 Types of root
Tap root : The tap root system develops from radicle of the germinating seed. It is also called the normal root system. The tap root system is present in dicotyledonous plants.
Adventitious root : The root system that develops from any part of the plant body other than the radicle is called the adventitious root system. It is mostly seen in monocotyledonous plants. In grasses, fibrous root system is present.
Types of root
Tap root : The tap root system develops from radicle of the germinating seed. It is also called the normal root system. The tap root system is present in dicotyledonous plants.
Adventitious root : The root system that develops from any part of the plant body other than the radicle is called the adventitious root system. It is mostly seen in monocotyledonous plants. In grasses, fibrous root system is present.  (ii) Sheathing leaf base : In grasses and many monocots, the leaf base is broad and surrounds the stem as an envelope, such a leafbase is called sheathing leaf base. e.g., Sorghum, Wheat and Palms. In grasses (Sorghum, Wheat etc.) the sheathing leaf base protects the intercalary meristem.
(iii) Amplexicaul : Leaf base completely encircles the stem. e.g., Polygonum.
(iv) Modified leaf base : The leaf bases in few plants perform accessory functions and show modifications. In Allium cepa (Onion), the leaf bases store food materials and become fleshy. In Platanus and Robenia, the leaf bases protect the axillary buds and grow around them to form cup like structures.
(v) Stipule : The stipules are the small lateral appendages present on either side of the leaf base. They protect the young leaf or leaf primordia. Leaves with stipules are called stipulate and those without them are called exstipulate. The stipules are commonly found in dicotyledons. In some grasses (Monocots) an additional outgrowth is present between leaf base and lamina. It is called ligule. The leaves having ligules are called ligulate.
Types of stipules : Depending upon the structure and position various kinds of stipules are recognized.
Free lateral stipules : A pair of freely arranged stipules present on either side of the leaf base are called free lateral stipules, e.g., Hibiscus and Cotton.
Adnate stipules : The two stipules that fuse with the leaf base or petiole on either side are called adnate stipules, e.g., Arachis and Rose.
Inter petiolar stipules : Stipules present in between the petioles of opposite leaves, e.g., Ixora and Hamelia.
Axillary stipules : more...
(ii) Sheathing leaf base : In grasses and many monocots, the leaf base is broad and surrounds the stem as an envelope, such a leafbase is called sheathing leaf base. e.g., Sorghum, Wheat and Palms. In grasses (Sorghum, Wheat etc.) the sheathing leaf base protects the intercalary meristem.
(iii) Amplexicaul : Leaf base completely encircles the stem. e.g., Polygonum.
(iv) Modified leaf base : The leaf bases in few plants perform accessory functions and show modifications. In Allium cepa (Onion), the leaf bases store food materials and become fleshy. In Platanus and Robenia, the leaf bases protect the axillary buds and grow around them to form cup like structures.
(v) Stipule : The stipules are the small lateral appendages present on either side of the leaf base. They protect the young leaf or leaf primordia. Leaves with stipules are called stipulate and those without them are called exstipulate. The stipules are commonly found in dicotyledons. In some grasses (Monocots) an additional outgrowth is present between leaf base and lamina. It is called ligule. The leaves having ligules are called ligulate.
Types of stipules : Depending upon the structure and position various kinds of stipules are recognized.
Free lateral stipules : A pair of freely arranged stipules present on either side of the leaf base are called free lateral stipules, e.g., Hibiscus and Cotton.
Adnate stipules : The two stipules that fuse with the leaf base or petiole on either side are called adnate stipules, e.g., Arachis and Rose.
Inter petiolar stipules : Stipules present in between the petioles of opposite leaves, e.g., Ixora and Hamelia.
Axillary stipules : more...  (ii) Biparous or Dichasial type : When the activity of terminal bud stops, further growth of plant takes place by two lateral branches, e.g., Viscum (Mistletoe), Silene, Stellaria, Mirabilis jalapa (Four O’ clock), Dianthus (Pink), Carissa carandas (Karonda), etc.
(iii) Multiparous or Polychasial type : When the activity of terminal bud stops, further growth of plant takes place by a whorl of three or more axillary branches. The axis is said to be multipodial, e.g., Euphorbia tirucalli, Croton, Nerium odoratum (Oleander).
(ii) Biparous or Dichasial type : When the activity of terminal bud stops, further growth of plant takes place by two lateral branches, e.g., Viscum (Mistletoe), Silene, Stellaria, Mirabilis jalapa (Four O’ clock), Dianthus (Pink), Carissa carandas (Karonda), etc.
