-
Write the structures of the following compounds.
(i) 2-chioro-3-rnethylpentane
(ii) 1-chloro-4-ethyi cyclohexane
(iii) 4-tert- butyl-3-iodoheptane
(iv) 1,4-dibromobut-2-ene
(v) 1 -bromo-4-sec-butyl-2-methylbenzene
View Answer play_arrow
-
Why is sulphuric acid not used during the reaction of alcohols with KI?
View Answer play_arrow
-
Write the structures of different dihalogen derivatives ofpropane.
View Answer play_arrow
-
Among the isomeric alkanes of molecular formula
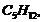
identify the one that one that on photochemical chlorination yields
(i) a single monochloride
(ii) three isomericmonochiorides
(iii) four isomericmonochiorides
-
Draw the structures of major monohalo products in each of the following reactions:
(i)
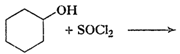
(ii)
(iii)
(iv)
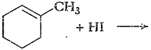
(v)
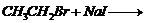
(vi)
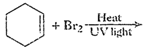
View Answer play_arrow
-
Arrange each set of compounds in order of increasingboiling points :
(i) Bromomethane, bromoform, chloromethane, dibromoniethane
(ii) 1-chloropropane, iso-propyl chloride, 1-chlorobutane
View Answer play_arrow
-
Which alkyl halide from the following pairs would youexpect to react more rapidly by an
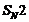
mechanism? Explain your answer.
(i)
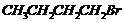
or
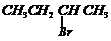
(ii)
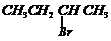
or
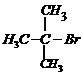
(iii)
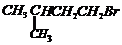
or
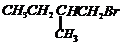
-
In the following pairs of halogen
compounds, which compound undergoes faster
reaction?
(i)and
(ii) and
-
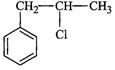
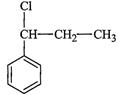
Identify A, B, C, D, E, R and R' m the following:
(i)
(ii)
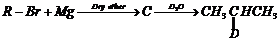
(iii)
-
Name the following halides according to IUPAC systemand classify them as alkyl, allyl, benzyl (primary, secondary, tertiary),vinyl or aryl halides :
(i) 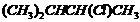 (ii)
(ii) 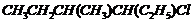 (iii)
(iii)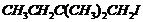 (iv)
(iv)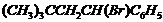 (v)
(v) 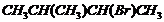 (vi)
(vi)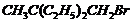 (vii)
(vii) 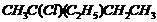 (viii)
(viii) 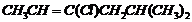 (ix)
(ix) 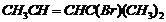 (x)
(x) 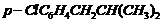 (xi)
(xi) 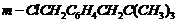 (xii)
(xii) 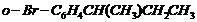
View Answer play_arrow
-
Give the IUPAC names of the following compounds.
(i)
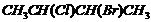
(ii)
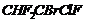
(iii)
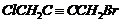
(iv)
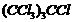
(v)
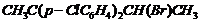
(vi)
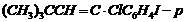
-
Write the structures of the following organic halogencompounds :
(i) 2-chloro-3-methylpentane
(ii) p-bromochlorobenzene
(iii) 1-chloro-4-ethylcyclohexane
(iv) 2-(2-chlorophenyl)-1-iodooctane
(v) 2-bromobutane
(vi) 4-tert-butyl-3-iodoheptane
(vii) 1-bromo-4-sec-butyl-2-methylbenzene
(viii) 1,4-dibromobut-2-ene
View Answer play_arrow
-
Which one of the following has the highest dipole moment?
(i) 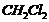 (ii)
(ii) 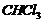 (iii)
(iii) 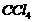
View Answer play_arrow
-
A hydrocarbon 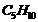 does not react with chlorine in dark but gives a single monochloro compound
does not react with chlorine in dark but gives a single monochloro compound 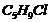 in bright sunlight.Identify the hydrocarbon.
in bright sunlight.Identify the hydrocarbon.
View Answer play_arrow
-
Write the
isomers of the compound having formula 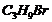 .
.
View Answer play_arrow
-
Writs the equations for the preparation of 1-iodobutanefrom
(i) 1-butanol (ii) 1-chlorobutane (iii) but-1-ene
View Answer play_arrow
-
What are ambident nucleophiles? Explain with an example.
View Answer play_arrow
-
Which compound in each of the following pairs will reactfaster in 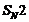 reaction with
reaction with 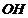 ?
(i)
?
(i) 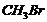 or
or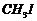 (ii)
(ii) 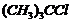 or
or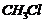
View Answer play_arrow
-
Predict all the alkanes that would be formed bydehydrohalogenation of the following halides with sodium ethoxide in ethanol and identify the major alkene:
(i) 1-bromo-1-methylcyclohexane
(ii) 2-chloro-2-methylbutane
(iii) 2,2,3-triniethyl-3-bromopentane
View Answer play_arrow
-
How will you bring about the following conversions?
(i) Ethanol to but-1-yne
(ii) Ethane to bromoethane
(iii) Propene to 1-nitropropane
(iv) Toluene to benzyl alcohol
(v) Propene to propyne
(vi) Ethanol to ethyl fluoride
(vii) Bromomethane to propanone
(viii) But-1-ene to but-2-ene
(ix) 1-chlorobutane to n-octane
(x) Benzene to biphenyl
View Answer play_arrow
-
Explain why
(i) the dipole moment of chlorobenzene is lower than that ofcyclohexyl chloride?
