Simple pendulum is executing simple harmonic motion with time period T. If the length of the pendulum is increased by \[21\,%\] then the increase in the time period of the pendulum of the increased length is
A)
\[22%\]
done
clear
B)
\[13%\]
done
clear
C)
\[50%\]
done
clear
D)
\[10%\]
done
clear
View Answer play_arrow
Which one of the following has the highest neutrons ratio?
A)
\[_{92}{{U}^{235}}\]
done
clear
B)
\[_{8}{{O}^{16}}\]
done
clear
C)
\[_{2}H{{e}^{4}}\]
done
clear
D)
\[_{26}F{{e}^{56}}\]
done
clear
View Answer play_arrow
A concave mirror having the focal length 15 cm forms an image having twice of the linear dimensions of the object. If the image is virtual, then the position of the object will be
A)
7.5 cm
done
clear
B)
22.5 cm
done
clear
C)
40 cm
done
clear
D)
30 cm
done
clear
View Answer play_arrow
Two materials having the dielectric constants \[{{K}_{1}}\,\,and\,\,{{K}_{2}}\] are filled between two parallel plates of a capacitor. Where area of each plate is a and the distance between the plates is d.
The capacity of the capacitor is
A)
\[\frac{A{{\varepsilon }_{0}}({{K}_{1}}\times {{K}_{2}})}{d({{K}_{1}}+{{K}_{2}})}\]
done
clear
B)
\[\frac{A{{\varepsilon }_{0}}({{K}_{1}}-{{K}_{2}})}{d}\]
done
clear
C)
\[\frac{A{{\varepsilon }_{0}}{{K}_{1}}{{K}_{2}}}{({{K}_{1}}+{{K}_{2}})}\]
done
clear
D)
\[\frac{A{{\varepsilon }_{0}}({{K}_{1}}+{{K}_{2}})}{d}\]
done
clear
View Answer play_arrow
A closely wound flat circular coil of 25 turns of wire has diameter of 10 cm which carries current of 4 A, the flux density at the centre of a coil will be
A)
\[1.256 \times 1{{0}^{-}}^{3}\,T\]
done
clear
B)
\[1.679 \times 1{{0}^{-}}^{5}\,T\]
done
clear
C)
\[1.512 \times 1{{0}^{-}}^{5}T\]
done
clear
D)
\[2.28 \times 1{{0}^{-}}^{4}\,T\]
done
clear
View Answer play_arrow
Four lenses having the focal length of +15 cm, 20 cm, +150 cm, and +250 cm respectively are provided to make an astronomical telescope. The focal length of the eyepiece to produce the largest magnification should be
A)
\[+250\,cm\]
done
clear
B)
\[+155\text{ }cm\]
done
clear
C)
25 cm
done
clear
D)
\[+15\text{ }cm\]
done
clear
View Answer play_arrow
When radioactive substance emits an a-particle, then its position in the periodic table is lowered by
A)
two places
done
clear
B)
three places
done
clear
C)
five places
done
clear
D)
one place
done
clear
View Answer play_arrow
When added an impurity into the silicon which one of the following produces n-type of semi- conductors?
A)
Iron
done
clear
B)
Magnesium
done
clear
C)
Aluminium
done
clear
D)
Phosphorus
done
clear
View Answer play_arrow
The point charges Q and -2Q are placed at some distance apart. If the electric field at the location of Q is E, the electric field at the location of -2Q will be
A)
\[-\frac{3\,E}{2}\]
done
clear
B)
\[-\,E\]
done
clear
C)
\[-\,\frac{E}{2}\]
done
clear
D)
\[-\,2\,E\]
done
clear
View Answer play_arrow
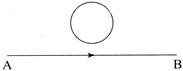
The current flows from A to B as shown in the figure, then the direction of the induced current in the loop will be
A)
straight line
done
clear
B)
anti-clockwise
done
clear
C)
clockwise
done
clear
D)
none of the above
done
clear
View Answer play_arrow
Which of the following pairs does not have similar dimensions?
A)
Tension and surface tension
done
clear
B)
Stress and pressure
done
clear
C)
Planck's constant and angular momentum
done
clear
D)
Angle and strain
done
clear
View Answer play_arrow
The current gain for a transistor working a common base amplifier is 0.96. If the emitter current is 7.2 mA, the base current will be
A)
0.42 mA
done
clear
B)
0.49 Ma
done
clear
C)
0.29 mA
done
clear
D)
0.35 mA
done
clear
View Answer play_arrow
In an atom bomb, the energy is released because of the
A)
chain reaction of neutrons and \[_{92}{{U}^{238}}\]
done
clear
B)
chain reaction of neutrons and \[_{92}{{U}^{235}}\]
done
clear
C)
chain reaction of neutrons and \[_{92}{{U}^{236}}\]
done
clear
D)
chain reaction of neutrons and \[_{92}{{U}^{240}}\]
done
clear
View Answer play_arrow
What change occurs, if the monochromatic light used in Young's double slit experiment is replaced by white light?
A)
Only the central fringe is white and all other fringes are observed coloured
done
clear
B)
No fringes are observed
done
clear
C)
All the bright fringes become white
done
clear
D)
All the bright fringes are coloured between violet and red
done
clear
View Answer play_arrow
The escape velocity from the earth is 11.2 km/s. The escape velocity from a planet having twice the radius and the same mean density as the earth is
A)
11.2 km/s
done
clear
B)
22.4 km/s
done
clear
C)
15.00 km/s
done
clear
D)
5.8 km/s
done
clear
View Answer play_arrow
A metal ball of mass 2 kg moving with speed of 36 km/h has a collision with a stationary ball of mass 3 kg. If after collision, both the ball move together, the loss in kinetic energy due to collision is
A)
80 J
done
clear
B)
40 J
done
clear
C)
60 J
done
clear
D)
160 J
done
clear
View Answer play_arrow
In a circuit, the current lags behind the voltage by a phase difference of \[\pi /2\], the circuit will contain which of the following
A)
only R
done
clear
B)
only C
done
clear
C)
R and C
done
clear
D)
only L
done
clear
View Answer play_arrow
The length and breadth of a metal sheet all 3.124 m and 3.002 m, respectively, the area of this sheet up to four correct significant figure
A)
\[9.378\,\,{{m}^{2}}\]
done
clear
B)
\[9.37{{m}^{2}}\]
done
clear
C)
\[9.378248\,\,{{m}^{2}}\]
done
clear
D)
\[9.3782{{m}^{2}}\]
done
clear
View Answer play_arrow
The tension in a piano wire is 10 N. The tension in a piano wire to produce a node of double frequency is
A)
20 N
done
clear
B)
40 N
done
clear
C)
10 N
done
clear
D)
120 N
done
clear
View Answer play_arrow
A sings with a frequency n and B sings with a frequency 1 /8 that of A. If the energy remains the same and the amplitude of A is a, then amplitude of B will be
A)
2a
done
clear
B)
8a
done
clear
C)
4a
done
clear
D)
a
done
clear
View Answer play_arrow
The velocity of a bullet is reduced from 200 m/s to 100 m/s, while travelling through a wooden block of thickness 10 cm. Assuming it to be uniform, the retardation will be
A)
\[15\,\,\times \,\,{{10}^{4}}m/{{s}^{2}}\]
done
clear
B)
\[10\,\,\times \,\,{{10}^{4}}m/{{s}^{2}}\]
done
clear
C)
\[12\,\,\times \,\,{{10}^{4}}m/{{s}^{2}}\]
done
clear
D)
\[14.5\text{ }m/{{s}^{2}}\]
