question_answer 1) The electrical permittivity and magnetic permeability of free space are respectively \[{{\varepsilon }_{0}}\]and \[{{\mu }_{0}}\]The corresponding value for a medium is :
A)
\[{{\tan }^{-1}}(5)\]
done
clear
B)
\[{{\tan }^{-1}}\left( \frac{1}{5} \right)\]
done
clear
C)
\[{{\sin }^{-1}}\left( \frac{1}{5} \right)\]
done
clear
D)
\[2.2\times {{10}^{3}}N\]
done
clear
View Answer play_arrow
question_answer 2) The frequency of a light ray is \[3.2\times {{10}^{3}}N\]Hz. Its frequency when it propagates in a medium of refractive index 1.5, will be :
A)
\[4.2\times {{10}^{3}}N\]
done
clear
B)
\[5.2\times {{10}^{3}}N\]
done
clear
C)
\[{{10}^{2}}\]
done
clear
D)
\[{{10}^{-2}}\]
done
clear
View Answer play_arrow
question_answer 3) In a Youngs experiment, if the distance between the slits is halved and the distance of screen from the slit is doubled, the fringe width will be :
A)
four times
done
clear
B)
doubled
done
clear
C)
halved
done
clear
D)
same
done
clear
View Answer play_arrow
question_answer 4) The magnetic moment of a magnet of length 10 cm and pole strong the 4.0 Am will be :
A)
\[{{10}^{-3}}\]
done
clear
B)
\[{{10}^{-4}}\]
done
clear
C)
\[J\]
done
clear
D)
\[\frac{I}{\omega }\]
done
clear
View Answer play_arrow
question_answer 5) Two point charges \[I{{\omega }^{2}}\]repel each other by a force of \[I\omega \]. If each of them is given an additional charge \[4\sqrt{3}\], the force between them will be :
A)
\[\frac{T}{\sqrt{3}}\]N(attractive)
done
clear
B)
\[\frac{T}{3}\] (attractive)
done
clear
C)
\[\frac{\sqrt{3}}{2}T\] (repulsive)
done
clear
D)
\[\sqrt{3}T\](attractive)
done
clear
View Answer play_arrow
question_answer 6) A man of weight 80 kg is standing in an elevator which is moving with an acceleration of 6 \[1.6\times {{10}^{-19}}\]in up ward direction. The apparent weight of the man will be (g = 10 \[1.12\times {{10}^{-19}}\] ) :
A)
1480 N
done
clear
B)
1280 N
done
clear
C)
1380 N
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 7) The power of pump, which can pump 200 kg of water to a height of 50 m in 10sec, will be :
A)
\[1.76\times {{10}^{-19}}\]
done
clear
B)
\[{{T}_{1}}k\]
done
clear
C)
\[{{T}_{2}}k\]
done
clear
D)
\[\mu \]
done
clear
View Answer play_arrow
question_answer 8) What is the value of linear velocity, if angular velocity is \[3\overset{\hat{\ }}{\mathop{i}}\,-4\overset{\hat{\ }}{\mathop{j}}\,+\overset{\hat{\ }}{\mathop{k}}\,\] and distance from the centre is \[5\overset{\hat{\ }}{\mathop{i}}\,-6\overset{\hat{\ }}{\mathop{j}}\,+6\overset{\hat{\ }}{\mathop{k}}\,\]
A)
\[6\overset{\hat{\ }}{\mathop{i}}\,+6\overset{\hat{\ }}{\mathop{j}}\,-3\overset{\hat{\ }}{\mathop{k}}\,\]
done
clear
B)
\[18\overset{\hat{\ }}{\mathop{i}}\,+13\overset{\hat{\ }}{\mathop{j}}\,-2\overset{\hat{\ }}{\mathop{k}}\,\]
done
clear
C)
\[4\overset{\hat{\ }}{\mathop{i}}\,-13\overset{\hat{\ }}{\mathop{j}}\,+6\overset{\hat{\ }}{\mathop{k}}\,\]
done
clear
D)
\[6\overset{\hat{\ }}{\mathop{i}}\,-2\overset{\hat{\ }}{\mathop{j}}\,+8\overset{\hat{\ }}{\mathop{k}}\,\]
done
clear
View Answer play_arrow
question_answer 9) Magnetic permeability is maximum for :
A)
paramagnetic substances
done
clear
B)
ferromagnetic substances
done
clear
C)
diamagnetic substances
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 10) An electron moves in a circular path of radius 15 cm in a magnetic field or\[2316.4\overset{0}{\mathop{A}}\,,0.1\times {{10}^{7}}{{m}^{-1}}\] tesla. The speed of electron will be :
A)
\[10\times {{10}^{5}}kWh\]
done
clear
B)
\[4\times {{10}^{-5}}kg\]
done
clear
C)
\[3\times {{10}^{-5}}kg\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 11) The effective length of a magnet is 31.4 cm and its pole strength is 0.5 Am. The magnetic moment, if it is bent in the form of a semicircle will be :
A)
0.1 \[5\times {{10}^{-5}}kg\]
done
clear
B)
0.01 \[8\times {{10}^{-5}}kg\]
done
clear
C)
0.2 \[{{n}^{2}}\]
done
clear
D)
1.2 \[\frac{1}{{{n}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 12) A magnet makes 30 oscillations per minute in the earths magnetic field. If the magnetic field is doubled. The period of oscillation of magnet is :
A)
\[{{n}^{3}}\]
done
clear
B)
2 s
done
clear
C)
\[\frac{1}{n}\]
done
clear
D)
\[{{10}^{7}}N/{{m}^{2}}\]
done
clear
View Answer play_arrow
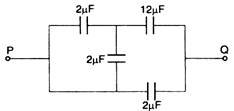
question_answer 13)
Four capacitors are connected in a circuit as shown in figure. The effective capacitance in fi F between P and Q will be :
A)
5 \[2.5\times {{10}^{6}}N/{{m}^{2}}\]
done
clear
B)
10 \[{{s}^{2}}\]
done
clear
C)
2 \[{{s}^{2}}\]
done
clear
D)
7.5 \[{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 14) If you are provided three resistances 2\[8.2\times {{10}^{6}}\], 3\[h=6.6\times {{10}^{-34}}Js\] and 6\[c=3\times {{10}^{8}}m/s\]. How will you connect them so as to obtain the equivalent resistance of 4\[e=1.6\times {{10}^{-19}}C\]:
A)
done
clear
B)
done
clear
C)
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 15) When the resistance of 9\[\overset{0}{\mathop{A}}\,\] is connected at the ends of a battery, its potential difference decreases from 40 volt to 30 volt. The internal resistance of the battery is:
A)
6\[\overset{0}{\mathop{A}}\,\]
done
clear
B)
3\[\overset{0}{\mathop{A}}\,\]
done
clear
C)
9\[\overset{0}{\mathop{A}}\,\]
done
clear
D)
15\[\left[ {{M}^{0}}{{L}^{0}}T{{A}^{0}} \right]\]
done
clear
View Answer play_arrow
question_answer 16) When a shunt of resistance 12\[\left[ M{{L}^{0}}T{{A}^{0}} \right]\] is connected with a galvanometer, its deflection reduces from 50 div to 10 div. The resistance of the coil of galvanometers :
A)
48\[\left[ M{{L}^{0}}T{{A}^{2}} \right]\]
done
clear
B)
24\[\left[ M{{L}^{0}}T{{A}^{-2}} \right]\]
done
clear
C)
56\[y=5\sin 2\pi \left( \frac{t}{0.04}-\frac{x}{40} \right)\]
done
clear
D)
36\[c=\frac{1}{\sqrt{{{\mu }_{0}}{{\varepsilon }_{0}}}}\]
done
clear
View Answer play_arrow
