-
question_answer1)
When an ideal monoatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
A)
2/5 done
clear
B)
3/5 done
clear
C)
3/7 done
clear
D)
5/7 done
clear
View Solution play_arrow
-
question_answer2)
The internal energy change in a system that has absorbed 2 Kcal of heat and done 500 J of work is
A)
8900 J done
clear
B)
6400 J done
clear
C)
5400 J done
clear
D)
7900 J done
clear
View Solution play_arrow
-
question_answer3)
In a given process on an ideal gas, \[dW=0\]and \[dQ\text{ }<\text{ }0.\]Then for the gas
A)
the temperature will decrease done
clear
B)
the volume will increase done
clear
C)
the pressure will remain constant done
clear
D)
the temperature will increase done
clear
View Solution play_arrow
-
question_answer4)
Unit mass of a liquid with volume \[{{V}_{1}}\] is completely changed into a gas of volume K, at a constant external pressure P and temperature T. If the latent heat of evaporation for the given mass is L, then the increase in the internal energy of the system is
A)
Zero done
clear
B)
\[P\left( {{V}_{2}}-{{V}_{1}} \right)\] done
clear
C)
\[L-P\left( {{V}_{2}}-{{V}_{1}} \right)\] done
clear
D)
\[L\] done
clear
View Solution play_arrow
-
question_answer5)
Two kg of water is converted into steam by boiling at atmospheric pressure. The volume changes from \[2\times {{10}^{-3}}{{m}^{3}}\] to \[3.34{{m}^{3}}.\]The work done by the system is about
A)
-340 kJ done
clear
B)
-170 kJ done
clear
C)
170U kJ done
clear
D)
340 kJ done
clear
View Solution play_arrow
-
question_answer6)
A gas is compressed from a volume of \[2{{m}^{3}}\] to a volume of \[1{{m}^{3}}\] at a constant pressure of\[100\text{ }N/{{m}^{2}}\]. Then it is heated at constant volume by supplying 150 J of energy. As a result, the internal energy of the gas:
A)
increases by 250 J done
clear
B)
decreases by 250 J done
clear
C)
increases by 50 J done
clear
D)
decreases by 50 J done
clear
View Solution play_arrow
-
question_answer7)
The internal energy of an ideal gas depends upon
A)
specific volume done
clear
B)
pressure done
clear
C)
temperature done
clear
D)
density done
clear
View Solution play_arrow
-
question_answer8)
Thermodynamics is concerned in part with transformations between
A)
different forms of heat energy done
clear
B)
internal energy at various temperatures done
clear
C)
one form of mechanical energy into other done
clear
D)
heat, internal energy and mechanical work done
clear
View Solution play_arrow
-
question_answer9)
110 joules of heat is added to a gaseous system, whose internal energy is 40J; then the amount of external work done is
A)
150 J done
clear
B)
70 J done
clear
C)
110 J done
clear
D)
40 J done
clear
View Solution play_arrow
-
question_answer10)
The change in internal energy of a thermo- dynamical system which has absorbed 2 kcal of heat and done 400 J of work is \[(1\text{ }cal=4.2J)\]
A)
2 kJ done
clear
B)
8 kJ done
clear
C)
3.5 kJ done
clear
D)
5.5 kJ done
clear
View Solution play_arrow
-
question_answer11)
If the amount of heat given to a system is 35 J and the amount of work done on the system is 15 J, then the change in internal energy of the system is
A)
-50 J done
clear
B)
20 J done
clear
C)
30 J done
clear
D)
50 J done
clear
View Solution play_arrow
-
question_answer12)
A perfect gas goes from a state A to another state B by absorbing \[8\times {{10}^{5}}J\] of heat and doing \[6.5\times {{10}^{5}}J\] of external work. It is now transferred between the same two states in another process in which it absorbs \[{{10}^{5}}J\] of heat. hi the second process
A)
work done by gas is \[{{10}^{5}}J\] done
clear
B)
work done on gas is \[{{10}^{5}}J\] done
clear
C)
work done by gas is \[0.5\times {{10}^{5}}J\] done
clear
D)
work done on the gas is \[0.5\times {{10}^{5}}\text{ }J\] done
clear
View Solution play_arrow
-
question_answer13)
Some of the thermodynamic parameters are state variables while some are process variables. Some grouping of the parameters are given. Choose the correct one
A)
State variables: Temperature, no. of moles Process variables: Internal energy, work done by the gas done
clear
B)
State variables: Volume, temperature Process variables: Internal energy, work done by the gas done
clear
C)
State variables: work done by the gas, heat rejected by the gas Process variables: Temperature, volume done
clear
D)
State variables: Internal energy, volume Process variables: Work done by the gas, heat absorbed by the gas done
clear
View Solution play_arrow
-
question_answer14)
An insulated container of gas has two chambers separated by an insulating partition. One of the chambers has volume \[{{V}_{1}}\] and contains ideal gas at pressure \[{{P}_{1}}\], and temperature \[{{T}_{1}}\] The other chamber has volume \[{{V}_{2}}\] and contains ideal gas at pressure \[{{P}_{2}}\] and temperature \[{{T}_{2}}\] If the partition is removed without doing any work on the gas, the final equilibrium temperature of the gas in the container will be