(ii) alkyl halides, though polar, are immiscible with water?
(iii) Grignard reagents should be prepared under anhydrousconditions?
(i) Find the hybridization of C-atoms in chlorobenzene and cyclohexylchloride and electronegativity to find the bond polarity' and thus,dipole moment.
(ii) Strong intermolecular-H bonding among water molecules,
(iii) Consider reactive nature of Grignard reagents with water.
View Answer play_arrow
-
Give the uses offreon 12, DDT, carbon tetrachloride andiodoform.
View Answer play_arrow
-
Write the structure for the major organic product in eachof
the following reactions.
(i)
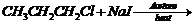
(ii)
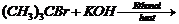
(iii)
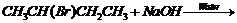
(iv)
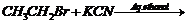
(v)
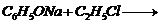
(vi)
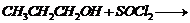
(vii)
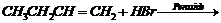
(viii)
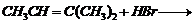
(i) Replacement of
by
.
(ii) Dehydrohalogenation (
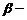
elimination).
(iii) Replacement of
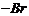
by
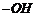
(iv) Replacement of
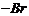
by
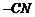
(v) Elimination of NaCl,
formation of ether. (Williamson?s synthesis)
(vi) Replacement of
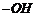
by
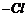
;
formation of alkyl halide.
(vii) Addition of HBr across the
double bond (peroxide effect)
(viii) Addition of HBr according to
Markownikoff?s rule.
-
Write the mechanism of the following reaction.

-
Arrange the
compounds of each set in order of reactivitytowards  displacement:
(i)
2-bromo-2-methylbutane, 1-bromopentane, 2-bromopentane
(ii)
1-bromo-3-methylbutane, 2-bromo-2-methylbutane, 2-bromo-3-methylbutane
(iii)
1-bromobutane, 1-bromo-2, 2-dimethylpropane, 1-bromo-2-methylbutane,
1-bromo-3-methylbutane.
displacement:
(i)
2-bromo-2-methylbutane, 1-bromopentane, 2-bromopentane
(ii)
1-bromo-3-methylbutane, 2-bromo-2-methylbutane, 2-bromo-3-methylbutane
(iii)
1-bromobutane, 1-bromo-2, 2-dimethylpropane, 1-bromo-2-methylbutane,
1-bromo-3-methylbutane.
 reactions involve the formation of transition state. Higher
the steric hindrance, lesser the stability of transition state and so lesser
the reactivity towards
reactions involve the formation of transition state. Higher
the steric hindrance, lesser the stability of transition state and so lesser
the reactivity towards  .
.
View Answer play_arrow
-
Out of
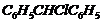
and
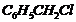
which
is more easily hydrolyzed by aqueous KOH?
In
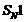
reactions,
stability of carbocations is responsible for the reactively. The reaction is
more likely to take place by
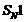
reaction.
-
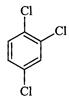
P-Dichlorobenzene has higher melting point and tower solubility than those of o-and m-isomers. Discuss.
Symmetry of the molecule is related to its crystal lattice structures and hence, to ifs melting point and solubility. A highly symmetrical structure has a higher melting point.
View Answer play_arrow
-
How the following conversions can be
carried out?
(i) Propene to propan-1-ol
(ii) Ethanol to but-1-yne
(iii) 1-bromopropane to
2-bromopropane
(iv) Toluene to benzyl alcohol
(v) Benzene to 4-bromonitrobenzene
(vi) Benzyl alcohol to 2-phenyl
ethanoic acid
(vii) Ethanol to propane nitrile
(viii) Aniline to chlorobenzene
(ix) 2-chlorobutane to 3,
4-dimethylhexane
(x) 2-methyl-1-propene to
2-chloro-2-methylpropane
(xi) Ethyl chloride to propanoic
acid
(xii) But-1-eiie to
n-butyliodide
(xiii) 2-chloropropane to
1-propanol
(xiv) Iso-propyl alcohol to iodoform
(xv) Chlorobenzene to p-nitrophenol
(xvi) 2-bromopropane to 1-bromopropane
(xvii) Chloroethane to butane
(xviii) Benzene to diphenyl
(xix) tert-butyl bromide to
iso-butyl bromide
(xx) Aniline to phenyl
isocyanide
(i) Addition of HBr (peroxide
effect); Replacement of ?Br by ?OH.
(ii) ? OH is substitudedby
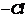
; then
substitution by alkynyl group to form higher alkyne.
(iii) Dehydrohalogenation by
alc. KOH (
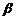
-elimination),
Addition of HBr (Markownikoff rule)
(iv) Chlorination of
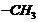
group;
Replacement
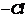
by ?
OH.
(v) Bromination of benzene;
Nitration of bromobenzene at p-position.
(vi) Replacement of ? OH by
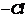
; then
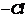
by
?CN and at last by ? COOH group.
(vii) Replacement of ?OH by?l
and then ?l by ?CN.
(viii) Diazotization; Formation
of chlrobenzene.
(ix) Preparation of alkane with
double number of C-atoms (Wurtz reaction)
(x) Addition of
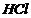
across
the double bond (Markownikoff rule)
(xi) Substitution of
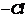
by
?CN; Acidic hydrolysis.
(xii) Addition of
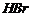
(peroxide
effect or Anti-Markownikoff?s rule); Substitution of ?Br by ?l.
(xiii) Dehydrohalogenation (
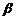
elimination);
Addition of
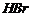
(peroxide
effect); Substituting of -Br by -OH by aqueous KOH.