done
clear
View Answer play_arrow
Two projectiles are projected with the same velocity. If one is projected at an angle of \[30{}^\circ \] and the other at \[60{}^\circ \] to the horizontal. The ratio of maximum heights reached is
A)
\[1:3\]
done
clear
B)
\[2:1\]
done
clear
C)
\[3:1\]
done
clear
D)
\[1:4\]
done
clear
View Answer play_arrow
In an adiabatic change, the pressure and temperature of a monoatomic gas are related with relation as \[P\propto {{T}^{C}}\], where C is equal to
A)
\[\frac{5}{4}\]
done
clear
B)
\[\frac{5}{3}\]
done
clear
C)
\[\frac{5}{2}\]
done
clear
D)
\[\frac{3}{5}\]
done
clear
View Answer play_arrow
A body A starts from rest with an acceleration\[{{a}_{1}}\]. After 2 s another body B starts from rest with an acceleration\[{{a}_{2}}\]. If they travel equal distances in 5 s, after the starts of A, the ratio \[{{a}_{1}}\,\,:\,\,{{a}_{2}}\] will be equal to
A)
\[9:5\]
done
clear
B)
\[5\,\,:\,\,7\]
done
clear
C)
\[5\,\,:\,\,9\]
done
clear
D)
\[7\,\,:\,\,9\]
done
clear
View Answer play_arrow
In a sinusoidal wave the time required for a particular point to move from equilibrium position to maximum displacement is 0.17 s, then the frequency of wave is
A)
1.47 Hz
done
clear
B)
0.36 Hz
done
clear
C)
2.94 Hz
done
clear
D)
2.48 Hz
done
clear
View Answer play_arrow
One mole of a monoatomic ideal gas is mixed with one mole of a diatomic ideal gas. The molar specific heat of the mixture at constant volume is
A)
8
done
clear
B)
\[\frac{3}{2}\,R\]
done
clear
C)
2R
done
clear
D)
\[2.5\,\,R\]
done
clear
View Answer play_arrow
A system is provided with 200 cal of heat and the work done by the system on the surrounding is 40 J. Then its internal energy
A)
Increases by 600 J
done
clear
B)
Decreases by 800 J
done
clear
C)
Increases by 800 J
done
clear
D)
Decreases by 50 J
done
clear
View Answer play_arrow
The coefficients of thermal conductivity of copper, mercury and glass are respectively \[{{K}_{m}}\,\,and\,\,{{K}_{g}}\] such that\[{{K}_{c}}>{{K}_{m}}>{{K}_{g}}\]. If the same quantity of heat is to flow per second per unit area of each and corresponding temperature gradients are \[{{X}_{c}},\,\,{{X}_{m}}\,\,and\,\,{{X}_{g}}\], then
A)
\[{{X}_{c}}={{X}_{m}}={{X}_{g}}\]
done
clear
B)
\[{{X}_{c}}>{{X}_{m}}>{{X}_{g}}\]c
done
clear
C)
\[{{X}_{c}}<{{X}_{m}}<{{X}_{g}}\]
done
clear
D)
\[{{X}_{m}}<{{X}_{c}}<{{X}_{g}}\]
done
clear
View Answer play_arrow
The amplitude of a particle executing SHM is 4 cm. At the mean position the speed of the particle is 16 cm/sec. The distance of the particle from the mean position at which the speed of the particle becomes \[8\sqrt{3}\,cm/s\] will be
A)
\[2\sqrt{3}\,cm\]
done
clear
B)
\[\sqrt{3}\,cm\]
done
clear
C)
1 cm
done
clear
D)
2 cm
done
clear
View Answer play_arrow
Velocity of sound waves in air is 330 m/sec. For a particular sound in air, a path difference of 40 cm is equivalent to a phase difference of\[1.6\,\pi \]. The frequency of this wave is
A)
165 Hz
done
clear
B)
150 Hz
done
clear
C)
660 Hz
done
clear
D)
330 Hz
done
clear
View Answer play_arrow
Between the plates of a parallel plate condenser, a plate of thickness \[{{t}_{1}}\] and dielectric constant \[{{k}_{1}}\]is placed. In the rest of the space, there is another plate of thickness \[{{t}_{2}}\] and dielectric constant\[{{k}_{2}}\]. The potential difference across the condenser will be
A)
\[\frac{Q}{A{{\varepsilon }_{0}}}\,\left( \frac{{{t}_{1}}}{{{k}_{1}}}+\frac{{{t}_{2}}}{{{k}_{2}}} \right)\]
done
clear
B)
\[\frac{{{\varepsilon }_{0}}Q}{A}\,\left( \frac{{{t}_{1}}}{{{k}_{1}}}+\frac{{{t}_{2}}}{{{k}_{2}}} \right)\]
done
clear
C)
\[\frac{Q}{A{{\varepsilon }_{0}}}\,\left( \frac{{{k}_{1}}}{{{t}_{1}}}+\frac{{{k}_{2}}}{{{t}_{2}}} \right)\]
done
clear
D)
\[\frac{{{\varepsilon }_{0}}Q}{A}\,\left( \frac{{{t}_{1}}}{{{k}_{1}}}+\frac{{{t}_{2}}}{{{k}_{2}}} \right)\]
done
clear
View Answer play_arrow
C Two charges each equal to \[nq({{\eta }^{-1}}<\sqrt{3})\] are placed at the corners of an equilateral triangle of side a. The electric field at the third corner is \[{{E}_{3}}\] where \[({{E}_{0}}=q/4\pi {{\varepsilon }_{0}}{{a}^{2}})\]
A)
\[{{E}_{3}}={{E}_{0}}\]
done
clear
B)
\[{{E}_{3}}<{{E}_{0}}\]
done
clear
C)
\[{{E}_{3}}>{{E}_{0}}\]
done
clear
D)
\[{{E}_{3}}\ge {{E}_{0}}\]
done
clear
View Answer play_arrow
The specific resistance of a wire is \[\rho \], its volume is \[3\text{ }{{m}^{3}}\] and its resistance is 3 ohms, then its length will be
A)
\[\sqrt{\frac{1}{\rho }}\]
done
clear
B)
\[\frac{3}{\sqrt{\rho }}\]
done
clear
C)
\[\frac{1}{\rho }\,\,\sqrt{3}\]
done
clear
D)
\[\rho \sqrt{\frac{1}{3}}\]
done
clear
View Answer play_arrow
An electric bulb is rated 220 volt and 100 watt. Power consumed by it when operated on 110 volt is
A)
50 watt
done
clear
B)
75 watt
done
clear
C)
90 watt
done
clear
D)
25 watt
done
clear
View Answer play_arrow
According to Joule?s law, if the potential difference across a conductor having a material of specific resistance remains constant, then the heat produced in the conductor is directly proportional to
A)
\[\rho \]
done
clear
B)
\[{{\rho }^{2}}\]
done
clear
C)
\[\frac{1}{\sqrt{\rho }}\]
done
clear
D)
\[\frac{1}{\rho }\]
done
clear
View Answer play_arrow
A helium nucleus makes a full rotation in a circle of radius 0.8 metre in two seconds. The value of the magnetic field B at the centre of the circle will be
A)
\[\frac{{{10}^{-19}}}{{{\mu }_{0}}}\]
done
clear
B)
\[{{10}^{-19}}\,{{\mu }_{0}}\]
done
clear
C)
\[2\,\,\times \,\,{{10}^{-10}}\,{{\mu }_{0}}\]
done
clear
D)
\[\frac{2\,\,\times \,\,{{10}^{-10}}}{{{\mu }_{0}}}\,\]
done
clear
View Answer play_arrow
Five very long, straight wires are bound together to form a small cable. Currents carried by the wires are\[{{I}_{1}}=20\,A,\,\,\,{{I}_{2}}=-6\,A,\,\,\,{{I}_{3}}=12\,A,\,\,{{I}_{4}}=-7A,\,\,{{I}_{5}}=18\,A\]. The magnetic induction at a distance of 10 cm from the cable is
A)
\[34\,\,\mu T\]
done
clear
B)
74 mT
done
clear
C)
34 mT
done
clear
D)
\[74\,\,\mu T\]
done
clear
View Answer play_arrow
A given object takes n times as much time to slide down a \[45{}^\circ \] rough incline as it takes to slide down a perfectly smooth \[45{}^\circ \] incline. The coefficient of kinetic friction between the object and the incline is given by
A)
\[\left( 1-\frac{1}{{{n}^{2}}} \right)\]
done
clear
B)
\[\frac{1}{1-{{n}^{2}}}\]
done
clear
C)
\[\sqrt{\left( 1-\frac{1}{{{n}^{2}}} \right)}\]