(iii) Multiparous or Polychasial type : When the activity of terminal bud stops, further growth of plant takes place by a whorl of three or more axillary branches. The axis is said to be multipodial, e.g., Euphorbia tirucalli, Croton, Nerium odoratum (Oleander). 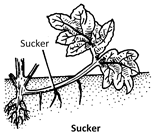 (2) Stem tuber : Stem tuber is the tuberous tip of an underground branch. It occurs beneath the soil at any depth. The axillary branches (stolons) that are produced near the soil surface grow into the soil and their tip become swollen due to accumulation of starch and proteins e.g., Solanum tuberosum (potato). In potato, the stem nature is evident by the presence of ‘eyes’ on its brownish corky surface. Each eye is a pit like structure and represents the node. Axillary bud is situated in the pit of the eye. The stem tubers are differentiated from the tuberous roots by the presence of vegetatively propagating eyes.
(3) Rhizome : The rhizome is a thickened, underground dorsiventral stem that grows horizontally at particular depth within the soil. The rhizome is brown in colour. It can be distinguished from the modified root by the presence of nodes, internodes, terminal bud, axillary bud and scale leaves. The terminal bud develops aerial shoot that bears inflorescence. Adventitious roots develop on the ventral surface of the rhizome. The rhizomes are perennial and vegetatively propagating structures. It is of following types :
(i) Rootstock : They are upright or oblique with their tips reaching the soil surface. e.g., Alocasia indica and Banana.
(ii) Straggling : They are horizontal in position and generally branched (Sympodial or Monopodial), e.g., Nelumbo nucifera (Lotus), Zingiber officinale (Ginger), Curcuma domestica (Turmeric), Saccharum etc.
(4) Corm : The corm is an underground modification of main stem. It grows vertically at particularly depth in the soil. The corm stores food materials and becomes tuberous. It is non green in colour and conical, cylindrical or flattened in shape. The corm bears scale leaves at each node. In the axils of these scale leaves axillary buds arise which grow into daughter corms. The terminal bud of the corm is large.
(2) Stem tuber : Stem tuber is the tuberous tip of an underground branch. It occurs beneath the soil at any depth. The axillary branches (stolons) that are produced near the soil surface grow into the soil and their tip become swollen due to accumulation of starch and proteins e.g., Solanum tuberosum (potato). In potato, the stem nature is evident by the presence of ‘eyes’ on its brownish corky surface. Each eye is a pit like structure and represents the node. Axillary bud is situated in the pit of the eye. The stem tubers are differentiated from the tuberous roots by the presence of vegetatively propagating eyes.
(3) Rhizome : The rhizome is a thickened, underground dorsiventral stem that grows horizontally at particular depth within the soil. The rhizome is brown in colour. It can be distinguished from the modified root by the presence of nodes, internodes, terminal bud, axillary bud and scale leaves. The terminal bud develops aerial shoot that bears inflorescence. Adventitious roots develop on the ventral surface of the rhizome. The rhizomes are perennial and vegetatively propagating structures. It is of following types :
(i) Rootstock : They are upright or oblique with their tips reaching the soil surface. e.g., Alocasia indica and Banana.
(ii) Straggling : They are horizontal in position and generally branched (Sympodial or Monopodial), e.g., Nelumbo nucifera (Lotus), Zingiber officinale (Ginger), Curcuma domestica (Turmeric), Saccharum etc.
(4) Corm : The corm is an underground modification of main stem. It grows vertically at particularly depth in the soil. The corm stores food materials and becomes tuberous. It is non green in colour and conical, cylindrical or flattened in shape. The corm bears scale leaves at each node. In the axils of these scale leaves axillary buds arise which grow into daughter corms. The terminal bud of the corm is large.
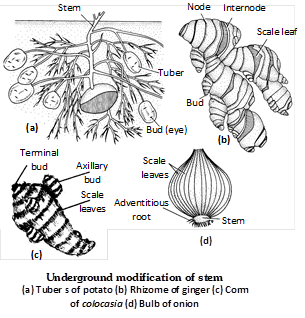 It grows into aerial shoot and bears leaves and flowers. Adventitious roots normally develop from the base or all over the body of the corm. With the help of some special adventitious roots called the contractile roots or pull roots, the corm remains constantly at a particular depth. The corm propagates vegetatively by daughter corms. e.g., Amorphophallus, Colocasia and Crocus (Saffron).
(5) Bulb : A bulb is a specialized underground stem which more...
It grows into aerial shoot and bears leaves and flowers. Adventitious roots normally develop from the base or all over the body of the corm. With the help of some special adventitious roots called the contractile roots or pull roots, the corm remains constantly at a particular depth. The corm propagates vegetatively by daughter corms. e.g., Amorphophallus, Colocasia and Crocus (Saffron).
(5) Bulb : A bulb is a specialized underground stem which more... You need to login to perform this action.
You will be redirected in
3 sec