(xiv) Oxidation of alcohol to
ketone, Iodoform reaction.
(xv) Nitration replacement of
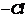
by
?OH;
(xvi) Dehydrohalogenation (
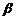
-elimination);
Addition of
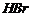
(peroxide
effect)
(xvii) Wurtz reaction-Formation
of hydrocarbon with double number of carbon atoms.
(xix) Dehydrohalogenation; Addition of
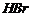
(peroxide
effect)
(xx) Carbylamine reaction
-
The treatment of alkyl chlorides with
aqueous KOH leadsto the formation of alcohols but in the presence of alcoholic
KOH, alkenesare major products. Explain.
Difference lies in the mode and extent of dissociation of KOH in thepresence of
different solvents.
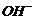
is a good
nucleophile while
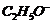
is a
strong base.
-
Primary alkyl halide
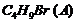
reacted
with alcoholicKOH to give compound (B). Compound (B) is reacted with
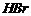
to
give (C) which is an isomer of (A). When (A) is reacted with sodium metal, it
gives compound (D),
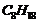
which
is different from the compound formedwhen n-butyl bromide is reacted with
sodium. Give the structural formulaof (A) and write the equations for all the
reactions.
(i) Write the possible isomers
of
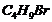
.
(ii) Write the equation for the
reaction of n-butyl bromide with sodium.
(iii) Write the equation for all
possible reactions and identify the compounds (A), (B). (C) and (D).
-
What happens when
(i) n- butyl chloride is treated
with alcoholic KOH.
(ii) bromobenzene is treated
with Mg in the presence of dry ether.
(iii) chlorobenzene is subjected
to hydrolysis.
(iv)ethyl chloride is treated
with aqueous KOH.
(v) methyl bromide is treated
with sodium in die presence of dry ether.
(vi) methyl chloride is treated
with KCN?
(i) Dehydrohalogenation (

elimination)
as alc. KOH is a dehydrohalogenating reagent.
(ii) Formation of Grignard
reagent.
(iii) No reaction, due to double
bond character in

bond.
(iv)

is
replaced by ? OH.
(v) Wurtz reaction
(vi)

replaced
by ? CN.
-
View Answer play_arrow
-
question_answer33)
The
order of reactivity of following alcohol with halogen acids is.............
(a)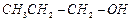 (b)
(b)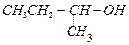 (c)
(c)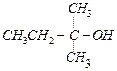 (a) (A) > (B) > (C) (b) (C) > (B)
> (A)
(c) (B) > (A) > (C) (d) (A) > (C)
> (B)
(a) (A) > (B) > (C) (b) (C) > (B)
> (A)
(c) (B) > (A) > (C) (d) (A) > (C)
> (B)
View Answer play_arrow
-
question_answer34)
Which
of the following alcohols will yield the
corresponding alkyl chloride on reaction with
concentrated HCl at room temperature ?
(a)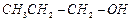 (b)
(b)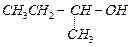 (c)
(c)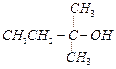 (d)
(d) 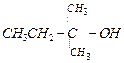
View Answer play_arrow
-
question_answer35)
Identify
the compound Y in the following reaction.
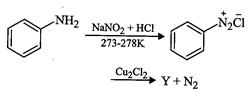 (a) (b)
(a) (b)
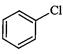
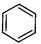 (c) (d)
(c) (d)
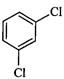
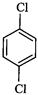
View Answer play_arrow
-
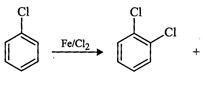
question_answer36)
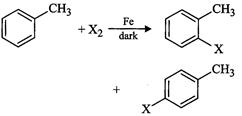
Toluene
reacts with a halogen in the presence of iron (HI) chloride giving ortho and
para halo compounds. The reaction is
(a) Electrophilic elimination reaction
(b) Electrophilic substitution reaction
(c) Free radical addition reaction
(d) Nucleophilic substitution reaction
View Answer play_arrow
-
question_answer37)
Which
of the following is halogen exchange reaction ?
(a)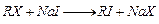 (b)
(b)
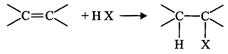 (c)
(c)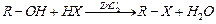 (d)
(d)
View Answer play_arrow
-
question_answer38)
Which
reagent will you use for the following
reaction?
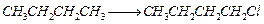
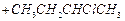 (a)
(a)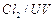 light (b)
light (b)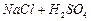 (c)
(c)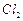 gas in dark
(d)
gas in dark
(d)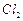 gas in the
presence of iron in dark
gas in the
presence of iron in dark
View Answer play_arrow
-
question_answer39)
Arrange
the following compounds in the
increasing order of their densities.
 (a)
(a)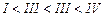 (b)
(b)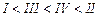 (c)
(c)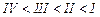 (d)
(d)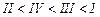
View Answer play_arrow
-
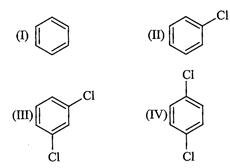
question_answer40)
Arrange
the following compounds in increasing order of their boiling points.
(I).. (II)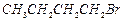
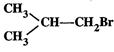 (III)
(III)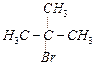 (a)
(a)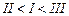 (b)
(b)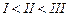 (c)
(c)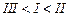 (d)
(d)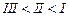
View Answer play_arrow
-
question_answer41)
In
which of the following molecules carbon atom marked with asterisk (*) is
asymmetric?