done
clear
D)
\[\sqrt{1-\frac{1}{{{n}^{2}}}}\]
done
clear
View Answer play_arrow
Two solid rubber balls A and B having masses 200 and 400 gm respectively are moving in opposite directions with velocity of A equal to 0.3 m/s. After collision the two balls come to rest, then the velocity of B is
A)
0.15 m/sec
done
clear
B)
1.5 m/sec
done
clear
C)
- 0.15 m/sec
done
clear
D)
None of the above
done
clear
View Answer play_arrow
A coil of \[Cu\] wire (radius-r, self-inductance-L) is bent in two concentric turns each having radius \[\frac{r}{2}\]. The self-inductance now
A)
2 L
done
clear
B)
L
done
clear
C)
4 L
done
clear
D)
L/2
done
clear
View Answer play_arrow
In making an alloy, a substance of specific gravity \[{{s}_{1}}\] and mass \[{{m}_{1}}\] is mixed with another substance of specific gravity \[{{s}_{2}}\] and mass \[{{m}_{2}}\], then the specific gravity of the alloy is
A)
\[\left( \frac{{{m}_{1}}+{{m}_{2}}}{{{s}_{1}}+{{s}_{2}}} \right)\]
done
clear
B)
\[\left( \frac{{{s}_{1}}{{s}_{2}}}{{{m}_{1}}+{{m}_{2}}} \right)\]
done
clear
C)
\[\frac{{{m}_{1}}+{{m}_{2}}}{\left( \frac{{{m}_{1}}}{{{s}_{1}}}+\frac{{{m}_{2}}}{{{s}_{2}}} \right)}\]
done
clear
D)
\[\frac{\left( \frac{{{m}_{1}}}{{{s}_{1}}}+\frac{{{m}_{2}}}{{{s}_{2}}} \right)}{{{m}_{1}}+{{m}_{2}}}\]
done
clear
View Answer play_arrow
The real coefficient of volume expansion of glycerine is \[0.000597\text{ }per\text{ }{}^\circ C\] and linear coefficient of expansion of glass is\[0.000009 per {}^\circ C\]. Then the apparent volume coefficient of expansion of glycerine is
A)
\[0.000558\text{ }per\text{ }{}^\circ C\]
done
clear
B)
\[0.00057\text{ }per\text{ }{}^\circ C\]
done
clear
C)
\[0.00027\text{ }per\text{ }{}^\circ C\]
done
clear
D)
\[0.00066\text{ }per\text{ }{}^\circ C\]
done
clear
View Answer play_arrow
The r.m.s. velocity of a gas at a certain temperature is \[\sqrt{2}\] times than that of the oxygen molecules at that temperature. The gas can be
A)
\[{{H}_{2}}\]
done
clear
B)
He
done
clear
C)
\[C{{H}_{4}}\]
done
clear
D)
\[S{{O}_{2}}\]
done
clear
View Answer play_arrow
The specific heat of hydrogen gas at constant pressure is \[{{\operatorname{C}}_{P}} =3.4 \times 1{{0}^{3}}cal/kg\,C\] and at constant volume is\[{{\operatorname{C}}_{V}} =2.4 \times 1{{0}^{3}}cal/kg C\]. If one kilogram hydrogen gas is heated from \[10{}^\circ C\,\,to\,\,20{}^\circ C\] at constant pressure, the external work done on the gas to maintain it at constant pressure is
A)
\[{{10}^{5}}\,cal\]
done
clear
B)
\[{{10}^{4}}\,cal\]
done
clear
C)
\[{{10}^{3}}\,cal\]
done
clear
D)
\[5\,\,\times \,\,{{10}^{3}}\,cal\]
done
clear
View Answer play_arrow
A thin copper wire of length metre increases in length by \[2%\] when heated through\[10{}^\circ C\]. While is the percentage increase in area when a square copper sheet of length / metre is heated through\[10{}^\circ C\]?
A)
\[4%\]
done
clear
B)
\[8%\]
done
clear
C)
\[16%~\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
In a fee arrangement of A and B atoms, where A atoms are at the comers of the unit cell, B atoms at the face centers, two atoms are missing from two comers in each unit cell, then the simplest formula of the compound is
A)
\[{{A}_{7}}{{B}_{6}}\]
done
clear
B)
\[{{A}_{6}}{{B}_{7}}\]
done
clear
C)
\[{{A}_{7}}{{B}_{24}}\]
done
clear
D)
\[A{{B}_{4}}\]
done
clear
View Answer play_arrow
A 0.2 molal aqueous solution of a weak acid (HX) is \[20%\] ionised. The freezing point of this solution is (Given \[{{\operatorname{K}}_{f}}=1.86{}^\circ C/m\] for water)
A)
\[-0.31{}^\circ C\]
done
clear
B)
\[-0.45{}^\circ C\]
done
clear
C)
\[-0.53{}^\circ C\]
done
clear
D)
\[-0.90{}^\circ C\]
done
clear
View Answer play_arrow
On adding solute to a solvent having vapour pressure 0.80 atm, vapour pressure reduces to 0.60 atm. Mole fraction of solute is
A)
0.25
done
clear
B)
0.75
done
clear
C)
0.50
done
clear
D)
0.33
done
clear
View Answer play_arrow
Fraction of total volume occupied by atoms in a simple cube is
A)
\[\frac{\pi }{2}\]
done
clear
B)
\[\frac{\sqrt{3\pi }}{8}\]
done
clear
C)
\[\frac{\sqrt{2\pi }}{6}\]
done
clear
D)
\[\frac{\pi }{6}\]
done
clear
View Answer play_arrow
A 0.001 molal solution of \[\left[ Pt{{(N{{H}_{3}})}_{4}}C{{l}_{3}} \right]C{{l}_{4}}\] in water had a freezing point depression of\[0.0054{}^\circ C\]. If \[{{K}_{f}}\] for water is 1.80, the correct formulation for the above molecule is
A)
\[\left[ Pt{{\left( NH{_{3}} \right)}_{4}}C{{l}_{3}} \right]Cl\]
done
clear
B)
\[\left[ Pt{{\left( NH{_{3}} \right)}_{4}}Cl \right]C{{l}_{2}}\]
done
clear
C)
\[\left[ Pt{{\left( NH{_{3}} \right)}_{4}}C{{l}_{2}} \right]Cl\]
done
clear
D)
\[\left[ Pt{{\left( NH{_{3}} \right)}_{4}}C{{l}_{4}} \right]\]
done
clear
View Answer play_arrow
For a binary ideal liquid solution, the total pressure of the solution is given as
A)
\[{{P}_{total}}\,=\,\,P_{A}^{*}+({{P}_{A}}^{*}-{{P}_{B}}^{*}){{X}_{A}}\]
done
clear
B)
\[{{P}_{total}}\,=\,\,P_{B}^{*}+({{P}_{B}}^{*}-{{P}_{A}}^{*}){{X}_{A}}\]
done
clear
C)
\[{{P}_{total}}\,=\,\,P_{A}^{*}+({{P}_{B}}^{*}-{{P}_{A}}^{*}){{X}_{A}}\]
done
clear
D)
\[{{P}_{total}}\,=\,\,P_{B}^{*}+({{P}_{B}}^{*}-{{P}_{A}}^{*}){{X}_{A}}\]
done
clear
View Answer play_arrow
Azeotropic mixture of HCl and water has
A)
\[84\,% HCl\]
done
clear
B)
\[22.2\,% HCl\]
done
clear
C)
\[63\,% HCl\]
done
clear
D)
\[20.2\,% HCl\]
done
clear
View Answer play_arrow
The relationship between the values of osmotic pressures of 0.1 M solutions of \[KN{{O}_{3}}\left( {{P}_{1}} \right)\] and \[C{{H}_{3}}COOH\,\,({{P}_{2}})\] is
A)
\[{{P}_{1}}>{{P}_{2}}\]
done
clear
B)
\[{{P}_{2}}>{{P}_{1}}\]
done
clear
C)
\[{{P}_{1}}={{P}_{2}}\]
done
clear
D)
\[{{P}_{1}}/\left( {{P}_{1}}+{{P}_{2}} \right)={{P}_{2}}/\left( {{P}_{1}}+{{P}_{2}} \right)\]
done
clear
View Answer play_arrow
The rate constant, the activation energy and Arrhenius parameter of a chemical reaction at \[25{}^\circ C\] are \[3.0 \,\times \, 1{{0}^{-}}^{4}{{s}^{-}}^{1}, \,1044 kJ mo{{l}^{-}}^{1}\] and \[6.0\,\,\times \,\,{{10}^{14}}\,{{s}^{-1}}\] respectively. The value of the rate constant as \[T\to \infty \] is
A)
\[2.0\times {{10}^{18}}\,{{s}^{-1}}\]
done
clear
B)
\[6.0\times {{10}^{14}}\,{{s}^{-1}}\]
done
clear
C)
Infinity
done
clear
D)
\[3.6\times {{10}^{30}}\,{{s}^{-1}}\]
done
clear
View Answer play_arrow
Which of the following is contributed towards the extra stability of lyophilic colloids?
