-
question_answer1)
Which of the following acids has the smallest dissociation constant [IIT-JEE (Screening) 2002]
A)
\[C{{H}_{3}}CHFCOOH\] done
clear
B)
\[FC{{H}_{2}}C{{H}_{2}}COOH\] done
clear
C)
\[BrC{{H}_{2}}C{{H}_{2}}COOH\] done
clear
D)
\[C{{H}_{3}}CHBrCOOH\] done
clear
View Solution play_arrow
-
question_answer2)
What is obtained, when propene is treated with N-bromo succinimide [MP PMT 2003]
A)
\[C{{H}_{3}}-\underset{\,\,}{\mathop{\underset{\underset{\,Br}{\mathop{|}}\,}{\mathop{C}}\,=C{{H}_{2}}\ \ }}\,\] done
clear
B)
\[BrC{{H}_{2}}-CH=C{{H}_{2}}\] done
clear
C)
\[BrC{{H}_{2}}-CH=CHBr\] done
clear
D)
\[BrC{{H}_{2}}-\underset{\underset{Br}{\mathop{|\,\,\,\,}}\,}{\mathop{CH}}\,-C{{H}_{2}}Br\] done
clear
View Solution play_arrow
-
question_answer3)
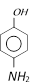
What will be the product, when carboxy phenol, obtained by Reimer Tiemann's process, is deoxidised with Zn powder [MP PMT 2003]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer4)
The vapour of a carboxylic acid HA when passed over \[Mn{{O}_{2}}\] at 573 K yields propanone. The acid HA is
A)
Methanoic acid done
clear
B)
Ethanoic acid done
clear
C)
Propanoic acid done
clear
D)
Butanoic acid done
clear
View Solution play_arrow
-
question_answer5)
Which acid is strongest or Which is most acidic [CPMT 1982, 89; BIT 1992; MP PET 1996; MP PMT/PET 1988; MP PMT 1995, 97;RPMT 1997]
A)
\[C{{l}_{2}}CH.COOH\] done
clear
B)
\[ClC{{H}_{2}}COOH\] done
clear
C)
\[C{{H}_{3}}COOH\] done
clear
D)
\[C{{l}_{3}}C.COOH\] done
clear
View Solution play_arrow
-
question_answer6)
Ethyl acetate at room temperature is a
A)
Solid done
clear
B)
Liquid done
clear
C)
Gas done
clear
D)
Solution done
clear
View Solution play_arrow
-
question_answer7)
Urea is a better fertilizer than ammonium sulphate because
A)
It has greater percentage of nitrogen done
clear
B)
It is more soluble done
clear
C)
It is weakly basic done
clear
D)
It does not produce acidity in soil done
clear
View Solution play_arrow
-
question_answer8)
The reaction of acetamide with water is an example of [Kurukshetra CEE 1998; RPMT 2000]
A)
Alcoholysis done
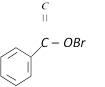
clear
B)
Hydrolysis done
clear
C)
Ammonolysis done
clear
D)
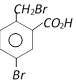
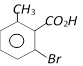
Saponification done
clear
View Solution play_arrow
-
question_answer9)
The acid which reduces Fehling solution is [KCET 1998]
A)
Methanoic acid done
clear
B)
Ethanoic acid done
clear
C)
Butanoic acid done
clear
D)
Propanoic acid done
clear
View Solution play_arrow
-
question_answer10)
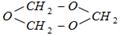
The above shown polymer is obtained when a carbon compound is allowed to stand. It is a white solid. The polymer is [CBSE PMT 1989]
A)
Trioxane done
clear
B)
Formose done
clear
C)
Paraformaldehyde done
clear
D)
Metaldehyde done
clear
View Solution play_arrow
-
question_answer11)
What will happen if \[LiAl{{H}_{4}}\] is added to an ester [CBSE PMT 2000]
A)
Two units of alcohol are obtained done
clear
B)
One unit of alcohol and one unit of acid is obtained done
clear
C)
Two units of acids are obtained done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer12)
When anisole is heated with HI, the product is [CET Pune 1998]
A)
Phenyl iodide and methyl iodide done
clear
B)
Phenol and methanol done
clear
C)
Phenyl iodide and methanol done
clear
D)
Methyl iodide and phenol done
clear
View Solution play_arrow
-
question_answer13)
When \[C{{H}_{3}}COOH\] reacts with \[C{{H}_{3}}-Mg-X\] [BVP 2003]
A)
\[C{{H}_{3}}COX\]is formed done
clear
B)
Hydrocarbon is formed done
clear
C)
Acetone is formed done
clear
D)
Alcohol is formed done
clear
View Solution play_arrow
-
question_answer14)
Which class of compounds shows H-bonding even more than in alcohols
A)
Phenols done
clear
B)
Carboxylic acids done
clear
C)
Ethers done
clear
D)
Aldehydes done
clear
View Solution play_arrow
-
question_answer15)
When propanamide reacts with \[B{{r}_{2}}\] and \[NaOH\] then which of the following compound is formed [Manipal 2001]
A)
Ethyl alcohol done
clear
B)
Propyl alcohol done
clear
C)
Propyl amine done
clear
D)
Ethylamine done
clear
View Solution play_arrow
-
question_answer16)
Hydrolysis of an ester gives a carboxylic acid which on Kolbe's electrolysis yields ethane. The ester is [EAMCET 1997; Manipal PMT 2001]
A)
Ethyl methonoate done
clear
B)
Methyl ethanoate done
clear
C)
Propylamine done
clear
D)
Ethylamine done
clear
View Solution play_arrow
-
question_answer17)
On prolonged heating of ammonium cyanate or urea, we get [DPMT 1982; CPMT 1979; MP PMT 1996]
A)
\[{{N}_{2}}\] done
clear
B)
\[C{{O}_{2}}\] done
clear
C)
Biurette done
clear
D)
Ammonium carbonate done
clear
View Solution play_arrow
-
question_answer18)
In the Gabriel's phthalimide synthesis, phthalimide is treated first with
A)
\[{{C}_{2}}{{H}_{5}}I/KOH\] done
clear
B)
Ethanolic Na done
clear
C)
Ethanol and \[{{H}_{2}}S{{O}_{4}}\] done
clear
D)
Ether and \[LiAl{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer19)
Which of the following is the strongest acid [NCERT 1984]
A)
\[C{{H}_{3}}COOH\] done
clear
B)
\[BrC{{H}_{2}}COOH\] done
clear
C)
\[ClC{{H}_{2}}COOH\] done
clear
D)
\[FC{{H}_{2}}COOH\] done
clear
View Solution play_arrow
-
question_answer20)
Which of the following reduces Tollen's reagent [MP PMT 1991]
A)
Acetic acid done
clear
B)
Citric acid done
clear
C)
Oxalic acid done
clear
D)
Formic acid done
clear
View Solution play_arrow
-
question_answer21)
Oxalic acid may be distinguished from tartaric acid by
