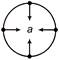
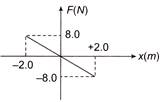
Which of the following is the unit vector perpendicular to \[\vec{A}\text{ }and\,\,\vec{B}\]?
A)
\[\frac{\hat{A}\,\,\times \,\,\hat{B}}{AB\,\,\sin \,\,\theta }\]
done
clear
B)
\[\frac{\hat{A}\,\,\times \,\,\hat{B}}{AB\,\,\cos \,\,\theta }\]
done
clear
C)
\[\frac{\vec{A}\,\,\times \,\,\vec{B}}{AB\,\,\sin \,\,\theta }\]
done
clear
D)
\[\frac{\vec{A}\,\,\times \,\,\vec{B}}{AB\,\,\cos \,\,\theta }\]
done
clear
View Answer play_arrow
The dimension of \[\frac{a}{b}\] in the equation\[P=\frac{a-{{t}^{2}}}{bx}\], where P is pressure, x is distance and t is time, are
A)
\[M{{T}^{-2}}\]
done
clear
B)
\[{{M}^{2}}L{{T}^{-\,3}}\]
done
clear
C)
\[M{{L}^{3}}{{T}^{-\,1}}\]
done
clear
D)
\[L{{T}^{-3}}\]
done
clear
View Answer play_arrow
A man standing on the roof of a house of height h throws one particle vertically downwards and another particle horizontally with the same velocity u. The ratio of their velocities when they reach the earth's surface will be
A)
\[\sqrt{2gh+{{u}^{2}}}\,\,:\,\,u\]
done
clear
B)
\[1\,\,:\,\,2\]
done
clear
C)
\[1\,\,:\,\,1\]
done
clear
D)
\[\sqrt{2gh+{{u}^{2}}}\,\,:\,\,\sqrt{2gh}\]
done
clear
View Answer play_arrow
The engine of a jet aircraft applies a thrust force of \[{{10}^{5}}N\] during take-off and causes the plane to attain a velocity of 1 km/sec in 10 sec. The mass of the plane is
A)
\[{{10}^{2}}\,kg\]
done
clear
B)
\[{{10}^{3}}\,kg\]
done
clear
C)
\[{{10}^{4}}\,kg\]
done
clear
D)
\[{{10}^{5}}\,kg\]
done
clear
View Answer play_arrow
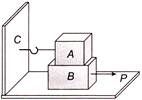
Block A weighing 100 kg rests on a block B and is tied with a horizontal string to the wall at C. Block B weighs 200 kg. The coefficient of friction between A and B is 0.25 and between B and the surface is \[1/3\]. The horizontal force P necessary to move the block B should be \[\left( g = 10m/{{s}^{2}} \right)\]
A)
1150 N
done
clear
B)
1250 N
done
clear
C)
1300 N
done
clear
D)
1420 N
done
clear
View Answer play_arrow
Two exactly similar wires of steel and copper are stretched by equal forces. If the difference in their elongations is 0.5 cm, the elongation (l) of each wire is
\[{{Y}_{s}}\,(steel)=2.0\times {{10}^{11}}\,N/{{m}^{2}}\] \[{{Y}_{c}}\,(copper)=1.2\times {{10}^{11}}\,N/{{m}^{2}}\]
A)
\[{{l}_{s}}=\,\,0.75\text{ }cm,\,\,\,{{l}_{c}}\,=\,\,1.25\text{ }cm\]
done
clear
B)
\[{{l}_{s}}=\,\,1.25\text{ }cm,\,\,\,{{l}_{c}}\,=\,\,0.75\text{ }cm\]
done
clear
C)
\[{{l}_{s}}=\,\,0.25\text{ }cm,\,\,\,{{l}_{c}}\,=\,\,0.75\text{ }cm\]
done
clear
D)
\[{{l}_{s}}=\,\,0.75\text{ }cm,\,\,\,{{l}_{c}}\,=\,\,0.25\text{ }cm\]
done
clear
View Answer play_arrow
Latent heat of ice is 80 cal/g. A man melts 60 g of ice by chewing in 1 min. His power is
A)
4800 W
done
clear
B)
336 W
done
clear
C)
1.33 W
done
clear
D)
0.75 W
done
clear
View Answer play_arrow
A gas expands with temperature according to the relation\[\operatorname{V} = k{{T}^{2/3}}\]. What is the work done Q when the temperature changes by\[30{}^\circ C\]?
A)
10 R
done
clear
B)
20 R
done
clear
C)
30 R
done
clear
D)
40 R
done
clear
View Answer play_arrow
The wavelength of maximum intensity of radiation emitted by a star is 289.8 nm. The radiation intensity for the star is (Stefan's constant\[5.67\times {{10}^{-8}}\,W{{m}^{-2}}\,{{K}^{-\,4}}\], constant\[\operatorname{b} =2898 \mu km\].
A)
\[5.67\times {{10}^{8}}\,W{{m}^{-2}}\]
done
clear
B)
\[5.67\times {{10}^{12}}\,W{{m}^{-2}}\]
done
clear
C)
\[5.67\times {{10}^{7}}\,W{{m}^{-2}}\]
done
clear
D)
\[5.67\times {{10}^{14}}\,W{{m}^{-2}}\]
done
clear
View Answer play_arrow
The kinetic energy and the potential energy of a particle executing S.H.M. are equal. The ratio, of its displacement and amplitude will be
A)
\[\frac{1}{\sqrt{2}}\]
done
clear
B)
\[\frac{\sqrt{3}}{2}\]
done
clear
C)
\[\frac{1}{2}\]
done
clear
D)
\[\sqrt{2}\]
done
clear
View Answer play_arrow
The equation of stationary wave along a stretched string is given by \[y=5\,\sin \,\frac{\pi x}{3}\,\cos \,40\pi t\] where x and y are in centimetre and t in second. The separation between two adjacent nodes is
A)
6 cm
done
clear
B)
4 cm
done
clear
C)
3 cm
done
clear
D)
1.5 cm
done
clear
View Answer play_arrow
Two identical balls having like charges and placed at a certain distance apart repel each other with a certain force. They are brought in contact and then moved apart to a distance equal to half their initial separation. The force of repulsion between them increases 4.5 times in comparison with the initial value. The ratio of the initial charges of the balls is
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
6
done
clear
View Answer play_arrow
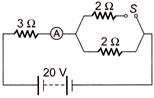
In the circuit shown, the reading of ammeter when switch S is open and when switch S is closed respectively are
A)
3 A and 4 A
done
clear
B)
4 A and 5 A
done
clear
C)
5 A and 6 A
done
clear
D)
6 A and 7 A
done
clear
View Answer play_arrow
Two bulbs consume same power when operate at 200 V and 300 V respectively. When these bulbs are connected in series across a D.C. source of 500 V, then
A)
Ratio of potential difference across them is 3/2
done
clear
B)
Ratio of potential difference across them is 9/4
done
clear
C)
Ratio of power consumed across them is 4/9
done
clear
D)
Ratio of power consumed across them is 2/3
done
clear
View Answer play_arrow
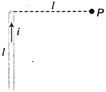
Figure shows a straight wire of length l current i. The magnitude of magnetic field produced by the current at point P is
A)
\[\frac{\sqrt{2}{{\mu }_{0}}i}{\pi l}\]
done
clear
B)
\[\frac{{{\mu }_{0}}i}{4\pi l}\]
done
clear
C)
\[\frac{\sqrt{2}{{\mu }_{0}}i}{8\pi l}\]
done
clear
D)
\[\frac{{{\mu }_{0}}i}{2\sqrt{2}\pi l}\]
done
clear
View Answer play_arrow
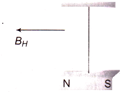
A magnet is suspended horizontally in the earth?s magnetic field. When it is displaced and then released it oscillates in a horizontal plane with a period T. If a place of wood of the same moment of inertia (about the axis of rotation) the magnet is attached to the magnet. What would the new period of oscillation of the system become?