question_answer 17) When the current flowing in a circular coil is doubled and the number of turns of the coil in it is halved, the magnetic field at its centre will become :
A)
four times
done
clear
B)
same
done
clear
C)
half
done
clear
D)
double
done
clear
View Answer play_arrow
question_answer 18) A proton is moving with a velocity v is subjected to an electric field E and magnetic field B. The proton will remain moving without any deflection, if :
A)
E and B both are parallel to v
done
clear
B)
E is parallel to v but perpendicular to B
done
clear
C)
E is perpendicular to B
done
clear
D)
E, v and B are mutually perpendicular to each other and \[\upsilon =\frac{1}{\sqrt{\mu \varepsilon }}\]
done
clear
View Answer play_arrow
question_answer 19) In an electric heater 4 amp current passes for 1 minute at potential difference of 250 volt, the power of heater and energy consumed will be respectively :
A)
1 kvs 60 kj
done
clear
B)
0.5 kw, 30 kj
done
clear
C)
10 kw, 600 kj
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 20) Magnetic flux in a circuit containing a coil of resistance 2\[\upsilon =\frac{1}{\sqrt{\mu \varepsilon }}\] changes from 2.0 wb to 10 wb in 0.2 sec. The charge passed through the coil in this time is :
A)
0.8 C
done
clear
B)
1.0 C
done
clear
C)
5.0 C
done
clear
D)
4.0 C
done
clear
View Answer play_arrow
question_answer 21) In the DC circuit, the current is expressed as\[\sqrt{\frac{{{\mu }_{0}}{{\varepsilon }_{0}}}{\mu \varepsilon }}\]. In this circuit the current rises from zero to peak value in time :
A)
\[\sqrt{\frac{{{\mu }_{0}}{{\varepsilon }_{0}}}{\mu \varepsilon }}\]
done
clear
B)
\[\sqrt{\frac{\mu \varepsilon }{{{\mu }_{0}}{{\varepsilon }_{0}}}}\]
done
clear
C)
\[\sqrt{\frac{{{\mu }_{0}}{{\varepsilon }_{0}}}{\mu \varepsilon }}\]
done
clear
D)
\[6\times {{10}^{16}}\]
done
clear
View Answer play_arrow
question_answer 22) In a LR circuit \[1.67\times {{10}^{14}}Hz\]and \[9.10\times {{10}^{14}}Hz\]. If the circuit has an alternating emf of 220 volt 50 cycles per sec. The impedance and current in the circuit will be :
A)
50\[6\times {{10}^{14}}Hz\], 4.4A
done
clear
B)
40.4\[4\times {{10}^{14}}Hz\], 5A
done
clear
C)
3.07\[0.4A{{m}^{2}}\], 6.0A
done
clear
D)
11.4\[1.6A{{m}^{2}}\], 17.5A
done
clear
View Answer play_arrow
question_answer 23) An electron initially at rest, is accelerated through a potential difference of 200 volt, so that it acquires a velocity\[20A{{m}^{2}}\] The value of e/m of electron will be :
A)
\[8.0A{{m}^{2}}\]
done
clear
B)
\[3\times {{10}^{-6}}C\]
done
clear
C)
\[6\times {{10}^{-3}}N\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 24) Work function of lithium and copper are respectively 2.3 eV and 4.0 eV. which one of the metal will be useful for the photoelectric cell working with visible light? \[-6\times {{10}^{6}}C,\]
A)
Lithium
done
clear
B)
Copper
done
clear
C)
Both
done
clear
D)
None
done
clear
View Answer play_arrow
question_answer 25) A proton and an \[\text{ }\!\!\alpha\!\!\text{ -}\]particle are accelerated through the same potential difference. The ratio of de-Broglie wavelength of proton to the de-Broglie wavelength of alpha particle will be :
A)
\[2.4\times {{10}^{-3}}\]
done
clear
B)
1 : 2
done
clear
C)
2 :1
done
clear
D)
1 : 1
done
clear
View Answer play_arrow
question_answer 26) The heat of 110 J is added to a gaseous system whose internal energy is 40 J, then the amount of external work done will be :
A)
140 J
done
clear
B)
70 J
done
clear
C)
40 J
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 27) Two rods having thermal conductivity in the ratio of 5 : 3 having equal lengths and equal cross-sectional area are joined by face to face. If the temperature of the free end of the first rod is \[100{}^\circ C\] and free end of second rod is \[20{}^\circ C\]. Then the temperature of the junction is :
A)
\[70{}^\circ C\]
done
clear
B)
\[50{}^\circ C\]
done
clear
C)
\[60{}^\circ C\]
done
clear
D)
\[90{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 28) A bomber plane is moving horizontally with a speed of 500 m/s and a bomb released from it, strikes the ground in 10 sec. Angle at which the bomb strikes the ground is, (g = 10 \[2.4\times {{10}^{-9}}N\]) :
A)
\[1.5\times {{10}^{-3}}N\]
done
clear
B)
\[1.5\times {{10}^{-3}}N\]
done
clear
C)
\[m/{{s}^{2}}\]
done
clear
D)
\[m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 29) The average force necessary to stop a bullet of 20 kg at speed of 250 m/s as it penetrates wood to a distance of 12 cm is :
A)
\[10\times {{10}^{3}}watt\]
done
clear
B)
\[20\times {{10}^{3}}watt\]
done
clear
C)
\[4\times {{10}^{3}}watt\]
done
clear
D)
\[6\times {{10}^{3}}watt\]
done
clear
View Answer play_arrow
question_answer 30) A black body is heated from \[27{}^\circ C\] to \[927{}^\circ C\]. The ratio of radiation emitted will be :
A)
1 : 256
done
clear
B)
1 : 64
done
clear
C)
1 : 4
done
clear
D)
1 : 16
done
clear
View Answer play_arrow
question_answer 31) Density of substance at \[0{}^\circ C\] is 10 g/cc and at \[100{}^\circ C\], its density is 9.7 g/cc. The coefficient of linear expansion of the substance is :
A)
\[3\hat{i}-4\hat{j}+\hat{k}\]
done
clear
B)
\[5\hat{i}-6\hat{j}+6\hat{k}\]
done
clear
C)
\[6\hat{i}-6\hat{j}+3\hat{k}\]
done
clear
D)
\[18\hat{i}-13\hat{j}+2\hat{k}\]
done
clear
View Answer play_arrow
question_answer 32) Angular momentum \[4\hat{i}-13\hat{j}+6\hat{k}\]of body with mass moment of inertia I and angular velocity ad/sec is equal to :
A)
\[6\hat{i}-2\hat{j}+8\hat{k}\]
done
clear
B)
\[4\times {{10}^{-4}}\]
done
clear
C)
\[1.07\times {{10}^{7}}m/s\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 33) The horizontal range of projectile is\[0.53\times {{10}^{7}}m/s\] times of its maximum height. The angle of projection will be :
A)
\[40{}^\circ C\]
done
clear
B)
\[90{}^\circ C\]
done
clear
C)
\[30{}^\circ C\]
done
clear
D)
\[45{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 34) At what temperature, the speed of sound in air will become double of its value at \[27{}^\circ C\] :
A)
\[927{}^\circ C\]
