A glass flask is filled up to a mark with 50 cc of mercury at \[9\times {{10}^{-6}}\text{/ }\!\!{}^\circ\!\!\text{ C}\] If the flask and contents are heated to \[38{}^\circ C,\] how much mercury-will be above the mark? (\[\alpha \]for glass is \[9\times {{10}^{-6}}\text{/ }\!\!{}^\circ\!\!\text{ C}\] and coefficient of real expansion of mercury is \[180\times {{10}^{-6}}/{}^\circ C)\]
A)
0.85 cc
done
clear
B)
0.46 cc
done
clear
C)
0.153 cc
done
clear
D)
0.05 cc
done
clear
View Answer play_arrow
Point masses of 1, 2, 3 and 4 kg are lying at the points \[(0,0,0),\] \[(2,0,0),\] \[(0,3,0)\] and \[(-2,-2,0)\] respectively The moment of inertia of this system about X-axis will be
A)
\[43\,\,kg\,{{m}^{2}}\]
done
clear
B)
\[34\,\,kg\,{{m}^{2}}\]
done
clear
C)
\[27\,\,kg\,{{m}^{2}}\]
done
clear
D)
\[72\,\,kg\,{{m}^{2}}\]
done
clear
View Answer play_arrow
A thin liquid film formed between a U-shaped wire and a light slider supports a weight of \[1.5\times {{10}^{-2}}\,N\](see figure). The length of the slider is 30 cm and its weight negligible. The surface tension of the liquid film is
A)
\[0.0125\,\,N{{m}^{-1}}\]
done
clear
B)
\[0.1\,\,N{{m}^{-1}}\]
done
clear
C)
\[0.05\,\,N{{m}^{-1}}\]
done
clear
D)
\[0.025\,\,N{{m}^{-1}}\]
done
clear
View Answer play_arrow
A bar magnet of magnetic moment \[\overrightarrow{M}\] is placed in the magnetic field \[\overrightarrow{B}.\] The torque acting on the magnet is
A)
\[\overrightarrow{M}\times \overrightarrow{B}\]
done
clear
B)
\[\overrightarrow{M}-\overrightarrow{B}\]
done
clear
C)
\[\frac{1}{2}\,\overrightarrow{M}\times \overrightarrow{B}\]
done
clear
D)
\[\overrightarrow{M}+\overrightarrow{B}\]
done
clear
View Answer play_arrow
A phase difference between two points separated by 0.8 m in a wave of frequency 120 Hz is \[\pi /2.\] The wave velocity is
A)
384 m/s
done
clear
B)
768 m/s
done
clear
C)
250 m/s
done
clear
D)
154 m/s
done
clear
View Answer play_arrow
A simple pendulum is executing simple harmonic motion with a time period T. If the length of the pendulum is increased by 21% then the increase in the time period of the pendulum due to increased length is
A)
50%
done
clear
B)
30%
done
clear
C)
21%
done
clear
D)
10%
done
clear
View Answer play_arrow
In a transformer, number of turns in the primary coil are 140 and that in the secondary coil are 280. If current in primary coil is 4A, then that in the secondary coil is
A)
4 A
done
clear
B)
2 A
done
clear
C)
6 A
done
clear
D)
10 A
done
clear
View Answer play_arrow
A triangular block of mass M with angles \[30{}^\circ ,\]\[60{}^\circ \]and \[90{}^\circ \] rest with its \[30{}^\circ -90{}^\circ \] side on a horizontal table. A cubical block of mass m rests on \[60{}^\circ -30{}^\circ \]side. The acceleration which M must have relative to the table to keep m stationary relative to the triangular block is (assuming frictionless contact)
A)
\[g\]
done
clear
B)
\[g/\sqrt{2}\]
done
clear
C)
\[g/\sqrt{3}\]
done
clear
D)
\[g/\sqrt{5}\]
done
clear
View Answer play_arrow
The K.E. of one mole of an ideal gas is E = (3/2) RT. Then \[{{C}_{p}}\] will be
A)
0.5 R
done
clear
B)
0.1 R
done
clear
C)
1.5 R
done
clear
D)
2.5 R
done
clear
View Answer play_arrow
A network of six identical capacitors, each of capacitance C is made as shown in the figure. The equivalent capacitance between points A and B is
A)
C/4
done
clear
B)
3C/4
done
clear
C)
4C/3
done
clear
D)
3C
done
clear
View Answer play_arrow
The condition for obtaining secondary maxima in the diffraction pattern due to single slit is
A)
\[a\,\,\sin \theta =n\lambda \]
done
clear
B)
\[a\,\,\sin \theta =(2n-1\frac{\lambda }{2})\]
done
clear
C)
\[a\,\,\sin \theta =(2n-1)\lambda \]
done
clear
D)
\[a\,\,\sin \theta =\frac{n\lambda }{2}\]
done
clear
View Answer play_arrow
If R is universal gas constant, the amount of heat needed to raise the temperature of 2 moles of an ideal monoatomic gas from 273 K to 373 K, when no work is done, is
A)
100 R
done
clear
B)
150 R
done
clear
C)
300 R
done
clear
D)
500 R
done
clear
View Answer play_arrow
How much energy is required for a body of mass 1000 kg to escape from the earth? \[(g=9.8\,m/{{s}^{2}},\] \[R=6400\,km)\]
A)
about \[3.2\times {{10}^{10}}\,J\]
done
clear
B)
about \[6.4\times {{10}^{10}}\,J\]
done
clear
C)
about \[6.4\times {{10}^{6}}\,J\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A wheel with ten metallic spokes each 0.50m long is rotated with a speed of 120 rev/min in a plane normal to the earth?s magnetic field at the place. If the magnitude of the field is 0.40 G, the induced emf between the axle and the rim of the wheel is equal to
A)
\[1.256\times {{10}^{-3}}V\]
done
clear
B)
\[6.28\times {{10}^{-4}}V\]
done
clear
C)
\[1.256\times {{10}^{-4}}V\]
done
clear
D)
\[6.28\times {{10}^{-5}}V\]
done
clear
View Answer play_arrow
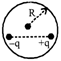
A closed spherical surface of radius R encloses an electric dipole. The net electric flux through the surface is
A)
zero
done
clear
B)
\[2q.4\pi {{R}^{2}}\]
done
clear
C)
\[\frac{q}{{{\varepsilon }_{0}}}\]
done
clear
D)
\[\frac{2q}{{{\varepsilon }_{0}}}\]
done
clear
View Answer play_arrow
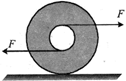
A spool is pulled horizontally by two equal and opposite forces on rough surface. The correct statement is:
A)
centre of mass of spool moves towards right
done
clear
B)
centre of mass of spool moves towards left
done
clear
C)
centre of mass remains at rest
done
clear
D)
none of these
done
clear
View Answer play_arrow
If the atom \[_{100}F{{m}^{257}}\] follows the Bohr model and the radius of \[_{100}F{{m}^{257}}\] is n times the Bohr radius, then find n.