A)
Hydration
done
clear
B)
Charge
done
clear
C)
Colour
done
clear
D)
Tyndall effect
done
clear
View Answer play_arrow
For an endothermic reaction, where \[\Delta H\] represents the enthalpy of the reaction in kJ/mol, the minimum value for the energy of activation will be
A)
\[less\,\,than\,\,\Delta H\]
done
clear
B)
Zero
done
clear
C)
\[more\,\,than\,\,\Delta H\]
done
clear
D)
\[Equal\,\,to\,\,\Delta H\]
done
clear
View Answer play_arrow
Which of the following methods is used for sol destruction?
A)
Condensation
done
clear
B)
Dialysis
done
clear
C)
Diffusion through animal membrane
done
clear
D)
Addition of an electrolyte
done
clear
View Answer play_arrow
Oxygen and hydrogen gases are produced at the anode and cathode respectively during electrolysis of dilute aq. solution of
A)
\[N{{a}_{2}}S{{O}_{4}}\]
done
clear
B)
\[AgN{{O}_{3}}\]
done
clear
C)
HCl
done
clear
D)
\[CuS{{O}_{4}}\]
done
clear
View Answer play_arrow
In Birkeland - Eyde process, the raw material used is
A)
Air
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[N{{O}_{2}}\]
done
clear
D)
\[HN{{O}_{3}}\]
done
clear
View Answer play_arrow
Consider the following statements in the extraction of iron from haematite:
(I) CO is the main reducing agent (II) \[F{{e}_{2}}{{O}_{3}}\] is used to oxidise the impurities in Bessemer process (III) In BOP process, air is used to oxidise the impurities (IV) When air is used to oxidise the impurities, iron nitride is fanned in iron Pick the correct set of statement(s):
A)
I, IV
done
clear
B)
I, II
done
clear
C)
I, II and III
done
clear
D)
I, II and IV
done
clear
View Answer play_arrow
Among the following nitrates, lead nitrate, silver nitrate and ammonium nitrate, the one that decomposes without leaving any solid residue is
A)
Lead nitrate
done
clear
B)
Ammonium nitrate
done
clear
C)
Silver nitrate
done
clear
D)
Sodium nitrate
done
clear
View Answer play_arrow
A coordination complex compound of cobalt has the molecular formula containing five ammonia molecules, one nitro group and two chlorine atoms for one cobalt atom. One mole of this compound produces three mole ions in an aqueous solution on reacting with excess of\[AgN{{O}_{3}}\], AgCl precipitates. The ionic formula for this complex would be
A)
\[\left[ Co{{\left( N{{H}_{3}} \right)}_{5}}N{{O}_{2}} \right)C{{l}_{2}}\]
done
clear
B)
\[\left[ Co{{\left( N{{H}_{3}} \right)}_{5}}Cl \right]\left[ Cl\left( N{{O}_{2}} \right) \right]\]
done
clear
C)
\[\left[ Co{{\left( N{{H}_{3}} \right)}_{4}}\left( N{{O}_{2}}Cl \right) \right]\left[ \left( N{{H}_{3}} \right)Cl \right]\]
done
clear
D)
\[\left[ Co{{\left( N{{H}_{3}} \right)}_{5}} \right]\left[ {{\left( {{N}_{2}} \right)}_{2}}C{{l}_{2}} \right]\]
done
clear
View Answer play_arrow
Ethyl bromide reacts with silver nitrite to form
A)
Nitroethane
done
clear
B)
Nitroethane and ethyl nitrite
done
clear
C)
Ethyl nitrite
done
clear
D)
Ethane
done
clear
View Answer play_arrow
In a reaction the ferrous \[(F{{e}^{++}})\] iron is oxidised to ferric \[(F{{e}^{+++}})\] ion. The equivalent weight of the ion in the above reaction is equal to
A)
Half of the atomic weight
done
clear
B)
1/5 of the atomic weight
done
clear
C)
The atomic weight
done
clear
D)
Twice the atomic weight
done
clear
View Answer play_arrow
Which would exhibit ionisation isomerism?
A)
\[\left[ Cr{{\left( N{{H}_{3}} \right)}_{6}} \right]C{{l}_{3}}\]
done
clear
B)
\[\left[ Co{{\left( N{{H}_{3}} \right)}_{5}}Br \right]S{{O}_{4}}\]
done
clear
C)
\[\left[ Cr{{\left( en \right)}_{2}}C{{l}_{2}} \right]\]
done
clear
D)
\[\left[ Cr\left( e{{n}_{3}} \right)C{{l}_{3}} \right]\]
done
clear
View Answer play_arrow
One of the following metals forms a volatile carbonyl compound and this property is taken advantage of for its extraction. This metal is
A)
Iron
done
clear
B)
Nickel
done
clear
C)
Cobalt
done
clear
D)
Tungsten
done
clear
View Answer play_arrow
Among the following, the molecule with the highest dipole moment is
A)
\[C{{H}_{3}}Cl\]
done
clear
B)
\[C{{H}_{2}}C{{l}_{2}}\]
done
clear
C)
\[CHC{{l}_{3}}\]
done
clear
D)
\[CC{{l}_{4}}\]
done
clear
View Answer play_arrow
The reagent used to convert
(i) (ii) (iii)
A)
\[{{O}_{3}}/\operatorname{Red}\] \[AlC{{O}_{3}}\] MeCOOH
done
clear
B)
\[{{H}_{2}}S{{O}_{4}}+\] \[{{H}_{2}}O,\,\,heat\] \[HgS{{O}_{4}}\]
done
clear
C)
\[{{O}_{3}}/Zn-\] \[{{H}_{2}}S{{O}_{4}}+\] \[{{H}_{2}}O,\,heat\] AcOH \[HgS{{O}_{4}}\]
done
clear
D)
\[C{{H}_{3}}COOH\] \[{{H}_{2}}{{O}_{2}}\] \[\overset{\Theta }{\mathop{O}}\,H/{{H}_{2}}O\]
done
clear
View Answer play_arrow
Acidic nature is more for
A)
o-amino phenol
done
clear
B)
m-amino phenol
done
clear
C)
p-amino phenol
done
clear
D)
All have equal \[{{K}_{a}}'s\]
done
clear
View Answer play_arrow
Which of the following is the stronger acid?
A)
Benzoic acid
done
clear
B)
4-Nitrobenzoic acid
done
clear
C)
4-Methoxy benzoic acid
done
clear
D)
4-Methyl benzoic acid
done
clear
View Answer play_arrow
Dipole moment of \[C{{H}_{3}}C{{H}_{2}}C{{H}_{3}}\], \[C{{H}_{3}}C{{H}_{2}}OH\] (II) and \[C{{H}_{3}}C{{H}_{2}}F\] (III) is in order
A)
\[I < II < III\]
done
clear
B)
\[I>II>III\]
done
clear
C)
\[I < III < II\]
done
clear
D)
\[III<I<II\]
done
clear
View Answer play_arrow
Which of the following substances on treatment with \[{{P}_{2}}{{O}_{5}}\] gives ethanenitrile?
A)
Propanamide
done
clear
B)
Ethanamide
done
clear
C)
Ethanoic acid
done
clear
D)
N-Methylethyl amine
done
clear
View Answer play_arrow
Which of the following statements is not correct?
A)
Symbols D- and L-before the n me of monosaccharides refer to the direction of rotation of the light
done
clear
B)
Pyranose has a six-membered ring structure
done
clear
C)
Furanose has a five-membered ring structure
done
clear
D)
Pyranose and furanose are the examples of hemiketal.
done
clear
View Answer play_arrow
The correct structure of the drug paracetamol is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Wash and wear clothes are manufactured using
A)
Nylon fibres
done
clear
B)
Cotton mixed with nylon
done
clear
C)
Terylene fibres
done
clear
D)
Wool fibres
done
clear
View Answer play_arrow
Which of the following statements is correct?