done
clear
B)
\[327{}^\circ C\]
done
clear
C)
\[54{}^\circ C\]
done
clear
D)
\[627{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 35) The efficiency of Carnots engine operating between reservoirs, maintained at temperatures \[27{}^\circ C\] and \[-123{}^\circ C\], is :
A)
50%
done
clear
B)
24%
done
clear
C)
0.75%
done
clear
D)
0.4%
done
clear
View Answer play_arrow
question_answer 36) The period of a simple pendulum measured inside a stationary lift is found to be T. If the lift starts accelerating upwards with acceleration of g/3, then the time period of the pendulum is :
A)
\[1.53\times {{10}^{7}}m/s\]
done
clear
B)
\[A{{m}^{2}}\]
done
clear
C)
\[A{{m}^{2}}\]
done
clear
D)
\[A{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 37) In germanium crystal, the forbidden energy gap in joule is :
A)
zero
done
clear
B)
\[A{{m}^{2}}\]
done
clear
C)
\[\sqrt{2}s\]
done
clear
D)
\[\frac{1}{\sqrt{2}}s\]
done
clear
View Answer play_arrow
question_answer 38) A semiconductor is cooled from \[\frac{1}{2}s\] to \[\mu F\], then its resistance :
A)
will not change
done
clear
B)
will increase
done
clear
C)
will decrease
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 39) The current gain of a transistor in common emitter mode is 49. The change in collector current and emitter current corresponding to the change in base current by 5.0 \[\mu F\]A, will be :
A)
\[\mu F\]
done
clear
B)
\[\mu F\]
done
clear
C)
\[\Omega \]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 40) If the wavelength of 1st line of Balmer series is 6563\[\Omega \]. The wavelength of first line of Lyman series and Rydbergs constant respectively, will be :
A)
\[\Omega \]
done
clear
B)
\[\Omega \]
done
clear
C)
\[\Omega \]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 41) The half-life of a radioactive substance is 40 years. How long will it take to reduce to one fourth of its original amount and what is the value of decay constant?
A)
40 year, 0.9173/year
done
clear
B)
90 year, 9.017/year
done
clear
C)
80 year, 0.0173/year
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 42) The mass equivalent to \[\Omega \]energy will be :
A)
\[\Omega \]
done
clear
B)
\[\Omega \]
done
clear
C)
\[\Omega \]
done
clear
D)
\[\Omega \]
done
clear
View Answer play_arrow
question_answer 43) Which one of the relation is correct between time period and number of orbits while an electron is revolving in a orbit?
A)
\[\Omega \]
done
clear
B)
\[\Omega \]
done
clear
C)
\[\Omega \]
done
clear
D)
\[\Omega \]
done
clear
View Answer play_arrow
question_answer 44) A cylinder contains 10 kg of gas at pressure of \[v=\frac{E}{B}\] , the quantity of gas taken out of the cylinder if final pressure is\[\Omega \], is :
A)
7.5 kg
done
clear
B)
15.2 kg
done
clear
C)
10.5 kg
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 45) A body of mass 1 kg tied to one end of string is revolved in a horizontal circle of radius 0.1 m with a speed of 3 revolution/sec, assuming the effect of gravity is negligible, then linear velocity, acceleration and tension in the string will be :
A)
1.88 m/s, 35.5 m/\[I=100\sin 200\pi t\], 35.5 N
done
clear
B)
2.88 m/s, 45.5 m/\[\frac{1}{400}s\], 45.5N
done
clear
C)
3.88 m/s, 55.5 m/\[\frac{1}{300}s\], 55.5N
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 46) If a source is transmitting electromagnetic waves of frequency \[\frac{1}{100}s\]Hz, the wavelength of electromagnetic wave transmitted from the source is :
A)
18.8 m
done
clear
B)
35.6 m
done
clear
C)
42.8 m
done
clear
D)
58 m
done
clear
View Answer play_arrow
question_answer 47) An X-ray tube operates on 30 kV. The minimum wavelength emitted is,\[\frac{1}{200}s\],\[L=\frac{0.4}{\pi }\]\[R=30\Omega \] :
A)
6.6 \[\Omega \]
done
clear
B)
1.2\[\Omega \]
done
clear
C)
0.133 \[\Omega \]
done
clear
D)
0.4 \[\Omega \]
done
clear
View Answer play_arrow
question_answer 48) In a nuclear reactor :
A)
moderator is used to slow down the neutrons
done
clear
B)
cadmium rods are used to slow down the neutrons
done
clear
C)
coolant is used to slow down the neutrons
done
clear
D)
moderator is used to control the neutrons
done
clear
View Answer play_arrow
question_answer 49) If C and R denote capacitance and resistance, then dimensions of CR will be :
A)
\[8.4\times {{10}^{6}}m/s.\]
done
clear
B)
\[2.76\times {{10}^{12}}C/kg\]
done
clear
C)
\[1.76\times {{10}^{11}}C/kg\]
done
clear
D)
\[0.76\times {{10}^{12}}C/kg\]
done
clear
View Answer play_arrow
question_answer 50) If the equation of transverse wave\[(h=6.6\times {{10}^{-34}}J.s,c=3\times {{10}^{8}}m/s)\]where distance is in cm and time in sec, then the wavelength of wave will be :
A)
20 cm
done
clear
B)
40 cm
done
clear
C)
60 cm
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 51) The colour of liquid \[{{O}_{2}}\] is:
A)
red
done
clear
B)
dark blue
done
clear
C)
pale yellow
done
clear
D)
pale blue
done
clear
View Answer play_arrow
question_answer 52) Ripening of fruits can be carried out in presence of:
A)
\[N{{a}_{2}}S{{O}_{4}}\]
done
clear
B)
\[NaCl\]
done
clear
C)
\[CaC{{l}_{2}}\]
done
clear
D)
\[Ca{{C}_{2}}\]
done
clear
View Answer play_arrow
question_answer 53) p-nitrophenol and o-nitrophenol are separated by:
A)
crystallization
done
clear
B)
fractional crystallization
done
clear
C)
distillation
done
clear
D)
steam distillation
done
clear
View Answer play_arrow
question_answer 54) Lead is maximum in:
A)
soda glass
done
clear
B)
jena glass
done
clear
C)
pyrex glass
done
clear
D)
flint glass
done
clear
View Answer play_arrow
question_answer 55) Which of the following is not an antibiotic?
A)
Penicillin
done
clear
B)
Sulphaguanidine
done
clear
C)
Chloramphenical
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 56) Which of the following salt undergoes hydrolysis?
A)
\[N{{a}_{2}}S{{O}_{4}}\]
done
clear
B)
\[N{{H}_{4}}Cl\]
done
clear
C)
\[NaCl\]
done
clear
D)
\[CaC{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 57) On hydrolysis, which produces only glucose?