A)
\[\frac{{{T}_{1}}{{T}_{2}}\left( {{P}_{1}}{{V}_{1}}+{{P}_{2}}{{V}_{2}} \right)}{{{P}_{1}}{{V}_{1}}+{{P}_{2}}{{V}_{2}}}\] done
clear
B)
\[\frac{{{P}_{1}}{{V}_{1}}{{T}_{1}}+{{P}_{2}}{{V}_{2}}{{T}_{2}}}{{{P}_{1}}{{V}_{1}}+{{P}_{2}}{{V}_{2}}}\] done
clear
C)
\[\frac{{{P}_{1}}{{V}_{1}}{{T}_{2}}+{{P}_{2}}{{V}_{2}}{{T}_{1}}}{{{P}_{1}}{{V}_{1}}+{{P}_{2}}{{V}_{2}}}\] done
clear
D)
\[\frac{{{T}_{1}}{{T}_{2}}\left( {{P}_{1}}{{V}_{1}}+{{P}_{2}}{{V}_{2}} \right)}{{{P}_{1}}{{V}_{1}}{{T}_{1}}+{{P}_{2}}{{V}_{2}}{{T}_{2}}}\] done
clear
View Solution play_arrow
-
question_answer15)
A cube of side 5 cm made of iron and having a mass of 1500 g is heated from \[25{}^\circ C\] to \[400{}^\circ C.\]The specific heat for iron is \[0.12\text{ }cal/g{}^\circ C\]and the coefficient of volume expansion is \[3.5\times {{10}^{-5}}/{}^\circ C,\]the change in the internal energy of the cube is (atm pressure \[1\times {{10}^{5}}N/{{m}^{2}}\])
A)
320 kJ done
clear
B)
282 kJ done
clear
C)
141 kJ done
clear
D)
423 kJ done
clear
View Solution play_arrow
-
question_answer16)
4 kg of oxygen gas is heated so as to raise its temperature from 20 to\[120{}^\circ C\]. If the heating is done at constant pressure, the external work done by the gas is (\[{{C}_{p}}=0.219cal/g{}^\circ C\] and )
A)
628 kJ done
clear
B)
104 kJ done
clear
C)
366 kJ done
clear
D)
206 kJ done
clear
View Solution play_arrow
-
question_answer17)
Two cylinders A and B fitted with pistons contain equal amounts of an ideal diatomic gas at 300 K. The piston of A is free to move while that of B is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in A is 30 K, then the rise in temperature of the gas in B is
A)
30 K done
clear
B)
18 K done
clear
C)
50 K done
clear
D)
42 K done
clear
View Solution play_arrow
-
question_answer18)
The specific heat of a gas at constant pressure is greater than the specific heat of the same gas at constant volume because
A)
work is done in the expansion of the gas at constant pressure. done
clear
B)
work is done in the expansion of the gas at constant volume. done
clear
C)
the attraction between the molecules increases at constant pressure. done
clear
D)
the molecular attraction increases at constant volume. done
clear
View Solution play_arrow
-
question_answer19)
The specific heat of a gas in an isothermal process is
A)
infinite done
clear
B)
zero done
clear
C)
negative done
clear
D)
remains constant done
clear
View Solution play_arrow
-
question_answer20)
The work done in an adiabatic change in a particular gas depends only upon
A)
change in volume done
clear
B)
change in temperature done
clear
C)
change in pressure done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer21)
Which of the following holds good for an isochoric process?
A)
No work is done on the gas done
clear
B)
No work is done by the gas done
clear
C)
Both [a] and [b] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer22)
The slopes of isothermal and adiabatic curves are related as
A)
isothermal curve slope = adiabatic curve slope done
clear
B)
isothermal curve slope =\[\gamma \]\[\times \]adiabatic curve slope done
clear
C)
adiabatic curve slope =\[\gamma \]\[\times \]isothermal curve slope done
clear
D)
adiabatic curve slope =\[\frac{1}{2}\]\[\times \]isothermal curve slope done
clear
View Solution play_arrow
-
question_answer23)
2 k mol of hydrogen at NTP expands isobarically to twice its initial volume. The change in its internal energy is \[({{C}_{v}}=10kJ/kg.K\]and atm pressure \[=1\times 105N/{{m}^{2}})\]
A)
10.9 MJ done
clear
B)
9.10 MJ done
clear
C)
109 MJ done
clear
D)
1.09 MJ done
clear
View Solution play_arrow
-
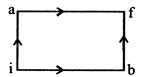
question_answer24)
When a system is taken a from state i to state f along the path iaf, it is found that \[Q=50\]cal and \[W=20cal.\], Along the path ibf \[Q=36\]cal. W along the path ibf is
A)
14 cal done
clear
B)
6 cal done
clear
C)
16 cal done
clear
D)
66 cal done
clear
View Solution play_arrow
-
question_answer25)
Choose the incorrect statement related to an isobaric process.
A)
\[\frac{V}{T}=\text{constant}\] done
clear
B)
\[W=P\Delta V\] done
clear
C)
Heat given to a system is used up in raising the temperature only. done
clear
D)
\[\Delta Q>W\] done
clear
View Solution play_arrow
-
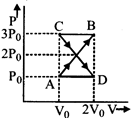
question_answer26)
A thermodynamic system undergoes cyclic process ABCDA as shown in fig. The work done by the system in the cycle is
A)
\[{{P}_{0}}{{V}_{0}}\] done
clear
B)
\[2{{P}_{0}}{{V}_{0}}\] done
clear
C)
\[\frac{{{P}_{0}}{{V}_{0}}}{2}\] done
clear
D)
Zero done
clear
View Solution play_arrow
-
question_answer27)
What will be the final pressure if an ideal gas in a cylinder is compressed adiabatically to \[\frac{1}{3}rd\] of its volume?