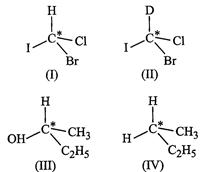 (a)
(a)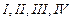 (b)
(b)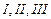 (c)
(c)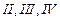 (d)
(d)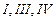
View Answer play_arrow
-
question_answer42)
Which
of the following structures is enantiomeric with the molecule (A) given below :
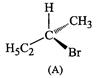 (a). (b)
(a). (b)
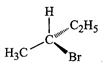
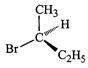 (c).. (d)
(c).. (d)
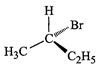
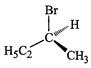
View Answer play_arrow
-
question_answer43)
Which
of the following is an example of vie- dihalide ?
(a) Dichloromethane (b) 1, 2-Dichloroethane
(c) Ethylidene chloride (d) Allyl chloride
View Answer play_arrow
-
question_answer44)
The
position of ?Br in the compound in 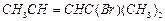 can
be classified as............ .
(a) Allyl (b) Aryl
(c) Vinyl (d) Secondary
can
be classified as............ .
(a) Allyl (b) Aryl
(c) Vinyl (d) Secondary
View Answer play_arrow
-
question_answer45)
Chlorobenzene
is formed by reaction of chlorine with benzene in the presence of 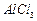 . Which of the following
species attacks the benzene ring in this reaction ?
(a)
. Which of the following
species attacks the benzene ring in this reaction ?
(a)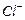 (b)
(b)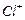 (c)
(c)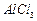 (d)
(d)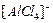
View Answer play_arrow
-
question_answer46)
Ethylidene
chloride is a/an................
(a) vic-dihalide (b) gew-dihalide
(c) allylic halide (d) vinylic halide
View Answer play_arrow
-
question_answer47)
What
is 'A' in the following reaction ?
(a)?.. (b)
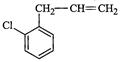
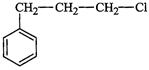 (c). (d)
(c). (d)
View Answer play_arrow
-
question_answer48)
A
primary alkyi halide would prefer to undergo..............
(a) 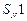 reaction (b)
reaction (b) 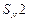 reaction
(c)
reaction
(c) 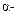 Elimination
(d) Racemisation
Elimination
(d) Racemisation
View Answer play_arrow
-
question_answer49)
Which
of the following alkyl halides will
undergo 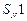 reaction most
readily ?
(a)
reaction most
readily ?
(a)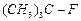 (b)
(b)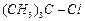 (c)
(c)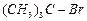 (d)
(d)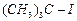
View Answer play_arrow
-
question_answer50)
Which
is the correct IUPAC name for
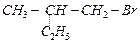 (a)l-Bromo-2-ethylpropane
(b) l-Bromo-2-ethyl-2-methylethane
(c) l-Bromo-2-methylbutane
(d) 2-Methyl-l-bromobutane
(a)l-Bromo-2-ethylpropane
(b) l-Bromo-2-ethyl-2-methylethane
(c) l-Bromo-2-methylbutane
(d) 2-Methyl-l-bromobutane
View Answer play_arrow
-
question_answer51)
What
should be the correct IUPAC name for
diethylbromomethane?
(a) 1-Bromo-l, 1-diethylmethane
(b) 3-Bromopentane
(c) 1-Bromo-l-ethylpropane
(d) 1-Bromopentane
View Answer play_arrow
-
question_answer52)
The
reaction of toluene with chlorine in the
presence of iron and in the absence of light
yields.............
(a)? (b)
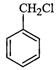
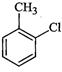 (c)
(c)
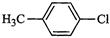 (d) Mixture of (b) and (c)
(d) Mixture of (b) and (c)
View Answer play_arrow
-
question_answer53)
Chloromethane
on treatment with excess of
ammonia yields mainly
(a) N, N-Dimethylmethanamine
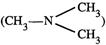 (b)N-methyhnethanamine
(b)N-methyhnethanamine 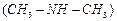 (c) Methanamine
(c) Methanamine 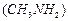 (d)Mixture containing all these in equal proportion
(d)Mixture containing all these in equal proportion
View Answer play_arrow
-
question_answer54)
Molecules
whose mirror image is non- Super imposable over them are known as chiral. Which
of the following molecules is chiral in
nature?
(a) 2-Bromobutane (b) 1-Bromobutane
(c)2-Bromopropane
(d) 2-Bromopropan-2-ol
View Answer play_arrow
-
question_answer55)
Reaction
of 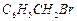 with aqueous sodium hydroxide
follows..............
(a)
with aqueous sodium hydroxide
follows..............
(a)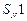 mechanism (b)
mechanism (b) 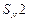 mechanism
(c) Any of the above two depending upon the temp- erature
of reaction
(d) Saytzeff rule
mechanism
(c) Any of the above two depending upon the temp- erature
of reaction
(d) Saytzeff rule
View Answer play_arrow
-
question_answer56)
Which
of the carbon atoms present in the molecule given below are asymmetric ?
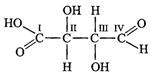 (a)
(a)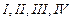 (b)
(b)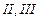 (c)
(c)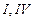 (d)
(d)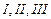
View Answer play_arrow
-
question_answer57)
Which
of the following compounds will give racemic mixture on nucleophilic
substitution by 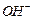 ion?
(I)
ion?