A)
Galactose
done
clear
B)
Maltose
done
clear
C)
Sucrose
done
clear
D)
None
done
clear
View Answer play_arrow
question_answer 58) Shortest \[C-C\] bond length is present in:
A)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\]
done
clear
C)
\[C{{H}_{2}}=CH-CH=C{{H}_{2}}\]
done
clear
D)
\[CH\equiv C-C\equiv CH\]
done
clear
View Answer play_arrow
question_answer 59)
\[C{{H}_{3}}-\underset{OH}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-C{{H}_{3}}\xrightarrow{PB{{r}_{3}}}\]
A)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-O-C{{H}_{2}}-C{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}-O-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-C{{H}_{2}}C{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-C{{H}_{2}}C{{H}_{2}}OH\]
done
clear
D)
\[C{{H}_{3}}-C{{H}_{2}}-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 60) In the reaction,\[HAs{{O}_{4}}+S{{n}^{2+}}\xrightarrow{{}}As+S{{n}^{4+}}+{{H}_{2}}O\]oxidising agent is:
A)
\[S{{n}^{2+}}\]
done
clear
B)
\[S{{n}^{4+}}\]
done
clear
C)
\[As\]
done
clear
D)
\[HAs{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 61) Noble gases are absorbed on :
A)
anhydrous \[CaC{{l}_{2}}\]
done
clear
B)
charcoal
done
clear
C)
cone. \[{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
coconut
done
clear
View Answer play_arrow
question_answer 62) Ammonia and \[HCl\] gases are allowed to pass in the glass tube from the opposite ends. Formation of \[N{{H}_{4}}Cl\] white ring, will take place:
A)
nearer the \[N{{H}_{3}}\] end
done
clear
B)
at the centre
done
clear
C)
nearer \[HCl\] end
done
clear
D)
anywhere
done
clear
View Answer play_arrow
question_answer 63) 0.833 mole of a carbohydrate, with empirical formula \[C{{H}_{2}}O\], has 10g of hydrogen. Molecular formula of carbohydrate is:
A)
\[{{C}_{3}}{{H}_{5}}{{O}_{3}}\]
done
clear
B)
\[{{C}_{6}}{{H}_{12}}{{O}_{6}}\]
done
clear
C)
\[{{C}_{3}}{{H}_{10}}{{O}_{5}}\]
done
clear
D)
\[{{C}_{3}}{{H}_{4}}{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 64) \[{{P}_{4}}+3NaOH+3{{H}_{2}}O\xrightarrow{{}}A+3Na{{H}_{2}}P{{O}_{2}}\]here, A is:
A)
\[N{{H}_{3}}\]
done
clear
B)
\[P{{H}_{3}}\]
done
clear
C)
\[{{H}_{3}}P{{O}_{4}}\]
done
clear
D)
\[{{H}_{3}}P{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 65) Solubility of \[Pb{{I}_{2}}\]. is 0.005 M. Then, the solubility product of \[Pb{{I}_{2}}\] is:
A)
\[6.8\times {{10}^{-6}}\]
done
clear
B)
\[6.8\times {{10}^{6}}\]
done
clear
C)
\[2.2\times {{10}^{-9}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 66) The number of \[C{{l}^{-}}\] ions around one \[N{{a}^{+}}\] in \[NaCl\]I crystal lattice is:
A)
\[10\]
done
clear
B)
\[12\]
done
clear
C)
\[8\]
done
clear
D)
\[8\]
done
clear
View Answer play_arrow
question_answer 67) Which element is found in chlorophyll?
A)
Iron
done
clear
B)
Cobalt
done
clear
C)
Magnesium
done
clear
D)
Chromium
done
clear
View Answer play_arrow
question_answer 68) Which of the following is not correct for carboxylic acids?
A)
The lower acids are soluble in water
done
clear
B)
Higher members are insoluble in water
done
clear
C)
The solubility increases with branching
done
clear
D)
They unite in water and take proton from solvent
done
clear
View Answer play_arrow
question_answer 69) A chemical compound is heated and dried. To get accurate weight of this compound, one should:
A)
cool in dessicator
done
clear
B)
weigh in warm conditions
done
clear
C)
weigh after cooling
done
clear
D)
blow air through it
done
clear
View Answer play_arrow
question_answer 70) Isocyanide can be prepared from alkyi halide on treatment with:
A)
\[KNC\]
done
clear
B)
\[KgNC\]
done
clear
C)
\[KCN\]
done
clear
D)
\[AgCN\]
done
clear
View Answer play_arrow
question_answer 71) What is obtained, when acetic acid is heated with chlorine in presence of red phosphorus?
A)
Acetyl chloride
done
clear
B)
a-chloro acetic acid
done
clear
C)
Trichloro acetic acid
done
clear
D)
Chloral
done
clear
View Answer play_arrow
question_answer 72) Which of the following has maximum boiling point?
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Cl\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\]
done
clear
C)
\[\underset{OH}{\mathop{\underset{|}{\mathop{C{{H}_{3}}CHC{{H}_{3}}}}\,}}\,\]
done
clear
D)
\[\underset{OH}{\mathop{\underset{|}{\mathop{C{{H}_{2}}}}\,}}\,-C{{H}_{2}}-C{{H}_{2}}-\underset{OH}{\mathop{\underset{|}{\mathop{C{{H}_{2}}}}\,}}\,\]
done
clear
View Answer play_arrow
question_answer 73) The number of oxygen atoms in 44g \[C{{O}_{2}}\] at S.T.P. is:
A)
\[6.023\times {{10}^{23}}\]
done
clear
B)
\[6.023\times {{10}^{22}}\]
done
clear
C)
\[12.046\times {{10}^{23}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 74) Weight of \[Ca{{(OH)}_{2}}\] needed to prepare 250 ml of solution with pH =13:
A)
\[0.225g\]
done
clear
B)
\[0.0125\text{ }g\]
done
clear
C)
\[0.25\text{ }g\]
done
clear
D)
\[1\text{ }g\]
done
clear
View Answer play_arrow
question_answer 75) \[2C+{{O}_{2}}\xrightarrow{{}}2CO;\,\Delta H=220kJ\]Which of the following statement is correct for this reaction?
A)
Heat of combustion of carbon is 110 kJ
done
clear
B)
Reaction is exothermic
done
clear
C)
Reaction needs no initiation
done
clear
D)
All of these are correct
done
clear
View Answer play_arrow
question_answer 76) Maximum bond angle is present in :
A)
\[BC{{l}_{3}}\]
done
clear
B)
\[BB{{r}_{3}}\]
done
clear
C)
\[B{{F}_{3}}\]
done
clear
D)
same for all
done
clear
View Answer play_arrow
question_answer 77) \[{{H}_{2}}S\] is more acidic than \[{{H}_{2}}O\], due to:
A)
O is more electronegative than S
done
clear
B)
\[O-H\] bond is stronger than \[S-H\] bond
done
clear
C)
\[O-H\] bond is weaker than \[S-H\] bond
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 78) Entropy is maximum in case of:
A)
steam
done
clear
B)
water at \[{{0}^{o}}C\]
done
clear
C)
water at \[{{4}^{o}}C\]
done
clear
D)
ice
done
clear
View Answer play_arrow
question_answer 79) What will be the volume of the mixture after the reaction? \[\underset{4\,litre}{\mathop{N{{H}_{3}}}}\,+\underset{1.5\,litre}{\mathop{HCl}}\,\xrightarrow{{}}\underset{(soilid)}{\mathop{N{{H}_{4}}Cl}}\,\]
A)
0.5 litre
done
clear
B)
1 litre
done
clear
C)
2.5 litre
done
clear
D)
0.1 litre
done
clear
View Answer play_arrow
question_answer 80) Co-ordination number of \[N{{a}^{+}}\] ion in rock salt is:
A)
\[12\]
done
clear
B)
\[4\]
done
clear
C)
\[8\]
done
clear
D)
\[6\]
done
clear
View Answer play_arrow
question_answer 81) For nitration in benzene, we require :
A)
cone. \[HN{{O}_{3}}\]+ cone. \[{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
cone. \[HN{{O}_{3}}\]
done
clear
C)
cone. \[{{H}_{2}}S{{O}_{4}}\]+ fuming \[HN{{O}_{3}}\]
done
clear
D)
\[KN{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 82) What will be the volume of a 12M solution, if it is equivalent to 240 ml 18 M solution?