A)
Final pressure will be three times less than initial pressure. done
clear
B)
Final pressure will be three times more than initial pressure. done
clear
C)
Change in pressure will be more than three times the initial pressure. done
clear
D)
Change in pressure will be less than three times the initial pressure. done
clear
View Solution play_arrow
-
question_answer28)
In the P-V diagram, I is the initial state and F is the final state. The gas goes from I to F by (i) IAF, (ii) IBF, (iii) ICE The heat absorbed by the gas is

A)
the same in all three processes done
clear
B)
the same in (i) and (ii) done
clear
C)
greater in (i) than in (ii) done
clear
D)
the-same in (i) and (iii) done
clear
View Solution play_arrow
-
question_answer29)
A closed system undergoes a process \[1\to 2\] for which the values \[{{W}_{1-2}}\] and \[{{Q}_{1-2}}\] are 50 kJ and - 20 kJ respectively. If the system is returned to state 1 and \[{{Q}_{1\to 2}}+10kJ\] the work done \[{{W}_{1\to 2}}\]is
A)
40 kJ done
clear
B)
50 kJ done
clear
C)
-60 kJ done
clear
D)
-50 kJ done
clear
View Solution play_arrow
-
question_answer30)
During an adiabatic process of an ideal gas, if P is proportional to \[\frac{1}{{{V}^{1.5}}}\], then the ratio of specific heat capacities at constant pressure to that at constant volume for the gas is
A)
1.5 done
clear
B)
0.25 done
clear
C)
0.75 done
clear
D)
0.4 done
clear
View Solution play_arrow
-
question_answer31)
The relation between U, P and V for an ideal gas in an adiabatic process is given by relation \[U=\text{ }a+bPV.\] Find the value of adiabatic exponent \[\left( \gamma \right)\]of this gas.
A)
\[\frac{b+1}{b}\] done
clear
B)
\[\frac{b+1}{a}\] done
clear
C)
\[\frac{a+1}{b}\] done
clear
D)
\[\frac{a}{a+b}\] done
clear
View Solution play_arrow
-
question_answer32)
An ideal gas is initially at \[{{P}_{1}}\], \[{{V}_{1}}\] is expanded to\[{{P}_{2}}\], \[{{V}_{2}}\] and then compressed adiabatically to the same volume f[ and pressure \[{{P}_{3}}\] If W is the net work done by the gas in complete process which of the following is true?
A)
\[W>0;{{P}_{3}}>{{P}_{1}}\] done
clear
B)
\[W<0;{{P}_{3}}>{{P}_{1}}\] done
clear
C)
\[W>0;{{P}_{3}}<{{P}_{1}}\] done
clear
D)
\[W<0;{{P}_{3}}<{{P}_{1}}\] done
clear
View Solution play_arrow
-
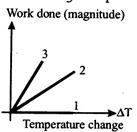
question_answer33)
For an ideal gas graph is shown for three processes, Process 1, 2 and 3 are respectively.
A)
Isobaric, adiabatic isochoric done
clear
B)
Adiabatic, isobaric, isochoric done
clear
C)
Isochoric, adiabatic, isobaric done
clear
D)
Isochoric, isobaric, adiabatic done
clear
View Solution play_arrow
-
question_answer34)
During an adiabatic process an object does 100 J of work and its temperature decreases by 5K. During another process it does 25J of work and its temperature decreases by 5K. Its heat capacity for 2nd process is
A)
20 J/K done
clear
B)
24 J/K done
clear
C)
15 J/K done
clear
D)
100 J/K done
clear
View Solution play_arrow
-
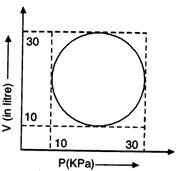
question_answer35)
Heat energy absorbed by a system in going through a cyclic process shown in the given figure is
A)
\[{{10}^{7}}\pi J\] done
clear
B)
\[{{10}^{4}}\pi J\] done
clear
C)
\[{{10}^{2}}\pi J\] done
clear
D)
\[{{10}^{-3}}\pi J\] done
clear
View Solution play_arrow
-
question_answer36)
The pressure inside a tyre is 4 times that of atmosphere. If the tyre bursts suddenly at temperature 300 K, what will be the new temperature?