(I)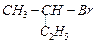 (II)
(II) (III)
(III)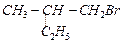 (a) I (b) I, II,
III
(c) II, III (d) I, III
(a) I (b) I, II,
III
(c) II, III (d) I, III
View Answer play_arrow
-
question_answer58)
(I)?. (II)
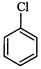
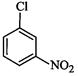
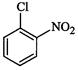 (III)
(III)
 (a)
(a)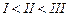 (b)
(b)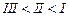 (c)
(c)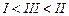 (d)
(d)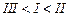
View Answer play_arrow
-
question_answer59)
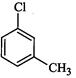
(I) (II)
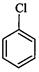
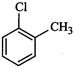 (III)
(III)
 (a)
(a)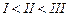 (b)
(b)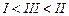 (c)
(c)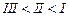 (d)
(d)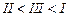
View Answer play_arrow
-
question_answer60)
(I)? (II)
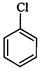
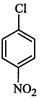 (III)
(III)
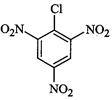 (a)
(a) (b)
(b) (c)
(c) (d)
(d)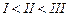
View Answer play_arrow
-
question_answer61)
(I).. (II)
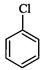
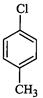 (III)
(III)
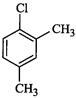 (a) I < II < III (b)II < I <
III
(c)III < II < I (d)I < III
<II
(a) I < II < III (b)II < I <
III
(c)III < II < I (d)I < III
<II
View Answer play_arrow
-
question_answer62)
Which
is the correct increasing order of boiling points of the following compounds ?
1-Iodobutane, 1-Bromobutane,
1-Chlorobutane, Butane
(a) Butane < 1-Chlorobutane < 1-Bromobutane <
1-Iodobutant
(b) 1-Iodobutane < 1-Bromobutane < 1-Chlorobutane
< Butane
(c) Butane < 1-Iodobutane < 1-Bromobutane <
1-Chlorobutail
(d) Butane < 1-Chlorobutane < 1-Iodobutane
< 1-Bromobutan
View Answer play_arrow
-
question_answer63)
Which
is the correct increasing order of boiling points of the following compounds ?
1-Bromoethane, 1-Bromopropane,
1-Bromobutane, Bromobenzene
(a) Bromobenzene < 1-Bromobutane < 1-Bromopropane
< 1-Bromoethaii
(b) Bromobenzene < 1-Bromoethane < 1-Bromopropane
< 1-Bromobutane
(c) 1-Bromopropane < 1-Bromobutane
< 1-Bromoethane < Bromobutane
(d)1-Bromoethane < 1-Bromopropane < 1-Bromobutane
< Bromobenzene
View Answer play_arrow
-
question_answer64)
Note : In the following questions two or more options
may be correct
Consider the following reaction and answer the questions
no. 32-34.
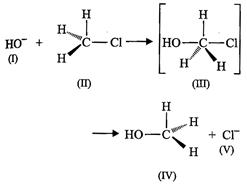
View Answer play_arrow
-
question_answer65)
Which
of the statements are correct about
above reaction ?
(I) and (V) both are nucleophiles.
(b) In (III) carbon atom is sp3 hybridised.
(c) In (IV) carbon atom is sp3 hybridised.
(d) and (V) both are electrophiles.
View Answer play_arrow
-
question_answer66)
Which
of the following statements are correct about this reaction ?
(a) The given reaction follows S^2 mechanism.
(b)(II) and (IV) have opposite configuration.
(c) (II) and (IV) have same configuration.
(d) The given reaction follows S^l mechanism.
View Answer play_arrow
-
question_answer67)
Which
of the following statements are correct about the reaction intermediate ?
(a) Intermediate (III) is unstable because in this carbon is attached to
5 atoms.
(b) Intermediate (HI) is unstable because carbon atom is sp2
hybridised.
(c) Intermediate (DI) is stable because carbon atom is sp2
hybridised.
(d) Intermediate (DI) is less stable than the reactant (II).
Answer Q. No. 35 and 36 on the basis of the following
reaction.
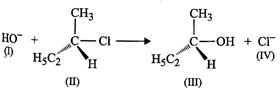
View Answer play_arrow
-
question_answer68)
Which
of the following statements are correct about the mechanism of this reaction?
(a) A carbocadon will be formed as an intermediate in the reaction.
(b)  will attach
the substrate (II) from one side and
will attach
the substrate (II) from one side and  will leave
it simultaneously from other side.
(c)An unstable intermediate will be formed in
which
will leave
it simultaneously from other side.
(c)An unstable intermediate will be formed in
which 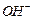 and
and 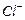 will be attached by weak bonds.
(d) Reaction proceeds through
will be attached by weak bonds.
(d) Reaction proceeds through 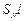 mechanism.
mechanism.
View Answer play_arrow
-
question_answer69)
Which
of the following statements are correct about the kinetics of this reaction ?
(a) The rate of reaction depends on the concentration of only (II)
(b) The rate of reaction depends on concentration of both
(I) and (H)
(c) Molecularity of reaction is one
(d) Molecularity of reaction is two
View Answer play_arrow
-
question_answer70)
Haloalkanes
contain halogen atom (s) attached to the sp3 hybridised
carbon atom of an alkyl group. Identify haloalkane from the following compounds.