A)
6 litre ,
done
clear
B)
600 litre
done
clear
C)
400 litre
done
clear
D)
0.36 litre
done
clear
View Answer play_arrow
question_answer 83) Which of the following is diamagnetic?
A)
Oxygen molecule
done
clear
B)
Boron molecule
done
clear
C)
\[N_{2}^{+}\]
done
clear
D)
None
done
clear
View Answer play_arrow
question_answer 84) Hydrolysis of DDT is a first order reaction, its halt life is 10 years. Time to hydrolyse 10 g DDT to half is:
A)
100 years
done
clear
B)
50 years
done
clear
C)
5 years
done
clear
D)
10 years
done
clear
View Answer play_arrow
question_answer 85) \[C{{r}_{2}}O_{7}^{2-}+{{I}^{-}}\xrightarrow{{}}{{I}_{2}}+C{{r}^{3+}}\]\[{{E}^{o}}_{cell}=0.79V\]\[{{E}^{o}}_{C{{r}_{2}}O_{7}^{2-}}=1.33V,\,{{E}^{o}}_{{{I}_{2}}}\]is:
A)
\[-0.10V\]
done
clear
B)
\[+0.18V\]
done
clear
C)
\[-0.54V\]
done
clear
D)
\[0.54V\]
done
clear
View Answer play_arrow
question_answer 86) \[Zn(s)+C{{l}_{2}}(1atm)\xrightarrow{{}}Z{{n}^{2+}}+2C{{l}^{-}}\].\[{{E}^{o}}_{cell}\] of the cell is 2.12 V. To increase E :
A)
\[[Z{{n}^{2+}}]\] should be increased
done
clear
B)
\[[Z{{n}^{2+}}]\] should be decreased
done
clear
C)
\[[C{{l}^{-}}]\] should be decreased
done
clear
D)
\[{{P}_{C{{l}_{2}}}}\] should be decreased
done
clear
View Answer play_arrow
question_answer 87) Which will have enantiomer?
A)
\[C{{H}_{3}}C{{H}_{2}}\underset{Cl}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-C{{H}_{3}}\]
done
clear
B)
\[C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}Cl\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}CHC{{l}_{2}}\]
done
clear
D)
none
done
clear
View Answer play_arrow
question_answer 88) Which of the following is called white vitriol?
A)
\[ZnC{{l}_{2}}\]
done
clear
B)
\[MgS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
C)
\[ZnS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
D)
\[A{{l}_{2}}{{(S{{O}_{4}})}_{3}}\]
done
clear
View Answer play_arrow
question_answer 89) Which gas is evolved, when ammonium dichromate is heated?
A)
Hydrogen
done
clear
B)
Ammonia
done
clear
C)
Oxygen
done
clear
D)
Nitrogen
done
clear
View Answer play_arrow
question_answer 90) Lead is soluble in:
A)
\[C{{H}_{3}}COOH\]
done
clear
B)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
\[HCl\]
done
clear
D)
\[HN{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 91) \[C{{H}_{3}}CH(OH).COOH\] shows:
A)
geometrical isomerism
done
clear
B)
optical isomerism
done
clear
C)
both
done
clear
D)
none
done
clear
View Answer play_arrow
question_answer 92) n-pentane and iso pentane can be distinguished by:
A)
\[B{{r}_{2}}\]
done
clear
B)
\[{{O}_{3}}\]
done
clear
C)
cone, \[{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
\[KMn{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 93) \[FeS{{O}_{4}}.7{{H}_{2}}O\] shows isomorphism with:
A)
\[ZnS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
B)
\[MnS{{O}_{4}}.4{{H}_{2}}O\]
done
clear
C)
\[CaS{{O}_{4}}.5{{H}_{2}}O\]
done
clear
D)
\[CaC{{l}_{2}}.2{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 94) \[N{{a}_{2}}C{{O}_{3}}\] cannot be used to identify:
A)
\[CO_{3}^{2-}\]
done
clear
B)
\[SO_{3}^{2-}\]
done
clear
C)
\[{{S}^{2-}}\]
done
clear
D)
\[SO_{4}^{2-}\]
done
clear
View Answer play_arrow
question_answer 95) Nitrolim is :
A)
\[Ca{{C}_{2}}+{{N}_{2}}\]
done
clear
B)
\[CaC{{N}_{2}}+C\]
done
clear
C)
\[Ca(C{{N}_{2}})+C\]
done
clear
D)
\[Ca{{(CN)}_{2}}+N{{H}_{4}}CN\]
done
clear
View Answer play_arrow
question_answer 96) What is the EAN of nickel in \[Ni{{(CO)}_{4}}\]?
A)
\[34\]
done
clear
B)
\[35\]
done
clear
C)
\[32\]
done
clear
D)
\[36\]
done
clear
View Answer play_arrow
question_answer 97) A weak acid is 0.1% ionised in 0.1 K solution. Its pH is:
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 98) Gold number is maximum for the lyophilic sol is :
A)
gelatin
done
clear
B)
haemoglobin
done
clear
C)
sodium oleate
done
clear
D)
potato starch
done
clear
View Answer play_arrow
question_answer 99) An alkene, on ozonolysis gives formaldehyde and acetaldehyde. The alkene is :
A)
ethene
done
clear
B)
propene
done
clear
C)
butene-1
done
clear
D)
butene-2
done
clear
View Answer play_arrow
question_answer 100) Which of the following polymer has este linkage?
A)
Nylon-66
done
clear
B)
PVC
done
clear
C)
Terylene
done
clear
D)
SBR
done
clear
View Answer play_arrow
question_answer 101) A person is injected with globulin against hepatitis. This is :
A)
artificially acquired passive immunity
done
clear
B)
artificially acquired active immunity
done
clear
C)
naturally acquired active immunity
done
clear
D)
naturally acquired passive immunity
done
clear
View Answer play_arrow
question_answer 102) Which one is mismatched?