A)
\[300\text{ }{{\left( 4 \right)}^{772}}\] done
clear
B)
\[300\text{ }{{\left( 4 \right)}^{2/7}}\] done
clear
C)
\[300\text{ }{{\left( 2 \right)}^{772}}\] done
clear
D)
\[300{{\left( 4 \right)}^{-2/7}}\] done
clear
View Solution play_arrow
-
question_answer37)
A monatomic ideal gas, initially at temperature \[{{T}_{1}}\] is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expand adiabatically to a temperature \[{{T}_{2}}\,\] by releasing the piston suddenly. If \[{{L}_{1}}\] and \[{{L}_{2}}\] are the length of the gas column before and after expansion respectively, then \[\frac{{{T}_{1}}}{{{T}_{2}}}\] is given by
A)
\[{{\left( \frac{{{L}_{1}}}{{{L}_{2}}} \right)}^{2/3}}\] done
clear
B)
\[\frac{{{L}_{1}}}{{{L}_{2}}}\] done
clear
C)
\[\frac{{{L}_{2}}}{{{L}_{1}}}\] done
clear
D)
\[{{\left( \frac{{{L}_{2}}}{{{L}_{1}}} \right)}^{2/3}}\] done
clear
View Solution play_arrow
-
question_answer38)
One mole of an ideal gas is taken from state A to state B by three different processes, (i) ACB (ii) ADB (iii) AEB as shown in the P-V diagram. The heat absorbed by the gas is-
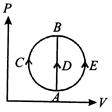
A)
greater in process (ii) than in (i) done
clear
B)
the least in process (ii) done
clear
C)
the same in (i) and (iii) done
clear
D)
less in (iii) than in (ii) done
clear
View Solution play_arrow
-
question_answer39)
During an isothermal expansion, a confined ideal gas does -150 J of work against its surroundings. This implies that
A)
150 J heat has been removed from the gas done
clear
B)
300 J of heat has been added to the gas done
clear
C)
no heat is transferred because the process is isothermal done
clear
D)
250 J of heat has been added to the gas done
clear
View Solution play_arrow
-
question_answer40)
A mass of diatomic gas \[(\gamma =1.4)\]at a pressure of 2 atmospheres is compressed adiabatically so that its temperature rises from \[27{}^\circ C\] to \[927{}^\circ C\]. The pressure of the gas in final state is
A)
28 atm done
clear
B)
68.7 atm done
clear
C)
256 atm done
clear
D)
8 atm done
clear
View Solution play_arrow
-
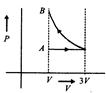
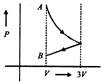
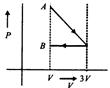
question_answer41)
One mole of an ideal gas goes from an initial stated to final state B via two processes: It first undergoes isothermal expansion from volume V to 3 V and then its volume is reduced from 3V to Fat constant pressure. The correct P-F diagram representing the two processes is:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer42)
One mole of an ideal gas at an initial temperature of T K does 6R joules of word adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is 5/3, the final temperature of gas will be
A)
\[(T-4)K\] done
clear
B)
\[(T+2.4)K\] done
clear
C)
\[(T-2.4)K\] done
clear
D)
\[(T+4)K\] done
clear
View Solution play_arrow
-
question_answer43)
In pressure-volume diagram, the isochoric, isothermal, isobaric and iso-entropic parts respectively, are
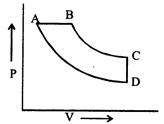
A)
BA, AD, DC, CB done
clear
B)
DC, CB, BA, AD done
clear
C)
AB, BC, CD, DA done
clear
D)
CD, DA, AB, BC done
clear
View Solution play_arrow
-
question_answer44)
A certain diatomic gas has the same specific heats as an ideal gas but a slightly different equation of state:\[PV=R(T+\alpha {{T}^{2}}),\]\[\alpha =0.001{{K}^{-1}}.\]The temperature of the gas is raised from \[{{T}_{1}}=300K\]to \[{{T}_{2}}\] at constant pressure. It is found that work done on the gas is 70% higher than what would be on an ideal gas. Choose the correct statement(s).
A)
\[{{T}_{2}}=400K,\]internal energy increases by 250R per mole. done
clear
B)
\[{{T}_{2}}=400K,\]internal energy increases by 350R per mole. done
clear
C)
Total heat absorbed in the process is 450R per mole. done
clear
D)
Total heat absorbed in the process in 520R per mole. done
clear
View Solution play_arrow
-
question_answer45)
Starting with the same initial conditions, an ideal gas expands from volume \[{{V}_{1}}\] to \[{{V}_{2}}\] in three different ways. The work done by the gas is \[{{W}_{1}}\] if the process is purely isothermal, \[{{W}_{2}}\] if purely isobaric and \[{{W}_{3}}\] if purely adiabatic. Then
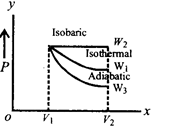
A)
\[{{W}_{2}}>{{W}_{1}}>{{W}_{3}}\] done
clear
B)
\[{{W}_{2}}>{{W}_{3}}>{{W}_{1}}\] done
clear
C)
\[{{W}_{1}}>{{W}_{2}}>{{W}_{3}}\] done
clear
D)
\[{{W}_{1}}>{{W}_{3}}>{{W}_{2}}\] done
clear
View Solution play_arrow
-
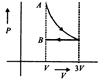
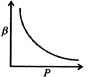
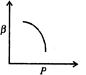
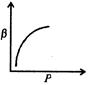
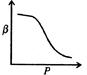
question_answer46)
Which of the following graphs correctly represents the variation of \[\beta =\frac{dV/dP}{V}\] with P for an ideal gas at constant temperature?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer47)
There are two processes ABC and DBF. In which of the process is the amount of work done by the gas greater?