(a) 2-Bromopentane
(b) Vinyl chloride (chloroethene)
(c) 2-chloroacetophenone
(d) Trichloromethane
View Answer play_arrow
-
question_answer71)
Ethylene
chloride and ethylidene chloride are isomers. Identify the correct statements.
(a) Both the compounds form same product on treatment with alcoholic KOH
(b) Both the compounds form same product on treatment with
alcoholic KOH
(c) Both the compounds form same product on reduction
(d) Both the compounds are optically active
View Answer play_arrow
-
question_answer72)
Which
of the following compounds are gem-
dihalides ?
(a) Ethylidene chloride
(b) Ethylene dichloride
(c) Methylene chloride
(d) Benzyl chloride
View Answer play_arrow
-
question_answer73)
Which
of the following are secondary bromides?
(a)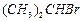 (b)
(b)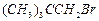 (c)
(c)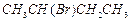 (d)
(d)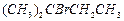
View Answer play_arrow
-
question_answer74)
Which
of the following compounds can be classified as aryl halides ?
(a)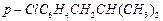 (b)
(b)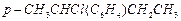 (c)
(c)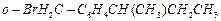 (d)
(d)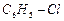
View Answer play_arrow
-
question_answer75)
Alkyl
halides are prepared from alcohols by
treating with
(a)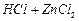 (b)
(b)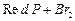 (c)
(c)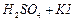 (d)All
of these
(d)All
of these
View Answer play_arrow
-
question_answer76)
Alkyl
fluorides are synthesised by heating a
alkyl chloride/bromide in presence of.........
or.............. .
(a)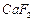 (b)
(b)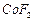 (c)
(c)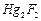 (d)
NaF
(d)
NaF
View Answer play_arrow
-
question_answer77)
Aryl chlorides and bromides can be
easily prepared by electrophilic substitution of arenes with chlorine and
bromine respectively in the presence of Lewis acid catalysts. But why does
preparation of aryl iodides require presence of an oxidising agent ?
View Answer play_arrow
-
question_answer78)
Out of o- and p-dibromobenzene which
one has higher melting point and why ?
View Answer play_arrow
-
question_answer79)
Which of the compounds will react
faster in 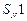 reaction with the
-OH ion?
CH3 ? CH2-Cl or C6H5
? CH2-Cl
reaction with the
-OH ion?
CH3 ? CH2-Cl or C6H5
? CH2-Cl
View Answer play_arrow
-
question_answer80)
Why indo form has appreciable
antiseptic property ?
View Answer play_arrow
-
question_answer81)
Haloarenes are less reactive than
haloalkanes and haloalkenes. Explain.
View Answer play_arrow
-
question_answer82)
Discuss
the role of Lewis acids in the preparation of aryl bromides and
chlorides in the dark.
View Answer play_arrow
-
question_answer83)
Which of the following compounds (a)
and (b) will not react with a mixture of NaBr and 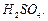 Explain why ?
(a)
Explain why ?
(a)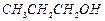 (b)
(b)
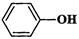
View Answer play_arrow
-
question_answer84)
Which of the products will be major
product in the reaction given below ? Explain.
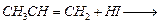
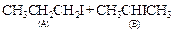
View Answer play_arrow
-
question_answer85)
Why is the solubility of haloalkanes
in water very low ?
View Answer play_arrow
-
question_answer86)
Draw other resonance structures related
to the following structure and find out whether the function group present in
the molecule is ortho, para directing or meta directing.
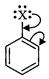
View Answer play_arrow
-
question_answer87)
Classify the following compounds as
primary, secondary and tertiary halides.
(i) l-Bromobut-2-ene
(ii) 4-Bromopent-2-ene
(iii) 2-Bromo-2-methylpropane
View Answer play_arrow
-
question_answer88)
Compound 'A' with molecular formula 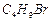 is treated with aq. KOH
solution. The rate of this reaction depends upon the concentration of the
compound 'A' only. When another optically active isomer 'B' of this compound
was treated with aq. KOH solution, the rate of reaction was found to be
dependent on concentration of compound and KOH both.
(i) Write down the structural formula of both
compounds 'A' and 'B'.
(ii) Out of these two compounds, which one will be
converted to the product with inverted configuration.
is treated with aq. KOH
solution. The rate of this reaction depends upon the concentration of the
compound 'A' only. When another optically active isomer 'B' of this compound
was treated with aq. KOH solution, the rate of reaction was found to be
dependent on concentration of compound and KOH both.
(i) Write down the structural formula of both
compounds 'A' and 'B'.
(ii) Out of these two compounds, which one will be
converted to the product with inverted configuration.
View Answer play_arrow
-
question_answer89)
Write the structures and names of
the compounds formed when compound 'A' with molecular formula, 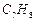 is treated with
is treated with 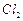 in the presence of
in the presence of 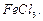
View Answer play_arrow
-
question_answer90)
Identify the products A and B formed
in the following reaction :
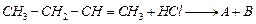
View Answer play_arrow
-
question_answer91)
Which of the following compounds
will have the highest melting point and why ?
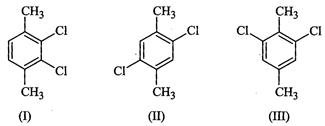
View Answer play_arrow
-
question_answer92)
Write down the structure and IUPAC
name for neopentyl bromide.
View Answer play_arrow
-
question_answer93)
A hydrocarbon of molecular mass 72 g
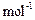 gives a single monochloro
derivative and two dichloro derivatives on photochlorination. Give the
structure of the hydrocarbon.
gives a single monochloro
derivative and two dichloro derivatives on photochlorination. Give the
structure of the hydrocarbon.
View Answer play_arrow
-
question_answer94)
Name the alkene which will yield
1-chloro-l-methylcyclohexane by its reaction with HCl. Write the reactions
involved.
View Answer play_arrow
-
question_answer95)
Which of the following haloalkanes
reacts with aqueous KOH most easily ? Explain giving reason.
(i) 1-Bromobutane (ii) 2-Bromobutane (iii) 2-Bromo-2-methylpropane (iv)
2-Chlorobutane
View Answer play_arrow
-
question_answer96)
Why can aryl halides not be prepared
by reaction of phenol with HC1 in the presence of 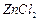 ?
?
View Answer play_arrow
-
question_answer97)
Which of the following compounds
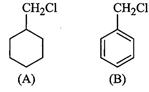
would undergo 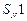 reaction faster
and why ?
reaction faster
and why ?
View Answer play_arrow
-
question_answer98)
Allyl chloride is hydrolysed more
readily than n-propyl chloride. Why ?
View Answer play_arrow
-
question_answer99)
Why is it necessary to avoid even
traces of moisture during the use of a Grignard reagent ?
View Answer play_arrow
-
question_answer100)
How do polar solvents help in the
first step in 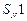 mechanism ?
mechanism ?
View Answer play_arrow
-
question_answer101)
Write a test to detect the presence
of double bond in a molecule.
View Answer play_arrow
-
question_answer102)
Diphenyls are potential threat to
the environment. How are these produced from aryl halides ?
View Answer play_arrow
-
question_answer103)
What are the IUPAC names of the
insecticide DDT and benzene hexachloride ? Why is their use banned in India and
other countries ?
View Answer play_arrow
-
question_answer104)
Elimination reactions (especially 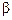 -elimination) are as common
as the nucleophilic substitution reaction in case of alkyl halides. Specify the
reagents used in both cases.
-elimination) are as common
as the nucleophilic substitution reaction in case of alkyl halides. Specify the
reagents used in both cases.
View Answer play_arrow
-
question_answer105)
How will you obtain monobromobenzene
from aniline ?
View Answer play_arrow
-
question_answer106)
Aryl halides are extremely less
reactive towards nucleophilic substitution. Predict and explain the order of
reactivity of the following compounds towards nucleophilic substitution
:
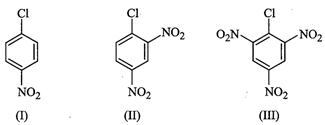
View Answer play_arrow
-
question_answer107)
tert-Butyl bromide reacts with aq.
NaOH by 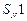 mechanism while w-butyl
bromide reacts by
mechanism while w-butyl
bromide reacts by 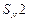 mechanism. Why
?
mechanism. Why
?
View Answer play_arrow
-
question_answer108)
Predict the major product formed
when HCl is added to isobutylene. Explain the mechanism involved.
View Answer play_arrow
-
question_answer109)
Discuss the nature of C-X bond in
the haloarenes.
View Answer play_arrow
-
question_answer110)
How can you obtain iodoethane from
ethanol when no other iodine containing reagent except Nal is available in the
laboratory ?
View Answer play_arrow
-
question_answer111)
Cyanide ion acts as an ambident
nucleophile. From which end it acts as a stronger nucleophile in aqueous medium
? Give reason for your answer.
View Answer play_arrow
-
question_answer112)
Match the compounds given in Column
I with the effects given in Column n.
Column I Column II
(a) Chloramphenicol (i) Malaria
(b) Thyroxine (ii)
Anaesthetic
(c) Chloroquine (iii) Typhoid fever
(d) Chloroform (iv) Goiter
(v) Blood substituent
View Answer play_arrow
-
question_answer113)
Match the items of Column I and
Column II.
Column I Column II
(a)  reaction (i)
vic-dibromides
(b) Chemicals in fire extinguisher
(ii) gem-dihalides
(c) Brommation of alkenes(iii) Racemisation
(d) Alkylidene halides (iv) Saytzeff rule
(e) Elimination of HX from alkyl halide
(v) Chlorobromocarbons
reaction (i)
vic-dibromides
(b) Chemicals in fire extinguisher
(ii) gem-dihalides
(c) Brommation of alkenes(iii) Racemisation
(d) Alkylidene halides (iv) Saytzeff rule
(e) Elimination of HX from alkyl halide
(v) Chlorobromocarbons
View Answer play_arrow
-
question_answer114)
Match the structures of compounds
given in Column I with the classes of compounds given in Column II.
Column
I Column II
(a) 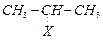 (i)
Aryl halide
(b)
(i)
Aryl halide
(b)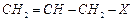 (ii) Alkyl
halide
(c) (iii)
Vinyl halide
(ii) Alkyl
halide
(c) (iii)
Vinyl halide
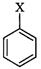 (d)
(d)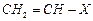 (iv)
Allyl halide
(iv)
Allyl halide
View Answer play_arrow
-
question_answer115)
Match the structures given in Column
I with the names given in Column II.
|
|
Column I
|
|
Column II
|
|
(a)
|
|
(i)
|
Nucleophilic aromatic substitution
|
|
(b)
|
|
(ii)
|
Electrophilic aromatic substitution
|
|
(c)
|
|
(iii)
|
Saytzeff elimination
|
|
(d)
|
|
(iv)
|
Electrophilic addition
|
|
(e)
|
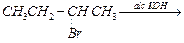 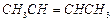
|
(v)
|
Nucleophilic substitution 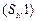
|
View Answer play_arrow
-
question_answer116)
Match the reactions given in Column
I with the names given in Column n.