A)
Bowmanns capsule- glomerular filtration
done
clear
B)
PCT-absorption of \[N{{a}^{+}}\]and \[{{K}^{+}}\]
done
clear
C)
DCT-absorption of glucose
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 103) How much amount of air can be inspired or expired during normal breathing :
A)
0.05
done
clear
B)
2.5
done
clear
C)
1.5
done
clear
D)
5.5
done
clear
View Answer play_arrow
question_answer 104) The largest variety of digestive enzymes is found in :
A)
carnivores
done
clear
B)
herbivores
done
clear
C)
omnivores
done
clear
D)
parasites
done
clear
View Answer play_arrow
question_answer 105) Alcohol is present in which of the following?
A)
Vitamin D
done
clear
B)
Vitamin 83
done
clear
C)
Vitamin 85
done
clear
D)
Vitamin C
done
clear
View Answer play_arrow
question_answer 106) In a double stranded DNA, the sequences of base pairs in one strand are AGCTAAGCC. Whats the complementary sequence on the other strand?
A)
TCGATTCGG
done
clear
B)
CGUAUUCG
done
clear
C)
AGCTAAGC
done
clear
D)
CAUTAUCG
done
clear
View Answer play_arrow
question_answer 107) The genes concerned with the production of cancer are called:
A)
cancer genes
done
clear
B)
carcinogens
done
clear
C)
carcinomas
done
clear
D)
oncogenes
done
clear
View Answer play_arrow
question_answer 108) A homozygous red flower is crossed with recessive white. Red is dominant over white. The progeny in the \[{{F}_{1}}\] generation will be :
A)
all red
done
clear
B)
all white
done
clear
C)
50% red and 50% white
done
clear
D)
75% red 50% white
done
clear
View Answer play_arrow
question_answer 109) Parasexuality was first discovered in :
A)
bacteria
done
clear
B)
virus
done
clear
C)
fungi
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 110) Sexual reproduction in algae results in the formation of :
A)
oospore
done
clear
B)
zoospore
done
clear
C)
zygote
done
clear
D)
zygospore
done
clear
View Answer play_arrow
question_answer 111) Which was first photosynthetic organism?
A)
Green algae
done
clear
B)
Red algae
done
clear
C)
Cyanobacteria
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 112) During protein denaturation which of the following is disrupted?
A)
2D structure
done
clear
B)
3D structure
done
clear
C)
Peptide bond
done
clear
D)
AA sequence
done
clear
View Answer play_arrow
question_answer 113) Which of the following pair is correct?
A)
Bats wing and insect wing are analogous
done
clear
B)
Seal flippers and bats paw are homologous
done
clear
C)
Insect wing and bird wing are homologous
done
clear
D)
Thorn of Bougainvillea and tendril of pea are analogous
done
clear
View Answer play_arrow
question_answer 114) ICBN is :
A)
International Culture Botanical Nomenclature
done
clear
B)
International Condition Botanical Nomenclature
done
clear
C)
International Code for Botanical Nomenclature
done
clear
D)
International Conversation for Botanical Nomenclature
done
clear
View Answer play_arrow
question_answer 115) If an organism has more rods it will:
A)
active during day
done
clear
B)
possess colour vision
done
clear
C)
active during night
done
clear
D)
both a and c are possible
done
clear
View Answer play_arrow
question_answer 116) The following cells cannot be grown under tissue culture conditions :
A)
hela cells
done
clear
B)
leucocytes
done
clear
C)
kidney cells
done
clear
D)
nerve cells
done
clear
View Answer play_arrow
question_answer 117) The largest gland in the human body is
A)
pancreas
done
clear
B)
brain
done
clear
C)
gall bladder
done
clear
D)
liver
done
clear
View Answer play_arrow
question_answer 118) Which of the phylum has none of fresh water form?
A)
Echinodennata
done
clear
B)
Mollusca
done
clear
C)
Chordata
done
clear
D)
Porifera
done
clear
View Answer play_arrow
question_answer 119) Bilaterally symmetrical but acoelomate animal is:
A)
liver fluke
done
clear
B)
jelly fish
done
clear
C)
round worms
done
clear
D)
crab
done
clear
View Answer play_arrow
question_answer 120) Maximum diversity is found in which phylum?
A)
Chordata
done
clear
B)
Arthropoda
done
clear
C)
Protozoa
done
clear
D)
Annelida
done
clear
View Answer play_arrow
question_answer 121) Which is not a feature of annelids:?
A)
Metameric segmentation
done
clear
B)
Nephridia
done
clear
C)
Pseudocoelom
done
clear
D)
Clitellum
done
clear
View Answer play_arrow
question_answer 122) Which of the following is a living fossil?
A)
Sphenodon
done
clear
B)
Heloderma
done
clear
C)
Rabbit
done
clear
D)
Frog
done
clear
View Answer play_arrow
question_answer 123) Which of the following required in translocation of food in plants?
A)
Xylem
done
clear
B)
Phloem
done
clear
C)
Sclerenchyma
done
clear
D)
Collenchyma
done
clear
View Answer play_arrow
question_answer 124) The organism which is used for gene transfer in higher organisms is?
A)
Agrobacterium tume faciens
done
clear
B)
E. coH
done
clear
C)
Acetobacter
done
clear
D)
Bacillus thuringenesis
done
clear
View Answer play_arrow
question_answer 125) In genetic engineering, the terms vector is applied for :
A)
plasmid
done
clear
B)
sources of DNA
done
clear
C)
cell which receives
done
clear
D)
virus
done
clear
View Answer play_arrow
question_answer 126) Heterozygous purple flower is crossed with recessive white flower. The progeny has the ratio :
A)
all purple
done
clear
B)
all white
done
clear
C)
50% purple, 50% white
done
clear
D)
75%
done
clear
View Answer play_arrow
question_answer 127) Progesterone pill helps in preventing pregnancy by not allowing
A)
ova formation
done
clear
B)
fertilization
done
clear
C)
implantation
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 128) Beadle and Tatum performed classical experiment on Neurospora crassa to explain:
A)
one gene produce one enzyme
done
clear
B)
one enzyme can correct one gene
done
clear
C)
both a and b
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 129) Which one of the following gases can deplete ozone layer in the upper atmosphere?
A)
Ammonia
done
clear
B)
Methane
done
clear
C)
Carbon monoxide
done
clear
D)
Sulphur dioxide
done
clear
View Answer play_arrow
question_answer 130) To remove seed dormancy by mechanically removing the seed coat, is called:
A)
stratification
done
clear
B)
scarification
done
clear
C)
vernalization
done
clear
D)
photoperiodism
done
clear
View Answer play_arrow
question_answer 131) Superority of hybrid over parent is :
A)
heterosis
done
clear
B)
selection
done
clear
C)
hybridized progeny
done
clear
D)
hybrid vigour
done
clear
View Answer play_arrow
question_answer 132) The jumping genes in maize were discovred by :
A)
H.G. Khorana
done
clear
B)
T.H. Morgan
done
clear
C)
Beadle and Tatum
done
clear
D)
Barbara McClintock
done
clear
View Answer play_arrow
question_answer 133) Which one among the following is likely to have the highest level of DDT deposition in its body?
A)
Phytoplanktons
done
clear
B)
Sea gull
done
clear
C)
Crab
done
clear
D)
Eel fish
done
clear
View Answer play_arrow
question_answer 134) Phytotoxins are secreted by plants in response to fungal reaction. These compounds are generally :
A)
proteins
done
clear
B)
glycoproteins
done
clear
C)
phenolic compounds
done
clear
D)
lipids
done
clear
View Answer play_arrow
question_answer 135) Which is a unit of evolution?