A)
Sodium bicarbonate solution done
clear
B)
Ammonical silver nitrate solution done
clear
C)
Litmus paper done
clear
D)
Phenolphthalein done
clear
View Solution play_arrow
-
question_answer22)
The reaction of HCOOH with conc. \[{{H}_{2}}S{{O}_{4}}\] gives [DPMT 1982, CPMT 1989; MP PET 1995; AIIMS 2000; Manipal 2001; Pb. CET 2002 ]
A)
\[C{{O}_{2}}\] done
clear
B)
CO done
clear
C)
Oxalic acid done
clear
D)
Acetic acid done
clear
View Solution play_arrow
-
question_answer23)
Sulphonation of benzoic acid produces mainly [CPMT 1982]
A)
o-sulphobenzoic acid done
clear
B)
m-sulphobenzoic acid done
clear
C)
p-sulphobenzoic acid done
clear
D)
o- and p-sulphobenzoic acid done
clear
View Solution play_arrow
-
question_answer24)
Which one is strongest acid [MP PMT 1992]
A)
\[C{{H}_{2}}FCOOH\] done
clear
B)
\[C{{H}_{2}}ClCOOH\] done
clear
C)
\[CHC{{l}_{2}}COOH\] done
clear
D)
\[CH{{F}_{2}}COOH\] done
clear
View Solution play_arrow
-
question_answer25)
Which does not give silver mirror with ammoniacal \[AgN{{O}_{3}}\] [MP PET 1992]
A)
HCHO done
clear
B)
\[C{{H}_{3}}CHO\] done
clear
C)
\[C{{H}_{3}}COOH\] done
clear
D)
HCOOH done
clear
View Solution play_arrow
-
question_answer26)
\[2C{{H}_{3}}COOH\underset{{{300}^{o}}C}{\mathop{\xrightarrow{MnO}}}\,A\], product 'A' in the reaction is [RPMT 2003]
A)
\[C{{H}_{3}}C{{H}_{2}}CHO\] done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-OH\] done
clear
C)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}-\underset{\underset{O}{\mathop{||}}\,}{\mathop{C}}\,-O-\underset{\underset{O}{\mathop{||}}\,}{\mathop{C}}\,-C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer27)
Acetic acid is weak acid than sulphuric acid because [CPMT 2003]
A)
It decompose on increasing temperature done
clear
B)
It has less degree of ionisation done
clear
C)
It has -COOH group done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer28)
In \[C{{H}_{3}}COOH\] and HCOOH, HCOOH will be [CPMT 1975; DPMT 1982]
A)
Less acidic done
clear
B)
Equally acidic done
clear
C)
More acidic done
clear
D)
None done
clear
View Solution play_arrow
-
question_answer29)
Acetic anhydride reacts with excess of ammonia to form [MP PET 1992]
A)
\[2C{{H}_{3}}COON{{H}_{4}}\] done
clear
B)
\[2C{{H}_{3}}CON{{H}_{2}}\] done
clear
C)
\[C{{H}_{3}}CON{{H}_{2}}+C{{H}_{3}}COON{{H}_{4}}\] done
clear
D)
\[2C{{H}_{3}}COOH\] done
clear
View Solution play_arrow
-
question_answer30)
In the following sequence of reactions, what is D

[UPSEAT 2002]
A)
Primary amine done
clear
B)
An amide done
clear
C)
Phenyl isocyanate done
clear
D)
A chain lengthened hydrocarbon done
clear
View Solution play_arrow
-
question_answer31)
Hydrolytic reaction of fats with caustic soda is known as [MP PMT/PET 1988; AMU 1988; KCET 2000; MP PET 2001]
A)
Esterification done
clear
B)
Saponification done
clear
C)
Acetylation done
clear
D)
Carboxylation done
clear
View Solution play_arrow
-
question_answer32)
In the reaction \[C{{H}_{3}}COOH\xrightarrow{LiAl{{H}_{4}}}(A)\xrightarrow{{{I}_{2}}+NaOH}(B)\xrightarrow{Ag\text{(Dust)}}(C)\] the final product C is
A)
\[{{C}_{2}}{{H}_{5}}I\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
C)
\[{{C}_{2}}{{H}_{2}}\] done
clear
D)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer33)
Reaction of ethyl formate with excess of \[C{{H}_{3}}MgI\] followed by hydrolysis gives [IIT (Screening) 1992]
A)
n-propyl alcohol done
clear
B)
Ethanal done
clear
C)
Propanal done
clear
D)
Isopropyl alcohol done
clear
View Solution play_arrow
-
question_answer34)
Of the following four reactions, formic and acetic acids differ in which respect [CPMT 1990, 93]
A)
Replacement of hydrogen by sodium done
clear
B)
Formation of ester with alcohol done
clear
C)
Reduction of Fehling solution done
clear
D)
Blue litmus reaction done
clear
View Solution play_arrow
-
question_answer35)
Formaldehyde and formic acid can be distinguished using [AFMC 1993]
A)
Tollen's reagent done
clear
B)
Fehling solution done
clear
C)
Ferric chloride done
clear
D)
Sodium bicarbonate done
clear
View Solution play_arrow
-
question_answer36)
Ester and acetamide are distinguished by [BHU 1996]
A)
Hydrolysis with strong acids or alkali done
clear
B)
Derivatives of fatty acids done
clear
C)
Both A and B done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer37)
Acetic acid exists as a dimer in benzene solution. This is due to [MP PMT 1989; CPMT 1982]
A)
Condensation done
clear
B)
Presence of \[-COOH\] group done
clear
C)
Presence of \[\alpha -\]hydrogen done
clear
D)
Hydrogen bonding done
clear
View Solution play_arrow
-
question_answer38)
Which of the following compounds will react with \[NaHC{{O}_{3}}\] solution to give sodium salt and carbon dioxide [CBSE PMT 1999; BHU 1983, 2002]
A)
Phenol done
clear
B)
n-hexanol done
clear
C)
Acetic acid done
clear
D)
Both A and B done
clear
View Solution play_arrow
-
question_answer39)
Acetic acid dissolved in benzene shows a molecular mass of [MP PET 1993]
A)
30 done
clear
B)
60 done
clear
C)
120 done
clear
D)
240 done
clear
View Solution play_arrow
-
question_answer40)
The reaction \[2C{{H}_{3}}-\underset{\underset{O}{\mathop{||}}\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}{\mathop{C-O{{C}_{2}}{{H}_{5}}\xrightarrow{{{C}_{2}}{{H}_{5}}ONa}}}\,\]\[C{{H}_{3}}-\underset{\underset{O}{\mathop{||}}\,}{\mathop{C}}\,-C{{H}_{2}}-\underset{\underset{O}{\mathop{||}}\,}{\mathop{C}}\,-O{{C}_{2}}{{H}_{5}}+{{C}_{2}}{{H}_{5}}OH\] is called [MP PMT 2003; KCET 1996]
A)
Etard reaction done
clear
B)
Perkin's reaction done
clear
C)
Claisen condensation done
clear
D)
Claisen Schmidt reaction done
clear
View Solution play_arrow
-
question_answer41)
Which is the strongest acid? (pKa value is given in the bracket) [MP PMT 1997; BHU 2003]
A)
\[HCOOH\,(3.77)\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}COOH\,(4.22)\] done
clear
C)
\[C{{H}_{3}}COOH\,(4.71)\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}COOH\,(4.88)\] done
clear
View Solution play_arrow
-
question_answer42)
In the presence of iodine catalyst, chlorine reacts with acetic acid to form [MP PMT 1997]
A)
\[\overset{\overset{\,\,\,\,O}{\mathop{\,\,\,\,\,||}}\,}{\mathop{C{{H}_{3}}-C-Cl}}\,\] done
clear
B)
\[\overset{\overset{\,\,\,\,\,\,O}{\mathop{\,\,\,\,\,\,\,\,\,||}}\,}{\mathop{C{{H}_{2}}Cl-C-OH}}\,\] done
clear
C)
\[\overset{\overset{\,\,\,Cl}{\mathop{\,\,|}}\,}{\mathop{\underset{\underset{\,\,Cl}{\mathop{\,\,\,|}}\,}{\mathop{C{{H}_{3}}-C-OH}}\,}}\,\] done
clear
D)
\[\overset{\overset{O\,\,}{\mathop{||\,\,\,\,}}\,}{\mathop{C{{H}_{3}}-C-O-Cl}}\,\] done
clear
View Solution play_arrow
-
question_answer43)
The acid showing salt-like character in aqueous solution is [MP PET/PMT 1998]
A)
Acetic acid done
clear
B)
Benzoic acid done
clear
C)
Formic acid done
clear
D)
\[\alpha -\]amino acetic acid done
clear
View Solution play_arrow
-
question_answer44)
\[C{{H}_{3}}COOH\underset{{{P}_{2}}{{O}_{5}}}{\mathop{\xrightarrow{\Delta }}}\,X\]. Identify X [JIPMER 2000; CPMT 2003]
A)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}CHO\] done
clear
C)
\[{{(C{{H}_{3}}CO)}_{2}}O\] done
clear
D)
\[C{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer45)
Formic acid [MP PET/PMT 1988]
A)
Is immiscible with water done
clear
B)
Reduces the ammonical silver nitrate done
clear
C)
Is a weak acid nearly three and a half time weaker than acetic acid done
clear
D)
Is prepared by heating potassium hydroxide done
clear
View Solution play_arrow
-
question_answer46)
Given below are some statements concerning formic acid, which of them is true [CPMT 1983]
A)
It is a weaker acid than acetic acid done
clear
B)
It is a reducing agent done
clear
C)
When its calcium salt is heated, it forms a ketone done
clear
D)
It is an oxidising agent done
clear
View Solution play_arrow
-
question_answer47)
Which decolourises the colour of acidic \[KMn{{O}_{4}}\] [CPMT 1991]
A)
\[C{{H}_{3}}COOH\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}COOH\] done
clear
C)
\[COOH.\,COOH\] done
clear
D)
\[C{{H}_{3}}COO{{C}_{2}}{{H}_{5}}\] done
clear
View Solution play_arrow
-
question_answer48)
A colourless water soluble organic liquid decomposes sodium carbonate and liberates carbon dioxide. It produces black precipitate with Tollen's reagent. The liquid is [KCET 1989]
A)
Acetaldehyde done
clear
B)
Acetic acid done
clear
C)
Formaldehyde done
clear
D)
Formic acid done
clear
View Solution play_arrow
-
question_answer49)
The end product B in the sequence of reactions \[R-X\xrightarrow{C{{N}^{-}}}A\xrightarrow{NaOH}B\] is [CPMT 1985]
A)
An alkane done
clear
B)
A carboxylic acid done
clear
C)
Sodium salt of carboxylic acid done
clear
D)
A ketone done
clear
View Solution play_arrow
-
question_answer50)
\[C{{H}_{3}}C{{H}_{2}}COOH\xrightarrow{C{{l}_{2}}/Fe}X\underset{K{{O}_{4}}}{\mathop{\xrightarrow{Alcoholic}}}\,Y\] Compound Y is [DPMT 1981; JIPMER 2000; AIEEE 2002]
A)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}CN\] done
clear
C)
\[C{{H}_{2}}=CHCOOH\] done
clear
D)
\[C{{H}_{3}}CHClCOOH\] done
clear
View Solution play_arrow
-
question_answer51)
In the precipitation of soap, which can be used instead of NaCl [CPMT 1979]
A)
Na done
clear
B)
\[C{{H}_{3}}COONa\] done
clear
C)
\[N{{a}_{2}}S{{O}_{4}}\] done
clear
D)
Sodium silicate done
clear
View Solution play_arrow
-
question_answer52)
Which of the following can possibly be used as analgesic without causing addiction and moon modification [CBSE PMT 1997]
A)
Morphine done
clear
B)
N-acetyl-para-aminophenol done
clear
C)
Drazepom done
clear
D)
Tetrahydrocatinol done
clear
View Solution play_arrow
-
question_answer53)
Which of the following esters cannot undergo Claisen self condensation [CBSE PMT 1998]
A)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-COO{{C}_{2}}{{H}_{5}}\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}COO{{C}_{2}}{{H}_{5}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}COO{{C}_{2}}{{H}_{5}}\] done
clear
D)
\[{{C}_{6}}{{H}_{11}}C{{H}_{2}}COO{{C}_{2}}{{H}_{5}}\] done
clear
View Solution play_arrow
-
question_answer54)
When acetic acid is dissolved in benzene its molecular mass [AFMC 1991]
A)
Decreases done
clear
B)
Increases done
clear
C)
Either increases or decreases done
clear
D)
Suffers no change done
clear
View Solution play_arrow
-
question_answer55)
Benzoic acid has higher molecular weight in benzene and less in water because
A)
Water has lower freezing point and higher boiling point than benzene done
clear
B)
It dissociates to a greater extent in benzene than in water done
clear
C)
It associates in water and dissociates in benzene done
clear
D)
It dissociates in water and associates in benzene done
clear
View Solution play_arrow
-
question_answer56)
What is the main reason for the fact that carboxylic acids can undergo ionization [MNR 1993; Pb. PMT 2004]
A)
Absence of \[\alpha -\]hydrogen done
clear
B)
Resonance stabilisation of the carboxylate ion done
clear
C)
High reactivity of \[\alpha -\]hydrogen done
clear
D)
Hydrogen bonding done
clear
View Solution play_arrow
-
question_answer57)