A)
Methyl amine is slightly acidic.
done
clear
B)
Methyl amine is less basic than ammonal
done
clear
C)
Methyl amine is less basic than dimethyl amine
done
clear
D)
methyl amine is less basic than aniline
done
clear
View Answer play_arrow
Which electronic level would allow the hydrogen to absorb a photon but not to emit a photon?
A)
3s
done
clear
B)
2p
done
clear
C)
2s
done
clear
D)
1s
done
clear
View Answer play_arrow
Which of the following statement is correct respect to the property of elements with an increase in atomic number in the carbon family (group 14)?
A)
Atomic size decrease
done
clear
B)
Ionization energy increase
done
clear
C)
Metallic character decrease
done
clear
D)
Stability of +2 oxidation state increase
done
clear
View Answer play_arrow
The number of neutrons in a drop of water \[(20 drops = 1\,mL)\,at\,4{}^\circ C\]
A)
\[6.023 \times \,\,1{{0}^{22}}\]
done
clear
B)
\[1.338 \times \,\,1{{0}^{22}}\]
done
clear
C)
\[6.023 \times 1{{0}^{20}}\]
done
clear
D)
\[7.338 \times 1{{0}^{22}}\]
done
clear
View Answer play_arrow
The uncertainty in the position of an electron (\[\operatorname{mass}\,\,=\,\,9.1\,\,\times \,\,1{{0}^{-}}^{28}\]) moving with a velocity of \[3.0\, \times \, 1{{0}^{4}}cm {{s}^{-}}^{1}\] accurate up to \[0.001\,%\] will be (Use \[\frac{h}{4\pi }\] in the uncertainty expression, where \[h=6.626\times \,\,{{10}^{-}}^{27}\])
A)
1.92 cm
done
clear
B)
7.68 cm
done
clear
C)
5.76 cm
done
clear
D)
3.84 cm
done
clear
View Answer play_arrow
The pair of amphoteric hydroxides is
A)
\[Al\left( O{{H}_{3}} \right),\,\,LiOH\]
done
clear
B)
\[Be{{\left( OH \right)}_{2}}, Mg{{\left( OH \right)}_{2}}\]
done
clear
C)
\[B{{\left( OH \right)}_{3}}, Be{{\left( OH \right)}_{2}}\]
done
clear
D)
\[Be{{\left( OH \right)}_{2}}, Zn{{\left( OH \right)}_{2}}\]
done
clear
View Answer play_arrow
Assuming that petrol is octane \[\left( {{C}_{8}}{{H}_{18}} \right)\] and has density 0.8 g/ml, 1.425 litre of petrol on complete combustion will consume
A)
\[50\text{ }moles\text{ }of\text{ }{{O}_{2}}\]
done
clear
B)
\[125\text{ }moles\text{ }of\text{ }{{O}_{2}}\]
done
clear
C)
\[100\text{ }moles\text{ }of\text{ }{{O}_{2}}\]
done
clear
D)
\[200\text{ }moles\text{ }of\text{ }{{O}_{2}}\]
done
clear
View Answer play_arrow
\[PC{{l}_{5}}\] exists, but \[NC{{l}_{5}}\] does not exist because
A)
Nitrogen has no vacant 2-d orbital
done
clear
B)
\[NC{{l}_{5}}\] is unstable
done
clear
C)
N-atom is much smaller than P
done
clear
D)
Nitrogen is highly inert
done
clear
View Answer play_arrow
Boyle's law may be expressed as
A)
\[{{\left( \frac{dP}{dV} \right)}_{T}}=\frac{K}{V}\]
done
clear
B)
\[{{\left( \frac{dP}{dV} \right)}_{T}}=\frac{K}{{{V}^{2}}}\]
done
clear
C)
\[{{\left( \frac{dP}{dV} \right)}_{T}}=-\frac{K}{V}\]
done
clear
D)
none
done
clear
View Answer play_arrow
When \[N{{H}_{3}}\] is treated with HCl, state of hybridization on central nitrogen
A)
\[Changes\text{ }from\,\,s{{p}^{3}}to\,\,s{{p}^{2}}\]
done
clear
B)
Remains unchanged
done
clear
C)
\[Changes\text{ }from\,\,s{{p}^{3}}\,to\,\,s{{p}^{3}}d\]
done
clear
D)
\[Changes\text{ }from\,\,s{{p}^{3}}\,to\,\,sp\]
done
clear
View Answer play_arrow
In the reversible reaction \[A+B\rightleftharpoons C+D\], the concentration of each C and D at equilibrium was 0.8 mole/litre, then the equilibrium constant K, will be
A)
6.4
done
clear
B)
0.64
done
clear
C)
1.6
done
clear
D)
16.0
done
clear
View Answer play_arrow
Smelting of iron ore takes place through this reaction
[a] \[2F{{e}_{2}}{{O}_{3}}\left( s \right) + 3C\left( s \right)\,\,\to \,4Fe\left( s \right) +3C{{O}_{2}}\left( g \right)\] \[\Delta H_{f}^{0}\,of\,\,F{{e}_{2}}{{O}_{3}}\,\,and\,\,C{{O}_{2}}\,are-8242\,\,kJ\,\,mol{{e}^{\text{-1}}}\] and \[-393.7 kJ mole{{\,}^{-}}^{1}\] The reaction is
A)
Endothermic
done
clear
B)
Exothermic
done
clear
C)
\[\Delta H=0\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
2 moles of \[PC{{l}_{5}}\] was heated in a closed vessel of 2 litre capacity. At equilibrium, \[40%\] of \[PC{{l}_{5}}\] is dissociated into\[PC{{l}_{3}}\,\,and\,\,C{{l}_{\text{2}}}\]. The value of equilibrium constant is
A)
0.266
done
clear
B)
0.53
done
clear
C)
2.66
done
clear
D)
5.3
done
clear
View Answer play_arrow
For the given heat of reaction,
(i) \[\operatorname{C}\left( s \right)+{{O}_{2}}\left( g \right)=C{{O}_{2}}\left( g \right)+97\,\,kcal\] (ii) \[\operatorname{C}{{O}_{2}}\left( g \right) + C\left( s \right)=2CO\left( g \right) - 39 kcal\] kcal the heat of combustion of CO(g) is:
A)
68 kcal
done
clear
B)
-68 kcal
done
clear
C)
+48 kcal
done
clear
D)
None
done
clear
View Answer play_arrow
A gas can be liquefied by pressure alone when its temperature is
A)
higher than its critical temperature
done
clear
B)
lower than its critical temperature
done
clear
C)
either of these
done
clear
D)
none of these
done
clear
View Answer play_arrow
Biological organisation starts with:
A)
Atomic level
done
clear
B)
Submicroscopic molecular level
done
clear
C)
Cellular level
done
clear
D)
Organismic level
done
clear
View Answer play_arrow
About 98 percent of the mass of every living organism is composed of just six elements including carbon, hydrogen, nitrogen, oxygen and:
A)
calcium and phosphorous
done
clear
B)
phosphorus and sulphur
done
clear
C)
sulphur and magnesium
done
clear
D)
magnesium and sodium
done
clear
View Answer play_arrow
Which one of the following is an example of negative feedback loop in humans?
A)
Secretion of sweat glands and constriction of skin blood vessels when it is too hot.
done
clear
B)
Constriction of skin blood vessels and contraction of skeletal muscles when it is too cold.
done
clear
C)
Secretion of tears after falling of sand particles into the eye
done
clear
D)
Salivation of mouth at the sight of delicious food.
done
clear
View Answer play_arrow
What is common to whale, seal and shark?
A)
Homoiothermy
done
clear
B)
Seasonal migration
done
clear
C)
Thick subcutaneous fat
done
clear
D)
Convergent evolution
done
clear
View Answer play_arrow
Which one of the following is not a constituent of cell membrane?
A)
Phospholipids
done
clear
B)
Cholesterol
done
clear
C)
Glycolipids
done
clear
D)
Proline
done
clear
View Answer play_arrow
Select the wrong statement from the following.