A)
Cell
done
clear
B)
Individual
done
clear
C)
Population
done
clear
D)
Species
done
clear
View Answer play_arrow
question_answer 136) Biological species concept emphasizes on :
A)
geographical isolation
done
clear
B)
reproductive isolation
done
clear
C)
physiological isolation
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 137) Cytokinins are formed in :
A)
roots
done
clear
B)
leaves
done
clear
C)
fruits
done
clear
D)
stems
done
clear
View Answer play_arrow
question_answer 138) Interferons are produced in response to :
A)
virus
done
clear
B)
bacteria
done
clear
C)
helminthes
done
clear
D)
malarial parasites
done
clear
View Answer play_arrow
question_answer 139) In which the pneumatophores are found?
A)
Tinospora
done
clear
B)
Pinus
done
clear
C)
Rhizophwra
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 140) Scorpion belongs to a class to which one of the following also belong?
A)
Ticks
done
clear
B)
Crabs
done
clear
C)
Branicles
done
clear
D)
Cockroaches
done
clear
View Answer play_arrow
question_answer 141) Which one of the following is antioxidant vitamin :
A)
C, E, A
done
clear
B)
Bi. 84
done
clear
C)
A,D,E
done
clear
D)
B3,B5
done
clear
View Answer play_arrow
question_answer 142) Acetylcholine helps in :
A)
synaptic transmission
done
clear
B)
membrane permeability
done
clear
C)
synaptic delay
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 143) The physiological capacity to produce off springs is called :
A)
birth rate
done
clear
B)
biotic potential
done
clear
C)
crude natality
done
clear
D)
mortality
done
clear
View Answer play_arrow
question_answer 144) Biogas produced by anaerobic decomposition used in combustion is mainly due to presence of :
A)
\[{{H}_{2}}\]
done
clear
B)
\[C{{H}_{4}}\]
done
clear
C)
\[{{H}_{2}}S\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 145) Which one of the gas is present today but was absent about 3.5 million years ago?
A)
\[{{O}_{2}}\]
done
clear
B)
\[{{N}_{3}}\]
done
clear
C)
\[{{H}_{2}}\]
done
clear
D)
\[C{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 146) In man, artificial pace-maker is implanted due to defects in :
A)
SA node
done
clear
B)
AV node
done
clear
C)
mitral valve
done
clear
D)
Purkinje fibres
done
clear
View Answer play_arrow
question_answer 147) Niacin deficiency leads to :
A)
pellagra
done
clear
B)
osteomalacia
done
clear
C)
marasmus
done
clear
D)
scurvy
done
clear
View Answer play_arrow
question_answer 148) Enzyme having different molecular arrangement but similar functions, is :
A)
isoenzyme
done
clear
B)
holoenzyme
done
clear
C)
apoenzyme
done
clear
D)
coenzyme
done
clear
View Answer play_arrow
question_answer 149) if the foetus is Rh+ and mother is Rh- then :
A)
foetus will transmit antigen to mother blood
done
clear
B)
foetus will transmit antibody to mother blood
done
clear
C)
foetus is attacked by antibodies to mother blood
done
clear
D)
foetus is attacked by antigen to mother blood
done
clear
View Answer play_arrow
question_answer 150) Theory of pangenesis was proposed by :
A)
F. Reedi
done
clear
B)
Darwin
done
clear
C)
Haeckel
done
clear
D)
Miller
done
clear
View Answer play_arrow
question_answer 151) Respiratory quotient (R.Q.) is :
A)
\[\frac{C{{O}_{2}}}{{{O}_{2}}}\]
done
clear
B)
\[\frac{{{O}_{2}}}{C{{O}_{2}}}\]
done
clear
C)
\[\frac{{{N}_{2}}}{C{{O}_{2}}}\]
done
clear
D)
\[\frac{C{{O}_{2}}}{{{N}_{2}}}\]
done
clear
View Answer play_arrow
question_answer 152) For DNA structure, Nobel prize was given to :
A)
Macria Wilkins
done
clear
B)
Franklin
done
clear
C)
Pauly
done
clear
D)
Watson and Crick
done
clear
View Answer play_arrow
question_answer 153) Formation of glucose from lipids is :
A)
gluconeogenesis
done
clear
B)
glycogenoiysis
done
clear
C)
glycogenesis
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 154) The edible part of litchi is :
A)
aril
done
clear
B)
endocarp
done
clear
C)
mesocarp
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 155) Which of the following is initiating cod on?
A)
AUG
done
clear
B)
AUU
done
clear
C)
UAG
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 156) Cyanobacteria are :
A)
mosses which attack bacteria
done
clear
B)
bacteria which attack Cyanophyccae
done
clear
C)
autotrophic organism with phycocyanin
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 157) Cryopreservation is done at temperature:
A)
\[-140{}^\circ C\]
done
clear
B)
\[-120{}^\circ C\]
done
clear
C)
\[-196{}^\circ C\]
done
clear
D)
\[-273{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 158) At which stage of mitosis chromatids separated and passes to different poles?
A)
Prophase
done
clear
B)
Metaphase
done
clear
C)
Anaphase
done
clear
D)
Telophase
done
clear
View Answer play_arrow
question_answer 159) Which one of the following is common in plant and animal?
A)
Mitochondria
done
clear
B)
Tonoplast
done
clear
C)
Centriole
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 160) Fungi lack :
A)
mitochondria
done
clear
B)
ribosomes
done
clear
C)
chloroplast
done
clear
D)
endoplasmic reticulum
done
clear
View Answer play_arrow
question_answer 161) Which one of the following organelles lack membranes?
A)
Ribosome
done
clear
B)
Mitochondria
done
clear
C)
Golgi complex
done
clear
D)
Nucleus
done
clear
View Answer play_arrow
question_answer 162) Rajaji national park is situated in :
A)
Tamil Nadu
done
clear
B)
Karnataka
done
clear
C)
Uttranchal
done
clear
D)
Rajasthan
done
clear
View Answer play_arrow
question_answer 163) Chlorophyll a is obsent in which of the following photosynthetic organism?
A)
Cyanobacteria
done
clear
B)
Red algae
done
clear
C)
Brown algae
done
clear
D)
Bacteria
done
clear
View Answer play_arrow
question_answer 164) Streptococcus is employed in the manufacturing of :
A)
wine
done
clear
B)
bread
done
clear
C)
cheese
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 165) water bloom is generally caused by
A)
green algae
done
clear
B)
blue-green algae
done
clear
C)
bacteria
done
clear
D)
Hydrilla
done
clear
View Answer play_arrow
question_answer 166) Who proposed the natural system of classification?