A)
\[\frac{T}{3}\]
done
clear
B)
\[\frac{T}{2}\]
done
clear
C)
\[\frac{T}{\sqrt{2}}\]
done
clear
D)
\[T\sqrt{2}\]
done
clear
View Answer play_arrow
The phase difference between the flux linkage and the induced e.m.f. in a rotating coil in a uniform magnetic field
A)
\[\pi \]
done
clear
B)
\[\pi /2\]
done
clear
C)
\[\pi /4\]
done
clear
D)
\[\pi /6\]
done
clear
View Answer play_arrow
In an LCR circuit\[\operatorname{R} =100 ohm\]. When capacitance C is removed, the current lags behind the voltage by \[\pi /3\]. When inductance L is removed, the current leads the voltage by \[\pi /3\]. The impedance of the circuit is
A)
50 ohm
done
clear
B)
100 ohm
done
clear
C)
200 ohm
done
clear
D)
400 ohm
done
clear
View Answer play_arrow
Light of wavelength \[2475\,\overset{{}^\circ }{\mathop{A}}\,\] is incident on barium. Photoelectrons emitted describe a circle of radius 100 cm by a magnetic field of flux density \[\frac{1}{\sqrt{17}}\,\times {{10}^{-5}}\] Tesla. Work function of the barium is \[\left( Given\,\,\frac{e}{m}=1.7\times {{10}^{11}} \right)\]
A)
1.8 eV
done
clear
B)
2.1 Ev
done
clear
C)
4.5 eV
done
clear
D)
3.3 eV
done
clear
View Answer play_arrow
What is the angular momentum of an electron in Bohr's hydrogen atom whose energy is-0.544eV.
A)
\[\frac{h}{\pi }\]
done
clear
B)
\[\frac{2h}{\pi }\]
done
clear
C)
\[\frac{5h}{2\pi }\]
done
clear
D)
\[\frac{7h}{2\pi }\]
done
clear
View Answer play_arrow
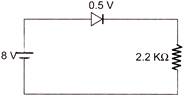
In the circuit, if the forward voltage drop for the diode is 0.5 V, the current will be
A)
3.4 mA
done
clear
B)
2 mA
done
clear
C)
2.5 mA
done
clear
D)
3 mA
done
clear
View Answer play_arrow
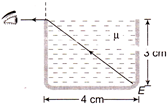
When the rectangular metal tank is filled to the top with an unknown liquid, as observer with eyes level with the top of the tank can just see the comer E; a ray that refracts towards the observer at the top surface of the liquid is shown. The refractive index of the liquid will be
A)
1.2
done
clear
B)
1.4
done
clear
C)
1.6
done
clear
D)
1.9
done
clear
View Answer play_arrow
A beam of light consisting of two wavelengths 650 nm and 520 nm is used to illuminate the slit of a Young's double slit experiment. Then the order of the bright fringe of the longer wavelength that coincide with a bright fringe of the shorter wavelength at the least distance from the central maximum is
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
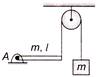
Uniform rod AB is hinged at end A in horizontal 5 position as shown in the figure. The other end is connected to a block through a massless string as shown. The pulley is smooth and massless. Masses of block and rod is same and is equal to m. Then acceleration of block just after release from this position is
A)
6g/13
done
clear
B)
g/4
done
clear
C)
3g/S
done
clear
D)
None
done
clear
View Answer play_arrow
An L-shaped thin uniform rod of total length 2l is free to rotate in a vertical plane about a horizontal axis at P as shown in the figure. The bar is released from rest. Neglect air and contact friction. The angular velocity at the instant it has rotated through \[90{}^\circ \] and reached the dotted position shown is
A)
zero
done
clear
B)
\[\sqrt{\frac{6g}{5l}}\]
done
clear
C)
\[\sqrt{\frac{3g}{5l}}\]
done
clear
D)
none
done
clear
View Answer play_arrow
If we use \[100\,\,\Omega \] and \[200\,\,\Omega \] resistance in place of R and S, we get null deflection at\[{{l}_{1}}=33.0\,cm\]. If we intercharge the resistance, the null deflection was found to be at\[{{l}_{2}}=67.0\,cm\]. The end correction \[\alpha \,\,and\,\,\beta \] should be
A)
\[\alpha =1 cm,\,\,\beta = 1 cm\]
done
clear
B)
\[\alpha =2 cm,\,\,\beta = 1 cm\]
done
clear
C)
\[\alpha =1 cm,\,\,\beta = 2 cm\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
The radioactivity of a sample is \[{{A}_{1}}\] at time \[{{t}_{1}}\] and \[{{A}_{2}}\] at time \[{{t}_{2}}\]. If the mean life of the specimen is T, the number of atoms that have disintegrated in the time interval of \[({{t}_{2}}-{{t}_{1}})\] is
A)
\[({{A}_{1}}-{{A}_{2}})\]
done
clear
B)
\[\frac{({{A}_{1}}-{{A}_{2}})}{T}\]
done
clear
C)
\[({{A}_{1}}-{{A}_{2}})\,T\]
done
clear
D)
\[{{A}_{1}}{{t}_{1}}\,-\,{{A}_{2}}{{t}_{2}}\]
done
clear
View Answer play_arrow
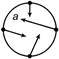
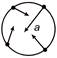
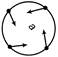
A car speeds up in a circular path. Which of the following figure illustrates the acceleration of the car?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Two condensers of capacity C and 2C are connected in parallel and these are charged up to V volt. If the battery is removed and dielectric medium of constant K is put between the plates of first condenser, then the potential at each condenser is
A)
\[\frac{V}{K+2}\]
done
clear
B)
\[2+\frac{K}{3V}\]
done
clear
C)
\[\frac{2V}{K+2}\]
done
clear
D)
\[\frac{3V}{K+2}\]
done
clear
View Answer play_arrow
A running man has half the kinetic energy of that of a boy of half of his mass. The man speeds up by 1 m/s so as to have same K.E. as that of boy. The original speed of the man will be
A)
\[\sqrt{2}\,\,m/s\]
done
clear
B)
\[(\sqrt{2}-1)\,\,m/s\]
done
clear
C)
\[\frac{1}{(\sqrt{2}-1)}\,m/s\]
done
clear
D)
\[\frac{1}{\sqrt{2}}\,m/s\]
done
clear
View Answer play_arrow
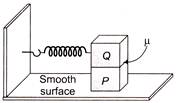
A block P of mass w is placed on a frictionless horizontal surface. Another block Q of same mass is kept on P and connected to the wall with the help of a spring of spring constant k as shown in the figure, fly is the coefficient of friction between P and Q. The blocks move together performing SHM of amplitude A. The maximum value of the friction force between P and Q is