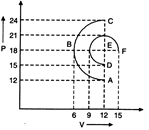
A)
ABC done
clear
B)
DEF done
clear
C)
Equal in both processes done
clear
D)
It cannot be predicted done
clear
View Solution play_arrow
-
question_answer48)
In a thermodynamic process, pressure of a fixed mass of gas is changed in such a manner that the gas molecules gives out 30 joules of heat and 10 J of work is done on the gas. If the initial internal energy of the gas was 40 J, then final internal energy will be
A)
zero done
clear
B)
80 J done
clear
C)
20 J done
clear
D)
-20 J done
clear
View Solution play_arrow
-
question_answer49)
Calculate the work done when 1 mole of a perfect gas is compressed adiabatically. The initial pressure and volume of the gas are \[{{10}^{5}}N/{{m}^{2}}\] and 6 liter respectively. The final volume of the gas is 2 liters. Molar specific heat of the gas at constant volume is 3R/2. [Given \[{{(3)}^{5/3}}=\text{ }6.19\]]
A)
-957 J done
clear
B)
+957 J done
clear
C)
-805 J done
clear
D)
+ 805 J. done
clear
View Solution play_arrow
-
question_answer50)
If a given mass of an ideal gas followed a relation VT = constant during a process, then which of the following graphs correctly represents the process?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer51)
The state of an ideal gas is changed through an isothermal process at temperature \[{{T}_{0}}\] as shown in figure. The work done by gas in going from state B to C is double the work done by gas in going from state A to B. If the pressure in the state B is \[{{P}_{0}}/2\] then the pressure of the gas in state C is
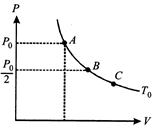
A)
\[{{P}_{0}}/2\] done
clear
B)
\[{{P}_{0}}/4\] done
clear
C)
\[{{P}_{0}}/6\] done
clear
D)
\[{{P}_{0}}/8\] done
clear
View Solution play_arrow
-
question_answer52)
Certain perfect gas is found to obey \[P{{V}^{3/2}}=\] constant during an adiabatic process. If such a gas at initial temperature T is adiabatically compressed to half the initial volume, its final temperature will be
A)
\[\sqrt{2}T\] done
clear
B)
\[~2T~~~\] done
clear
C)
\[2\sqrt{2}T\] done
clear
D)
\[4T\] done
clear
View Solution play_arrow
-
question_answer53)
Figure shows the variation of internal energy (U) with the pressure (P) of 2.0 mole gas in cyclic process abcda. The temperature of gas at c and d are 300 K and 500 K. calculate the heat absorbed by the gas during the process.

A)
400 R In 2 done
clear
B)
200 R In 2 done
clear
C)
100 R In 2 done
clear
D)
300 R In 2 done
clear
View Solution play_arrow
-
question_answer54)
A one mole sample of an ideal gas is carried around the thermodynamic cycle shown in the figure. The cycle consists of three steps: (i) an isothermal expansion \[(a\to b),\] (ii) an isobaric compression \[(b\to c),\] and (iii) a constant volume increases in pressure \[(c\to a).\] If \[{{T}_{a}}=400K,\] \[{{P}_{a}}=4atm,\]and \[{{P}_{b}}={{P}_{c}},=1atm,\] the work done by the gas per cycle is

A)
211.6 J done
clear
B)
1158 J done
clear
C)
705.3 J done
clear
D)
2116 J done
clear
View Solution play_arrow
-
question_answer55)
A 500 ml sealed cylinder contains nitrogen at a pressure of 1 atm. A tiny glass tube lies at the bottom of the cylinder. Its volume is 0.50 ml and it contains hydrogen at a pressure of 4.5 atm. The glass tube is broken so that hydrogen also fills the cylinder. The new pressure in the cylinder is \[(1\text{ }atm=1\times 105N/{{m}^{2}})\]
A)
76.34 cm Hg done
clear
B)
82.40 cm Hg done
clear
C)
94.24 cm Hg done
clear
D)
104.34 cm Hg done
clear
View Solution play_arrow
-
question_answer56)
In a reversible cyclic process of a gaseous system
A)
\[\Delta Q=\Delta U\] done
clear
B)
\[\Delta U=\Delta W\] done
clear
C)
\[\Delta W=0\] done
clear
D)
\[\Delta U=0\] done
clear
View Solution play_arrow
-
question_answer57)
Which of the following processes is irreversible?
A)
Transfer of heat by radiation done
clear
B)
Adiabatic changes performed slowly done
clear
C)
Extremely slow extension of a spring done
clear
D)
Isothermal changes performed slowly done
clear
View Solution play_arrow
-
question_answer58)
A Carnot engine works between a source and a sink maintained at constant temperatures \[{{T}_{1}}\] and \[{{T}_{2}}\], For efficiency to be the greatest
A)
\[{{T}_{1}}\] and \[{{T}_{2}}\], should be high done
clear
B)
\[{{T}_{1}}\] and \[{{T}_{2}}\] should be low done
clear
C)
\[{{T}_{1}}\] should be low and \[{{T}_{2}}\] should be high done
clear
D)
\[{{T}_{1}}\] should be high and \[{{T}_{2}}\] should be low done
clear
View Solution play_arrow
-
question_answer59)
The temperature of source and sink of a heat engine are \[127{}^\circ C\] and \[27{}^\circ C\] respectively. An inventor claims its efficiency to be 26%, then:
A)
it is impossible done
clear
B)
it is possible with high probability done
clear
C)
it is possible with low probability done
clear
D)
data are insufficient. done
clear
View Solution play_arrow
-
question_answer60)
A steam engine takes 0.1 kg of steam at \[100{}^\circ C\] per minute and cools it down to \[20{}^\circ C.\]What is the heat rejected by the steam engine per second? [Latent heat of vaporization of steam = 540 cal
A)
\[6.2\times {{10}^{4}}cal/\sec \] done
clear
B)
\[1.03\times {{10}^{3}}cal/\sec \] done
clear
C)
\[62\times {{10}^{3}}cal/sec\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer61)
If an air conditioner is put in the middle of a room and started working
A)
the room can be cooled slightly done
clear
B)
the temperature of the room will not change done
clear
C)
the room will become slightly warmer done
clear
D)
the same temperature will be attained in the room as by putting it on the window in the standard position done
clear
View Solution play_arrow
-
question_answer62)
The pressure-volume diagram shows six curved paths that can be followed by the gas (connected by vertical paths). Which two of them should be part of a closed cycle if the net work done by the gas is to be its maximum positive value?