A)
The chloroplasts are generally much larger than mitochondria
done
clear
B)
Both chloroplasts and mitochondria contain an inner membrane and an outer membrane
done
clear
C)
Both chloroplasts and mitochondria have an internal compartment, the thylakoid space bounded by the thylakoid membrane
done
clear
D)
Both chloroplasts and mitochondria contain DNA
done
clear
View Answer play_arrow
The overall goal of glycolysis, Krebs cycle and the electron transport system is the formation of:
A)
Nucleic acids
done
clear
B)
ATP is small stepwise units
done
clear
C)
ATP in one large oxidation reaction
done
clear
D)
Sugars
done
clear
View Answer play_arrow
If the mean and the median pertaining to a certain character of a population are of the same value, the following is most likely to occur:
A)
a skewed curve
done
clear
B)
a normal distribution
done
clear
C)
a bi-modal distribution
done
clear
D)
a T-shaped curve
done
clear
View Answer play_arrow
Which one of the following is a slime mould?
A)
Anabaena
done
clear
B)
Rhizopus
done
clear
C)
Physarum
done
clear
D)
Thiobacillus
done
clear
View Answer play_arrow
For a critical study of secondary growth in planar which one of the following pairs is suitable?
A)
Wheat and maiden hair fern
done
clear
B)
Sugarcane and sunflower
done
clear
C)
Teak and pine
done
clear
D)
Deodar and fern
done
clear
View Answer play_arrow
Which one of the following statements about Mycoplasma is wrong?
A)
They cause disease in plants
done
clear
B)
They are also called PPLO
done
clear
C)
They are pleomorphic
done
clear
D)
They are sensitive to penicillin
done
clear
View Answer play_arrow
In the prothallus of vascular cryptogam, the antherozoids and eggs mature at different times, As a result
A)
self-fertilization is prevented
done
clear
B)
there is no change in success rate of fertilization
done
clear
C)
there is high degree of sterility
done
clear
D)
one can conclude that the plant is apomictic
done
clear
View Answer play_arrow
Two plants can be conclusively said to belong to the same species if they
A)
have same number of chromosomes
done
clear
B)
can reproduce freely with each other and form seeds
done
clear
C)
have more than 90 per cent similar genes
done
clear
D)
look similar and possess identical secondary metabolites.
done
clear
View Answer play_arrow
If you are asked to classify the various algae into distinct groups, which of the following characters you should choose?
A)
Chemical composition of the cell wall
done
clear
B)
Types of pigments present in the cell
done
clear
C)
Nature of stored food materials in the cell
done
clear
D)
Structural organization of thallus.
done
clear
View Answer play_arrow
Flagellated male gametes are present in all the three of which one of the following sets?
A)
Riccia, Dryopteris and Cycas
done
clear
B)
Anthoceros, Funaria and Spirogyra
done
clear
C)
Zygnema, Saprolegnia and Hydrilla
done
clear
D)
Fucus, Marsilea and Calotropis
done
clear
View Answer play_arrow
In gymnosperms, the pollen chamber represents
A)
the microsporangium in which pollen grains develop
done
clear
B)
a cell in the pollen grain in which the sperms are formed
done
clear
C)
a cavity in the ovule in which pollen grains are stored after pollination
done
clear
D)
an opening in the mega gametophyte through which the pollen tube approaches the egg
done
clear
View Answer play_arrow
Spore dissemination in some liverworts is aided by
A)
peristome teeth
done
clear
B)
elaters
done
clear
C)
indusium
done
clear
D)
calyptra
done
clear
View Answer play_arrow
Which pair of the following belongs to Basidiomycetes?
A)
Morchella and Mushrooms
done
clear
B)
Birds? nest fungi and Puffballs
done
clear
C)
Puffballs and Claviceps
done
clear
D)
Peziza and Stinkhoms
done
clear
View Answer play_arrow
ICBN stands for
A)
Indian Code of Botanical Nomenclature
done
clear
B)
Indian Congress of Biological Names
done
clear
C)
International Code of Botanical Nomenclature
done
clear
D)
International Congress of Biological Names
done
clear
View Answer play_arrow
Ergot of rye is caused by a species of
A)
Claviceps
done
clear
B)
Phytophthora
done
clear
C)
Uncinula
done
clear
D)
Ustilago
done
clear
View Answer play_arrow
When two species of different genealogy come to resemble each other as a result of adaptation, the phenomenon is termed:
A)
Convergent evolution
done
clear
B)
Divergent evolution
done
clear
C)
Microevolution
done
clear
D)
Co-evolution
done
clear
View Answer play_arrow
Adaptive radiation refers to
A)
Power of adaptation in an individual to a variety of environments
done
clear
B)
Adaptations due to geographical isolation
done
clear
C)
Evolution of different species from a common ancestor
done
clear
D)
Migration of members of a species to different geographical areas
done
clear
View Answer play_arrow
The living organisms can be unexceptionally distinguished from the non-living things on the basis of their ability for
A)
growth and movement
done
clear
B)
responsiveness to touch
done
clear
C)
interaction with the environment and progressive evolution
done
clear
D)
reproduction
done
clear
View Answer play_arrow
The finches of Galapagos islands provide an evidence in favour of
A)
Biogeographical evolution
done
clear
B)
Special creation
done
clear
C)
Evolution due to mutation
done
clear
D)
Retrogressive evolution
done
clear
View Answer play_arrow
One of the important consequences of geographical isolation is:
A)
Random creation of new species
done
clear
B)
No change in the isolation fauna
done
clear
C)
Preventing speciation
done
clear
D)
Speciation through reproductive isolation
done
clear
View Answer play_arrow
Industrial melanism as observed in peppered moth proves that:
A)
Melanism is a pollution-generated feature
done
clear
B)
The true black melanic forms arise by a recurring random mutation
done
clear
C)
The melanic form of the moth has no selective advantage over lighter form in industrial area
done
clear
D)
The lighter form moth has no selective advantage either in polluted industrial area or in non-polluted area.
done
clear
View Answer play_arrow
The concept of chemical evolution is based on:
A)
Possible origin of life by combination of chemicals
done
clear
B)
Crystallization of chemicals under suitable environmental conditions
done
clear
C)
Interaction of water, air and clay under
done
clear
D)
Effect of solar radiation of chemicals
done
clear
View Answer play_arrow
Among the human ancestors the brain size was more than 1000 CC in:
A)
Homo habilis
done
clear
B)
Homo neanderthalensis
done
clear
C)
Homo erectus
done
clear
D)
Ramapithecus
done
clear
View Answer play_arrow
Which of the following pairs are correctly matched?
A. Crocodile 4-Chambered heart B. Sea Urchin Parapodia C. Obella Metagenesis D. Lemur Thecodont
A)
Only A and B
done
clear
B)
A, C and D
done
clear
C)
B, C and D
done
clear
D)
Only A and D
done
clear
View Answer play_arrow
Select the correct statement from the following.
A)
Mutations are random and directional
done
clear
B)
Darwinian variations are small and directionless
done
clear
C)
Fitness is the end result of the ability to adapt and gets selected by nature
done
clear
D)
All mammals except whales and camels have seven cervical vertebrates
done
clear
View Answer play_arrow
Which one of the following is a matching pair of a body feature and the animal possessing it?
A)
Ventral heart - Scorpion
done
clear
B)
Post-anal tail - Octopus
done
clear
C)
Ventral central nervous system - Leech
done
clear
D)
Pharyngeal gill slits absent in - Chamaeleon embryo
done
clear
View Answer play_arrow
What is common between parrot, plaiypus and kangaroo?
A)
Oviparity
done
clear
B)
Homoiothermy
done
clear
C)
Toothless jaws
done
clear
D)
Functional post-anal tail
done
clear
View Answer play_arrow
What is true about Nereis, Scorpion, Cockroach and Silver fish?
A)
They all belong to the same phylum
done
clear
B)
They all have jointed paired appendages
done
clear
C)
They all possess dorsal heart
done
clear
D)
None of them is aquatic
done
clear
View Answer play_arrow
Which one of the following statement is correct?