A)
100
done
clear
B)
200
done
clear
C)
4
done
clear
D)
¼
done
clear
View Answer play_arrow
Two bodies of masses 10 kg and 100 kg are separated by a distance of 2m \[(G=6.67\times {{10}^{-11}}N{{m}^{2}}k{{g}^{-2}}).\] The gravitational potential at the mid-point on the line joining the two is
A)
\[7.3\times {{10}^{-7}}J/kg\]
done
clear
B)
\[7.3\times {{10}^{-9}}J/kg\]
done
clear
C)
\[-7.3\times {{10}^{-9}}J/kg\]
done
clear
D)
\[7.3\times {{10}^{-6}}J/kg\]
done
clear
View Answer play_arrow
For a given material, the Young's modulus is 2.4 times that of rigidity modulus, the Poission's ratio is
A)
0.2
done
clear
B)
0.4
done
clear
C)
12
done
clear
D)
2.4
done
clear
View Answer play_arrow
The logic behind 'NOR' gate is that it gives
A)
high output when both inputs are low
done
clear
B)
high output when both inputs are high
done
clear
C)
low output when both inputs are low
done
clear
D)
None of these
done
clear
View Answer play_arrow
A Laser light of wavelength 660 nm is used to weld Retina detachment. If a Laser pulse of width 60 ms and power 0.5 kW is used the approximate number of photons in the pulse are : [Take Planck's constant h \[=6.62\times {{10}^{-34}}Js\]]
A)
1020
done
clear
B)
1018
done
clear
C)
1022
done
clear
D)
1019
done
clear
View Answer play_arrow
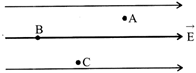
A, B and C are three points in a uniform electric field. The electric potential is
A)
maximum at B
done
clear
B)
maximum at C
done
clear
C)
same at all the three points A, B and C
done
clear
D)
maximum at A
done
clear
View Answer play_arrow
A block B of mass 4 kg is placed on a rough horizontal plane. A time dependent horizontal force \[f=kt\] acts on the block, k \[=2\,N/{{s}^{2}}.\] The frictional force between the block and the plane at \[t=2\]sec is \[(\mu =0.2)\]
A)
4 N
done
clear
B)
8 N
done
clear
C)
12 N
done
clear
D)
zero
done
clear
View Answer play_arrow
Four charges equal to \[-\,Q\] are placed at the four corners of a square and a charge q is at its centre. If the system is in equilibrium the value of q is
A)
\[-\frac{Q}{4}\left( 1+2\sqrt{2} \right)\]
done
clear
B)
\[\frac{Q}{4}\left( 1+2\sqrt{2} \right)\]
done
clear
C)
\[-\frac{Q}{2}\left( 1+2\sqrt{2} \right)\]
done
clear
D)
\[\frac{Q}{2}\left( 1+2\sqrt{2} \right)\]
done
clear
View Answer play_arrow
Using equipartition of energy, the specific heat (in \[J\,k{{g}^{-1}}{{K}^{-1}}\]) of aluminium at room temperature can be estimated to be (atomic weight of aluminium = 27)
A)
410
done
clear
B)
25
done
clear
C)
1850
done
clear
D)
925
done
clear
View Answer play_arrow
A bag of mass M hangs by a long thread and a bullet (mass m) comes horizontally with velocity V and gets caught in the bag. Then for the combined (bag + bullet) system
A)
momentum \[=\frac{mvM}{M+m}\]
done
clear
B)
kinetic energy \[=\frac{m\,{{V}^{2}}}{2}\]
done
clear
C)
momentum \[=\frac{m\,V(M+m)}{M}\]
done
clear
D)
kinetic energy \[=\frac{{{m}^{2}}\,\,{{v}^{2}}}{2(M+m)}\]
done
clear
View Answer play_arrow
The efficiency of an ideal gas with adiabatic exponent Y for the shown cyclic process would be
A)
\[\frac{(2\,\,\text{ln}\,2-1)}{\gamma /(\gamma -1)}\]
done
clear
B)
\[\frac{(1-2\,\,\text{ln}\,2)}{\gamma /(\gamma -1)}\]
done
clear
C)
\[\frac{(2\,\,\text{ln}\,2+1)}{\gamma /(\gamma -1)}\]
done
clear
D)
\[\frac{(2\,\,\text{ln}\,\,2-1)}{\gamma /(\gamma +1)}\]
done
clear
View Answer play_arrow
The electric potential V(x) in a region around the origin is given by \[V(x)=4{{x}^{2}}\] volts. The electric charge enclosed in a cube of 1 m side with its centre at the origin is (in coulomb)
A)
\[8{{\varepsilon }_{0}}\]
done
clear
B)
\[-4{{\varepsilon }_{0}}\]
done
clear
C)
\[0\]
done
clear
D)
\[-8{{\varepsilon }_{0}}\]
done
clear
View Answer play_arrow
A gas is compressed from a volume of \[2{{m}^{3}}\] to a volume of \[1\,{{m}^{3}}\] at a constant pressure of \[100\,N/{{m}^{2}}.\] Then it is heated at constant volume by supplying 150 J of energy. As a result, the internal energy of the gas:
A)
increases by 250 J
done
clear
B)
decreases by 250 J
done
clear
C)
increases by 50 J
done
clear
D)
decreases by 50 J
done
clear
View Answer play_arrow
As intensity of incident light increases
A)
photoelectric current increase
done
clear
B)
K.E. of emitted photoelectron increases
done
clear
C)
photo electric current decreases
done
clear
D)
K.E. of emitted photoelectrons decreases
done
clear
View Answer play_arrow
A block of mass 60 kg just slides over a horizontal distance of 0.9 m. If the coefficient of friction between their surface is 0.15 then work done against friction will be
A)
79.4 J
done
clear
B)
97.54 J
done
clear
C)
105.25 J
done
clear
D)
None of these
done
clear
View Answer play_arrow
A projectile is projected at an angle of \[45{}^\circ \] with speed u. The radius of curvature of its trajectory at the maximum height is
A)
\[\frac{{{u}^{2}}}{g}\]
done
clear
B)
\[\frac{{{u}^{2}}}{4g}\]
done
clear
C)
\[\frac{2{{u}^{2}}}{g}\]
done
clear
D)
\[\frac{{{u}^{2}}}{2g}\]
done
clear
View Answer play_arrow
A wheel having moment of inertia \[2\,kg-{{m}^{2}}\] about its vertical axis, rotates at the rate of 60 rpm about this axis, The torque which can stop the wheel's rotation in one minute would be
A)
\[\frac{\pi }{18}Nm\]
done
clear
B)
\[\frac{2\pi }{15}Nm\]
done
clear
C)
\[\frac{\pi }{12}Nm\]
done
clear
D)
\[\frac{\pi }{15}Nm\]
done
clear
View Answer play_arrow
Two long parallel wires P and Q are held perpendicular to the plane of paper with distance of 5m between them. If P and Q carry current of 2.5 A. and 5 A. respectively in the same direction, then the magnetic field at a point half-way between the wires is
A)
\[{{\mu }_{0}}/17\]
done
clear
B)
\[\sqrt{3}{{\mu }_{0}}/2\pi \]
done
clear
C)
\[{{\mu }_{0}}/2\pi \]
done
clear
D)
\[3{{\mu }_{0}}/2\pi \]
done
clear
View Answer play_arrow
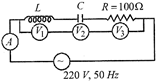
In the given circuit the reading of voltmeter \[{{V}_{1}}\] and \[{{V}_{2}}\] are 300 volt each. The reading of the voltmeter \[{{V}_{3}}\] and ammeter A are respectively
A)
150 V and 2.2 A
done
clear
B)
220 V and 2.2 A
done
clear
C)
220 V and 2.0 A
done
clear
D)
100 V and 2.0 A
done
clear
View Answer play_arrow
The photoelectric work function for a metal surface is 4.125 eV. The cut-off wavelength for this surface is
A)
\[4125\,A{}^\circ \]
done
clear
B)
\[3000\,A{}^\circ \]
done
clear
C)
\[6000\,A{}^\circ \]
done
clear
D)
\[2062\,A{}^\circ \]
done
clear
View Answer play_arrow
Which is true of the following in a purely resistive ac circuit?
A)
The current leads the driving voltage
done
clear
B)
The driving voltage leads the current.
done
clear
C)
The current and driving voltage are in the same phase.
done
clear
D)
Any of the above may be true depending on the value of resistance
done
clear
View Answer play_arrow
The position x of a particle at time t is given by\[x=\frac{{{V}_{0}}}{a}(1-{{e}^{-at}},)\] where \[{{V}_{0}}\] is constant and \[a>0.\] The dimensions of \[{{V}_{0}}\] and a are respectively
A)
\[{{M}^{0}}L\,{{T}^{-1}}\] and \[{{T}^{-1}}\]
done
clear
B)
\[{{M}^{0}}L\,{{T}^{0}}\] and \[{{T}^{-1}}\]
done
clear
C)
\[{{M}^{0}}L\,{{T}^{-1}}\] and \[L{{T}^{-2}}\]
done
clear
D)
\[{{M}^{0}}L\,{{T}^{-1}}\] and \[T\]
done
clear
View Answer play_arrow
A soap bubble has radius r and volume V. If the excess pressure inside the bubble is P, then PV is proportional to
A)
\[r\]
done
clear
B)
\[{{r}^{2}}\]
done
clear
C)
\[{{r}^{3}}\]
done
clear
D)
\[{{r}^{4}}\]
done
clear
View Answer play_arrow
A particle executes S.H.M. having time period T. Time period with which the potential energy changes is
A)
\[T\]
done
clear
B)
\[2T\]
done
clear
C)
\[T/2\]
done
clear
D)
\[\infty \]
done
clear
View Answer play_arrow
If red light and violet light rays are of focal lengths \[{{F}_{R}}\] and \[{{F}_{V}}\] respectively, then which one of the following is true?