A)
Carolus Linnaeus
done
clear
B)
lone Hutchinson
done
clear
C)
Bentham and Hooker
done
clear
D)
Oswald Tippo
done
clear
View Answer play_arrow
question_answer 167) Which one of the following is a major pollutant of automobile gases
A)
Carbon monoxides
done
clear
B)
Oxides of nitrogen
done
clear
C)
Oxides of sulphur
done
clear
D)
Carbon dioxide
done
clear
View Answer play_arrow
question_answer 168) During meiosis, crossing over occurs between:
A)
sister chromatids of homologous chromosome
done
clear
B)
non-sister chromatids of homologous chromosome
done
clear
C)
sister chromatids of non-homologous chromosome
done
clear
D)
non-homologous chromatids of homologous chromosome
done
clear
View Answer play_arrow
question_answer 169) Node of Ranvier is found in :
A)
right auricle
done
clear
B)
muscle bundles
done
clear
C)
dendrite
done
clear
D)
axon
done
clear
View Answer play_arrow
question_answer 170) The layer of cells that secrete enamel of tooth is :
A)
dentoblast
done
clear
B)
ameloblast
done
clear
C)
osteoblast
done
clear
D)
odontoblast
done
clear
View Answer play_arrow
question_answer 171) Monosomy and trisomy can be represented as :
A)
2n+1,2n+3
done
clear
B)
2n-1,2n-2
done
clear
C)
2n, 2n +1
done
clear
D)
2n -1,2n +1
done
clear
View Answer play_arrow
question_answer 172) Trisomy of which chromosome is involved in Downs syndrome?
A)
15th
done
clear
B)
21st
done
clear
C)
20th
done
clear
D)
19th
done
clear
View Answer play_arrow
question_answer 173) Choose the correct statement about the direction of DNA strand
A)
\[5\to 3\]takes place on template strand
done
clear
B)
\[3\to 5\] takes place on new strand
done
clear
C)
\[5\to 3\] takes place on leading strand
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 174) \[{{O}_{2}}\] evolution is directly associated with :
A)
PS-I
done
clear
B)
PS-II
done
clear
C)
phytochrome
done
clear
D)
phycocyanin
done
clear
View Answer play_arrow
question_answer 175) The hormone involved in metabolism of food material in cereal grains during germination, is :
A)
auxin
done
clear
B)
cytokinin
done
clear
C)
gibberellin acid
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 176) Cycas circinalis is a source of :
A)
resin
done
clear
B)
timber
done
clear
C)
essential oil
done
clear
D)
starch
done
clear
View Answer play_arrow
question_answer 177) Maximum productivity is found in :
A)
Grassland
done
clear
B)
tropical rain forest
done
clear
C)
Ocean
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 178) Which of the following is absent in DNA?
A)
U
done
clear
B)
A
done
clear
C)
T
done
clear
D)
G
done
clear
View Answer play_arrow
question_answer 179) Vernalization is done at:
A)
lower temperature
done
clear
B)
low light intensity
done
clear
C)
higher temperature
done
clear
D)
high light intensity
done
clear
View Answer play_arrow
question_answer 180) Ginseng is an important drug of :
A)
America
done
clear
B)
Egypt
done
clear
C)
India
done
clear
D)
China
done
clear
View Answer play_arrow
question_answer 181) Triticum aestizrium the common bread wheat is :
A)
triploid with 21 chromosome
done
clear
B)
hexaploid with 42 chromosome
done
clear
C)
tetraploid with 30 chromosome
done
clear
D)
diploid with 14 chromosome
done
clear
View Answer play_arrow
question_answer 182) CFCs are not recomended to be used in refrigerators because they :
A)
increase temperature
done
clear
B)
deplete ozone
done
clear
C)
affect environment
done
clear
D)
affect human body
done
clear
View Answer play_arrow
question_answer 183) Single stranded nucleic acid is found in :
A)
E. coli
done
clear
B)
\[\phi \times \,174\]
done
clear
C)
\[\lambda \]
done
clear
D)
\[{{T}_{4}}\]
done
clear
View Answer play_arrow
question_answer 184) Black rust of wheat is caused by :
A)
Puccinia graminis
done
clear
B)
Ustifago
done
clear
C)
Pythium
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 185) A cell active in protein synthesis will rich in :
A)
ribosome
done
clear
B)
Golgi bodies
done
clear
C)
mitochondria
done
clear
D)
lysosome
done
clear
View Answer play_arrow
question_answer 186) Lateral root originates from :
A)
endodermis
done
clear
B)
pericycle
done
clear
C)
cortex
done
clear
D)
phloem
done
clear
View Answer play_arrow
question_answer 187) Use and disuse Theory was propsed by :
A)
Lamarck
done
clear
B)
Darwin
done
clear
C)
Hugo de Vries
done
clear
D)
Malthus
done
clear
View Answer play_arrow
question_answer 188) Cell organelle responsible for autolysis :
A)
dictyosome
done
clear
B)
lysosome
done
clear
C)
peroxisome
done
clear
D)
glyoxysome
done
clear
View Answer play_arrow
question_answer 189) Eggs of cockroach are :
A)
alecithal
done
clear
B)
microlecithal
done
clear
C)
telolecithal
done
clear
D)
centrolecithal
done
clear
View Answer play_arrow
question_answer 190) The endoskeleton of cell is made up of :
A)
deutoplasm
done
clear
B)
protoplasm
done
clear
C)
endoplasmic reticulum
done
clear
D)
cell wall
done
clear
View Answer play_arrow
question_answer 191) Ley ding cells are found in
A)
heart
done
clear
B)
lungs
done
clear
C)
spleen
done
clear
D)
testis
done
clear
View Answer play_arrow
question_answer 192) The instrument by which B.P. of man is determined :
A)
Sphygmomanometer
done
clear
B)
B.P. meter
done
clear
C)
altra sound
done
clear
D)
stethoscope
done
clear
View Answer play_arrow
question_answer 193) In recent years which disease of digestive system in India has received great attention and against that active vaccination is being done even in schools?
A)
Polimyelitis
done
clear
B)
Hepatitis
done
clear
C)
Jaundice
done
clear
D)
Enterocolitis
done
clear
View Answer play_arrow
question_answer 194) Cystoliths are composed of :
A)
glucosides
done
clear
B)
calcium oxalate
done
clear
C)
calcium carbonate
done
clear
D)
magnesium oxalate
done
clear
View Answer play_arrow
question_answer 195) Which one of the following element is essential for the life of animal and not for plants?
A)
Calcium
done
clear
B)
Iodine
done
clear
C)
Phosphorus
done
clear
D)
Potassium
done
clear
View Answer play_arrow
question_answer 196) Haversian canal are found in :
A)
internal ear of mammal
done
clear
B)
external ear of mammal
done
clear
C)
long bone of rabbit
done
clear
D)
spinal chord
done
clear
View Answer play_arrow
question_answer 197) The AIDS test is known as :
A)
ELISA
done
clear
B)
Australian antigen
done
clear
C)
HIV test
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 198) Hematology is the study of :
A)
bone
done
clear
B)
blood
done
clear
C)
cartilage
done
clear
D)
nerves
done
clear
View Answer play_arrow
question_answer 199) Osmotic pressure of a solution is :
A)
greater than pure solvent
done
clear
B)
less than pure solvent
done
clear
C)
equal to pure solvent
done
clear
D)
less than or greater than pure solvent
done
clear
View Answer play_arrow
question_answer 200) Number of linkage group in Pisum sativum is :
A)
2
done
clear
B)
5
done
clear
C)
7
done
clear
D)
9
done
clear
View Answer play_arrow
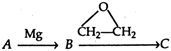 \[\xrightarrow{{{H}_{2}}O}\]D Here ,D is:
\[\xrightarrow{{{H}_{2}}O}\]D Here ,D is:
