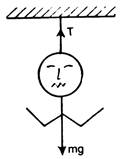
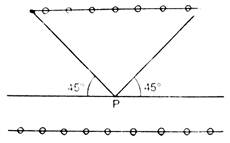
question_answer 1) A two metre wire is moving with a velocity of 1 m/s perpendicularly to a magnetic field of 0.5 weber/\[{{m}^{2}}\]. The emf induced in it will be:
A)
0.1 volt
done
clear
B)
0.25 volt
done
clear
C)
0.75 volt
done
clear
D)
1 volt
done
clear
View Answer play_arrow
question_answer 2) A uniform chain of mass m and length f is held on a horizontal frictionless table with \[\frac{1}{3}\]rd of its length hanging vertically over the edge of the table. The work done in pulling hanging part of the chain up on the table will be:
A)
\[\frac{gml}{4}\]
done
clear
B)
\[\frac{gm{{l}^{2}}}{2}\]
done
clear
C)
\[\frac{gml}{\sqrt{2}}\]
done
clear
D)
\[\frac{gml}{18}\]
done
clear
View Answer play_arrow
question_answer 3) A body, moving in a straight line with an initial velocity of 5 m/s and a constant acceleration, covers a distance of 30 m in the 3rd sec. How much distance will it cover in the next two seconds:
A)
60 m
done
clear
B)
85 m
done
clear
C)
90 m
done
clear
D)
100 m
done
clear
View Answer play_arrow
question_answer 4) A circular ring of mass M and radius R is rotating about it axis at an angular frequency\[\omega \]. Two blocks each of mass mare gently placed on the opposite ends of a diameter of the ring. The angular frequency becomes\[\omega \]. Then the ratio of\[\frac{\omega }{\omega }\]will be:
A)
\[\frac{M}{M+2m}\]
done
clear
B)
\[\frac{m}{2M}\]
done
clear
C)
\[\frac{m}{2M+M}\]
done
clear
D)
\[\frac{2M}{(2m+M)}\]
done
clear
View Answer play_arrow
question_answer 5) A man walks for 1 min at a speed of 1 m/s and then run for 1 min at 3 m/s along a straight track. What is the average speed of the man?
A)
2 m/s
done
clear
B)
4 m/s
done
clear
C)
3 m/s
done
clear
D)
1 m/s
done
clear
View Answer play_arrow
question_answer 6) A force \[(3\hat{i}+2\hat{j})\]N displaces an object through a distance \[(2\hat{i}+3\hat{j})\] m. The work done will be:
A)
15 J
done
clear
B)
11 J
done
clear
C)
4 J
done
clear
D)
zero J
done
clear
View Answer play_arrow
question_answer 7) The maximum range of a gun on a horizontal terrain is 16 km. If g = 10 m/s , what must be the muzzle velocity of the shell ?
A)
50 m/s
done
clear
B)
200 m/s
done
clear
C)
400 m/s
done
clear
D)
100 m/s
done
clear
View Answer play_arrow
question_answer 8)
With what minimum acceleration can a fireman slide down a rope whose bricking strength is two third of his weight?
A)
\[\frac{g}{3}\]
done
clear
B)
\[\frac{g}{6}\]
done
clear
C)
\[\frac{g}{2}\]
done
clear
D)
\[\frac{g}{9}\]
done
clear
View Answer play_arrow
question_answer 9) A vehicle of mass 100 kg is moving with a velocity of 5 m/s. To stop it in \[\frac{1}{10}\] sec, the required force in opposite direction is :
A)
5000 N
done
clear
B)
2500 N
done
clear
C)
500 N
done
clear
D)
100 N
done
clear
View Answer play_arrow
question_answer 10) The kinetic friction is always :
A)
greater than rolling friction
done
clear
B)
may be less than, greater or equal to rolling friction
done
clear
C)
equal to rolling friction
done
clear
D)
less than rolling friction
done
clear
View Answer play_arrow
question_answer 11) A body weighs W newton on the surface of the earth. Its weight at a height equal to half the radius of the earth will be :
A)
\[\frac{5W}{6}\]
done
clear
B)
\[\frac{W}{6}\]
done
clear
C)
\[\frac{3W}{2}\]
done
clear
D)
\[\frac{4W}{9}\]
done
clear
View Answer play_arrow
question_answer 12) A small solid sphere rolls down without slipping a \[30{}^\circ \] inclined plane. Its acceleration is \[(g=10\text{ }m/{{s}^{2}})\]:
A)
\[\frac{3}{2}m/{{s}^{2}}\]
done
clear
B)
\[\frac{9}{15}m/{{s}^{2}}\]
done
clear
C)
\[\frac{3}{25}m/{{s}^{2}}\]
done
clear
D)
\[\frac{25}{7}m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 13) The escape velocity of a body from the earth is 11.2 km/s. If the radius of a planet be half the radius of earth and its mass be one fourth that of earth. The escape velocity for the planet will be :
A)
8 km/s
done
clear
B)
4 km/s
done
clear
C)
6 km/s
done
clear
D)
12 km/s
done
clear
View Answer play_arrow
question_answer 14) A simple harmonic oscillator oscillates with a frequency f. Then the potential energy varies with the frequency:
A)
\[2f\]
done
clear
B)
\[\frac{f}{2}\]
done
clear
C)
\[\frac{f}{3}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 15) A 2 MeV proton is moving perpendicular to a uniform magnetic field of 2.5 tesla. What is the magnetic force on the proton? \[({{M}_{p}}=1.6\times {{10}^{-27}}kg):\]
A)
\[8\times {{10}^{-12}}N\]
done
clear
B)
\[4\times {{10}^{-12}}N\]
done
clear
C)
\[6\times {{10}^{-12}}N\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 16)
The magnetic field at P on the axis of a solenoid having 100 turns/metre and carrying a current of 5 A is :
A)
\[500\sqrt{2}{{\mu }_{0}}\]
done
clear
B)
\[300\sqrt{2}{{\mu }_{0}}\]
done
clear
C)
\[200\sqrt{2}{{\mu }_{0}}\]
done
clear
D)
\[250\sqrt{2}{{\mu }_{0}}\]
done
clear
View Answer play_arrow
question_answer 17) If 2% of the main current is to be passed through the galvanometer of resistance G. The resistance of the shunt requires is :
A)
49G
done
clear
B)
\[\frac{3G}{50}\]
done
clear
C)
50G
done
clear
D)
\[\frac{G}{49}\]
done
clear
View Answer play_arrow
question_answer 18) The earths magnetic field at a certain place has a horizontal component 0.4 gauss where angle of dip is 60°. Then the total intensity of earth will be :
A)
\[0.2\sqrt{3}\]gauss
done
clear
B)
0.8 gauss
done
clear
C)
0.2 gauss
done
clear
D)
\[\frac{0.8}{\sqrt{3}}\] gauss
done
clear
View Answer play_arrow
question_answer 19) A dynamo dissipates 20 watt when it supplies a current of 4 amp through it. If the terminal potential difference is 220 V. What is the emf produced?
A)
225 volt
done
clear
B)
240 volt
done
clear
C)
220 volt
done
clear
D)
200 volts
done
clear
View Answer play_arrow
question_answer 20) Self-inductance of a coil is 5 mH. If a current of 2 A is flown. What is the magnetic flux through the coil?
A)
0.04 weber
done
clear
B)
0.05 weber
done
clear
C)
0.2 weber
done
clear
D)
0.01 weber
done
clear
View Answer play_arrow
question_answer 21) Two rods of different materials having coefficients of the thermal expansion \[{{\alpha }_{1}},{{\alpha }_{2}}\]and Youngs moduli,\[{{\gamma }_{1}},{{\gamma }_{2}}\] respectively are fixed between two rigid massive walls. The rods are heated such that they undergo the same increase in temperature. There is no bending of the rods. If \[{{\alpha }_{1}}:{{\alpha }_{2}}=2:3\]. The thermal, stresses developed in the two rods are equal provided, \[{{\gamma }_{1}}:{{\gamma }_{2}}\]is equal to :
A)
5 : 3
done
clear
B)
1 : 2
done
clear
C)
9 : 4
done
clear
D)
3 : 2
done
clear
View Answer play_arrow
question_answer 22) A tuning fork of frequency 90 Hz is sounded and moved towards an stationary observer with a velocity equal to one tenth of sound. The frequency of note heard by the observer is :
A)
200 Hz
done
clear
B)
100 Hz
done
clear
C)
50 Hz
done
clear
D)
150 Hz
done
clear
View Answer play_arrow
question_answer 23) The focal length of the objective and eye piece of a telescope are respectively 200 cm and 5 cm. The maximum magnifying power of telescope will be :
A)
40
done
clear
B)
48
done
clear
C)
80
done
clear
D)
20
done
clear
View Answer play_arrow
question_answer 24) If we consider electrons and photons of the same wavelength, then they will have the same:
A)
velocity
done
clear
B)
angular momentum
done
clear
C)
energy
done
clear
D)
linear momentum
done
clear
View Answer play_arrow
question_answer 25) The dispersive power of the material of the prism if \[{{\mu }_{v}}=\]1.524 and \[{{\mu }_{R}}=\]1.514, will be:
A)
0.19
done
clear
B)
1.19
done
clear
C)
0.29
done
clear
D)
0.019
done
clear
View Answer play_arrow
question_answer 26) Which series of hydrogen atom lie in infrared region ?