Which of the following compounds will evolve hydrogen on treatment with metal [CPMT 1974]
A)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
B)
\[C{{H}_{3}}COOH\] done
clear
C)
A and B both done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer58)
When urea is heated, it forms biurette, alkaline solution of which forms ..... with \[CuS{{O}_{4}}\] solution [AFMC 1980]
A)
Violet colour done
clear
B)
Red colour done
clear
C)
Green colour done
clear
D)
Black colour done
clear
View Solution play_arrow
-
question_answer59)
Which of the following would be expected to be most highly ionised in water [AIIMS 1982]
A)
\[C{{H}_{2}}ClC{{H}_{2}}C{{H}_{2}}COOH\] done
clear
B)
\[C{{H}_{3}}CHCl.C{{H}_{2}}.COOH\] done
clear
C)
\[C{{H}_{3}}.C{{H}_{2}}.CC{{l}_{2}}.COOH\] done
clear
D)
\[C{{H}_{3}}.C{{H}_{2}}.CHCl.COOH\] done
clear
View Solution play_arrow
-
question_answer60)
Alkaline hydrolysis of esters is known as [CPMT 1986, 88, 93; MNR 1986; MP PET 1993]
A)
Saponification done
clear
B)
Hydration done
clear
C)
Esterification done
clear
D)
Alkalisation done
clear
View Solution play_arrow
-
question_answer61)
Which of the following undergoes hydrolysis when dissolved in water [CPMT 1989]
A)
\[C{{H}_{3}}COONa\] done
clear
B)
\[C{{H}_{3}}CON{{H}_{2}}\] done
clear
C)
Both A and B done
clear
D)
\[{{C}_{6}}{{H}_{5}}C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer62)
Name the end product in the following series of reactions \[C{{H}_{3}}COOH\xrightarrow{N{{H}_{3}}}A\underset{{{P}_{2}}{{O}_{5}}}{\mathop{\xrightarrow{\Delta }}}\,B\] [DPMT 1984]
A)
\[C{{H}_{4}}\] done
clear
B)
\[C{{H}_{3}}OH\] done
clear
C)
Acetonitrile done
clear
D)
Ammonium acetate done
clear
View Solution play_arrow
-
question_answer63)
Reduction of carboxylic acids gives
A)
Alcohol with hydrogen in presence of palladium done
clear
B)
Alcohol with \[LiAl{{H}_{4}}\] done
clear
C)
Aldehyde with \[LiAl{{H}_{4}}\] done
clear
D)
Alcohol with \[2HI(P)\] done
clear
View Solution play_arrow
-
question_answer64)
Which of the following substances when boiled with caustic soda solution will evolve ammonia [BHU 1983]
A)
Ethylamine done
clear
B)
Aniline done
clear
C)
Acetamide done
clear
D)
Acetoxime done
clear
View Solution play_arrow
-
question_answer65)
\[C{{H}_{2}}=CH-{{(C{{H}_{2}})}_{5}}COOH\underset{HBr}{\mathop{\xrightarrow{\text{Peroxide}}}}\,Z\] where Z is [CPMT 1996]
A)
\[\underset{\underset{Br\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}{\mathop{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}}\,}{\mathop{C{{H}_{3}}-CH-{{(C{{H}_{2}})}_{5}}COOH}}\,\] done
clear
B)
\[BrC{{H}_{2}}-{{(C{{H}_{2}})}_{6}}COOH\] done
clear
C)
\[C{{H}_{2}}=CH-{{(C{{H}_{2}})}_{5}}-C{{H}_{2}}OH\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}COOH\] done
clear
View Solution play_arrow
-
question_answer66)
HCOOH shows all tests of aldehyde because [CPMT 1996]
A)
It has one aldehyde group done
clear
B)
It is member of aldehyde done
clear
C)
All acids show tests of aldehyde done
clear
D)
Does not show any test done
clear
View Solution play_arrow
-
question_answer67)
Which one of the following orders of acid strength is correct [CBSE PMT 2003]
A)
\[RCOOH>HC\equiv CH>HOH>ROH\] done
clear
B)
\[RCOOH\ >ROH\ >HOH\ >HC\equiv CH\] done
clear
C)
\[RCOOH\ >HOH\ >ROH\ >HC\equiv CH\] done
clear
D)
\[RCOOH\ >HOH\ >HC\equiv CH\ >ROH\] done
clear
View Solution play_arrow
-
question_answer68)
The order of decreasing rate of reaction with ammonia is [Pb. PMT 1998]
A)
Anhydrides, esters, ethers done
clear
B)
Anhydrides, ethers, esters done
clear
C)
Ethers, anhydrides, esters done
clear
D)
Esters, ethers, anhydrides done
clear
View Solution play_arrow
-
question_answer69)
Oxidation of toluene with \[Cr{{O}_{3}}\] in the presence of \[(C{{H}_{3}}CO){{O}_{2}}\] gives a product 'A' which on treatment with aqueous NaOH produces [CBSE PMT 1995]
A)
\[{{C}_{6}}{{H}_{5}}CHO\] done
clear
B)
\[{{({{C}_{6}}{{H}_{5}}CO)}_{2}}O\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}COONa\] done
clear
D)
2, 4-diacetyl toluene done
clear
View Solution play_arrow
-
question_answer70)
\[C{{H}_{3}}COOC{{H}_{3}}+\] excess \[PhMgBr\]\[\to \text{product}\xrightarrow{{{H}^{+}}}X\] The product \[X\] is [Orissa JEE 2005]
A)
1, 1-diphenylethanol done
clear
B)
1, 1-diphenylmethanol done
clear
C)
Methyl phenylethanol done
clear
D)
Methyl phenylketone done
clear
View Solution play_arrow
-
question_answer71)
Which of the following is most acidic [MP PMT 1995]
A)
Formic acid done
clear
B)
Chloroacetic acid done
clear
C)
Propionic acid done
clear
D)
Acetic acid done
clear
View Solution play_arrow
-
question_answer72)
Urea on slow heating gives
A)
\[N{{H}_{2}}CON.HN{{O}_{2}}\] done
clear
B)
\[N{{H}_{2}}CONHCON{{H}_{2}}\] done
clear
C)
HCNO done
clear
D)
\[N{{H}_{2}}CON{{H}_{2}}.HN{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer73)
The principal organic product formed in the following reaction is \[C{{H}_{2}}=CH{{(C{{H}_{2}})}_{8}}COOH+HBr\xrightarrow{\text{peroxide}}\] [Pb. PMT 1998]
A)
\[C{{H}_{3}}CHBr{{(C{{H}_{2}})}_{8}}COOH\] done
clear
B)
\[C{{H}_{2}}=CH{{(C{{H}_{2}})}_{8}}COBr\] done
clear
C)
\[C{{H}_{2}}BrC{{H}_{2}}{{(C{{H}_{2}})}_{8}}COOH\] done
clear
D)
\[C{{H}_{2}}=CH{{(C{{H}_{2}})}_{7}}CHBrCOOH\] done
clear
View Solution play_arrow
-
question_answer74)
Which one of the following compound gives aspirin on reacting with acetic anhydride in presence of \[{{H}_{2}}S{{O}_{4}}\] [EAMCET 2003]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer75)
An acyl halide is formed when \[PC{{l}_{5}}\] reacts with an [CBSE PMT 1994; AIIMS 1998; CBSE PMT 2002]