A)
\[{{\lambda }_{R}}\le {{\lambda }_{\,V}}\]
done
clear
B)
\[{{\mu }_{R}}>{{\mu }_{\,V}}\]
done
clear
C)
\[{{\mu }_{R}}={{\mu }_{\,V}}\]
done
clear
D)
\[{{\mu }_{R}}<{{\mu }_{\,V}}\]
done
clear
View Answer play_arrow
Hysteresis loops for two magnetic materials A and B are given below:
These materials are used to make magnets for elecric generators, transformer core and electromagnet core. Then it is proper to use:
A)
A for transformers and B for electric generators.
done
clear
B)
B for electromagnets and transformers.
done
clear
C)
(e) A for electric generators and trasformers.
done
clear
D)
A for electromagnets and B for electric generators.
done
clear
View Answer play_arrow
A ray of monochromatic light suffers minimum deviation of \[38{}^\circ ,\] while passing through a prism of refracting angle \[60{}^\circ .\] Refractive index of the prism material is
A)
2.4
done
clear
B)
0.8
done
clear
C)
1.3
done
clear
D)
1.5
done
clear
View Answer play_arrow
Two resistors A and B have resistances \[{{R}_{A}}\] and \[{{R}_{B}}\] respectively with \[{{R}_{A}}<{{R}_{B}}.\] The resistivities of their materials are \[{{\rho }_{A}}\] and \[{{\rho }_{B}}.\] Then
A)
\[{{\rho }_{A}}>{{\rho }_{B}}\]
done
clear
B)
\[{{\rho }_{A}}={{\rho }_{B}}\]
done
clear
C)
\[{{\rho }_{A}}<{{\rho }_{B}}\]
done
clear
D)
The information is not sufficient to find the relation between \[{{\rho }_{A}}\] and \[{{\rho }_{B}}\]
done
clear
View Answer play_arrow
The escape velocity for a body projected vertically upwards from the surface of earth is 11 km/s. If the body is projected at an angle of \[45{}^\circ \] with the vertical, the escape velocity will be
A)
\[22\,km/s\]
done
clear
B)
\[11\,km/s\]
done
clear
C)
\[\frac{11}{\sqrt{2}}\,km/s\]
done
clear
D)
\[11\sqrt{2}\,km/s\]
done
clear
View Answer play_arrow
Which of the following statements is incorrect regarding dehydrohalogenation of alkenes?
A)
During the reaction hydrogen atom is eliminated from the p - carbon atom.
done
clear
B)
Rate of reaction for same alkyl group; Iodine > Bromine > Chlorine
done
clear
C)
Rate of reaction; \[{{(C{{H}_{3}})}_{3}}C->{{(C{{H}_{3}})}_{2}}CH->C{{H}_{3}}C{{H}_{2}}-\]
done
clear
D)
Only nature of halogen atom determine rate of the reaction.
done
clear
View Answer play_arrow
Which of the following pairs of compounds are positional isomers?
A)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-\underset{\underset{O}{\mathop{\parallel }}\,}{\mathop{C}}\,-C{{H}_{3}}\] and \[C{{H}_{3}}-C{{H}_{2}}-\underset{\underset{O}{\mathop{\parallel }}\,}{\mathop{C}}\,-C{{H}_{2}}-C{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-CHO\] and \[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-\overset{O}{\mathop{\overset{\parallel }{\mathop{C}}\,}}\,-C{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-\underset{\underset{O}{\mathop{\parallel }}\,}{\mathop{C}}\,-C{{H}_{3}}\] and \[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,H}}\,-C{{H}_{2}}-CHO\]
done
clear
D)
\[C{{H}_{3}}-C{{H}_{2}}-\underset{\underset{O}{\mathop{\parallel }}\,}{\mathop{C}}\,-C{{H}_{2}}-C{{H}_{3}}\] and
done
clear
View Answer play_arrow
Which one of the following complexes will consume more equivalents of aqueous solution of \[AgN{{O}_{3}}?\]
A)
\[N{{a}_{2}}[CrC{{l}_{5}}({{H}_{2}}O)]\]
done
clear
B)
\[N{{a}_{3}}[CrC{{l}_{6}}]\]
done
clear
C)
\[[Cr{{({{H}_{2}}O)}_{5}}Cl]C{{l}_{2}}\]
done
clear
D)
\[[Cr{{({{H}_{2}}O)}_{6}}Cl]C{{l}_{3}}\]
done
clear
View Answer play_arrow
A)
A-(q), B-(s), C-(p), D-(r)
done
clear
B)
A-(r), B-(p), C-(s), D-(q)
done
clear
C)
A-(r), B-(s), C-(p), D-(q)
done
clear
D)
A-(s), B-(r), C-(q), D-(p)
done
clear
View Answer play_arrow
If 0.1 M of a weak acid is taken, and its percentage of degree of ionization is 1.34%, then its ionization constant will be :
A)
\[0.8\times {{10}^{-5}}\]
done
clear
B)
\[1.79\times {{10}^{-5}}\]
done
clear
C)
\[0.182\times {{10}^{-5}}\]
done
clear
D)
none of the above
done
clear
View Answer play_arrow
An organic compound X (molecular formula \[{{C}_{6}}{{H}_{7}}{{O}_{2}}N\]) has six carbon atoms in a ring system, two double bonds and a nitro group as substituent, X is :
A)
Homocyclic but not aromatic
done
clear
B)
Aromatic but not homocyclic
done
clear
C)
Homocyclic and aromatic
done
clear
D)
Heterocyclic and aromatic
done
clear
View Answer play_arrow
In which of the following regions hydrogen and helium are found:
A)
Stratosphere
done
clear
B)
Mesosphere
done
clear
C)
Exosphere
done
clear
D)
Troposphere
done
clear
View Answer play_arrow
Which of the following statements) is/are correct?
(i) The atomic and ionic radii of alkaline earth metals are smaller than those of the corresponding alkali metals in the same periods. (ii) Second ionisation enthalpies of the alkaline earth metals are smaller than those of the corresponding alkali metals. (iii) Compounds of alkaline earth metals are more extensively hydrated than those of alkali metals
A)
(i) and (ii)
done
clear
B)
(ii) and (iii)
done
clear
C)
(i) and (iii)
done
clear
D)
(i), (ii) and (iii)
done
clear
View Answer play_arrow
The chemical system that is non-aromatic is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Among the following compounds (I - III), the ease of their reaction with electrophiles is,
A)
II > III > I
done
clear
B)
III > II > I
done
clear
C)
II > I > III
done
clear
D)
I > II > III
done
clear
View Answer play_arrow
Haemoglobin contains 0.334%ofiron by weight. The molecular weight of haemoglobin is approximately 67200. The number of iron atoms (at. wt. of Fe is 56) present in one molecule of haemoglobin are
A)
1
done
clear
B)
6
done
clear
C)
4
done
clear
D)
2
done
clear
View Answer play_arrow
The cation that will not be precipitated by \[{{H}_{2}}S\] in the presence of dil. HCl is :
A)
\[P{{b}^{2+}}\]
done
clear
B)
\[C{{u}^{2+}}\]
done
clear
C)
\[C{{o}^{2+}}\]
done
clear
D)
done
clear
View Answer play_arrow
The correct order of thermal stability of hydroxides is:
A)
\[Ba{{(OH)}_{2}}<Ca{{(OH)}_{2}}<Sr{{(OH)}_{2}}<Mg{{(OH)}_{2}}\]
done
clear
B)
\[Mg{{(OH)}_{2}}<Sr{{(OH)}_{2}}<Ca{{(OH)}_{2}}<Ba{{(OH)}_{2}}\]
done
clear
C)
\[Mg{{(OH)}_{2}}<Ca{{(OH)}_{2}}<Sr{{(OH)}_{2}}<Ba{{(OH)}_{2}}\]
done
clear
D)
\[Ba{{(OH)}_{2}}<Sr{{(OH)}_{2}}<Ca{{(OH)}_{2}}<Mg{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
AB crystallizes in a body centred cubic lattice with edge length 'a' equal to 387 pm. The distance between two oppositely charged ions in the lattice is:
A)
335 pm
done
clear
B)
250 pm
done
clear
C)
200 pm
done
clear
D)
300 pm
done
clear
View Answer play_arrow
The reagent (s) which can be used to distinguish acetophenone from benzophenone is (are):
A)
\[2,\]4-Dinitrophenylhydrazine
done
clear
B)
Aqueous solution of \[NaHS{{O}_{3}}\]
done
clear
C)
Benedict reagent
done
clear
D)
\[{{I}_{2}}\]and \[NaOH.\]
done
clear
View Answer play_arrow
For the cell \[Zn|Z{{n}^{2+}}(1M)\parallel C{{u}^{2+}}(1M)|Cu,\]\[E_{cell}^{{}^\circ }\,is\,1.10\,V,\] \[E_{C{{u}^{2+}}/Cu}^{{}^\circ }=0.34\,V\] and for the cell \[Cu|C{{u}^{2+}}(1M)\parallel A{{g}^{+}}(1M)|Ag,\] \[E_{cell}^{{}^\circ }=0.46\,V\] hence, \[E_{cell}^{{}^\circ }\] of the cell \[Zn|Z{{n}^{2+}}(1M)\parallel A{{g}^{+}}(1M)|Ag\] is
A)
\[-0.04\,V\]
done
clear
B)
\[+0.04\,V\]
done
clear
C)
\[+0.30\,V\]
done
clear
D)
\[+1.56\,V\]
done
clear
View Answer play_arrow
Among the following complexes, optical activity is possible in
A)
\[{{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
done
clear
B)
\[{{[Co{{({{H}_{2}}O)}_{2}}{{(N{{H}_{3}})}_{2}}C{{l}_{2}}]}^{+}}\]
done
clear
C)
\[{{[Cr{{({{H}_{2}}O)}_{2}}C{{l}_{2}}]}^{+}}\]
done
clear
D)
\[[Co{{(CN)}_{5}}NC]\]
done
clear
View Answer play_arrow
The r.m.s velocity of hydrogen is \[\sqrt{7}\] times the r.m.s velocity of nitrogen. If T is the temperature of the gas , then
A)
\[{{T}_{({{H}_{2}})}}={{T}_{({{N}_{2}})}}\]
done
clear
B)
\[{{T}_{({{H}_{2}})}}>{{T}_{({{N}_{2}})}}\]
done
clear
C)
\[{{T}_{({{H}_{2}})}}<{{T}_{({{N}_{2}})}}\]
done
clear
D)
\[{{T}_{({{H}_{2}})}}=\sqrt{7}{{T}_{({{N}_{2}})}}\]
done
clear
View Answer play_arrow
The correct order of ionic radii of \[{{Y}^{3+}},\]\[L{{a}^{3+}},\]\[E{{u}^{3+}}\] and \[L{{u}^{3+}}\] is
A)
\[{{Y}^{3+}}<L{{a}^{3+}}<E{{u}^{3+}}<L{{u}^{3+}}\]
done
clear
B)
\[L{{u}^{3+}}<E{{u}^{3+}}<L{{a}^{3+}}<{{Y}^{3+}}\]
done
clear
C)
\[L{{a}^{3+}}<E{{u}^{3+}}<L{{u}^{3+}}<{{Y}^{3+}}\]
done
clear
D)
\[{{Y}^{3+}}<L{{u}^{3+}}<E{{u}^{3+}}<L{{a}^{3+}}\]
done
clear
View Answer play_arrow
The\[p{{K}_{a}}\]of an amino acid is 9.15. At what pH amino acid is 5% dissociated?
