide to the lungs
done
clear
C)
return RBCs to the lymph nodes
done
clear
D)
return interstitial fluid to the blood
done
clear
View Answer play_arrow
question_answer 175) The lining of intestine and kidneys in humans is
A)
keratinized
done
clear
B)
brush border
done
clear
C)
ciliated
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 176) The yellow colour of urine is due to presence of
A)
urea
done
clear
B)
uric acid
done
clear
C)
urochrome
done
clear
D)
bilirubin
done
clear
View Answer play_arrow
question_answer 177) The Leydig cells secrete
A)
oestrogen
done
clear
B)
testosterone
done
clear
C)
progesterone
done
clear
D)
corticosterone
done
clear
View Answer play_arrow
question_answer 178) The function of pineal body is to :
A)
lighten the skin colours
done
clear
B)
control sexual behavior
done
clear
C)
regulates the period of puberty
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 179) Which of the following nerve is purely moia nerve?
A)
Vagus
done
clear
B)
Facial
done
clear
C)
Abducens
done
clear
D)
Trigeminal
done
clear
View Answer play_arrow
question_answer 180) Which of the following part of a neuron is covered by fatty sheath?
A)
Axon
done
clear
B)
Cyton
done
clear
C)
Dendrite
done
clear
D)
Node of Ranvier
done
clear
View Answer play_arrow
question_answer 181) Synsacrum of fowl is consist of about
A)
29 vertebrae
done
clear
B)
3 vertebrae
done
clear
C)
16 vertebrae
done
clear
D)
single vertebrae
done
clear
View Answer play_arrow
question_answer 182) In rabbit, head of the epididymis present at the head of the testes, is called
A)
vas deferens
done
clear
B)
gubemaculum
done
clear
C)
cauda epididymis
done
clear
D)
caput epididymis
done
clear
View Answer play_arrow
question_answer 183) The extra embryonic membranes of mammalian embryo are derived from
A)
trophoblast
done
clear
B)
follicle cells
done
clear
C)
formative cells
done
clear
D)
inner cell mass
done
clear
View Answer play_arrow
question_answer 184) The eggs of silk moth are
A)
homolecithal
done
clear
B)
telolecithal
done
clear
C)
mesolecithal
done
clear
D)
centrolecithal
done
clear
View Answer play_arrow
question_answer 185) Which of the following was formed in S. Millers experiment?
A)
Amino acids
done
clear
B)
Nucleic acids
done
clear
C)
UV radiations
done
clear
D)
Microspheres
done
clear
View Answer play_arrow
question_answer 186) Trilobites were evolved during, which of the following period?
A)
Silurian
done
clear
B)
Cambrian
done
clear
C)
Ordovician
done
clear
D)
Precambrian
done
clear
View Answer play_arrow
question_answer 187) Which of the following variations are temporary and have nothing to do with the last or next generation ?
A)
Hereditary variations
done
clear
B)
Discontinuous variations
done
clear
C)
Environmental variations
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 188) The modem man differs from the apes in
A)
protruding eyes
done
clear
B)
spare body hair
done
clear
C)
wearing of clothes
done
clear
D)
arms shorter than legs
done
clear
View Answer play_arrow
question_answer 189) The highest cranial capacity is/was present in
A)
Java man
done
clear
B)
Peking man
done
clear
C)
handy man
done
clear
D)
modem man
done
clear
View Answer play_arrow
question_answer 190) Wild life is
A)
any living organism in any habitat
done
clear
B)
predatory animals in their natural habitat
done
clear
C)
any living organisms in its natural habitat
done
clear
D)
economically important animals and plants
done
clear
View Answer play_arrow
question_answer 191) A marriage between normal visioned man and colour blind woman will produce, which of the following type of offsprings ?
A)
Normal sons and carrier daughters
done
clear
B)
Colourblind sons and carrier daughters
done
clear
C)
Colourblind sons and S0% carrier daughters
done
clear
D)
50% colourblind sons and 50% carrier daughters
done
clear
View Answer play_arrow
question_answer 192) Haemophilia is a
A)
deficiency disorder
done
clear
B)
Y-linked disorder
done
clear
C)
X-linked disorder
done
clear
D)
autosomal sex disorder
done
clear
View Answer play_arrow
question_answer 193) UV radiation from sun causes, which of the following disorder of eyes ?
A)
Cataract
done
clear
B)
Glaucoma
done
clear
C)
Dilation pupil
done
clear
D)
Some defect of retina
done
clear
View Answer play_arrow
question_answer 194) The study of relationship between size and shape is called
A)
allopatric
done
clear
B)
allelopathy
done
clear
C)
allomecry
done
clear
D)
allogamy
done
clear
View Answer play_arrow
question_answer 195) Which of the following is the most sparsely populated state of India ?
A)
Manipur
done
clear
B)
Rajasthan
done
clear
C)
Meghalaya
done
clear
D)
Arunachal Pradesh
done
clear
View Answer play_arrow
question_answer 196) The natural parthenogenesis is found in
A)
sharks
done
clear
B)
housefly
done
clear
C)
Drosophilia
done
clear
D)
honey bee
done
clear
View Answer play_arrow
question_answer 197) The interferons are
A)
antiviral proteins
done
clear
B)
antibiotic proteins
done
clear
C)
antigen proteins
done
clear
D)
antibacterial particle
done
clear
View Answer play_arrow
question_answer 198) An organic substance bound to an enzyme and essential for its activity is called
A)
Co-enzyme
done
clear
B)
holoenzyme
done
clear
C)
apoenzyme
done
clear
D)
isoenzyme
done
clear
View Answer play_arrow
question_answer 199) Who coined the term antibiotics?
A)
Darwin
done
clear
B)
Woodruff
done
clear
C)
Fleming
done
clear
D)
Selman Waksman
done
clear
View Answer play_arrow
question_answer 200) A complex polysaccharide produced from sucrose by the bacterium Leuconostoc mesenteroides is
A)
chitin
done
clear
B)
starch
done
clear
C)
cellulose
done
clear
D)
dextr
done
clear
View Answer play_arrow
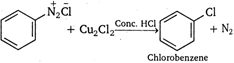 Above reaction is known as
Above reaction is known as