A)
kA
done
clear
B)
\[kA/2\]
done
clear
C)
zero
done
clear
D)
\[{{\mu }_{s}}mg\]
done
clear
View Answer play_arrow
A body of mass 0.01 kg executes simple harmonic motion (S.H.M.) about \[x=0\] under the influence of a force shown below. The period of the S.H.M. is
A)
1.05 s
done
clear
B)
0.52 s
done
clear
C)
0.25 s
done
clear
D)
0.30 s
done
clear
View Answer play_arrow
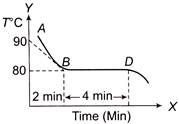
The figure given below shows the cooling curve of pure wax material after heating. It cools from A to B and solidifies along BD. If L and C are respective values of latent heat and the specific heat of the liquid wax, the ratio L/C is
A)
40
done
clear
B)
80
done
clear
C)
100
done
clear
D)
20
done
clear
View Answer play_arrow
Two capillary tubes of same diameter are put vertically one each in two liquids whose relative densities are 0.8 and 0.6 and surface tensions are 60 and 50 dyne/cm respectively Ratio of heights of liquids in the two tubes \[{{h}_{1}}/{{h}_{2}}\] is
A)
\[\frac{10}{9}\]
done
clear
B)
\[\frac{3}{10}\]
done
clear
C)
\[\frac{10}{3}\]
done
clear
D)
\[\frac{9}{10}\]
done
clear
View Answer play_arrow
If the linear momentum is increased by \[50%\], the kinetic energy will increase by
A)
\[50%\]
done
clear
B)
\[100\,%\]
done
clear
C)
\[125\,%\]
done
clear
D)
\[25\,%\]
done
clear
View Answer play_arrow
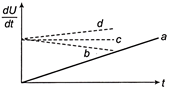
A constant current i is passed through a resistor. Taking the temperature coefficient of resistance into account, indicate which of the plots shown in figure best represents the rate of production of thermal energy in the resistor
A)
a
done
clear
B)
b
done
clear
C)
c
done
clear
D)
d
done
clear
View Answer play_arrow
Atomic power station at Tarapore has a generating capacity of 200 MW. The energy generated in a day by this station is
A)
200 MW
done
clear
B)
200 J
done
clear
C)
\[4800\,\,\times \,\,\text{1}{{\text{0}}^{6}}\,J\]
done
clear
D)
\[1728\,\,\times \,\,\text{1}{{\text{0}}^{10}}\,J\]
done
clear
View Answer play_arrow
The focal length of objective and eye lens of an astronomical telescope are respectively 2 m and 5 cm. Final image is formed at (i) least distance of distinct vision (ii) infinity. The magnifying power in both cases will be
A)
-48, -40
done
clear
B)
-40, -48
done
clear
C)
-40, 48
done
clear
D)
-48, 40
done
clear
View Answer play_arrow
Atomic weight of boron is 10.81 and it has two isotopes \[_{5}{{B}^{10}}\] and\[_{5}{{B}^{11}}\]. Then ratio of \[_{5}{{B}^{10}}:\,{{\,}_{5}}{{B}^{11}}\] in nature would be
A)
\[19:81\]
done
clear
B)
\[10: 11\]
done
clear
C)
\[15 : 16\]
done
clear
D)
\[81 : 19\]
done
clear
View Answer play_arrow
Two parallel slits 0.6 mm apart are illuminated by light source of wavelength\[6000\,\overset{{}^\circ }{\mathop{A}}\,\]. The distance between two consecutive dark fringes on a screen 1 m away from the slits is
A)
1 mm
done
clear
B)
0.01 mm
done
clear
C)
0.1 m
done
clear
D)
10 m
done
clear
View Answer play_arrow
If the dimensions of a physical quantity are given by \[{{M}^{a}}{{L}^{b}}{{T}^{c}}\], then the physical quantity will be
A)
\[pressure\,\,if\,\,a=1,\,\,b=-1,\,\,c=-2\]
done
clear
B)
\[velocity\,\,if\,\,a=1,\,\,b=0,\,\,c=-1\]
done
clear
C)
\[acceleration\,\,if\,\,a=1,\,\,b=1,\,\,c=-2\]
done
clear
D)
\[force\,\,if\,\,a=0,\,\,b=-1,\,\,c=-2\]
done
clear
View Answer play_arrow
In dimension of critical velocity \[{{\nu }_{c}}\], of liquid following through a tube are expressed as \[({{\eta }^{x}}\,{{\rho }^{y}}\,{{r}^{z}})\], where \[\eta ,\,\,\rho \,\,and\,\,r\] are the coefficient of viscosity of liquid, density of liquid and radius of the tube respectively, then the values of x, y and z are given by:
A)
\[1,\,\,1,\,\,1\]
done
clear
B)
\[1,\,\,-1,\,\,-1\]
done
clear
C)
\[-1,\,\,-1,\text{ }1\]
done
clear
D)
\[-1,\,\,-1,\,\,-1\]
done
clear
View Answer play_arrow
A ball is thrown vertically upward. It has a speed of 10 m/s when it has reached one half of its maximum height. How high does the ball rise? \[(Taking\,\,g=10m/{{s}^{2}})\]
A)
15 m
done
clear
B)
10 m
done
clear
C)
20 m
done
clear
D)
5 m
done
clear
View Answer play_arrow
A bus is moving with a speed of \[10\text{ }m{{s}^{-}}^{1}\] on a straight road. A scooterist wishes to overtake the bus in 100 s. If the bus is at a distance of 1 km from the scooterist, with what speed should the scooterist chase the bus?
A)
\[20\,\,m{{s}^{-}}^{1}\]
done
clear
B)
\[40\,\,m{{s}^{-}}^{1}\]
done
clear
C)
\[25\,\,m{{s}^{-}}^{1}\]
done
clear
D)
\[10\,\,m{{s}^{-}}^{1}\]
done
clear
View Answer play_arrow
A stone falls freely under gravity. It covers distances \[{{h}_{1}},\,\,{{h}_{2}}\,and\,\,{{h}_{3}}\] in the first 5 seconds, the next 5 seconds and the next 5 seconds respectively. The relation between \[{{h}_{1}},\,\,{{h}_{2}}\,and\,\,{{h}_{3}}\] is:
A)
\[{{h}_{1}}\,=\,\,2{{h}_{2}}=3{{h}_{3}}\]
done
clear
B)
\[{{h}_{1}}\,=\,\,\frac{{{h}_{2}}}{3}\,\,=\,\,\frac{{{h}_{3}}}{5}\]
done
clear
C)
\[{{h}_{2}}\,=\,\,3{{h}_{1}}\,and\,\,{{h}_{3}}=3{{h}_{2}}\]
done
clear
D)
\[{{h}_{1}}\,\,=\,\,{{h}_{2}}\,=\,{{h}_{3}}\]
done
clear
View Answer play_arrow
Lattice energy of an ionic compound depends upon
A)
charge on the ion only
done
clear
B)
size of the ion only
done
clear
C)
packing of ions only
done
clear
D)
charge on the ion and size of the ion
done
clear
View Answer play_arrow
Electrovalent compounds do not have
A)
high M.P. and low B.P.
done
clear
B)
high dielectric constant
done
clear
C)
high M.P. and high B.P.