A)
9.15
done
clear
B)
4.85
done
clear
C)
9.44
done
clear
D)
7.87
done
clear
View Answer play_arrow
In the froth floatation process for the purification of ores, the ore particles float because :
A)
They are light.
done
clear
B)
Their surface is hydrophobic i.e. not easily wetted by water.
done
clear
C)
They bear electrostatic charge.
done
clear
D)
They are insoluble.
done
clear
View Answer play_arrow
\[{{T}_{50}}\] of first -order reaction is 10 min. Starting with \[10\,mol\,{{L}^{-1}}\] rate after 20 min is
A)
\[0.0693\,mol\,{{L}^{-1}}\,{{\min }^{-1}}\]
done
clear
B)
\[0.0693\times 2.5\,mol\,{{L}^{-1}}\,{{\min }^{-1}}\]
done
clear
C)
\[0.0693\times 5\,mol\,{{L}^{-2}}\,{{\min }^{-1}}\]
done
clear
D)
\[0.0693\times 10\,mol\,{{L}^{-1}}\,{{\min }^{-1}}\]
done
clear
View Answer play_arrow
Which of the following organometallic compound is \[\sigma \] and \[\pi \] bonded?
A)
\[[Fe{{({{\eta }^{5}}-{{C}_{5}}{{H}_{5}})}_{2}}]\]
done
clear
B)
\[Fe{{(C{{H}_{3}})}_{3}}\]
done
clear
C)
\[K[PtC{{l}_{3}}({{\eta }^{2}}-{{C}_{2}}{{H}_{4}})]\]
done
clear
D)
\[{{[Co{{(CO)}_{5}}N{{H}_{3}}]}^{2+}}\]
done
clear
View Answer play_arrow
Which of the following statement is false?
A)
For 1 mole of an ideal gas, \[{{C}_{p}}-{{C}_{v}}=R\]
done
clear
B)
\[{{\left( \frac{\partial E}{\partial T} \right)}_{T}}=0\] for an ideal gas
done
clear
C)
\[\Delta q=\Delta w+P\Delta V\]
done
clear
D)
For reversible isothermal expansion of 1 mole of an ideal gas from volume \[{{V}_{1}}\] to \[{{V}_{2}},\]work done is equal to RT In\[({{V}_{2}}/{{V}_{1}})\]
done
clear
View Answer play_arrow
The correct order of the decreasing ionic radii among the following isoelectronic species are :
A)
\[C{{a}^{2+}}>{{K}^{+}}>{{S}^{2-}}>C{{l}^{-}}\]
done
clear
B)
\[C{{l}^{-}}>{{S}^{2-}}>C{{a}^{2+}}>{{K}^{+}}\]
done
clear
C)
\[{{S}^{2-}}>C{{l}^{-}}>{{K}^{+}}>C{{a}^{2+}}\]
done
clear
D)
\[{{K}^{+}}>C{{a}^{2+}}>C{{l}^{-}}>{{S}^{2-}}\]
done
clear
View Answer play_arrow
A compound A with molecular formula \[{{C}_{10}}{{H}_{13}}Cl\] gives a white precipitate on adding silver nitrate solution. A on reacting with alcoholic KOH gives compound B as the main product. B on ozonolysis gives C and D. C gives Cannizzaro reaction but not aldol condensation. D gives aldol condensation but not Cannizzaro reaction. A is:
A)
\[{{C}_{6}}{{H}_{5}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-Cl\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}-C{{H}_{2}}-C{{H}_{2}}-\underset{Cl}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{3}}\]
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
\[Sn{{O}_{2}}\] is taken in basic medium and current is passed. Colloidal sol migrates towards
A)
anode (\[+\] plate)
done
clear
B)
cathode (\[-\] plate)
done
clear
C)
both [a] and [b]
done
clear
D)
none of these
done
clear
View Answer play_arrow
0.45 g of acid of molecular weight 90 was neutralized by 20 ml. of a 0.5N caustic potash. The basicity of an acid is
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Which of the following is a broad spectrum drug?
A)
Plasmoquine
done
clear
B)
Chloroquine
done
clear
C)
Chloramphenicol
done
clear
D)
D.D.T.
done
clear
View Answer play_arrow
A compound on treatment with NaOH followed by addition of \[Ag\,N{{O}_{3}}\]produces white precipitate at room temperature. The precipitate is soluble in \[N{{H}_{4}}OH.\] The compound is identified as
A)
vinyl chloride
done
clear
B)
benzyl chloride
done
clear
C)
chlorobenzene
done
clear
D)
ethyl bromide
done
clear
View Answer play_arrow
Which of the following statements(s) is/are incorrect?
(i) Only \[1/{{8}^{th}}\] portion of an atom located at comer of a cubic unit cell is its neighbouring unit cell. (ii) Total number of atoms per unit cell for a face centered cubic unit cell is 3. (iii) Atom located at the body center is shared between two adjacent unit cells.
A)
(iii) only
done
clear
B)
(ii) only
done
clear
C)
(i) and (ii)
done
clear
D)
(ii) and (iii)
done
clear
View Answer play_arrow
Equivalent conductance of an electrolyte containing NaF at infinite dilution is \[90.1\,Oh{{m}^{-1}}c{{m}^{2}}.\] If NaF is replaced by KF what is the value of equivalent conductance?
A)
\[90.1\,Oh{{m}^{-1}}c{{m}^{2}}\]
done
clear
B)
\[111.2\,Oh{{m}^{-1}}c{{m}^{2}}\]
done
clear
C)
\[0\]
done
clear
D)
\[222.4\,Oh{{m}^{-1}}c{{m}^{2}}\]
done
clear
View Answer play_arrow
The enthalpy of combustion of \[{{C}_{6}}{{H}_{6}}\] is -3250 kJ, when 0.39 gm of \[{{C}_{6}}{{H}_{6}}\] is burnt in excess of oxygen in an open vessel, the amount of heat evolved is
A)
8.32 kJ
done
clear
B)
12.36 kJ
done
clear
C)
16.25 kJ
done
clear
D)
20.74 kJ
done
clear
View Answer play_arrow
In which of the following is there a consistent decrease in atomic radius as the atomic number increases?
A)
halogens
done
clear
B)
representative elements
done
clear
C)
transition elements
done
clear
D)
lanthanides
done
clear
View Answer play_arrow
The basicity of aniline is less than that of cyclohexylamine. This is due to
A)
\[+\]R effect of \[-\]\[N{{H}_{2}}\] group
done
clear
B)
\[-\]1 effect of \[-\]\[N{{H}_{2}}\] group
done
clear
C)
\[-\]R effect of \[-\]\[N{{H}_{2}}\] group
done
clear
D)
hyperconjugation effect
done
clear
View Answer play_arrow
Given \[F{{e}^{3+}}(aq)+{{e}^{-}}\to F{{e}^{2+}}(aq);\]\[E{}^\circ =+0.77\,V\] \[A{{l}^{3+}}(aq)+3{{e}^{-}}\to Al(s);\]\[E{}^\circ =-1.66\,V\] \[B{{r}_{2}}(aq)+2{{e}^{-}}\to 2b{{r}^{-}};\,\,E{}^\circ =+1.09\,V\] Considering the electrode potentials, which of the following represents the correct order of reducing power?