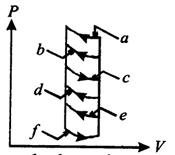
A)
af done
clear
B)
ae done
clear
C)
ac done
clear
D)
ce done
clear
View Solution play_arrow
-
question_answer63)
In the following P-V adiabatic cut two isothermals at temperatures \[{{T}_{1}}\]and \[{{T}_{2}}\](fig). The value of \[\frac{{{V}_{a}}}{{{V}_{c}}}\] will be
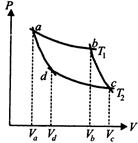
A)
\[\frac{{{V}_{b}}}{{{V}_{c}}}\] done
clear
B)
\[\frac{{{V}_{c}}}{{{V}_{b}}}\] done
clear
C)
\[\frac{{{V}_{d}}}{{{V}_{a}}}\] done
clear
D)
\[{{V}_{b}}{{V}_{c}}\] done
clear
View Solution play_arrow
-
question_answer64)
A diatomic ideal gas undergoes a thermodynamic change according to the P-V diagram shown in the figure. The total heat given to the gas is nearly (use ln \[2=0.7\])
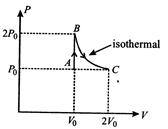
A)
\[2.5{{P}_{0}}{{V}_{0}}\] done
clear
B)
\[1.4{{P}_{0}}{{V}_{0}}\] done
clear
C)
\[1.1{{P}_{0}}{{V}_{0}}\] done
clear
D)
\[3.9{{P}_{0}}{{V}_{0}}\] done
clear
View Solution play_arrow
-
question_answer65)
In P-V diagram shown in figure AB C is a semicircle. The work done in the process ABC is
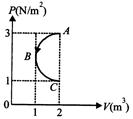
A)
4 J done
clear
B)
\[-\frac{\pi }{2}J\] done
clear
C)
\[\frac{\pi }{2}J\] done
clear
D)
zero done
clear
View Solution play_arrow
-
question_answer66)
On P-V coordinates, the slope of an isothermal curve of a gas at a pressure \[P=1MPa\] and volume \[F=0.0025{{m}^{3}}\] is equal to\[-400\text{ }MPa/{{m}^{3}}\] . If \[{{C}_{p}}/{{C}_{v}}=1.4,\] the slope of the adiabatic curve passing through this point is:
A)
\[-56\text{ }MPa/{{m}^{3}}\] done
clear
B)
\[-\,400\text{ }MPa/{{m}^{3}}\] done
clear
C)
\[-560\text{ }MPa/{{m}^{3}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer67)
A refrigerator with coefficient of performance releases 200 J of heat to a hot reservoir. Then the work done on the working substance is
A)
\[\frac{100}{3}J\] done
clear
B)
\[100J\] done
clear
C)
\[\frac{200}{3}J\] done
clear
D)
\[150J\] done
clear
View Solution play_arrow
-
question_answer68)
A Carnot engine takes \[3\times {{10}^{6}}cal.\] of heat from a reservoir at \[\,627{}^\circ C,\]and gives it to a sink at \[27{}^\circ C.\] The work done by the engine is
A)
\[4.2\times {{10}^{6}}J\] done
clear
B)
\[8.4\times {{10}^{6}}J\] done
clear
C)
\[16.8\times {{10}^{6}}J\] done
clear
D)
zero done
clear
View Solution play_arrow
-
question_answer69)
A diatomic ideal gas is used in a car engine as the working substance. If during the adiabatic expansion part of the cycle, volume of the gas increases from V to 32 V, the efficiency of the engine is
A)
0.5 done
clear
B)
0.75 done
clear
C)
0.99 done
clear
D)
0.25 done
clear
View Solution play_arrow
-
question_answer70)
A Carnot engine operating between temperatures \[{{T}_{1}}\]and \[{{T}_{2}}\] has efficiency\[\frac{1}{6}\]. When \[{{T}_{2}}\] is lowered by 62 K its efficiency increases to \[\frac{1}{3}\]. Then \[{{T}_{1}}\]and \[{{T}_{2}}\] are, respectively
A)
372 K and 330 K done
clear
B)
330 K and 268 K done
clear
C)
310 K and 248 K done
clear
D)
372 K and 310 K done
clear
View Solution play_arrow
-
question_answer71)
If the co-efficient of performance of a refrigerator is 5 and operates at the room temperature \[\]the temperature inside the refrigerator is
A)
240 K done
clear
B)
250 K done
clear
C)
230 K done
clear
D)
260 K done
clear
View Solution play_arrow
-
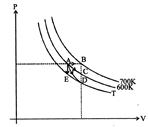
question_answer72)
The temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is

A)
\[\frac{1}{4}\] done
clear
B)
\[\frac{1}{2}\] done
clear
C)