A)
Ontogeny repeats phylogeny
done
clear
B)
Stem cells are specialized cells
done
clear
C)
There is no evidence of the existence of gills during embryogenesis of mammals
done
clear
D)
All plant and animal cells are totipotent.
done
clear
View Answer play_arrow
"Foolish Seedling" disease of rice led to the discovery of
A)
IAA
done
clear
B)
GA
done
clear
C)
ABA
done
clear
D)
2, 4 - D
done
clear
View Answer play_arrow
Passage cells are thin-walled cells found in
A)
central region of style through which the pollen tube grows towards the ovary.
done
clear
B)
endodermis of roots facilitating rapid transport of water from cortex to pericycle
done
clear
C)
phloem elements that serve as entry points for substances for transport to other parts.
done
clear
D)
testa of seed to enable emergence of growing embryonic axis during seed germination.
done
clear
View Answer play_arrow
The first acceptor of electrons from an excited chlorophyll molecule of photosystem II is
A)
Quinone
done
clear
B)
Cytochrome
done
clear
C)
Iron-sulphur protein
done
clear
D)
Ferredoxin.
done
clear
View Answer play_arrow
All enzymes of TCA cycle are located in the mitochondrial matrix except one which is located in inner mitochondrial membranes in eukaryotes and in cytosol in prokaryotes. This enzyme
A)
succinate dehydrogenase
done
clear
B)
lactate dehydrogenase
done
clear
C)
isocitrate dehydrogenase
done
clear
D)
malate dehydrogenase
done
clear
View Answer play_arrow
The wavelength of light absorbed by Pr form of phytochrome is
A)
620 nm
done
clear
B)
640 nm
done
clear
C)
680 nm
done
clear
D)
720 nm
done
clear
View Answer play_arrow
Opening of floral buds into flowers is type of
A)
Autonomic movement of growth
done
clear
B)
Autonomic movement of locomotion
done
clear
C)
Autonomic movement of variation
done
clear
D)
Paratonic movement of growth
done
clear
View Answer play_arrow
Which one of the following pairs is not correctly matched?
A)
IAA - Cell wall elongation
done
clear
B)
Abscisic Acid - Stomatal closure
done
clear
C)
Gibberellic Acid - Leaf fall
done
clear
D)
Cytokinin - Cell division
done
clear
View Answer play_arrow
One gene-one enzyme relationship was established for the first time in:
A)
Diplococcus pneumonia
done
clear
B)
Neurospora crassa
done
clear
C)
Salmonella typhimurium
done
clear
D)
Escherichia coli
done
clear
View Answer play_arrow
Male gametes in angiosperms are formed by the division of
A)
Microspore mother cell
done
clear
B)
Microspore
done
clear
C)
Generative cell
done
clear
D)
Vegetative cell
done
clear
View Answer play_arrow
Two cells A and B are contiguous. Cell A has osmotic pressure 10 atm, turgor pressure 7 atm and diffusion pressure deficit 3 atm. Cell B has osmotic pressure 8 atm, turgor pressure 3 atm and diffusion pressure deficit 5 atm. The result will be
A)
Movement of water of Cell A to B
done
clear
B)
Movement of water from Cell B to A
done
clear
C)
No movement of water
done
clear
D)
Equilibrium between the two
done
clear
View Answer play_arrow
In the leaves of C4 plants, malic acid formation during \[C{{O}_{2}}\] fixation occurs in the cells of
A)
Epidermis
done
clear
B)
Mesophyll
done
clear
C)
Bundle sheath
done
clear
D)
Phloem
done
clear
View Answer play_arrow
Which of the following is a flowering plant with nodules containing filamentous nitrogen-fixing microorganism?
A)
Cicer arietinum
done
clear
B)
Casuarina equisetifolia
done
clear
C)
Crotalaria juncea
done
clear
D)
Cycas revoluta
done
clear
View Answer play_arrow
Which one of the following is surrounded by a callose wall?
A)
Pollen grain
done
clear
B)
Microspore mother cell
done
clear
C)
Male gamete
done
clear
D)
Egg
done
clear
View Answer play_arrow
Which one of the following elements is not an essential micronutrient for plant growth?
A)
Ca
done
clear
B)
Mn
done
clear
C)
Zn
done
clear
D)
Cu
done
clear
View Answer play_arrow
If you suspect major deficiency of antibodies in person, to which of the following would you look for confirmatory evidence?
A)
Haemocytes
done
clear
B)
Serum albumins
done
clear
C)
Serum globulins
done
clear
D)
Fibrinogen in the plasma
done
clear
View Answer play_arrow
Which one of the following is a fat-soluble vitamin and its related deficiency disease?
A)
Calciferol - Pellagra
done
clear
B)
Ascorbic acid - Scurvy
done
clear
C)
Retinol - Xerophthalmia
done
clear
D)
Cobalamin - Beriberi
done
clear
View Answer play_arrow
Which one of the following mammalian cells is not capable of metabolising glucose to carbon dioxide aerobically?
A)
Red blood cells
done
clear
B)
White blood cells
done
clear
C)
Unstriated muscle cells
done
clear
D)
Liver cells
done
clear
View Answer play_arrow
Compared to a bull a bullock is docile because of
A)
lower levels of adrenalin/noradrenalin in its blood
done
clear
B)
higher levels of thyroxin
done
clear
C)
higher levels of cortisone
done
clear
D)
lower levels of blood testosterone
done
clear
View Answer play_arrow
In the human female, menstruation can be deferred by the administration of
A)
FSH only
done
clear
B)
LH only
done
clear
C)
Combination of FSH and LH
done
clear
D)
Combination of estrogen and progesterone
done
clear
View Answer play_arrow
In human body, which one of the following is anatomically correct?
A)
Cranial nerves - 10 pairs
done
clear
B)
Floating ribs - 2 pairs
done
clear
C)
Collar bones - 3 pairs
done
clear
D)
Salivary glands - 1 pairs
done
clear
View Answer play_arrow
In which one of the following preparations are you likely to come across cell junctions most frequently?
A)
Hyaline cartilage
done
clear
B)
Ciliated epithelium
done
clear
C)
Thrombocytes
done
clear
D)
Tendon
done
clear
View Answer play_arrow
A drop of each of the following is placed separately on four slides. Which of them will not coagulate?
A)
Whole blood from pulmonary vein
done
clear
B)
Blood plasma
done
clear
C)
Blood serum
done
clear
D)
Sample from the thoracic duct of lymphatic system
done
clear
View Answer play_arrow
Feeling the tremors of an earthquake a scared resident of seventh floor of a multistoried building starts climbing down the stairs rapidly. Which hormone initiated this action?
A)
Gastrin
done
clear
B)
Thyroxin
done
clear
C)
Adrenaline
done
clear
D)
Glucagon
done
clear
View Answer play_arrow
A person who is on a long hunger strike and is surviving only on water will have
A)
less urea in his urine
done
clear
B)
more sodium in his urine
done
clear
C)
less amino acids in his urine
done
clear
D)
more glucose in his blood.
done
clear
View Answer play_arrow
Which one of the following pairs of structures distinguishes a nerve cell from other types of cell?
A)
Nucleus and mitochondria
done
clear
B)
Perikaryon and dendrites
done
clear
C)
Vacuoles and fibers
done
clear
D)
Flagellum and medullary sheath
done
clear
View Answer play_arrow
Which part of ovary in mammals acts as an endocrine gland after evolution?
A)
Vitelline membrane
done
clear
B)
Graafian follicle
done
clear
C)
Stroma
done
clear
D)
Germinal epithelium
done
clear
View Answer play_arrow
During the transmission of nerve impulse through a nerve fibre, the potential on the inner side of the plasma membrane has which type of electric charge?
A)
First positive, then negative and again back to positive
done
clear
B)
First negative, then positive and again back to negative
done
clear
C)
First positive, then negative and continue to be negative
done
clear
D)
First negative, then positive and continue to be positive.
done
clear
View Answer play_arrow
A person is having problems with calcium and phosphorous metabolism in his body. Which one of the following glands may not be functioning properly?
A)
Thyroid
done
clear
B)
Parathyroid
done
clear
C)
Parotid
done
clear
D)
Pancreas
done
clear
View Answer play_arrow
Identify the odd combination of the habitat and the particular animal concerned:
A)
Rann of Kutch-Wild Ass
done
clear
B)
Dachigam National Park- Snow Leopard
done
clear
C)
Sunderbans - Bengal Tiger
done
clear
D)
Periyar-Elephant
done
clear
View Answer play_arrow
In which one of the following the BOD (bio- chemical oxygen demand) of sewage (S), distillery effluent (DE), paper mill effluent (PE) sugar mill effluent (SE) have been arranged ascending order?