done
clear
D)
high polarity
done
clear
View Answer play_arrow
2.0 molar solution is obtained, when 0.5 mole solute is dissolved in
A)
250 ml solvent
done
clear
B)
250 g solvent
done
clear
C)
250 ml solution
done
clear
D)
1000 ml solvent
done
clear
View Answer play_arrow
36 g water and 828 g ethyl alcohol form an ideal solution. The mole fraction of water in it, is
A)
1.0
done
clear
B)
0.7
done
clear
C)
0.4
done
clear
D)
0.1
done
clear
View Answer play_arrow
Given \[\operatorname{P}= 0.0030 m,\,\,Q =2.40 m,\,\,R= 3000 m\], Significant figures in P, Q and R are respectively
A)
2, 2, 1
done
clear
B)
2, 3, 4
done
clear
C)
4, 2, 1
done
clear
D)
4, 2, 3
done
clear
View Answer play_arrow
The number of significant figures in 60.0001 is
A)
5
done
clear
B)
6
done
clear
C)
3
done
clear
D)
2
done
clear
View Answer play_arrow
Which is correct statement about proton
A)
Proton is nucleus of deuterium
done
clear
B)
Proton is ionized hydrogen molecule
done
clear
C)
Proton is ionized hydrogen atom
done
clear
D)
Proton is \[\alpha -particle\]
done
clear
View Answer play_arrow
Cathode rays are made up of
A)
positively charged particles
done
clear
B)
negatively charged particles
done
clear
C)
neutral particles
done
clear
D)
none of these
done
clear
View Answer play_arrow
\[X\xrightarrow{-\alpha }Y\xrightarrow{-\beta }Z\xrightarrow{-\beta }W\] In the above sequence of reaction, the elements which are isotopes of each other are
A)
X and W
done
clear
B)
Y and Z
done
clear
C)
X and Z
done
clear
D)
none of these
done
clear
View Answer play_arrow
A particle having the same charge and 200 times greater mass than that of electron is
A)
positron
done
clear
B)
proton
done
clear
C)
neutrino
done
clear
D)
meson
done
clear
View Answer play_arrow
In chemical reaction \[A\rightleftharpoons B\], the system will be known in equilibrium when
A)
A completely changes to B
done
clear
B)
\[50\,%\] of A changes to B.
done
clear
C)
The rate of change of A to B and B to A on both the sides are same
done
clear
D)
Only \[10\,%\] of A changes to B
done
clear
View Answer play_arrow
In the chemical reaction \[{{N}_{2}}+3{{H}_{2}}\rightleftharpoons 2N{{H}_{3}}\] at equilibrium point, state whether
A)
equal volumes of \[{{N}_{2}}\text{ }and\text{ }{{H}_{2}}\] are reacting
done
clear
B)
equal masses of \[{{N}_{2}}\text{ }and\text{ }{{H}_{2}}\] are reacting
done
clear
C)
the reaction has stopped
done
clear
D)
the same amount of ammonia is formed as is decomposed into \[{{N}_{2}}\text{ }and\text{ }{{H}_{2}}\]
done
clear
View Answer play_arrow
In which of the following, dissociation \[N{{H}_{4}}OH\] will be minimum?
A)
NaOH
done
clear
B)
\[{{H}_{2}}O\]
done
clear
C)
\[N{{H}_{4}}Cl\]
done
clear
D)
NaCl
done
clear
View Answer play_arrow
van?t Hoff factor of \[BaC{{l}_{2}}\] of cone. 0.10 M is 1.98. Percentage dissociation of \[BaC{{l}_{2}}\] on this conc. will be
A)
49
done
clear
B)
69
done
clear
C)
89
done
clear
D)
98
done
clear
View Answer play_arrow
The velocity of the chemical reaction doubles every \[10{}^\circ \,C\] rise of temperature. If the temperature is raised by \[50{}^\circ \,C\], the velocity of the reaction increases to about
A)
32 times
done
clear
B)
16 times
done
clear
C)
20 times
done
clear
D)
50 times
done
clear
View Answer play_arrow
The temperature coefficient for reaction in which food deteriorates is 2. Then food deteriorates ? times as rapidly at \[25{}^\circ \,C\] as it does at\[5{}^\circ \,C\].
A)
two
done
clear
B)
four
done
clear
C)
six
done
clear
D)
twenty
done
clear
View Answer play_arrow
One gas bleaches the colour of flowers by reduction while the other by oxidation
A)
\[CO\,\,and\,\,C{{l}^{2}}\]
done
clear
B)
\[S{{O}_{2}}\,\,and\,\,C{{l}^{2}}\]
done
clear
C)
\[{{H}_{2}}S\,\,and\,\,B{{r}_{2}}\]
done
clear
D)
\[N{{H}_{3}}\,\,and\,\,S{{O}_{2}}\]
done
clear
View Answer play_arrow
Which one of the following reactions does not involve either oxidation or reduction
A)
\[VO_{2}^{+}\,\to \,{{V}_{2}}{{O}_{3}}\]
done
clear
B)
\[Na\,\to \,N{{a}^{+}}\]
done
clear
C)
\[CrO_{4}^{2-}\,\to \,\,C{{r}_{2}}O_{7}^{2-}\]
done
clear
D)
\[Z{{n}^{2+}}\,\to \,\,Zn\]
done
clear
View Answer play_arrow
Which of the following has highest octane number?
A)
n-hexane
done
clear
B)
n-heptane
done
clear
C)
n-pentane
done
clear
D)
2, 2, 4-trimethyl pentane
done
clear
View Answer play_arrow
Which of the following reactions will not give propane?
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Cl\,\,\xrightarrow[{{H}_{2}}O]{Mg/ether}\]
done
clear
B)
\[C{{H}_{3}}COCl\,\,\xrightarrow[{{H}_{2}}O]{C{{H}_{3}}MgX}\]
done
clear
C)
\[C{{H}_{3}}CH=C{{H}_{2}}\,\,\xrightarrow[C{{H}_{3}}COOH]{{{B}_{2}}{{H}_{6}}}\]
done
clear
D)
done
clear
View Answer play_arrow
A crystalline solid has
A)
long range order
done
clear
B)
short range order
done
clear
C)
disordered arrangement
done
clear
D)
none of these
done
clear
View Answer play_arrow
Crystalline solids has
A)
glass
done
clear
B)
rubber
done
clear
C)
plastic
done
clear
D)
sugar
done
clear
View Answer play_arrow
The coordination number of a metal in coordination compounds is
A)
same as primary valency
done
clear
B)
sum of primary and secondary valencies
done
clear
C)
same as secondary valency
done
clear
D)
none of these
done
clear
View Answer play_arrow
The number of ions formed when cuprammonium, sulphate is dissolved in water is
A)
1
done
clear
B)
2
done
clear
C)
4
done
clear
D)
zero
done
clear
View Answer play_arrow
Hydrogen combines with other elements by
A)
losing an electron
done
clear
B)
gaining an electron
done
clear
C)
sharing an electron
done
clear
D)
losing, gaining or sharing electron
done
clear
View Answer play_arrow
When electric current is passed through an ionic hydride in the molten state
A)
hydrogen is liberated at the anode
done
clear
B)
hydrogen is liberated at the cathode
done
clear
C)
no reaction takes place
done
clear
D)
hydride ion migrates towards cathode
done
clear
View Answer play_arrow
Air at sea level is dense. This is a practical application of
A)
Boyle's law
done
clear
B)
Charle's law
done
clear
C)
Avogadro's law
done
clear
D)
Dalton's law
done
clear
View Answer play_arrow
Densities of two gases are in the ratio \[1\,\,:\, 2\] and their temperatures are in the ratio \[2\,\,:\, 1\], then the ratio of their respective pressures is
A)
\[1 : 1\]
done
clear
B)
\[1 : 2\]
done
clear
C)
\[2 : 1\]
done
clear
D)
\[4 : 1\]
done
clear
View Answer play_arrow
Adsorption due to strong chemical forces is called
A)
chemisorption
done
clear
B)
physisorption
done
clear
C)
reversible adsorption
done
clear
D)
both [b] and [c]
done
clear
View Answer play_arrow
50 ml of 1 M oxalic acid is shaken with 0.5 g of wood charcoal. The final concentration of the solution after adsorption is 0.5 M. Amount of oxalic acid absorbed per gram of charcoal is
A)
3.45 g
done
clear
B)
3.15 g
done
clear
C)
6.30 g
done
clear
D)
None
done
clear
View Answer play_arrow
The coinage metals are
A)
Iron, Cobalt, Nickel
done
clear
B)
Copper and Zinc
done
clear
C)
Copper, Silver and Gold
done
clear
D)
Gold and Platinum
done
clear
View Answer play_arrow
Which of the following transition metal is present in misch metal?