A)
Acid done
clear
B)
Alcohol done
clear
C)
Amide done
clear
D)
Ester done
clear
View Solution play_arrow
-
question_answer76)
Which one of the following orders is wrong with respect to the property indicated [CBSE PMT 1994]
A)
Formic acid > acetic acid > propanoic acid (acid strength) done
clear
B)
Fluoroacetic acid > chloroacetic acid > bromoacetic acid (acid strength) done
clear
C)
Benzoic acid > phenol > cyclohexanol (acid strength) done
clear
D)
Aniline > cyclohexylamine > benzamide (basic strength) done
clear
View Solution play_arrow
-
question_answer77)
A fruity smell is produced by the reaction of \[{{C}_{2}}{{H}_{5}}OH\] with [AFMC 2000]
A)
\[PC{{l}_{5}}\] done
clear
B)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}COOH\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer78)
Which of the following orders of relative strengths of acids is correct [CPMT 2000]
A)
\[FC{{H}_{2}}COOH\,>\,ClC{{H}_{2}}COOH>BrC{{H}_{2}}COOH\] done
clear
B)
\[ClC{{H}_{2}}COOH>\,BrC{{H}_{2}}COOH>FC{{H}_{2}}COOH\] done
clear
C)
\[BrC{{H}_{2}}COOH>ClC{{H}_{2}}COOH>FC{{H}_{2}}COOH\] done
clear
D)
\[ClC{{H}_{2}}COOH>FC{{H}_{2}}COOH>BrC{{H}_{2}}COOH\] done
clear
View Solution play_arrow
-
question_answer79)
When acetamide is treated with \[NaOBr,\] the product formed is [Haryana CEET 2000]
A)
\[C{{H}_{3}}CN\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\] done
clear
C)
\[C{{H}_{3}}N{{H}_{2}}\] done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer80)
The fatty acid which shows reducing property is [Kerala CET 2000]
A)
Acetic acid done
clear
B)
Ethanoic Acid done
clear
C)
Oxalic acid done
clear
D)
Formic acid done
clear
View Solution play_arrow
-
question_answer81)
The reagent that can be used to distinguish between methanoic acid and ethanoic acid is [Kerala CET 2001, 02]
A)
Ammoniacal silver nitrate solution done
clear
B)
Neutral ferric Chloride solution done
clear
C)
Sodium carbonate solution done
clear
D)
Phenolphthalein done
clear
View Solution play_arrow
-
question_answer82)
Hydrolysis of an ester gives acid A and alcohol B. A reduces Fehling solution and oxidation of B gives A. The ester is [MP PMT 1999]
A)
Methyl formate done
clear
B)
Ethyl formate done
clear
C)
Methyl acetate done
clear
D)
Ethyl acetate done
clear
View Solution play_arrow
-
question_answer83)
Order of reactivity is [RPMT 2003]
A)
\[R-\overset{\overset{O}{\mathop{||}}\,}{\mathop{C}}\,-X\ \ >RCON{{H}_{2}}\ \ >RCOOCOR\ \ >RCOOR\] done
clear
B)
\[RCOX\ >RCOOCOR\ >RCOOR\ >RCON{{H}_{2}}\] done
clear
C)
\[RCOOR\ >RCON{{H}_{2}}\ >RCOX\ >RCOOCOR\] done
clear
D)
\[RCOOCOR\ >RCOOR\ >RCOX\ >RCON{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer84)
Right order of acidic strength is [RPMT 2003]
A)
\[C{{H}_{2}}ClCOOH>HCOOH>\]\[{{C}_{2}}{{H}_{5}}COOH>C{{H}_{3}}COOH\] done
clear
B)
\[C{{H}_{2}}ClCOOH\ \ >HCOOH\ \ >\]\[C{{H}_{3}}COOH\ \ >{{C}_{2}}{{H}_{5}}COOH\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}COOH\ \ >\ C{{H}_{3}}COOH\ \ >\]\[HCOOH\ \ >C{{H}_{2}}ClCOOH\] done
clear
D)
\[HCOOH\ \ >C{{H}_{2}}ClCOOH\ \ >\]\[C{{H}_{3}}COOH\ \ >{{C}_{2}}{{H}_{5}}COOH\] done
clear
View Solution play_arrow
-
question_answer85)
Saponification of ethyl benzoate with caustic soda as alkali gives [Kerala (Med.) 2001]
A)
Benzyl alcohol and ethanoic acid done
clear
B)
Sodium benzoate and ethanol done
clear
C)
Benzoic acid and sodium ethoxide done
clear
D)
Phenol and ethanoic acid done
clear
E)
Sodium benzoxide and ethanoic acid done
clear
View Solution play_arrow
-
question_answer86)
Lactic acid on oxidation by alkaline potassium permanganate gives [Tamil Nadu CET 2002]
A)
Tartaric acid done
clear
B)
Pyruvic acid done
clear
C)
Cinnamic acid done
clear
D)
Propionic acid done
clear
View Solution play_arrow
-
question_answer87)
\[RCOOH\xrightarrow{{}}RC{{H}_{2}}OH\] This mode of reduction of an acid to alcohol can be affected only by [CBSE PMT 1989]
A)
\[Zn/HCl\] done
clear
B)
Na-alcohol done
clear
C)
Aluminium isopropoxide and isopropyl alcohol done
clear
D)
\[LiAl{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer88)
Which one of the following compounds forms a red coloured solution on treatment with neutral \[FeC{{l}_{3}}\]solution [EAMCET 2003]
A)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}OC{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
D)
\[C{{H}_{3}}COOH\] done
clear
View Solution play_arrow
-
question_answer89)
Urea can be tested by [UPSEAT 1999; BVP 2003]
A)
Benedict test done
clear
B)
Mullicken test done
clear
C)
Ninhydrin done
clear
D)
Biuret test done
clear
View Solution play_arrow
-
question_answer90)
What are the organic products formed in the following reaction \[{{C}_{6}}{{H}_{5}}-COO-C{{H}_{3}}\underset{2.\,{{H}_{2}}O}{\mathop{\xrightarrow{1.\,LiAl{{H}_{4}}}}}\,\] [IIT 1995]
A)
\[{{C}_{6}}{{H}_{5}}-COOH\] and \[C{{H}_{4}}\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}-C{{H}_{2}}-OH\] and \[C{{H}_{4}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}-C{{H}_{3}}\] and \[C{{H}_{3}}-OH\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}-C{{H}_{2}}-OH\] and \[C{{H}_{3}}-OH\] done
clear
View Solution play_arrow
-
question_answer91)
Reaction between an acid and alcohol will give [Roorkee 1995]
A)
Higher C containing acid done
clear
B)
Secondary alcohol done
clear
C)
Alkane done
clear
D)
Ester done
clear
View Solution play_arrow
-
question_answer92)
Benzoic acid gives benzene on being heated with X and phenol gives benzene on being heated with Y. Therefore X and Y are respectively [CBSE PMT 1992]
A)
Sodalime and copper done
clear
B)
Zn dust and NaOH done
clear
C)
Zn dust and sodalime done
clear
D)
Sodalime and zinc dust done
clear
View Solution play_arrow
-
question_answer93)
The product obtained when acetic acid is treated with phosphorus trichloride is [CPMT 1989, 93, 94; RPMT 1997; AIIMS 1998; EAMCET 1998]
A)
\[C{{H}_{3}}COOPC{{l}_{3}}\] done
clear
B)
\[C{{H}_{3}}COOCl\] done
clear
C)
\[C{{H}_{3}}COCl\] done
clear
D)
\[ClC{{H}_{2}}COOH\] done
clear
View Solution play_arrow
-
question_answer94)
Acetyl chloride is reduced with \[LiAl{{H}_{4}}\] the product formed is [SCRA 1990]
A)
Methyl alcohol done
clear
B)
Ethyl alcohol done
clear
C)
Acetaldehyde done
clear
D)
Acetone done
clear
View Solution play_arrow
-
question_answer95)
In the preparation of an ester, the commonly used dehydrating agent is [KCET 1992]
A)
Phosphorus pentaxide done
clear
B)
Anhydrous calcium carbide done
clear
C)
Anhydrous aluminium chloride done
clear
D)
Concentrated sulphuric acid done
clear
View Solution play_arrow
-
question_answer96)
In the esterification reaction of alcohols [KCET 1984]
A)
\[O{{H}^{-}}\] is replaced by \[{{C}_{6}}{{H}_{5}}OH\] done
clear
B)
\[{{H}^{+}}\] is replaced by sodium metal done
clear
C)
\[O{{H}^{-}}\] is replaced by chlorine done
clear
D)
\[O{{H}^{-}}\] is replaced by \[C{{H}_{3}}CO{{O}^{-}}\] group done
clear
View Solution play_arrow
-
question_answer97)
Lower carboxylic acids are soluble in water due to [MP PET 1999]
A)
Low molecular weight done
clear
B)
Hydrogen bonding done
clear
C)
Dissociation into ions done
clear
D)
Easy hydrolysis done
clear
View Solution play_arrow
-
question_answer98)
Acetamide reacts with \[{{P}_{2}}{{O}_{5}}\] (phosphorus pentaoxide) to give [AFMC 1997]
A)
Methyl cyanide done
clear
B)
Methyl cyanate done
clear
C)
Ethyl cyanide done
clear
D)
Ethyl isocyanate done
clear
View Solution play_arrow
-
question_answer99)
The reaction\[C{{H}_{3}}COOH+C{{l}_{2}}\xrightarrow{P}ClC{{H}_{2}}COOH+HCl\] is called [NSE 2001; MP PET 2003]
A)
Hell-Volhard-Zelinsky reaction done
clear
B)
Birch reaction done
clear
C)
Rosenmund reaction done
clear
D)
Hunsdiecker reaction done
clear
View Solution play_arrow
-
question_answer100)
An aqueous solution of urea [CPMT 1983]
A)
Is neutral done
clear
B)
Is acidic done
clear
C)
Is basic done
clear
D)
Can act as an acid and a base done
clear
View Solution play_arrow
-
question_answer101)
Nitration of benzoic acid gives [MP PMT 1997]
A)
3-nitrobenzoic acid done
clear
B)
2-nitrobenzoic acid done
clear
C)
2, 3-dinitrobenzoic acid done
clear
D)
2, 4-dinitrobenzoic acid done
clear
View Solution play_arrow
-
question_answer102)
The reagent used for converting ethanoic acid to ethanol is [KCET 1996; EAMCET 1998]
A)
\[LiAl{{H}_{4}}\] done
clear
B)
\[KMn{{O}_{4}}\] done
clear
C)
\[PC{{l}_{3}}\] done
clear
D)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}/{{H}^{+}}\] done
clear
View Solution play_arrow
-
question_answer103)
Which one of the following has the maximum acid strength [NCERT 1983]
A)
o-nitrobenzoic acid done
clear
B)
m-nitrobenzoic acid done
clear
C)
p-nitrobenzoic acid done
clear
D)
p-nitrophenol done
clear
View Solution play_arrow
-
question_answer104)
When benzoic acid is treated with \[PC{{l}_{5}}\]at 100°C, it gives [Orissa JEE 2003]
A)
Benzoyl chloride done
clear
B)
o-chlorobenzoic acid done
clear
C)
p-chlorobenzoic acid done
clear
D)
Benzyl chloride done
clear
View Solution play_arrow
-
question_answer105)
Oxalic acid on being heated upto \[{{90}^{o}}C\] with conc. \[{{H}_{2}}S{{O}_{4}}\] forms [AFMC 1989; MP PET 1994; MP PMT 1989]
A)
\[HCOOH+C{{O}_{2}}\] done
clear
B)
\[C{{O}_{2}}+{{H}_{2}}O\] done
clear
C)
\[C{{O}_{2}}+CO+{{H}_{2}}O\] done
clear
D)
\[HCOOH+CO\] done
clear
View Solution play_arrow
-
question_answer106)
Benzoic acid is less acidic than salicylic acid because of [Bihar MEE 1997]
A)
Hydrogen bond done
clear
B)
Inductive effect done
clear
C)
Resonance done
clear
D)
All of these done
clear
E)
None of these done
clear
View Solution play_arrow
-
question_answer107)
Lactic acid on heating with conc. \[{{H}_{2}}S{{O}_{4}}\] gives [MP PET 1996]
A)
Acetic acid done
clear
B)
Propionic acid done
clear
C)
Acrylic acid done
clear
D)
Formic acid done
clear
View Solution play_arrow
-
question_answer108)
Acetamide is [MP PET 1990; RPMT 1999]
A)
Acidic done
clear
B)
Basic done
clear
C)
Neutral done
clear
D)
Amphoteric done
clear
View Solution play_arrow
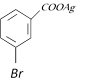
-
question_answer109)
Silver benzoate reacts with bromine to form [KCET 1996]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer110)
Acetic anhydride reacts with diethyl ether in presence of anhydrous \[AlC{{l}_{3}}\] to form [MP PMT 1992]
A)
Ethyl acetate done
clear
B)
Methyl propionate done
clear
C)
Methyl acetate done
clear
D)
Propionic acid done
clear
View Solution play_arrow
-
question_answer111)
Treatment of benzoic acid with \[C{{l}_{2}}/FeC{{l}_{3}}\] will give [KCET 1998; CET Pune 1998]
A)
p-chlorobenzoic acid done
clear
B)
o-chlorobenzoic acid done
clear
C)
2, 4-dichlorobenzoic acid done
clear
D)
m-chlorobenzoic aicd done
clear
View Solution play_arrow
-
question_answer112)
Hinsberg's reagent is [MP PMT 2003]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer113)
Which of the following is the correct order of increasing strengths of carboxylic acids
A)
\[C{{H}_{2}}FCOOH<C{{H}_{3}}COOH\]\[<C{{H}_{2}}ClCOOH<CC{{l}_{3}}COOH\] done
clear
B)
\[C{{H}_{3}}COOH<C{{H}_{2}}ClCOOH\]\[<C{{H}_{2}}FCOOH<CC{{l}_{3}}COOH\] done
clear
C)
\[C{{H}_{2}}ClCOOH<C{{H}_{2}}FCOOH\]\[<CC{{l}_{3}}COOH<C{{H}_{3}}COOH\] done
clear
D)
\[CC{{l}_{3}}COOH<C{{H}_{2}}ClCOOH\]\[<C{{H}_{2}}FCOOH<C{{H}_{3}}COOH\] done
clear
View Solution play_arrow
-
question_answer114)
The weakest acid among the following is [CPMT 1976, 82, 89; BHU 1982; CBSE PMT 1991; MP PMT 1989; Roorkee 1992; RPET 1999]
A)
\[C{{H}_{3}}COOH\] done
clear
B)
\[C{{l}_{2}}CHCOOH\] done
clear
C)
\[ClC{{H}_{2}}COOH\] done
clear
D)
\[C{{l}_{3}}CCOOH\] done
clear
View Solution play_arrow
-
question_answer115)
Consider the acidity of the carboxylic acids \[PhCOOH\] \[o-N{{O}_{2}}{{C}_{6}}{{H}_{4}}COOH\] \[p-N{{O}_{2}}{{C}_{6}}{{H}_{4}}COOH\] \[m-N{{O}_{2}}{{C}_{6}}{{H}_{4}}COOH\] Which of the following order is correct? [AIEEE 2004]
A)
\[b>d>a>c\] done
clear
B)
\[b>d>c>a\] done
clear
C)
\[a>b>c>d\] done
clear
D)
\[b>c>d>a\] done
clear
View Solution play_arrow
-
question_answer116)
On mixing ethyl acetate with aqueous sodium chloride, the composition of the resultant solution is [AIEEE 2004]
A)
\[C{{H}_{3}}COCl+{{C}_{2}}{{H}_{5}}OH+NaOH\] done
clear
B)
\[C{{H}_{3}}COONa+{{C}_{2}}{{H}_{5}}OH\] done
clear
C)
\[C{{H}_{3}}COO{{C}_{2}}{{H}_{5}}+NaCl\] done
clear
D)
\[C{{H}_{3}}Cl+{{C}_{2}}{{H}_{5}}COONa\] done
clear
View Solution play_arrow
-
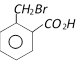
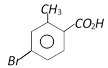
question_answer117)
o-Toluic acid on reaction with \[B{{r}_{2}}+Fe,\]gives [AIIMS 2004]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer118)
The reaction of an ester \[RCOO{R}'\] with an alcohol \[{R}''OH\] in the presence of an acid gives [Kerala PMT 2004]
A)
\[RCOOH\] done
clear
B)
\[{R}'COOH\] done
clear
C)
\[{R}''COOR\] done
clear
D)
\[RCOO{R}''\] done
clear
E)
\[{R}'COO{R}''\] done
clear
View Solution play_arrow
-
question_answer119)
\[RCOOH\]on treatment with \[PC{{l}_{5}}\] and \[KCN,\] is subjected to hydrolysis followed by Clemmensen?s reduction, the product obtained is [Kerala PMT 2004]
A)
\[RC{{H}_{2}}-COCl\] done
clear
B)
\[RC{{H}_{2}}-COOH\] done
clear
C)
\[RCOCN\] done
clear
D)
\[RCN\] done
clear
E)
\[R-OH\] done
clear
View Solution play_arrow
-
question_answer120)
The reagent which does not give acid chloride on treating with a carboxylic acid is [KCET 2004]
A)
\[PC{{l}_{5}}\] done
clear
B)
\[C{{l}_{2}}\] done
clear
C)
\[SOC{{l}_{2}}\] done
clear
D)
\[PC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer121)
An organic compound is boiled with alcoholic potash. The product is cooled and acidified with \[HCl.\] A white solid separates out. The starting compound may be [KCET 2004]
A)
Ethyl benzoate done
clear
B)
Ethyl formate done
clear
C)
Ethyl acetate done
clear
D)
Methyl acetate done
clear
View Solution play_arrow
-
question_answer122)
The \[OH\] group of an alcohol or the \[-COOH\] group of a carboxylic acid can be replaced by \[-Cl\]using [CBSE PMT 2004]
A)
Chlorine done
clear
B)
Hydrochloric acid done
clear
C)
Phosphorus pent chloride done
clear
D)
Hypochlorous acid done
clear
View Solution play_arrow
-
question_answer123)
Which of the following is most acidic [MP PET 2004]
A)
Picric acid done
clear
B)
p-nitrophenol done
clear
C)
m-nitrophenol done
clear
D)
o-p dinitrophenol done
clear
View Solution play_arrow
-
question_answer124)
Benedict's solution is not reduced by [CPMT 2004]
A)
Formaldehyde done
clear
B)
Acetaldehyde done
clear
C)
Glucose done
clear
D)
Acetic anhydride done
clear
View Solution play_arrow
-
question_answer125)
\[C{{H}_{3}}COOH\] is reacted with \[CH\equiv CH\] in presence of \[H{{g}^{++}},\] the product is [DPMT 2004; BHU 1998]
A)
\[\underset{\underset{C{{H}_{2}}(OOC{{H}_{3}})\,\,\,\,}{\mathop{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}}\,}{\mathop{C{{H}_{3}}(OOCC{{H}_{3}})}}\,\] done
clear
B)
\[\underset{\begin{smallmatrix} \,\,| \\ C{{H}_{2}}-(OOC-C{{H}_{3}})\,\, \end{smallmatrix}}{\mathop{C{{H}_{3}}\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}}\,\] done
clear
C)
\[\underset{\begin{smallmatrix} \,\,| \\ CH{{(OOC-C{{H}_{3}})}_{2}} \end{smallmatrix}}{\mathop{C{{H}_{3}}\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}}\,\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer126)
Acetic acid reacts with \[PC{{l}_{5}}\]to form [Pb. CET 2001]
A)
\[C{{H}_{3}}COCl\] done
clear
B)
\[CHC{{l}_{2}}COOH\] done
clear
C)
\[C{{H}_{2}}ClCOOH\] done
clear
D)
\[C{{H}_{3}}COOCl\] done
clear
View Solution play_arrow
-
question_answer127)
\[C{{H}_{3}}COO{{C}_{2}}{{H}_{5}}\] with excess of \[{{C}_{2}}{{H}_{5}}MgBr\]and hydrolysis gives [MH CET 2004]
A)
\[\underset{\begin{smallmatrix} \ \ \ \ \ \ \ \ \ \,| \\ \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,{{C}_{2}}{{H}_{5}}\,\,\,\,\,\,\,\,\,\,\,\,\,\,\, \end{smallmatrix}}{\mathop{C{{H}_{3}}-C=O\,\,\,\,\,\,\,\,\,\,\,\,\,\,}}\,\] done
clear
B)
\[\overset{\,\,\,\,\,\,\,\overset{\,\,\,\,\,\,\,\,\,{{C}_{2}}{{H}_{5}}}{\mathop{|}}\,}{\mathop{\underset{\underset{\,\,\,\,\,\,\,\,\,\,\,\,{{C}_{2}}{{H}_{5}}}{\mathop{\,\,\,\,\,\,\,|}}\,}{\mathop{C{{H}_{3}}-C-OH}}\,}}\,\] done
clear
C)
\[\underset{\underset{\,\,\,\,\,\,\,\,\,\,C{{H}_{3}}}{\mathop{\,\,\,\,\,\,\,|}}\,}{\mathop{C{{H}_{3}}-C=O}}\,\] done
clear
D)
\[\overset{\overset{\,\,\,\,\,\,\,\,\,\,\,\,\,{{C}_{2}}{{H}_{5}}}{\mathop{\,\,\,\,\,\,\,\,\,|}}\,}{\mathop{\underset{\underset{\,\,\,\,\,\,\,\,\,\,\,C{{H}_{3}}}{\mathop{\,\,\,\,\,\,\,|}}\,}{\mathop{C{{H}_{3}}-C=O}}\,}}\,\] done
clear
View Solution play_arrow
-
question_answer128)
Urea upon hydrolysis yields [Pb. CET 2001]
A)
Acetamide done
clear
B)
Carbonic acid done
clear
C)
Ammonium hydroxide done
clear
D)
\[N{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer129)
\[C{{H}_{3}}CHO\xrightarrow{HCN}A\xrightarrow{HOH}B.\] The product B is [Pb. CET 2003]
A)
Malonic acid done
clear
B)
Glycolic acid done
clear
C)
Lactic acid done
clear
D)
Malic acid done
clear
View Solution play_arrow
-
question_answer130)
What is the % of acetic acid present in vinegar? [AFMC ; 2004; MH CET 2003; CPMT 1974, 75]
A)
\[6-10%\] done
clear
B)
\[70-80%\] done
clear
C)
\[7-8%\] done
clear
D)
\[90-100%\] done
clear
View Solution play_arrow
-
question_answer131)
Fruity smell is given by [MH CET 2004]
A)
Esters done
clear
B)
Alcohols done
clear
C)
Chloroform done
clear
D)
Acid anhydrides done
clear
View Solution play_arrow
-
question_answer132)
Lactic acid molecule has [MH CET 2004]
A)
One chiral carbon atom done
clear
B)
Two chiral carbon atoms done
clear
C)
No chiral carbon atom done
clear
D)
As asymetric molecule done
clear
View Solution play_arrow
-
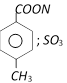
question_answer133)
4-methyl benzene sulphonic acid reacts with sodium acetate to give [IIT-JEE (Screening) 2005]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer134)
In a set reactions acid yielded a product D \[C{{H}_{3}}COOH\xrightarrow{SOC{{l}_{2}}}A\underset{Anhr.\,AlC{{l}_{3}}}{\mathop{\xrightarrow{Benzene}}}\,B\xrightarrow{HCN}C\xrightarrow{HOH}D.\] [CBSE PMT 2005]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer135)
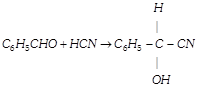
The product would be [Pb. PMT 1998]
A)
A racemate done
clear
B)
Optically active done
clear
C)
A meso compound done
clear
D)
A mixture of diastereomers done
clear
View Solution play_arrow
-
question_answer136)
What happens when 2-hydroxy benzoic acid is distilled with zinc dust, it gives [MP PET/PMT 1998]
A)
Phenol done
clear
B)
Benzoic acid done
clear
C)
Benzaldehyde done
clear
D)
A polymeric compound done
clear
View Solution play_arrow
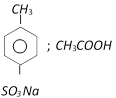
-
question_answer137)
\[C{{H}_{3}}C{{O}_{2}}{{C}_{2}}{{H}_{5}}\] on reaction with sodium ethoxide in ethanol gives A, which on heating in the presence of acid gives B compound B is [AIIMS 2005]
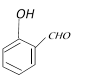
A)
\[C{{H}_{3}}COC{{H}_{2}}COOH\] done
clear
B)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
C)
D)
\[C{{H}_{2}}=C\ <\begin{matrix} O{{C}_{2}}{{H}_{5}} \\ O{{C}_{2}}{{H}_{5}} \\ \end{matrix}\] done
clear
View Solution play_arrow
-
question_answer138)
\[{{C}_{6}}{{H}_{5}}CONHC{{H}_{3}}\] can be converted into \[{{C}_{6}}{{H}_{5}}C{{H}_{2}}NHC{{H}_{3}}\] by [AIIMS 2005]
A)
\[NaB{{H}_{4}}\] done
clear
B)
\[{{H}_{2}}-Pd/C\] done
clear
C)
\[LiAl{{H}_{4}}\] done
clear
D)
\[Zn-Hg/HCl\] done
clear
View Solution play_arrow
-
question_answer139)
Among the following acids which has the lowest \[pKa\] value [AIEEE 2005]
A)
\[C{{H}_{3}}COOH\] done
clear
B)
\[HCOOH\] done
clear
C)
\[{{(C{{H}_{3}})}_{2}}CH-COOH\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}COOH\] done
clear
View Solution play_arrow
-
question_answer140)
X is heated with soda lime and gives ethane. X is [AFMC 2005]
A)
Ethanoic acid done
clear
B)
Methanoic acid done
clear
C)
Propanoic acid done
clear
D)
Either A or C done
clear
View Solution play_arrow
-
question_answer141)
Which of the following is an amphoteric acid [KCET 2005]
A)
Glycine done
clear
B)
Salicylic acid done
clear
C)
Benzoic acid done
clear
D)
Citric acid done
clear
View Solution play_arrow
-
question_answer142)
Colouration of \[B{{r}_{2}}/CC{{l}_{4}}\] will be discharged by [Orissa JEE 2005]
A)
Cinnamic acid done
clear
B)
Benzoic acid done
clear
C)
o-phthalic acid done
clear
D)
Acetophenone done
clear
View Solution play_arrow
-
question_answer143)
Order of hydrolysis for the following (I) \[RCOCl\] (II) \[RCOOR\] (III) \[RCON{{H}_{2}}\] (IV) \[{{(RCO)}_{2}}O\] [DPMT 2005]
A)
I>IV>II>III done
clear
B)
I>II>III>IV done
clear
C)
I>III>II>IV done
clear
D)
IV>III>II>I done
clear
View Solution play_arrow
-
question_answer144)
If the enolate ion combines with carbonyl group of ester, we get [DPMT 2005]
A)
Aldol done
clear
B)
\[\alpha ,\,\beta \]-unsaturated ester done
clear
C)
\[\beta \]-keto aldehyde done
clear
D)
Acid done
clear
View Solution play_arrow
-
question_answer145)
Which of the following compounds will react with \[NaHC{{O}_{3}}\] solution to give sodium salt and carbon dioxide [DPMT 2005]
A)
Acetic acid done
clear
B)
\[n\]-hexanol done
clear
C)
Phenol done
clear
D)
Both A and C done
clear
View Solution play_arrow
-
question_answer146)
A carboxylic acid is converted into its anhydride using [J & K 2005]
A)
Thionyl chloride done
clear
B)
Sulphur chloride done
clear
C)
Sulphuric acid done
clear
D)
Phosphorus pent oxide done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
![]() [UPSEAT 2002]
[UPSEAT 2002] 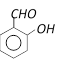 done
clear
done
clear
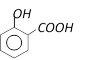 done
clear
done
clear
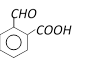 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
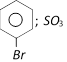 done
clear
done
clear
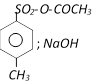 done
clear
done
clear
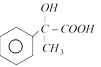 done
clear
done
clear
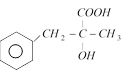 done
clear
done
clear
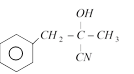 done
clear
done
clear
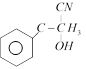 done
clear
done
clear
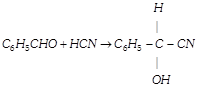 The product would be [Pb. PMT 1998]
The product would be [Pb. PMT 1998] 