A)
\[F{{e}^{2+}}<Al<B{{r}^{-}}\]
done
clear
B)
\[B{{r}^{-}}<F{{e}^{2+}}<Al\]
done
clear
C)
\[Al<B{{r}^{-}}<F{{e}^{2+}}\]
done
clear
D)
\[Al<F{{e}^{2+}}<B{{r}^{-}}\]
done
clear
View Answer play_arrow
Mac Arthur process is used for the extraction of:
A)
Au
done
clear
B)
Pt
done
clear
C)
Cu
done
clear
D)
Zn
done
clear
View Answer play_arrow
\[{{H}_{3}}B{{O}_{2}}\]is
A)
monobasic and weak Lewis acid
done
clear
B)
monobasic and weak Bronsted acid
done
clear
C)
monobasic and strong Lewis acid
done
clear
D)
tribasic and weak Bronsted acid
done
clear
View Answer play_arrow
The following equilibrium constants are given: \[{{N}_{2}}+3{{H}_{2}}2N{{H}_{3}};\]\[{{K}_{1}}\] \[{{N}_{2}}+{{O}_{2}}2NO;\]\[{{K}_{2}}\] \[{{H}_{2}}+\frac{1}{2}{{O}_{2}}{{H}_{2}}O;\]\[{{K}_{2}}\] The equilibrium constant for the oxidation of \[N{{H}_{3}}\] by oxygen to give NO is
A)
\[\frac{{{K}_{2}}K_{3}^{2}}{{{K}_{1}}}\]
done
clear
B)
\[\frac{K_{2}^{2}{{K}_{3}}}{{{K}_{1}}}\]
done
clear
C)
\[\frac{{{K}_{1}}{{K}_{2}}}{{{K}_{3}}}\]
done
clear
D)
\[\frac{{{K}_{2}}K_{3}^{3}}{{{K}_{1}}}\]
done
clear
View Answer play_arrow
The ortho/para directing group among the following is:
A)
\[COOH\]
done
clear
B)
\[CN\]
done
clear
C)
\[COC{{H}_{3}}\]
done
clear
D)
\[NHCON{{H}_{2}}\]
done
clear
View Answer play_arrow
Benzoic acid gives benzene on being heated with X and phenol gives benzene on being heated with Y. Therefore X and Y are respectively
A)
Soda-lime and copper
done
clear
B)
Zn dust and NaOH
done
clear
C)
Zn dust and soda-lime
done
clear
D)
Soda-lime and zinc dust.
done
clear
View Answer play_arrow
The enthalpies of the following reactions are shown below. \[\frac{1}{2}{{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\to OH(g);\] \[\Delta H=42.09\,kJ\,mo{{l}^{-1}}\] \[{{H}_{2}}(g)\to 2H(g);\]\[\Delta H=435.89\,kJ\,mo{{l}^{-1}}\] \[{{O}_{2}}(g)\to 2O(g);\]\[\Delta H=495.05\,kJ\,mo{{l}^{-1}}\] Calculate the \[O-H\] bond energy for the hydroxyl radical.
A)
\[223.18\,kJ\,mo{{l}^{-1}}\]
done
clear
B)
\[423.38\,kJ\,mo{{l}^{-1}}\]
done
clear
C)
\[513.28\,kJ\,mo{{l}^{-1}}\]
done
clear
D)
\[113.38\,kJ\,mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
Elements X, Y and Z have atomic numbers 19, 37 and 55 respectively. Which of the following statements is true about them?
A)
Their ionization potential would increase with increasing atomic number
done
clear
B)
'Y' would have an ionisation potential between those of 'X' and 'Z'
done
clear
C)
'Z' would have the highest ionization potential
done
clear
D)
'Y' would have the highest ionization potential.
done
clear
View Answer play_arrow
\[Mn{{O}_{4}}^{2-}\] (1mole) in neutral aqueous medium disproportionates to
A)
2/3 mole of \[Mn{{O}_{4}}^{-}\] and 1 /3 mole of \[Mn{{O}_{2}}\]
done
clear
B)
113 mole of \[Mn{{O}_{4}}^{-}\] and 2/3 mole of \[Mn{{O}_{2}}\]
done
clear
C)
1/3 mole of \[M{{n}_{2}}{{O}_{7}}\] and 1/3 mole of \[Mn{{O}_{2}}\]
done
clear
D)
2/3 mole of \[M{{n}_{2}}{{O}_{7}}\] and 1/3 mole of \[Mn{{O}_{2}}\]
done
clear
View Answer play_arrow
Formation of a solution from two components can be considered as
(i) pure solvent \[\to \] separated solvent molecules,\[\Delta {{H}_{1}}\] (ii) pure solute \[\to \] separated solute molecules,\[\Delta {{H}_{2}}\] (iii) separated solvent and solute molecules\[\to \] solution,\[\Delta {{H}_{3}}\] Solution so formed will be ideal if
A)
\[\Delta {{H}_{\text{soln}}}=\Delta {{H}_{1}}+\Delta {{H}_{2}}-\Delta {{H}_{3}}\]
done
clear
B)
\[\Delta {{H}_{\text{soln}}}=\Delta {{H}_{1}}-\Delta {{H}_{2}}-\Delta {{H}_{3}}\]
done
clear
C)
\[\Delta {{H}_{\text{soln}}}=\Delta {{H}_{3}}-\Delta {{H}_{1}}-\Delta {{H}_{2}}\]
done
clear
D)
\[\Delta {{H}_{\text{soln}}}=\Delta {{H}_{1}}+\Delta {{H}_{2}}+\Delta {{H}_{3}}\]
done
clear
View Answer play_arrow
Match column-I (Biological name) with column- II (Class) and choose the correct option.
Column-I (Biological name) Column-II (Class) A. Homo sapiens I. Dicotyledonae B. Musca domestica II. Mammalia C. Mangifera indica III. Monocotyledonae D. Triticum IV. Insects
A)
A-IV; B-II; C-I; D-III
done
clear
B)
A-II; B-IV; C-III; D-I
done
clear
C)
A-II; B-IV; C-I; D-III
done
clear
D)
A-II; B-I; C-IV; D-III
done
clear
View Answer play_arrow
Which meristem helps in increasing girth?
A)
Lateral meristem
done
clear
B)
Intercalary meristem
done
clear
C)
Primary meristem
done
clear
D)
Apical meristem
done
clear
View Answer play_arrow
A human bone marrow cell, in prophase of mitosis, contains 46 chromosomes. How many chromatids does it contain altogether?
A)
46
done
clear
B)
92
done
clear
C)
23
done
clear
D)
23 or 46
done
clear
View Answer play_arrow
In Chlorophyceae, sexual reproduction occurs by
A)
isogamy and anisogamy
done
clear
B)
isogamy, anisogamy and oogamy
done
clear
C)
oogamy only
done
clear
D)
anisogamy and oogamy
done
clear
View Answer play_arrow
Yeast is not included in protozoans but are placed fungi because
A)
it has no chlorophyll.
done
clear
B)
some fungal hyphae grow in such a way that they give the appearance of pseudomycelium.
done
clear
C)
it has eukaryotic organization.
done
clear
D)
cell wall is made up of cellulose and reserve food material is starch.
done
clear
View Answer play_arrow
According to widely accepted "fluid mosaic model" cell membranes are semi-fluid, where lipids and integral proteins can diffuse randomly. In recent years, this model has been modified in several respects. In this regard, which of the following statements is incorrect?
A)
Proteins in cell membranes can travel within the lipid bilayer.
done
clear
B)
Proteins can also undergo flip-flop movements in the lipid bilayer.
done
clear
C)
Proteins can remain confined within certain domains of the membrane.
done
clear
D)
Many proteins remain completely embedded within the lipid bilayer.
done
clear
View Answer play_arrow
Which one of the following animals is correctly matched with its particular named taxonomic category?
A)
Tiger - tigris, the species
done
clear
B)
Cuttle fish - mollusca, a class
done
clear
C)
Humans - primata, the family
done
clear
D)
Housefly - musca, an order
done
clear
View Answer play_arrow
The endosperm of gymnosperm is
A)
triploid
done
clear
B)
haploid
done
clear
C)
diploid
done
clear
D)
polyploidy
done
clear
View Answer play_arrow
Which of the following statement is incorrect about emasculation?
A)
During emasculation process, stigma is removed.
done
clear
B)
Emasculated flowers are bagged in order to prevent self-pollination.
done
clear
C)
Emasculation is the removal of stamens before the maturation of selected bisexual flowers.
done
clear
D)
It is one of the steps for artificial hybridization.
done
clear
View Answer play_arrow
Which one of the following statements is correct for secondary succession?
A)
It begins on a bare rock
done
clear
B)
It occurs on a deforested site
done
clear
C)
It follows primary succession
done
clear
D)
It is similar to primary succession except that it has a relatively fast pace
done
clear
View Answer play_arrow
Identified A, B, C and D.