A)
\[S<DE<PE<SE~\]
done
clear
B)
\[SE<S<PE<DE\]
done
clear
C)
\[SE<PE<S<DE\]
done
clear
D)
\[PE<S<SE<DE\]
done
clear
View Answer play_arrow
Which one of the following ecosystem types has the highest annual net primary productivity?
A)
Temperate deciduous forest
done
clear
B)
Tropical rain forest
done
clear
C)
Tropical deciduous forest
done
clear
D)
Temperate evergreen forest
done
clear
View Answer play_arrow
Which one of the following is being utilized as a source of biodiesel in the Indian countryside?
A)
Pongamia
done
clear
B)
Euphorbia
done
clear
C)
Beetroot
done
clear
D)
Sugarcane
done
clear
View Answer play_arrow
In a coal fires power plant electrostatic precipitators are installed to control emission of
A)
CO
done
clear
B)
\[S{{O}_{2}}\]
done
clear
C)
NOX
done
clear
D)
SPM
done
clear
View Answer play_arrow
Which one of the following is not a bioindiol of water pollution?
A)
Sewage fungus
done
clear
B)
Sludge worms
done
clear
C)
Blood worms
done
clear
D)
Stone flies
done
clear
View Answer play_arrow
A high density of elephant population in an area can result in
A)
Predation on one another
done
clear
B)
Mutualism
done
clear
C)
Intra-specific competition
done
clear
D)
Inter-specific competition
done
clear
View Answer play_arrow
Geometric representation of age structure is a characteristic of
A)
Ecosystem
done
clear
B)
Biotic community
done
clear
C)
Population
done
clear
D)
Landscape
done
clear
View Answer play_arrow
Which one of the following pairs of organisms are exotic species introduced in India?
A)
Nile perch, Ficus religiosa
done
clear
B)
Ficus religiosa, Lantana camara
done
clear
C)
Lantana camara. Water hyacinth
done
clear
D)
Water hyacinth, Prosopis cineraria
done
clear
View Answer play_arrow
One of endangered species of Indian medicinal plants is that of
A)
Nepenthes
done
clear
B)
Podophyllum
done
clear
C)
Ocimum
done
clear
D)
Garlic
done
clear
View Answer play_arrow
A genetically engineered microorganism used successfully in bioremediation of oil spills is a species of
A)
Bacillus
done
clear
B)
Pseudomonas
done
clear
C)
Trichoderma
done
clear
D)
Xanthomonas
done
clear
View Answer play_arrow
A sequential expression of a set of human genes occurs when a steroid molecule binds to the
A)
Ribosome
done
clear
B)
Transfer RNA
done
clear
C)
Messenger RNA
done
clear
D)
DNA sequence
done
clear
View Answer play_arrow
The Okazaki fragments in DNA chain growth polymerize in the direction and explain
A)
DNA replication
done
clear
B)
result in transcription
done
clear
C)
polymerize in the direction and forms replication fork
done
clear
D)
prove semi-conservative nature of DNA replication
done
clear
View Answer play_arrow
In the hexaploid wheat, the haploid (n) and basic (x) numbers of chromosomes are
A)
\[n = 21 and x = 7\]
done
clear
B)
\[n=7and x=21\]
done
clear
C)
\[n=21 and x= 21\]
done
clear
D)
\[n=21 and x=14\]
done
clear
View Answer play_arrow
Molecular basis of organ differentiation depends on the modulation in transcription by:
A)
Anticodon
done
clear
B)
RNA polymerase
done
clear
C)
Ribosome
done
clear
D)
Transcription factor
done
clear
View Answer play_arrow
Telomere repetitive DNA sequence control the function of eukaryote chromosomes because they
A)
prevent chromosome loss
done
clear
B)
act as replicons
done
clear
C)
are RNA transcription initiator
done
clear
D)
help chromosome pairing
done
clear
View Answer play_arrow
Inheritance of skin colour in humans is an example of
A)
Codominance
done
clear
B)
chromosomal aberration
done
clear
C)
point mutation
done
clear
D)
polygenic inheritance
done
clear
View Answer play_arrow
A common test to find the genotype of a hybrid is by
A)
crossing of one \[{{F}_{1}}\] progeny with male parent
done
clear
B)
crossing of one \[{{F}_{2}}\] progeny with male parent
done
clear
C)
crossing of one \[{{F}_{2}}\] progeny with female parent
done
clear
D)
studying the sexual behaviour of \[{{F}_{1}}\] progenies
done
clear
View Answer play_arrow
During transcription, RNA polymerase holoenzyme binds to a gene promoter and assumes a saddle-like structure. What is its DNA-binding sequence?
A)
TATA
done
clear
B)
TTAA
done
clear
C)
AATT
done
clear
D)
CACC
done
clear
View Answer play_arrow
Two genes R and Y are located very close on the chromosomal linkage map of maize plant. When RRYY and rryy genotypes are hybridized, the F2 segregation will show
A)
Higher number of the parental types
done
clear
B)
Higher number of the recombinant types
done
clear
C)
Segregation in the expected \[9:3:3:1\] ratio
done
clear
D)
Segregation in \[3:1\] ratio
done
clear
View Answer play_arrow
In maize, hybrid vigour is exploited by
A)
Inducing mutations
done
clear
B)
Bombarding the seeds with DNA
done
clear
C)
Crossing of two inbred parental lines
done
clear
D)
Harvesting seeds from the most productive plants
done
clear
View Answer play_arrow
Differentiation of organs and tissues in a developing organism is associated with
A)
Deletion of genes
done
clear
B)
Developmental mutations
done
clear
C)
Differential expression of genes
done
clear
D)
Lethal mutations
done
clear
View Answer play_arrow
In pea plants, yellow seeds are dominant to green. If a heterozygous yellow seeded plant is crossed with a green seeded plant, what ratio of yellow and green seeded plants would you expect in \[{{F}_{1}}\] generation?
A)
\[3:1\]
done
clear
B)
\[50 : 50\]
done
clear
C)
\[9:1~\]
done
clear
D)
\[1:3\]
done
clear
View Answer play_arrow
The length of DNA molecule greatly exceeds the dimensions of the nucleus in eukaryotic cells. How is this DNA accommodated?
A)
Through elimination of repetitive DNA
done
clear
B)
Deletion of non-essential genes
done
clear
C)
Super-coiling in nucleosomes
done
clear
D)
DNAse digestion
done
clear
View Answer play_arrow
In cloning of cattle a fertilized egg is taken out of the mother's womb and
A)
from this up to eight identical twins can be produced
done
clear
B)
the egg is divided into 4 pairs of cells which are implanted into the womb of others cows
done
clear
C)
in the eight cell stage, cells are separated and cultured until small embryos are formed which are implanted into the womb of other cows
done
clear
D)
in the eight cell stage the individual cells are separated under electrical field far further development in culture media
done
clear
View Answer play_arrow
Which one of the following statements is correct?
A)
At present it is not possible to grow maize without chemical fertilizers.
done
clear
B)
Extensive use of chemical fertilizers may lead to eutrophication of nearby water bodies.
done
clear
C)
Both Azotobacter and Rhizobium fix atmospheric nitrogen in root nodules of plants
done
clear
D)
Cyanobacteria such as Anabaena m Nostoc are important mobilizers of phosphates and potassium for plant nutrition in soil.
done
clear
View Answer play_arrow
The population of an insect species shows explosive increase in numbers during ran season followed by its disappearance at the a of the season. What does this show?
A)
The population of its predators increase enormously.
done
clear
B)
S-shaped or sigmoid growth of this insect.
done
clear
C)
The food plants mature and die at the end of the rainy season.
done
clear
D)
Its population growth curve is of
done
clear
View Answer play_arrow
The two polynucleotide chains in DNA are
A)
semiconservative
done
clear
B)
parallel
done
clear
C)
discontinuous
done
clear
D)
antiparallel
done
clear
View Answer play_arrow
 The capacity of the capacitor is
The capacity of the capacitor is