|
|
Column I
|
|
Column II
|
|
(a)
|
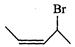
|
(i)
|
4-Bromopent-2-ene
|
|
(b)
|
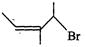
|
(ii)
|
4-Bromo-3-methylpent-2-ene
|
|
(c)
|
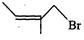
|
(iii)
|
1-Bromo-2-methylbut-2-ene
|
|
(d)
|
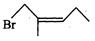
|
(iv)
|
1-Bromo-2-methylpent-2-ene
|
View Answer play_arrow
-
question_answer117)
Match the reactions given in Column
I with the names given in Column II.
|
|
Column I
|
|
Column II
|
|
(a)
|
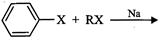 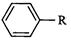
|
(i)
|
Fitting reaction
|
|
(b)
|
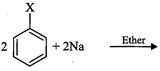 
|
(ii)
|
Wurtz Fitting reaction
|
|
(c)
|
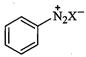 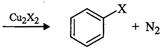
|
(iii)
|
Finkelstein reaction
|
|
(d)
|
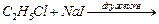 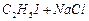
|
(iv)
|
Sandmeyer reaction
|
View Answer play_arrow
-
question_answer118)
Assertion
: Phosphorus chlorides (tri and penta) are preferred over thionyl chloride
for the preparation of alkyl chlorides from alcohols.
View Answer play_arrow
-
question_answer119)
Assertion
: The boiling points of alkyil halides decrease in the order :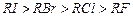
View Answer play_arrow
-
question_answer120)
Assertion
: KCN reacts with methyl chloride to give methyl isocyanide.
View Answer play_arrow
-
question_answer121)
Assertion
; tert-Butyl bromide undergoes Wurtz reaction to give 2, 2, 3,
3-tetramethylbutane.
View Answer play_arrow
-
question_answer122)
Assertion:
Presence of a nitro group at ortho or para position increases the reactivity of
haloarenes towards nucleophilic substitution.
View Answer play_arrow
-
question_answer123)
Assertion
: In monohaloarenes, further electrophilic substitution occurs at ortho and
para positions.
View Answer play_arrow
-
question_answer124)
Assertion
: Aryl iodides can be prepared by reaction of arenes with iodine in the
presence of an oxidizing agent.
View Answer play_arrow
-
question_answer125)
Assertion:
It is difficult to replace chlorine by -OH in chlorobenzene in comparison to
that in chloroethane
View Answer play_arrow
-
question_answer126)
Assertion
: Hydrolysis of (-)-2-bromooctane proceeds with inversion of configuration.
View Answer play_arrow
-
question_answer127)
Assertion
: Nitration of chlorobenzene leads to the formation of
m-nitrochlorobenzene.
View Answer play_arrow
-
question_answer128)
Some
alkyl halides undergo substitution whereas some undergo elimination reaction on
treatment with bases. Discuss the structural features of alkyl halides with the
help of examples which are responsible for this difference.
View Answer play_arrow
-
question_answer129)
Some
halogen containing compounds are useful in daily life. Some compounds of this
class are responsible for exposure of flora and fauna to more and more of UV
light which causes destruction to a great extent Name the class of these Halo
compounds. In your opinion, what should be done to minimize harmful effects of
these compounds.
View Answer play_arrow
-
question_answer130)
Why
are aryl halides less reactive towards nucleophilic substitution reactions than
alkyi halides ?
How can we enhance the reactivity of aryl halides ?
View Answer play_arrow
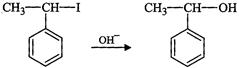
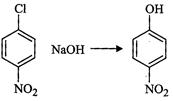
 (ii)
(ii) (iii)
(iii) (iv)
(iv)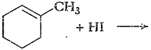 (v)
(v)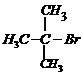 (iii)
(iii) 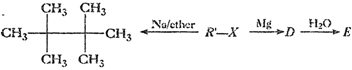
 (a) (b)
(a) (b)

![]() (c) (d)
(c) (d)

![]()
![]() (b)
(b)
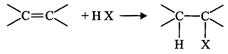 (c)
(c)![]() (d)
(d)


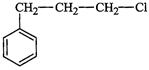 (c). (d)
(c). (d)


![]()
 (III)
(III)
 (a)
(a)![]() (b)
(b)![]() (c)
(c)![]() (d)
(d)![]()
![]()
 (III)
(III)
 (a)
(a)![]() (b)
(b)![]() (c)
(c)![]() (d)
(d)![]()
![]()
![]() (III)
(III)
 (a)
(a)![]() (b)
(b)![]() (c)
(c)![]() (d)
(d)![]()
![]()
![]() (III)
(III)
 (a) I < II < III (b)II < I <
III
(c)III < II < I (d)I < III
<II
(a) I < II < III (b)II < I <
III
(c)III < II < I (d)I < III
<II
![]() (b)
(b)![]() (c)
(c)![]() (d)
(d)![]()
![]()

![]() reaction faster
and why ?
reaction faster
and why ?

![]() mechanism ?
mechanism ?




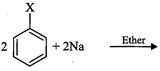


![]()