A)
La
done
clear
B)
Sc
done
clear
C)
Ni
done
clear
D)
Cr
done
clear
View Answer play_arrow
If a refrigerator's door is opened, then we get
A)
room heated
done
clear
B)
room cooled
done
clear
C)
more amount of heat is passed out
done
clear
D)
no effect on room
done
clear
View Answer play_arrow
In an isochoric process the increase in internal energy is
A)
equal to the heat absorbed
done
clear
B)
equal to the heat evolved
done
clear
C)
equal to the work done
done
clear
D)
equal to the sum of the heat absorbed and work done
done
clear
View Answer play_arrow
When a solution of an electrolyte is heated the conductance of the solution
A)
increases because of the electrolyte conducts better
done
clear
B)
decreases because of the increased heat
done
clear
C)
decreases because of the dissociation of the electrolyte is suppressed
done
clear
D)
increases because the electrolyte is dissociated more
done
clear
View Answer play_arrow
The passage of current liberates \[{{H}_{2}}\] at cathode and \[C{{l}_{2}}\] at anode. The solution is
A)
Copper chloride in water
done
clear
B)
\[NaCl\] in water
done
clear
C)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
Water
done
clear
View Answer play_arrow
An element with atomic number 20 will be placed in which period of the periodic table
A)
4
done
clear
B)
3
done
clear
C)
2
done
clear
D)
1
done
clear
View Answer play_arrow
Elements of atomic number 6 is placed in
A)
IV group
done
clear
B)
IV period
done
clear
C)
VI group
done
clear
D)
III group
done
clear
View Answer play_arrow
Which of the following is not an ore of lead
A)
Galena
done
clear
B)
Anglesite
done
clear
C)
Calamine
done
clear
D)
Cerrusite
done
clear
View Answer play_arrow
Which one of the following is correct?
A)
A mineral cannot be an ore
done
clear
B)
An ore cannot be a mineral
done
clear
C)
All minerals are ores
done
clear
D)
All ores are minerals
done
clear
View Answer play_arrow
Concentrated aqueous sulphuric acid is\[98\,%\]...r, \[{{H}_{2}}S{{O}_{4}}\] by mass and has a density of\[1.80\text{ }g\,m{{L}^{-\,1}}\]. Volume of acid required to make one litre of 0.1. \[M{{H}_{2}}S{{O}_{4}}\] solution is:
A)
16.65 mL
done
clear
B)
22.20 mL
done
clear
C)
5.55 mL
done
clear
D)
11.10 mL
done
clear
View Answer play_arrow
25.3 g sodium carbonate, \[N{{a}_{2}}C{{O}_{3}}\] was dissolved in enough water to make 250 mL of solution. If sodium carbonate dissociates completely, molar concentration of \[N{{a}^{+}}\] and carbonate ions are respectively: \[(Molar\,\,mass\,\,of\,\,N{{a}_{2}}C{{O}_{3}}\,=\,\,106\,\,g\,mo{{l}^{-1}})\]
A)
0.9555 M and 1.910 M
done
clear
B)
1.910 M and 0.955 M
done
clear
C)
1.90 M and 1.090 M
done
clear
D)
0.477 M and 0.477 M
done
clear
View Answer play_arrow
Consider the following sets of quantum numbers
N l m s (i) 3 0 0 +1/2 (ii) 2 2 1 +/1/2 (iii) 4 3 -2 -1/2 (iv) 1 0 -1 -1/2 (v) 3 2 3 +1/2
Which of the following sets of quantum number is not possible?
A)
(ii), (iii) and (iv)
done
clear
B)
(i), (ii), (iii) and (iv)
done
clear
C)
(ii), (iv) and (v)
done
clear
D)
(i) and (iii)
done
clear
View Answer play_arrow
Maximum number of electrons in a sub-shell with\[\operatorname{l} = 3 and\,\,n =4\] is
A)
10
done
clear
B)
12
done
clear
C)
14
done
clear
D)
16
done
clear
View Answer play_arrow
The number of d-electrons in \[F{{e}^{2+}}\] \[\left( Z = 26 \right)\] not equal to the number of electrons in which one of the following
A)
\[d-electrons in Fe \left( Z = 26 \right)\]
done
clear
B)
\[p-electrons in Ne \left( Z = 10 \right)\]
done
clear
C)
\[s-electrons in Mg \left( Z = 12 \right)\]
done
clear
D)
\[p-electrons in Cl \left( Z=17 \right)\]
done
clear
View Answer play_arrow
The compound, which is soluble in water but does not impede the oxygen transportation, is
A)
NO
done
clear
B)
\[S{{O}_{2}}\]
done
clear
C)
CO
done
clear
D)
\[S{{O}_{3}}\]
done
clear
View Answer play_arrow
The branch of biology dealing with the process of improvement of human race by selective breeding is called
A)
euphenics
done
clear
B)
euthenics
done
clear
C)
obstetrics
done
clear
D)
eugenics
done
clear
View Answer play_arrow
Waste products of adenine and guanine metabolism are excreted by man as
A)
uric acid
done
clear
B)
urea
done
clear
C)
allantoin
done
clear
D)
ammonia
done
clear
View Answer play_arrow
Scientists pin-pointed the location of colour processing perception centres in human visual cortex by a method named
A)
CT scanning
done
clear
B)
PET scanning
done
clear
C)
ultra-sound imaging
done
clear
D)
NMR imaging
done
clear
View Answer play_arrow
The disorders such as alkaptonuria and phenylketonuria are referred to as
A)
infectious disease
done
clear
B)
acquired disease
done
clear
C)
congenital disease
done
clear
D)
all of these
done
clear
View Answer play_arrow
There is an irregular mating population. If the frequency of an autosomal recessive lethal gene is 0.4, then the frequency of the carriers in a population of 200 individuals is
A)
96
done
clear
B)
36
done
clear
C)
104
done
clear
D)
72
done
clear
View Answer play_arrow
If the rate of addition of new members increases with respect to the individual host of the same population, then the graph obtained has
A)
zero population growth
done
clear
B)
declined growth
done
clear
C)
exponential growth
done
clear
D)
none of these
done
clear
View Answer play_arrow
To yield milk, cow is given
A)
stilbesterol
done
clear
B)
sorbitol
done
clear
C)
gonadotropin
done
clear
D)
prolactin
done
clear
View Answer play_arrow
The disease, in which thick cough, stops the passage of throat and form a layer of mucous membrane, is called
A)
tuberculosis
done
clear
B)
tetanus
done
clear
C)
diphtheria
done
clear
D)
pertussis
done
clear
View Answer play_arrow
Ora serrata is
A)
gland present in oral cavity of frog
done
clear
B)
a part of third wall of retina of eye
done
clear
C)
present in utriculus of ear
done
clear
D)
oral cavity of protochordates
done
clear
View Answer play_arrow
Which of the following is an example of sex- linked inheritance?
A)
night-blindness
done
clear
B)
anaemia
done
clear
C)
colour-blindness
done
clear
D)
cretinism
done
clear
View Answer play_arrow
Chloragogen cells of earthworm are similar to the vertebrate organ
A)
kidney
done
clear
B)
liver
done
clear
C)
spleen
done
clear
D)
lung
done
clear
View Answer play_arrow
The type of immunoglobulin present in the colostrum secreted from mammary gland is
A)
IgD
done
clear
B)
IgC
done
clear
C)
IgM
done
clear
D)
IgE
done
clear
View Answer play_arrow
A pregnant woman, who has done amniocentesis test, finds an extra barr body in her embryo. The syndrome which is likely to be associated with embryo is
A)
Edward's syndrome
done
clear
B)
Down's syndrome
done
clear
C)
Klinfelter's syndrome
done
clear
D)
Patau's syndrome
done
clear
View Answer play_arrow
The preparation of sperm before penetration of ovum is called
A)
insemination
done
clear
B)
coition
done
clear
C)
spermiation
done
clear
D)
capacitation
done
clear
View Answer play_arrow
Which proteolytic enzyme induces lysis of fibrin during fibrinolysis?
A)
fibrin
done
clear
B)
plasmin
done
clear
C)
thrombin
done
clear
D)
all of these
done
clear
View Answer play_arrow
The lining of bone marrow cavity is called
A)
endoneurium
done
clear
B)
endosteum
done
clear
C)
endothelium
done
clear
D)
endomyosium
done
clear
View Answer play_arrow
The endocrine gland of insects, which secretes the juvenile hormone, is
A)
corpora albicans
done
clear
B)
corpora allata
done
clear
C)
corpora myecaena
done
clear
D)
all of these
done
clear
View Answer play_arrow
Quill feathers at the base of quill wings are called
A)
coverts
done
clear
B)
remiges
done
clear
C)
down feathers
done
clear
D)
barbules
done
clear
View Answer play_arrow
Structure which remains unchanged during metamorphosis of frog's tadpole is
A)
intestine
done
clear
B)
lung
done
clear
C)
nervous system
done
clear
D)
heart
done
clear
View Answer play_arrow
How much amount of oxygen is present in one gram of haemoglobin?
A)
20 ml
done
clear
B)
1.34 ml
done
clear
C)
40 ml
done
clear
D)
13.4 ml
done
clear
View Answer play_arrow
Otoconium is found in
A)
synovial fluid
done
clear
B)
perilymph
done
clear
C)
otolithic membrane
done
clear
D)
haemolymph
done
clear
View Answer play_arrow
Exaggerated dread is a death disease which is known as
A)
haemotophobia
done
clear
B)
algophobia
done
clear
C)
pathophobia
done
clear
D)
myophobia
done
clear
View Answer play_arrow
Antiserum contains
A)
leucocytes
done
clear
B)
antigens
done
clear
C)
antibodies
done
clear
D)
all of these
done
clear
View Answer play_arrow
The branch, which is associated with diagnosis, prevention and cure of mental disorders is called
A)
neurology
done
clear
B)
psychiatry
done
clear
C)
neuropsychiatry
done
clear
D)
psychology
done
clear
View Answer play_arrow
Translocation of organic materials is best explained by
A)
imbibition theory
done
clear
B)
active transport
done
clear
C)
mass flow hypothesis
done
clear
D)
transpiration pull
done
clear
View Answer play_arrow
Which of the following is the largest and edible bud?
A)
cabbage
done
clear
B)
onion
done
clear
C)
cauliflower
done
clear
D)
Agave
done
clear
View Answer play_arrow
When a tall and red flowered individual is crossed with a dwarf and white flowered individual, phenotype in the progeny is dwarf and white. What will be the genotype of tall and red flowered individual?
A)
TtRr
done
clear
B)
TTRR
done
clear
C)
TTRr
done
clear
D)
TtRR
done
clear
View Answer play_arrow
An insectivorous plant having glandular structures on its leaf secreting stickly fluid is
A)
sundew plant
done
clear
B)
aldrovanda
done
clear
C)
venus fly trap
done
clear
D)
all of these
done
clear
View Answer play_arrow
Positive pollution of soil is due to
A)
excessive use of fertilizers
done
clear
B)
reduction in soil productivity
done
clear
C)
addition of wastes on soil
done
clear
D)
all of these
done
clear
View Answer play_arrow
Perisperm is
A)
peripheral part of endosperm
done
clear
B)
remnant of endosperm
done
clear
C)
disintegrated secondary nucleus
done
clear
D)
persistence of nucellus
done
clear
View Answer play_arrow
The biome, which is characterised by broad- leaved vegetation, life-resistant resinous plants. and drought-evading plants, is known as
A)
steppes
done
clear
B)
chapparal
done
clear
C)
deciduous forest
done
clear
D)
savannah
done
clear
View Answer play_arrow
Which of the following compound has very important role in prebiotic evolution?
A)
\[C{{H}_{4}}\]
done
clear
B)
\[S{{O}_{2}}\]
done
clear
C)
\[S{{O}_{3}}\]
done
clear
D)
NO
done
clear
View Answer play_arrow
Genes present on Y-chromosome are called
A)
polygenic gene
done
clear
B)
basic gene
done
clear
C)
pleiotropic gene
done
clear
D)
holandric gene
done
clear
View Answer play_arrow
The carnivorous fish, Gambusia, which is introduced in the lakes and ponds to control a deadly disease in India, feeds on the larva of
A)
dragon fly
done
clear
B)
Nepenthes
done
clear
C)
Anopheles
done
clear
D)
all of these
done
clear
View Answer play_arrow
The \[{{C}_{4}}-plants\] differ from \[{{C}_{3}}-plants\] with reference to the
A)
substrate that accepts CO in carbon as- similation
done
clear
B)
type of end product
done
clear
C)
type of pigment involved in photosynthesis J
done
clear
D)
number of ATPs that are consumed in preparing sugar
done
clear
View Answer play_arrow
The plants in desert, in order to tolerate water stress, have
A)
stipular spines
done
clear
B)
no stomata
done
clear
C)
stems which are converted into leaf type
done
clear
D)
long root system reach the water level
done
clear
View Answer play_arrow
The sphere of living matter together with water air and soil on the surface of earth is called
A)
lithosphere
done
clear
B)
atmosphere
done
clear
C)
biosphere
done
clear
D)
hydrosphere
done
clear
View Answer play_arrow
Which of the following prevents the photo- oxidation and destruction of pigments?