\[\frac{2}{3}\] done
clear
D)
\[\frac{1}{3}\] done
clear
View Solution play_arrow
-
question_answer73)
A Carnot engine whose efficiency is 50% has an exhaust temperature of 500 K. If the efficiency is to be 60% with the same intake temperature, the exhaust temperature must be (in K)
A)
800 done
clear
B)
200 done
clear
C)
400 done
clear
D)
600 done
clear
View Solution play_arrow
-
question_answer74)
For an ideal gas four processes are marked as 1, 2, 3 and 4 on P-V diagram as shown in figure. The amount of heat supplied to the gas in the process 1, 2, 3 and 4 are \[{{Q}_{1}}\], \[{{Q}_{2}}\], \[{{Q}_{3}}\] and \[{{Q}_{4}}\] respectively, then correct order of heat supplied to the gas is [AB is process-1, AC is process-2, AD is adiabatic process-3 and AE is process-4]
A)
\[{{Q}_{1}}>{{Q}_{2}}>{{Q}_{3}}>{{Q}_{4}}\] done
clear
B)
\[{{Q}_{1}}>{{Q}_{2}}>{{Q}_{4}}>{{Q}_{3}}\] done
clear
C)
\[{{Q}_{1}}>{{Q}_{4}}>{{Q}_{2}}>{{Q}_{3}}\] done
clear
D)
\[{{Q}_{1}}<{{Q}_{2}}<{{Q}_{3}}<{{Q}_{4}}\] done
clear
View Solution play_arrow
-
question_answer75)
An ideal gas can be expanded from an initial state to a certain volume through two different processes, \[P{{V}^{2}}=K\] and \[P=K{{V}^{2}},\] where K is a positive constant. Then, choose the correct option from the following.
A)
Final temperature in will be greater than in (B) done
clear
B)
Final temperature in will be greater than in (A) done
clear
C)
Work done by the gas in both the processes would be equal done
clear
D)
Total heat given to the gas in is greater than in (B) done
clear
View Solution play_arrow
-
question_answer76)
Suppose 0.5 mole of an ideal gas undergoes an isothermal expansion as energy is added to its heat Q. Graph shows the final volume \[{{V}_{f}}\] versus Q. The temperature of the gas is (use ln 9 = 2 and\[R=\frac{25}{3}J/mol-K\])
A)
360 K done
clear
B)
293 K done
clear
C)
386 K done
clear
D)
412 K done
clear
View Solution play_arrow
-
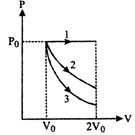
question_answer77)
A gas is expanded from volume \[{{V}_{0}}\] to \[2{{V}_{0}}\] under three different processes. Process 1 is isobaric process, process 2 is isothermal and process is adiabatic. Let\[\Delta {{U}_{1}}\], \[\Delta {{U}_{2}}\]and \[\Delta {{U}_{3}}\]be the change in internal energy of the gas is these three processes. Then-
A)
\[\Delta {{U}_{1}}>\Delta {{U}_{2}}>\Delta {{U}_{3}}\] done
clear
B)
\[\Delta {{U}_{1}}<\Delta {{U}_{2}}<\Delta {{U}_{3}}\] done
clear
C)
\[\Delta {{U}_{2}}<\Delta {{U}_{1}}<\Delta {{U}_{3}}\] done
clear
D)
\[\Delta {{U}_{2}}<\Delta {{U}_{3}}<\Delta {{U}_{1}}\] done
clear
View Solution play_arrow
-
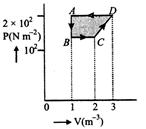
question_answer78)
The P-V diagram of a gas system undergoing cyclic process is shown here. The work done during isobaric compression is
A)
100 J done
clear
B)
200 J done
clear
C)
600 J done
clear
D)
400 J done
clear
View Solution play_arrow
-
question_answer79)
1 gm of water at a pressure of \[1.01\times {{10}^{5}}\] Pa is converted into steam without any change of temperature. The volume of 1 g of steam is 1671 cc and the latent heat of evaporation is 540 cal. The change in internal energy due to evaporation of 1 gm of water is
A)
\[\approx 167\text{ }cal\] done
clear
B)
\[\approx 500\text{ }cal\] done
clear
C)
540 cal done
clear
D)
58 cal done
clear
View Solution play_arrow
-
question_answer80)
A system changes from the state \[({{P}_{1}},\text{ }{{V}_{1}})\] to \[({{P}_{2}},\text{ }{{V}_{2}})\] as shown in the figure. What is the work done by the system?