A)
A - Mesocarp; B - Endocarp; C - Seed; D-Thalamus
done
clear
B)
A - Seed; B - Thalamus; C - Mesocarp; D - Endocarp
done
clear
C)
A - Thalamus; B - Seed; C - Endocarp; D - Mesocarp
done
clear
D)
A - Mesocarp; B - Endocarp; C - Seed; D -Thalamus
done
clear
View Answer play_arrow
Syngamy means
A)
fusion of similar spores.
done
clear
B)
fusion of gametes.
done
clear
C)
fusion of dissimilar spores.
done
clear
D)
fusion of cytoplasm.
done
clear
View Answer play_arrow
Biochemical Oxygen Demand (BOD) in a river water
A)
has no relationship with concentration of oxygen in the water.
done
clear
B)
gives a measure of Salmonella in the water.
done
clear
C)
increases when sewage gets mixed with river water.
done
clear
D)
remains unchanged when algal bloom occurs.
done
clear
View Answer play_arrow
A bract can be differentiated from bracteole by its:
A)
Size
done
clear
B)
Position
done
clear
C)
Function
done
clear
D)
Colour
done
clear
View Answer play_arrow
The parenchyma tissue which forms the bulk of ovule where the sporogenous tissue is produced is-
A)
Megaspore mother cell
done
clear
B)
Nucellus
done
clear
C)
Ovule
done
clear
D)
Embryo sac
done
clear
View Answer play_arrow
Which of the following mineral is associated with the characters/functions given below?
(i) Helps in formation of middle lamella. (ii) Needed in mitotic spindle formation. (iii) Accumulates in older leaves. (iv) Involves in normal functioning of the cell membranes. (v) Activate certain enzymes. (vi) Plays an important role in regulating metabolic activities.
A)
\[{{K}^{+}}\]
done
clear
B)
\[F{{e}^{3+}}\]
done
clear
C)
\[N{{O}^{3-}}\]
done
clear
D)
\[C{{a}^{2+}}\]
done
clear
View Answer play_arrow
Movement of ions or molecules in a direction opposite to that of prevailing electrochemical gradient is known as
A)
diffusion
done
clear
B)
active transport
done
clear
C)
pinocytosis
done
clear
D)
brownian movement
done
clear
View Answer play_arrow
Which one is the correct summary equation of photosynthesis?
A)
\[{{C}_{6}}{{H}_{12}}{{O}_{6}}+6{{O}_{2}}\xrightarrow[{}]{}6C{{O}_{2}}+6{{H}_{2}}O+energy\]
done
clear
B)
\[{{C}_{6}}{{H}_{12}}{{O}_{6}}+6{{O}_{2}}+6{{H}_{2}}O\xrightarrow{{}}6C{{O}_{2}}+12{{H}_{2}}O+energy\]
done
clear
C)
\[6C{{O}_{2}}+6{{H}_{2}}O\xrightarrow{{}}6{{H}_{2}}O+{{C}_{6}}{{H}_{12}}{{O}_{6}}\]
done
clear
D)
\[6C{{O}_{2}}+12{{H}_{2}}O\xrightarrow[Chlorophyll]{Light}6{{O}_{2}}+{{C}_{6}}{{H}_{12}}{{O}_{6}}+6{{H}_{2}}O\]
done
clear
View Answer play_arrow
Which one is a long day plant?
A)
Tobacco
done
clear
B)
Glycinemax
done
clear
C)
Mirabilis jalapa
done
clear
D)
Spinach
done
clear
View Answer play_arrow
Opium is obtained from
A)
Papaver somniferum
done
clear
B)
Rauwolfia serpentine
done
clear
C)
Cannabis sativus
done
clear
D)
Claviceps purpurea
done
clear
View Answer play_arrow
Which of the following statements concerning rubisco are froe.
A)
Rubisco is an enzyme
done
clear
B)
Rubisco catalyzes both the beginning steps of photorespiration and the Calvin-Benson cycle.
done
clear
C)
Rubisco is the most abundant protein on earth
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Turgor pressure become equal to the wall pressure when
A)
water leaves the cell
done
clear
B)
water enters the cell
done
clear
C)
no exchange of water takes place
done
clear
D)
solute goes from cell into water
done
clear
View Answer play_arrow
The figure given below shows the conversion of a substrate into product by an enzyme. In which one of the four options (a-d) the components or reaction labelled as A, B, C and D are identified correctly?
A)
A-Potential energy, B-Transition state, C-Activation energy with enzyme, D-Activation energy without enzyme
done
clear
B)
A-Transition state, B-Potential energy, C-Activation energy without enzyme, D-Activation energy with enzyme
done
clear
C)
A-Potential energy, B-Transition state, C-Activation energy with enzyme, D-Activation energy with enzyme
done
clear
D)
A-Activation energy with enzyme, B-Transition state, C-Activation energy without enzyme, D-Potential energy
done
clear
View Answer play_arrow
Which of the following statements about Rhizobium legume nodule formation is not true?
A)
Rhizobium can only fix nitrogen after it becomes a bacteroid within a root cortex cell.
done
clear
B)
Rhizobium induces invagination of root hairs.
done
clear
C)
Within an infection thread, Rhizobium is still extracellular to the plant.
done
clear
D)
The infection thread can fuse with any root cell of an appropriate legume species.
done
clear
View Answer play_arrow
Which of the following statement is incorrect?
A)
\[{{C}_{3}}\] plants respond to higher temperature, show higher photosynthetic rate while \[{{C}_{4}}\] plants have lower optimum temperature.
done
clear
B)
Tropical plants have higher temperature optimum than the plants adapted to temperate climate.
done
clear
C)
Light reaction is less temperature sensitive than dark reaction.
done
clear
D)
The effect of water as a factor is more through its effect on plant, rather than directly on photosynthesis.
done
clear
View Answer play_arrow
Nucleotide arrangement in DNA can be seen by
A)
X-ray crystallography
done
clear
B)
electron microscope
done
clear
C)
ultracentrifuge
done
clear
D)
light microscope
done
clear
View Answer play_arrow
Genetic map is one that
A)
shows the distribution of various species in a region
done
clear
B)
establishes sites of the genes on a chromosome
done
clear
C)
establishes the various stages in gene evolution
done
clear
D)
show the stages during the cell division
done
clear
View Answer play_arrow
DNA template sequence of CTGATAGC is transcribed over mRNA as
A)
GUCTUTCG
done
clear
B)
GACUAUCG
done
clear
C)
GAUTATUG
done
clear
D)
UACTATCU
done
clear
View Answer play_arrow
Humus is essential for plant growth because:
A)
it is rich in nutrients and increases the water holding capacity of soil
done
clear
B)
it increases aeration of soil
done
clear
C)
it increases porocity of soil
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Which one of the following pesticides is banned now a-days?
A)
DDT
done
clear
B)
Eldrin
done
clear
C)
Aldrin
done
clear
D)
Toxaphene
done
clear
View Answer play_arrow
Ozone hole means
A)
hole in the ozone layer in stratosphere.
done
clear
B)
decrease in the thickness of ozone in the stratosphere.
done
clear
C)
decrease in concentration of ozone in trophosphere.
done
clear
D)
increase in the concentration of ozone in trophosphere.
done
clear
View Answer play_arrow
The new varieties of plants are produced by
A)
selection and hybridization
done
clear
B)
mutation and selection
done
clear
C)
introduction and mutation
done
clear
D)
selection and introduction
done
clear
View Answer play_arrow
A functional piece of mRNA has 66 codons. What is the maximum number of amino acids that could be present in the protein coded for by this mRNA?
A)
22
done
clear
B)
64
done
clear
C)
65
done
clear
D)
66
done
clear
View Answer play_arrow
What is the optimum pH for growth in most bacteria?
A)
2.5 - 3.5
done
clear
B)
3.5 - 4.5
done
clear
C)
4.5 - 5.5
done
clear
D)
6.5. - 7.5
done
clear
View Answer play_arrow
The following diagram is the polynucleotide chain. Identify A, B, C, D and E.
A)
A - Hydrogen bonds, B - Pyrimidine, C - Hexose (deoxyribose) sugar, D - 5' end, E - Purine base.
done
clear
B)
A - Hydrogen bonds, B - Purine base, C - Hexose (deoxyribose) sugar, D - 5' end, E - Pyrimidine.
done
clear
C)
A - Hydrogen bonds, B - Pyrimidine, C - Pentose (deoxyribose) sugar, D - 5' end, E - Purine base
done
clear
D)
A - Hydrogen bonds, B - Purine base, C - Pentose (deoxyribose) sugar, D - 5' end, E - Pyrimidine
done
clear
View Answer play_arrow
Waves of muscle contractions that move the intestinal content are:
A)
caused by contraction of skeletal muscle.
done
clear
B)
regulated by liver secretions.
done
clear
C)
called peristalsis.
done
clear
D)
voluntary.
done
clear
View Answer play_arrow
The method that renders the seed coat permeable to water so that embryo expansion is not physically retarded is
A)
vernalization
done
clear
B)
stratification
done
clear
C)
denudation
done
clear
D)
scarification
done
clear
View Answer play_arrow
In Kreb's cycle, the FAD precipitates as electron acceptor during the conversion of
A)
succinyl CoA to succinic acid
done
clear
B)
\[\alpha \]- ketoglutarate to succinyl CoA
done
clear
C)
fumaric acid to malic acid
done
clear
D)
succinic acid to fumaric acid
done
clear
View Answer play_arrow
In the DNA molecule:
A)
there are two strands which run antiparallel- one in 5'\[\to \]3' direction and other in 3'\[\to \]5' direction.
done
clear
B)
the total amount of purine nucleotides and pyrimidine nucleotides is not always equal.
done
clear
C)
there are two strands which run parallel in the 5'\[\to \]3' direction.
done
clear
D)
the proportion of Adenine in relation to thymine varies with the organism.
done
clear
View Answer play_arrow
Dormant lettuce seeds were exposed to the following succession of brief periods of red light (R) and far- red light (FR) light: FR-R-FR-R-FR-R-FR Which of the following is most likely to occur?