A)
phycoerythrin
done
clear
B)
phytochrome
done
clear
C)
phytohormone
done
clear
D)
phycocyanin
done
clear
View Answer play_arrow
Meiosis in Dryopteris takes place during
A)
gamete formation
done
clear
B)
spore formation
done
clear
C)
sex organ formation
done
clear
D)
spore germination
done
clear
View Answer play_arrow
Recent reports of acid rains in big industrial cities are due to the effect of atmospheric pollution by excessive release of
A)
\[N{{H}_{3}}\] by coal gas industries
done
clear
B)
\[N{{O}_{2}}\] and \[S{{O}_{2}}\] by burning of fossil fuels
done
clear
C)
\[C{{O}_{2}}\] by incomplete combustion of carbon fuel
done
clear
D)
\[C{{O}_{2}}\] by burning of coal/wood, cutting of forests
done
clear
View Answer play_arrow
The process in which the amount of DNA, RNA and protein can be known at a time is called
A)
cellular fractioning
done
clear
B)
autoradiography
done
clear
C)
phase-contrast microscopy
done
clear
D)
tissue culture
done
clear
View Answer play_arrow
The hexaploid wheat is obtained by
A)
chromosomes map
done
clear
B)
hybridomas
done
clear
C)
hybridization
done
clear
D)
both [b] and [c]
done
clear
View Answer play_arrow
In sweet peas, genes C and P are necessary for colour in flowers. The flowers are white in the absence of either or both the genes. What will be the percentage of coloured flowers in the offspring of the cross\[\operatorname{Ccpp} \times ccPp\]?
A)
\[75\,%\]
done
clear
B)
\[25\,%\]
done
clear
C)
\[100\,%\]
done
clear
D)
\[50\,%\]
done
clear
View Answer play_arrow
Which plant species is on the verge of extinction due to over-exploitation?
A)
Gloriosa
done
clear
B)
Podophyllum
done
clear
C)
Ceritella
done
clear
D)
all of these
done
clear
View Answer play_arrow
The asexual production of seed is called
A)
fragmentation
done
clear
B)
apomixes
done
clear
C)
self-fertilization
done
clear
D)
both [a] and [b]
done
clear
View Answer play_arrow
If mitotic division is restricted in \[{{G}_{1}}-phase\] of a cell cycle, then the condition is known as
A)
S-phase
done
clear
B)
\[{{G}_{2}}-phase\]
done
clear
C)
M-phase
done
clear
D)
\[{{G}_{0}}-phase\]
done
clear
View Answer play_arrow
Epistatic effect, in which the dihybrid cross \[9:3:3:1\] between \[\operatorname{AaBb} \times AaBb\] is modified as
A)
interaction between two alleles of the same loci
done
clear
B)
dominance of one allele on another allele of the same loci
done
clear
C)
interaction between two alleles of different loci
done
clear
D)
dominance of one allele on another allele of both loci
done
clear
View Answer play_arrow
Potato is included in solanaceae family because
A)
it is epipetalous
done
clear
B)
it is pentamerous
done
clear
C)
ovary is slightly diverted from its position
done
clear
D)
all the above
done
clear
View Answer play_arrow
The chemical compounds which are produced by host plants due to infection as a defence reaction to pathogen, are called
A)
phytotron
done
clear
B)
phytotoxin
done
clear
C)
phytoalexins
done
clear
D)
phytol
done
clear
View Answer play_arrow
Chromosomes in a bacterial cell can be 1-3 in number and:
A)
are always circular
done
clear
B)
are always linear
done
clear
C)
can be either circular or linear, but never both within the same cell
done
clear
D)
can be circular as well as linear within the same cell
done
clear
View Answer play_arrow
The thalloid body of a slime mould (Myxomy- cetes) is known as
A)
Protonema
done
clear
B)
Plasmodium
done
clear
C)
Fruiting body
done
clear
D)
Mycelium
done
clear
View Answer play_arrow
Mannitol is the stored food in
A)
Gracilaria
done
clear
B)
Chara
done
clear
C)
Porphyra
done
clear
D)
Fucus
done
clear
View Answer play_arrow
Which one of the following is a vascular cryptogram?
A)
Cedrus
done
clear
B)
Equisetum
done
clear
C)
Ginkgo
done
clear
D)
Marchantia
done
clear
View Answer play_arrow
Which one of the following is common to multi- cellular fungi, filamentous algae and protonema of mosses?
A)
Mode of Nutrition
done
clear
B)
Multiplication by fragmentation
done
clear
C)
Diplontic life cycle
done
clear
D)
Members of kingdom plantae
done
clear
View Answer play_arrow
Metameric segmentation is the characteristic of:
A)
Platyhelminthes and Arthropoda
done
clear
B)
Echinodermata and Annelida
done
clear
C)
Annelida and Arthropoda
done
clear
D)
Mollusca and Chordata
done
clear
View Answer play_arrow
Which one of the following groups of animals is bilaterally symmetrical and triploblastic?
A)
Sponges
done
clear
B)
Coelenterates (Cnidarians)
done
clear
C)
Aschelminthes (round worms)
done
clear
D)
Ctenophores
done
clear
View Answer play_arrow
Planaria possess high capacity of:
A)
metamorphosis
done
clear
B)
regeneration
done
clear
C)
alternation of generation
done
clear
D)
bioluminescence
done
clear
View Answer play_arrow
The technical term used for the androecium in a flower of China rose (Hibiscus rosasinensis) is:
A)
Monoadelphous
done
clear
B)
Diadelphous
done
clear
C)
Polyandrous
done
clear
D)
Polyadelphous
done
clear
View Answer play_arrow
An aggregate fruit is one which developed from
A)
Multicarpellary syncarppous gynoecium
done
clear
B)
Multicarpellary apocarups gynoecium
done
clear
C)
Complete inflorescence
done
clear
D)
Multicarpellary superior ovary
done
clear
View Answer play_arrow
In a longitudinal section of a root, starting from the tip upward, the four zones occur in the following order:
A)
Root cap, cell division, cell maturation, cell enlargement
done
clear
B)
Cell division, cell enlargement, cell maturation, root cap
done
clear
C)
Cell division, cell maturation, cell enlargement, root cap
done
clear
D)
Root cap, cell division, cell enlargement, cell maturation
done
clear
View Answer play_arrow
The annular and spirally thickened conducting elements generally develop in the protoxylem when the root or stem is
A)
Differentiating
done
clear
B)
Maturing
done
clear
C)
Elongating
done
clear
D)
Widening
done
clear
View Answer play_arrow
Gymnosperms are also called soft wood spermatophytes because they lack
A)
Thick-walled tracheids
done
clear
B)
Xylem fibres
done
clear
C)
Cambium
done
clear
D)
Phloem fibres
done
clear
View Answer play_arrow
Age of a tree can be estimated by:
A)
Its height and girth
done
clear
B)
Biomass
done
clear
C)
Number of annual rings
done
clear
D)
Diameter of its heartwood
done
clear
View Answer play_arrow
Select the correct statement from the ones given below with respect to Periplaneta americancr.
A)
There are 16 very long malpighian tubules present at the junctions of midgut and hindgut
done
clear
B)
Grinding of food is carried out only by the mouth parts
done
clear
C)
Nervous system located dorsally consists of segmentally arranged ganglia joined by a pair of longitudinal connective
done
clear
D)
Males bear a pair of short thread like anal styles
done
clear
View Answer play_arrow
Plasmodesmata are:
A)
Connection between adjacent cells
done
clear
B)
Lignified cemented layers between cells m
done
clear
C)
Locomotory structures
done
clear
D)
Membranes connecting the nucleus with plasmalemma
done
clear
View Answer play_arrow
Nuclear membrane is absent in:
A)
Volvox
done
clear
B)
Nostoc
done
clear
C)
Penicillium
done
clear
D)
Agaricus
done
clear
View Answer play_arrow
The solid linear cytoskeletal elements having a diameter of 6 nm and made up of a single type of monomer are known as:
A)
Microtubules
done
clear
B)
Microfilaments
done
clear
C)
Intermediate filaments
done
clear
D)
Lamins
done
clear
View Answer play_arrow
A phosphoglyceride is always made up of:
A)
Only a saturated fatty acid esterified to a glycerol molecule to which a phosphate B group is also attached
done
clear
B)
Only an unsaturated fatty acid esterified to a glycerol molecule to which a phosphate group is also attached
done
clear
C)
A saturated or unsaturated fatty acid esterified to a glycerol molecule to which a phosphate group is also attached
done
clear
D)
A saturated or unsaturated fatty acid esterified to a phosphate group which is also attached to a glycerol molecule
done
clear
View Answer play_arrow
The enzyme recombinase is required at which stage of meiosis:
A)
Pachytene
done
clear
B)
Zygotene
done
clear
C)
Diplotene
done
clear
D)
Diakinesis
done
clear
View Answer play_arrow
The recessive genes located on X-chromosomes in humans are always:
A)
Sub-lethal
done
clear
B)
Expressed in males
done
clear
C)
Expressed in females
done
clear
D)
Lethal
done
clear
View Answer play_arrow
At a particular locus, frequency of 'A' allele is 0.6 and that of 'a' is 0.4. What would be the frequency of heterozygotes in a random mating population at equilibrium?
A)
0.24
done
clear
B)
0.16
done
clear
C)
0.48
done
clear
D)
0.36
done
clear
View Answer play_arrow
Which one of the following conditions in humans is correctly matched with its chromosomal abnormality/linkage?
A)
Erythroblastosis foetalis - X-linked
done
clear
B)
Down syndrome - 44 autosomes + XO
done
clear
C)
Klinefelter syndrome - 44 autosomes + XXY
done
clear
D)
Colourblindness - Y-linked
done
clear
View Answer play_arrow
The genotype of a plant showing the dominant phenotype can be determined by:
A)
Test cross
done
clear
B)
Dihybrid cross
done
clear
C)
Pedigree analysis
done
clear
D)
Back cross
done
clear
View Answer play_arrow
The incorrect statement with regard to, haemophilia is:
A)
It is a sex-linked disease
done
clear
B)
It is a recessive disease
done
clear
C)
It is a dominant disease
done
clear
D)
A single protein involved in the clotting of blood is affected
done
clear
View Answer play_arrow
When a cluster of genes show linkage behavior, they:
A)
do not show a chromosome map
done
clear
B)
show recombination during meiosis
done
clear
C)
do not show independent assortment
done
clear
D)
induce cell division
done
clear
View Answer play_arrow
DNA fingerprinting refers to:
A)
Analysis of DNA samples using imprinting devices
done
clear
B)
Techniques used for molecular analysis of different specimens of DNA
done
clear
C)
Techniques used for identification of fingerprints of individuals
done
clear
D)
Molecular analysis of profiles of DNA Samples
done
clear
View Answer play_arrow
The Okazaki fragments in DNA chain growth:
A)
polymerize in the 5'-to-3' direction and explain 3'-to-5' DNA replication
done
clear
B)
result in transcription
done
clear
C)
polymerize in the 3'-to-5' direction and forms replication fork
done
clear
D)
prove semi-conservative nature of DNA replication
done
clear
View Answer play_arrow
In a random mating population in equilibrium, which of the following brings about a change in gene frequency in a non-directional manner:
A)
Mutations
done
clear
B)
Random drift
done
clear
C)
Selection
done
clear
D)
Migration
done
clear
View Answer play_arrow
Which one of the following phenomena supports Darwin's concept of natural selection in organic evolution?
A)
production of 'Dolly', the sheep by cloning
done
clear
B)
Development of organs from 'stem' cells for organ transplantation
done
clear
C)
Development of transgenic animals
done
clear
D)
Prevalence of pesticide resistant insects
done
clear
View Answer play_arrow
Jurassic period of the Mesozoic era characterized by
A)
Gymnosperms are dominant plants and first birds appear
done
clear
B)
Radiation of reptiles and origin of mammal like reptiles
done
clear
C)
Dinosaurs become extinct and angiosperms appear
done
clear
D)
Flowering plants and first dinosaurs appear
done
clear
View Answer play_arrow
What is common to whale, seal and shark?
A)
Homoiothermy
done
clear
B)
Seasonal migration
done
clear
C)
Thick subcutaneous fat
done
clear
D)
Convergent evolution
done
clear
View Answer play_arrow
The process by which organisms with different evolutionary history evolve similar phenotypic adaptations in response to a common environmental challenge is called:
A)
natural selection
done
clear
B)
convergent evolution
done
clear
C)
non-random evolution
done
clear
D)
adaptive radiation
done
clear
View Answer play_arrow
Antibodies in our body are complex
A)
Lipoproteins
done
clear
B)
Steroids
done
clear
C)
Prostaglandins
done
clear
D)
Glycoproteins
done
clear
View Answer play_arrow
Which one of the following is the correct statement regarding the particular psychotropic drug specified?
A)
Morphine leads to delusions and disturbed emotions.
done
clear
B)
Barbiturates cause relaxation and temporary euphoria.
done
clear
C)
Hashish causes after thought perceptions and hallucinations.
done
clear
D)
Opium stimulates nervous system and causes hallucinations.
done
clear
View Answer play_arrow
Which one of the following statements is correct with respect to AIDS?
A)
The HIV can be transmitted through eating food together with an infected person
done
clear
B)
Drug addicts are least susceptible to HIV infection
done
clear
C)
AIDS patients are being fully cured cent percent with proper care and nutrition
done
clear
D)
The causative HIV retrovirus enters helper T-lymphocytes thus reducing their numbers.
done
clear
View Answer play_arrow
Reason of fast speciation in present day crop plant is
A)
Mutation
done
clear
B)
Isolation
done
clear
C)
Polyploidy
done
clear
D)
Sexual reproduction
done
clear
View Answer play_arrow
Compared to a bull a bullock docile because of:
A)
Lower levels of adrenalin/ noradrenalin in its blood
done
clear
B)
Higher levels of thyroxin
done
clear
C)
Higher levels of cortisone
done
clear
D)
Lower levels of blood testosterone
done
clear
View Answer play_arrow
Which of the following is not used as a biopesticide?
A)
Xanthomonas campestris
done
clear
B)
Bacillus thuringiensis
done
clear
C)
Trichoderma harzianum
done
clear
D)
Nuclear polyhedral virus (NPV)
done
clear
View Answer play_arrow
Which of the following enhances or induces fusion of protoplasts?
A)
IAA and gibberellins
done
clear
B)
Sodium chloride and potassium chloride
done
clear
C)
Polyethyulene glycol and sodium nitrate
done
clear
D)
IAA and kinetin
done
clear
View Answer play_arrow