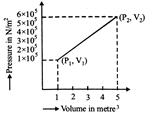
A)
\[7.5\times {{10}^{5}}joule~\] done
clear
B)
\[7.5\times {{10}^{5}}erg\] done
clear
C)
\[12\times {{10}^{5}}joule\] done
clear
D)
\[6\times {{10}^{5}}joule\] done
clear
View Solution play_arrow
-
question_answer81)
When the state of a gas adiabatically changed from an equilibrium state A to another equilibrium state B an amount of work done on the system is 35 J. If the gas is taken from state A to B via process in which the net heat absorbed by the system is 12 cal, then the net work done by the system is \[\left( 1\text{ }cal=4.19J \right)\]
A)
13.2 J done
clear
B)
15.4 J done
clear
C)
12.6 J done
clear
D)
16.8 J done
clear
View Solution play_arrow
-
question_answer82)
A Carnot engine is working between \[127{}^\circ C\] and \[27{}^\circ C.~\]The increase in efficiency will be maximum when the temperature of
A)
the source is increased by \[50{}^\circ C\] done
clear
B)
the sink is decreased by\[50{}^\circ C\] done
clear
C)
source is increased by \[25{}^\circ C\]and that of sink is decreased by \[25{}^\circ C\] done
clear
D)
both source and sink are decreased by \[25{}^\circ C\] done
clear
View Solution play_arrow
-
question_answer83)
When 1 kg of ice at \[0{}^\circ C\] melts to water at \[0{}^\circ C,\]the resulting change in its entropy, taking latent heat of ice to be 80 cal/\[{}^\circ C\], is
A)
273 cal/K done
clear
B)
253 cal/K done
clear
C)
263 cal/K done
clear
D)
293 cal/K done
clear
View Solution play_arrow
-
question_answer84)
If the energy input to a Carnot engine is thrice the work it performs then, the fraction of energy rejected to the sink is
A)
\[\frac{1}{3}\] done
clear
B)
\[\frac{1}{4}\] done
clear
C)
\[\frac{2}{5}\] done
clear
D)
\[\frac{2}{3}\] done
clear
View Solution play_arrow
-
question_answer85)
The efficiency of an ideal gas with adiabatic exponent \['\gamma '\] for the shown cyclic process would be
A)
\[\frac{\left( 2\,l\text{n}2-1 \right)}{\gamma /\left( \gamma -1 \right)}\] done
clear
B)
\[\frac{\left( 1-2\,l\text{n}2 \right)}{\gamma /\left( \gamma -1 \right)}\] done
clear
C)
\[\frac{\left( 2l\text{n}2+1 \right)}{\gamma /\left( \gamma -1 \right)}\] done
clear
D)
\[\frac{\left( 2l\text{n}2-1 \right)}{\gamma /\left( \gamma +1 \right)}\] done
clear
View Solution play_arrow
-
question_answer86)
In a Carnot engine, the temperature of reservoir is \[927{}^\circ C\] and that of sink is\[27{}^\circ C\]. If the work done by the engine when it transfers heat from reservoir to sink is \[12.6\times {{10}^{6}}J,\] the quantity of heat absorbed by the engine from the reservoir is
A)
\[16.8\times {{10}^{6}}J\] done
clear
B)
\[4\times {{10}^{6}}J\] done
clear
C)
\[7.6\times {{10}^{6}}J\] done
clear
D)
\[4.2\times {{10}^{6}}J\] done
clear
View Solution play_arrow
-
question_answer87)
A monoatomic ideal gas goes through a process \[p={{p}_{0}}-\alpha V\] where \[{{p}_{0}}\] and \[\alpha \] are positive constants and V is its volume. At what volume will the entropy of gas be maximum?
A)
\[\frac{5{{p}_{0}}}{6\alpha }\] done
clear
B)
\[\frac{{{p}_{0}}}{2\alpha }\] done
clear
C)
\[\frac{{{p}_{0}}}{4\alpha }\] done
clear
D)
\[\frac{5{{p}_{0}}}{8\alpha }\] done
clear
View Solution play_arrow
-
question_answer88)
An ideal gas has temperature\[{{T}_{1}}\], at the initial state shown in the P- V diagram. The gas has a higher ternperature \[\,{{T}_{2}}\] at the final states a and b, which it can reach the paths shown. The change in entropy:
A)
greatest in a done
clear
B)
greatest in b done
clear
C)
same in a and b done
clear
D)
nothing can be said done
clear
View Solution play_arrow
-
question_answer89)
In Carnot engine efficiency is 40% at hot reservoir temperature T. For efficiency 50% what will be temperature of hot reservoir?
A)
\[\frac{T}{5}\] done
clear
B)
\[\frac{2T}{5}\] done
clear
C)
\[6T\] done
clear
D)
\[\frac{6T}{5}\] done
clear
View Solution play_arrow
-
question_answer90)
An ideal gas is subjected to cyclic process involving four thermodynamic states, the amounts of heat (Q) and work (W) involved in each of these states \[{{Q}_{1}}=6000J;{{Q}_{2}}=-5500J;{{Q}_{3}}=-3000J\] \[{{Q}_{4}}=+\,3500J\] \[{{W}_{1}}=2500J;{{W}_{2}}=-1000J;{{W}_{3}}=-1200J\] \[{{W}_{4}}=xJ\] The ratio of the net work done by the gas to the total heat absorbed by the gas is \[\eta \]. The values of x and n respectively are
A)
500; 7.5% done
clear
B)
700; 10.5% done
clear
C)
1000; 21% done
clear
D)
1500; 15% done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