A)
Most of the seeds will germinate
done
clear
B)
Most of the seeds will not germinate
done
clear
C)
About one-half will germinate
done
clear
D)
The seeds will germinate but development will cease unless the epicotyl is exposed to red light.
done
clear
View Answer play_arrow
Which of the following would generally be considered the healthiest habitat?
A)
A sink habitat populated by many endemic species
done
clear
B)
A source habitat populated by many endemic species
done
clear
C)
A sink habitat populated by many exotic species
done
clear
D)
A source habitat populated by many exotic species
done
clear
View Answer play_arrow
Which one of the following help in absorption of phosphorus from soil by plants?
A)
Glomus
done
clear
B)
Rhizobium
done
clear
C)
Frankia
done
clear
D)
Anabaena
done
clear
View Answer play_arrow
The area where wild populations, traditional life styles and genetic resources are protected is :
A)
Core Zone
done
clear
B)
Buffer Zone
done
clear
C)
Biosphere reserve
done
clear
D)
Manipulation Zone
done
clear
View Answer play_arrow
The figure below is the diagrammatic representation of the E.coli vector pBR 322. Which one of the given options correctly identifies its certain component (s)?
A)
ori-original restriction enzyme
done
clear
B)
rop-reduced osmotic pressure
done
clear
C)
Hind HI, EcoRI-selectable markers
done
clear
D)
\[am{{p}^{R}},\]\[te{{t}^{R}}\]-antibiotic resistance genes
done
clear
View Answer play_arrow
Which one of the following option is correct for A, B, C and D?
A)
A - Exonuclease, B - Endonuclease, C - DNA ligase, D - Transformation
done
clear
B)
A - Exonuclease, B - Exonuclease, C - DNA ligase, D - Transformation
done
clear
C)
A - Exonuclease, B - Endonuclease, C - Hydrolase, D - Transduction
done
clear
D)
A - Restriction endonuclease, B - Restriction endonuclease, C - DNA ligase, D - transformation
done
clear
View Answer play_arrow
Which of the following groups of plants play an important role in plant succession on bare rocks/ soil?
A)
Algae
done
clear
B)
Bryophytes
done
clear
C)
Pteridophytes
done
clear
D)
Gymnosperms
done
clear
View Answer play_arrow
Which of the following cations is required for the conversion of prothrombin into active thrombin by thromboplastin?
A)
\[C{{u}^{2+}}\]
done
clear
B)
\[F{{e}^{3+}}\]
done
clear
C)
\[F{{e}^{2+}}\]
done
clear
D)
\[C{{a}^{2+}}\]
done
clear
View Answer play_arrow
Which one of the following statement regarding coelom of given animals is correct?
A)
Round worms (aschelminthes) are pseudocoelomates.
done
clear
B)
Molluscs are acoelomates.
done
clear
C)
Insects are pseudocoelomates.
done
clear
D)
Flatworms (platyhelminthes) are coelomates.
done
clear
View Answer play_arrow
Lymph differs from blood in
A)
absence of RBC
done
clear
B)
absence of WBC
done
clear
C)
excess of water
done
clear
D)
absence of protein
done
clear
View Answer play_arrow
Hardening of the arteries due to deposition of cholesterol is called
A)
thrombosis
done
clear
B)
atherosclerosis
done
clear
C)
rhinitis
done
clear
D)
angina
done
clear
View Answer play_arrow
Which one of the following is NOT a characteristic of phylum Annelida?
A)
Closed circulatory system
done
clear
B)
Segmentation
done
clear
C)
Pseudocoelom
done
clear
D)
Ventral nerve cord
done
clear
View Answer play_arrow
Fish which can be used in biological control of mosquitoes/Larvicidal fish is
A)
Eel
done
clear
B)
Carp
done
clear
C)
Cat Fish
done
clear
D)
Gambusia
done
clear
View Answer play_arrow
A person which shows the secondary sexual characters of both male and female is called -
A)
Intersex
done
clear
B)
Hermaphrodite
done
clear
C)
Bisexual
done
clear
D)
Gynandromorph
done
clear
View Answer play_arrow
Amniocentesis is a technique
A)
by which the essential amino acids in the body can be estimated
done
clear
B)
by which any chromosomal anomalies in the foetus can be detected
done
clear
C)
in which the sex of the foetus can be reversed
done
clear
D)
that can be used for correcting genetic disorders of the foetus
done
clear
View Answer play_arrow
The diagram shows an antibody molecule. Identify A top.
A)
A - Antigen binding site; B - Variable region (of L-Chain); C - Constant region (of L-Chain); D - Light polypeptide chain (L-Chain); E - Heavy polypeptide chain (H-Chain); F - Disulfide (bond)
done
clear
B)
A - Antigen binding site; B - Constant region (of L-Chain); C - Variable region (of L-Chain); D - Light polypeptide chain (L-Chain); E - Heavy polypeptide chain (H-Chain); F - Disulfide (bond)
done
clear
C)
A - Antigen binding site; B - Variable region (of L-Chain); C - Constant region (of L-Chain); D - Heavy polypeptide chain (L-Chain); E - Light polypeptide chain (H-Chain); F - Hydrogen (bond)
done
clear
D)
A - Antigen binding site; B - Variable region (of L-Chain); C - Constant region (of L-Chain); D - Light polypeptide chain (L-Chain); E - Heavy polypeptide chain (H-Chain); F - Hydrogen (bond)
done
clear
View Answer play_arrow
The copper ions of IUDs
A)
Suppress the motility and fertilization capacity of sperms
done
clear
B)
Make the uterus unsuitable for implantation
done
clear
C)
Increase phagocytosis of sperms
done
clear
D)
Make cervix hostile to sperms
done
clear
View Answer play_arrow
The transfer of zygote or early embryo (up to 8 blastomere) into fallopian tube is
A)
IVF and ET
done
clear
B)
ZIFT
done
clear
C)
GIFT
done
clear
D)
IUT
done
clear
View Answer play_arrow
The embryonic membrane involved in the formation of placenta in human is
A)
yolk sac
done
clear
B)
allantois
done
clear
C)
amnion
done
clear
D)
chorion
done
clear
View Answer play_arrow
Identical twins are produced when
A)
One fertilized egg divides and two blastomeres separate
done
clear
B)
One sperm fertilizes two eggs
done
clear
C)
One egg is fertilized with two sperms
done
clear
D)
Two eggs are fertilized
done
clear
View Answer play_arrow
Which of the following induces parturition?
A)
Vasopressin
done
clear
B)
Oxytocin
done
clear
C)
Growth hormone
done
clear
D)
Thyroid stimulating hormone
done
clear
View Answer play_arrow
Jaundice is a disorder of
A)
Excretory system
done
clear
B)
Skin and eyes
done
clear
C)
Digestive system
done
clear
D)
Circulatory system
done
clear
View Answer play_arrow
Child death may occur in the marriage between
A)
\[R{{h}^{+}}\] man and \[R{{h}^{+}}\] woman
done
clear
B)
\[R{{h}^{+}}\] man and \[R{{h}^{-}}\] woman
done
clear
C)
\[R{{h}^{-}}\] man and \[R{{h}^{-}}\] woman
done
clear
D)
\[R{{h}^{-}}\] man and \[R{{h}^{+}}\] woman
done
clear
View Answer play_arrow
Continued consumption of a diet rich in butter, red meat and eggs for a long period may lead to
A)
vitamin A toxicity
done
clear
B)
kidney stones
done
clear
C)
hypercholesterolemia
done
clear
D)
urine laden with ketone bodies
done
clear
View Answer play_arrow
A person who is one along hunger strike and is surviving only on water, will have
A)
less amino acids in his urine
done
clear
B)
more glucose in his blood
done
clear
C)
less urea in his urine
done
clear
D)
more sodium in his urine
done
clear
View Answer play_arrow
Haemoglobin is having maximum affinity with:
A)
\[N{{H}_{3}}\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[C{{O}_{2}}\]
done
clear
D)
\[CO\]
done
clear
View Answer play_arrow
Which one of the following is a matching pair?
A)
Lubb-Sharp closure of AV valves at the beginning of ventricular systole
done
clear
B)
Dup-Sudden opening of semilunar valves at the beginning of ventricular diastole
done
clear
C)
Pulsation of the radial artery-Valves in the blood vessels
done
clear
D)
Initiation of the heart beat-Purkinje fibres
done
clear
View Answer play_arrow
Which region of the kidney nephron is the main site of amino acid reabsorption?
A)
glomerulus
done
clear
B)
Bowman's capsule
done
clear
C)
proximal convoluted tubule
done
clear
D)
distal convoluted tubule
done
clear
View Answer play_arrow
Both cri du chat syndrome and Down's syndrome
A)
are X-linked recessive genetic disorders.
done
clear
B)
result from inherited chromosomal abnormalities.
done
clear
C)
result from the loss of one entire chromo- some.
done
clear
D)
result from the gain of one entire chromo- some.
done
clear
View Answer play_arrow
In livestock breeding experiments the following stage is transferred to surrogate mothers
A)
Unfertilized eggs
done
clear
B)
Fertilized eggs
done
clear
C)
8 to 32 celled embryo
done
clear
D)
Frozen semen
done
clear
View Answer play_arrow
The presence of gill slits, in the embryos of all vertebrates, supports the theory of
A)
biogenesis
done
clear
B)
recapitulation
done
clear
C)
metamorphosis
done
clear
D)
organic evolution
done
clear
View Answer play_arrow
Which of the following has been recently used for increasing productivity of super milk cows?
A)
Artificial insemination by a pedigreed bull only
done
clear
B)
Superovulation of a high production cow only
done
clear
C)
Embryo transplantation only
done
clear
D)
A combination of Superovulation, artificial insemination and embryo transplantation into a 'carrier cow' (surrogate mother)
done
clear
View Answer play_arrow
The change of the light-coloured variety of peppered moth (Biston betularia) to its darker variety (Biston carbonaria) is due to
A)
mutation
done
clear
B)
regeneration
done
clear
C)
genetic isolation
done
clear
D)
temporal isolation
done
clear
View Answer play_arrow
A person with the sex chromosomes XXY suffers from
A)
Down's syndrome
done
clear
B)
Klinefelter's syndrome
done
clear
C)
Turner's syndrome
done
clear
D)
Gynandromorphism
done
clear
View Answer play_arrow
Productivity at the second trophic level is always
A)
greater than the productivity at the first trophic level.
done
clear
B)
less than the productivity at the first trophic level.
done
clear
C)
equal to the productivity at the first trophic level.
done
clear
D)
extremely variable compared to the productivity at the first trophic level.
done
clear
View Answer play_arrow
The density of a population in a given habitat during a given period, fluctuates due to changes in four basic process. On this basis fill up A and B boxes in the given diagram with correct options
A)
A = Natality + Immigration, B = Mortality + Emigration
done
clear
B)
A = Natality + Mortality, B = Immigration + Emigration
done
clear
C)
A = Birth rate + Death rate, B= Migration+ Emigration
done
clear
D)
A = Natality + Emigration, B = Mortality + Immigration
done
clear
View Answer play_arrow
Two of the body parts which do not appear in MRI may be
A)
molar teeth and eye lens
done
clear
B)
scapula and canines
done
clear
C)
ligaments and ribs
done
clear
D)
tendons and premolars
done
clear
View Answer play_arrow
Hormones generally cause a response in a cell by
A)
interacting directly with the cell's DNA.
done
clear
B)
binding with a receptor and stimulating protein production.
done
clear
C)
changing the polarity of the cell membrane and causing a cascade of events within the cell.
done
clear
D)
halting all other cellular activity except the required response
done
clear
View Answer play_arrow
The largest quantity of air that can be expired after a maximal inspiratory effort is called
A)
residual volume
done
clear
B)
tidal volume
done
clear
C)
vital capacity
done
clear
D)
total lung volume
done
clear
View Answer play_arrow
The black pigment in the eye which reduces the internal reflection is located in
A)
retina
done
clear
B)
iris
done
clear
C)
cornea
done
clear
D)
sclerotic
done
clear
View Answer play_arrow
Which one of the following does NOT change in length when a muscle fibre contracts?
A)
I band
done
clear
B)
H zone
done
clear
C)
A band
done
clear
D)
Sarcomere
done
clear
View Answer play_arrow
Which of the following correctly describes the role of myelin in the nervous system?
A)
Myelin insulates and speeds transmission along dendrites.
done
clear
B)
Myelin insulates and speeds transmission along axons.
done
clear
C)
Myelin slows down depolarization of an axon.
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Following feedback relationship is seen between three glands and their hormones.
The secretion of 'b is decreased by 'a', secretion of 'c' is increased by 'V while the secretion of 'a' is decreased by 'c'. Increased or decreased amounts of a hormone cause an increase or decrease in the amount of the following hormone in the system. Now if 'a' is added to the system, the secretion of 'c' is
A)
decreased
done
clear
B)
increased
done
clear
C)
first decreased and then increased
done
clear
D)
first increased and then decreased
done
clear
View Answer play_arrow
Match the following (w.r.t. insert size)
A. Plasmid (i) 9 - 23 kb B. \[\lambda \]phage (ii) 0.5 - 8 kb C. Cosmid (iii) 30 - 40 kb D. BAC (iv) 50 - 300 kb
A)
A-(ii), B-(i), C-(iii), D-(iv)
done
clear
B)
A-(i), B-(ii), C-(iii), D-iv)
done
clear
C)
A-(ii), B-(i), C-(iv), D-(iii)
done
clear
D)
A-(iii), B-(i), C-(ii), D-(iv)
done
clear
View Answer play_arrow
Which one of the following pairs is incorrect?
A)
Plasmid - small piece of extrachromosomal DNA in bacteria
done
clear
B)
Interferon - an enzyme that interferes with DNA replication
done
clear
C)
Cosmid - A vector for carrying large DNA fragments into host cells
done
clear
D)
Myeloma - antibody producing tumor cells.
done
clear
View Answer play_arrow
In which one of the following pairs of diseases both are caused by viruses?
A)
Tetanus and typhoid
done
clear
B)
Whooping cough and sleeping sickness
done
clear
C)
Syphilis and AIDS
done
clear
D)
Measles and Rabies
done
clear
View Answer play_arrow
Electroporation procedure involves
A)
fast passage of food through sieve pores in phloem elements with the help of electric stimulation.
done
clear
B)
opening of stomatal pores during night by artificial light.
done
clear
C)
making transient pores in the cell membrane to introduce gene constructs.
done
clear
D)
purification of saline water with the help of a membrane system.
done
clear
View Answer play_arrow
Black water fever is a severe complication of
A)
Plasmodium falcifarum
done
clear
B)
P. vivax
done
clear
C)
P. malariae
done
clear
D)
All the above
done
clear
View Answer play_arrow
Increased asthmatics attacks in certain seasons are related to
A)
eating fruits preserved in tin containers
done
clear
B)
inhalation of seasonal pollen
done
clear
C)
low temperature
done
clear
D)
hot and humid environment.
done
clear
View Answer play_arrow
For transformation, micro-particles coated with DNA to be bombarded with gene gun are made up of:
A)
Silver or Platinum
done
clear
B)
Platinum or Zinc
done
clear
C)
Silicon or Platinum
done
clear
D)
Gold or Tungsten
done
clear
View Answer play_arrow
Which one of the following statements is correct with respect to AIDS?
A)
The HIV can be transmitted through eating food together with an infected person
done
clear
B)
Drug addicts are least susceptible to HIV infection.
done
clear
C)
AIDS patients are being fully cured with proper care and nutrition
done
clear
D)
The causative HIV retro-virus enters helper T-lymphocytes thus reducing their numbers.
done
clear
View Answer play_arrow

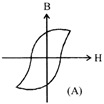
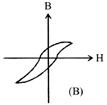 These materials are used to make magnets for elecric generators, transformer core and electromagnet core. Then it is proper to use:
These materials are used to make magnets for elecric generators, transformer core and electromagnet core. Then it is proper to use: 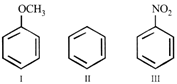
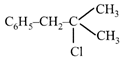
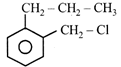
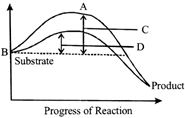
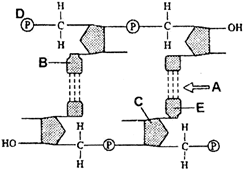
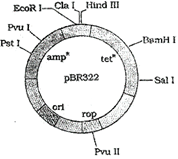
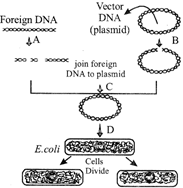
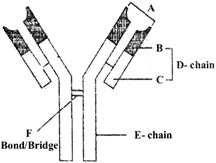

 The secretion of 'b is decreased by 'a', secretion of 'c' is increased by 'V while the secretion of 'a' is decreased by 'c'. Increased or decreased amounts of a hormone cause an increase or decrease in the amount of the following hormone in the system. Now if 'a' is added to the system, the secretion of 'c' is
The secretion of 'b is decreased by 'a', secretion of 'c' is increased by 'V while the secretion of 'a' is decreased by 'c'. Increased or decreased amounts of a hormone cause an increase or decrease in the amount of the following hormone in the system. Now if 'a' is added to the system, the secretion of 'c' is 