A)
Brackett, Paschen, Pound
done
clear
B)
Lyman
done
clear
C)
Balmer
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 27) The wavelength of X-rays is of the order of:
A)
1 Fermi
done
clear
B)
\[1\overset{0}{\mathop{A}}\,\]
done
clear
C)
1 mm
done
clear
D)
1 micron
done
clear
View Answer play_arrow
question_answer 28) \[\alpha \]-particles are :
A)
electromagnetic wave
done
clear
B)
negatively charged particles
done
clear
C)
protons
done
clear
D)
ionised helium atom
done
clear
View Answer play_arrow
question_answer 29) An electron and a positron each of mass \[\frac{1}{2000}\]amu annihilate each other and produce two photons of equal energy The minimum energy of each photon produced, will be :
A)
0.0465 MeV
done
clear
B)
4.65 MeV
done
clear
C)
0.465 MeV
done
clear
D)
0.565 MeV
done
clear
View Answer play_arrow
question_answer 30) A coil develops heat of 800 cal/sec when 20 volts is applied across its ends. The resistance of the coil is : (1cal = 4.2 J)
A)
0.14\[\Omega \]
done
clear
B)
0.12 \[\Omega \]
done
clear
C)
1.4\[\Omega \]
done
clear
D)
1.2\[\Omega \]
done
clear
View Answer play_arrow
question_answer 31) The equation of stationary wave is\[y=0.8\cos \left( \frac{\pi x}{20} \right)\sin 200\pi t\] where x is in cm and t is in sec. The separation between two consecutive nodes will be :
A)
5 cm
done
clear
B)
1.5 cm
done
clear
C)
10 cm
done
clear
D)
20 cm
done
clear
View Answer play_arrow
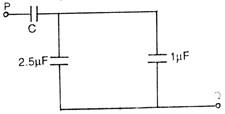
question_answer 32)
The equivalent capacitance between P and Q in the given figure is lf if. Then the value of capacitance C will be :
A)
\[1.2\mu F\]
done
clear
B)
\[3.6\mu F\]
done
clear
C)
\[2\mu F\]
done
clear
D)
\[1.4\mu F\]
done
clear
View Answer play_arrow
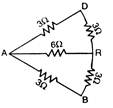
question_answer 33)
The effective resistances between the points P and R in the given figure is :
A)
4 \[\Omega \]
done
clear
B)
3 \[\Omega \]
done
clear
C)
2.5\[\Omega \]
done
clear
D)
2\[\Omega \]
done
clear
View Answer play_arrow
question_answer 34) n-type semiconductor is prepared by doping Si in a semiconductor by a :
A)
pentavalent impurity
done
clear
B)
trivalent impurity
done
clear
C)
divalent impurity
done
clear
D)
monovalent impurity
done
clear
View Answer play_arrow
question_answer 35) In any Bohrs orbit of the hydrogen atom, the ratio of kinetic energy to potential energy of the electron is :
A)
-2
done
clear
B)
\[-\frac{1}{2}\]
done
clear
C)
2
done
clear
D)
\[\frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 36) When\[_{92}{{U}^{235}}\] undergoes fission 0.1% of its original mass is changed into energy. How much energy is released if 1 kg of \[_{92}{{U}^{235}}\] undergoes fission?
A)
\[9\times {{10}^{10}}J\]
done
clear
B)
\[9\times {{10}^{12}}J\]
done
clear
C)
\[9\times {{10}^{11}}J\]
done
clear
D)
\[9\times {{10}^{10}}J\]
done
clear
View Answer play_arrow
question_answer 37) The magnetic field due to a small bar magnet at a large distance varies as :
A)
\[\frac{1}{d}\]
done
clear
B)
\[\frac{1}{{{d}^{3}}}\]
done
clear
C)
\[\frac{1}{{{d}^{3/2}}}\]
done
clear
D)
\[\frac{1}{{{d}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 38) Mass number of a nucleus is equal to the number of:
A)
nucleons
done
clear
B)
neutrons
done
clear
C)
protons
done
clear
D)
electrons
done
clear
View Answer play_arrow
question_answer 39) The momentum of a photon in an X-ray beam of \[{{10}^{-10}}\]metre wavelength is
A)
\[2.2\times {{10}^{-50}}kg\,m/s\]
done
clear
B)
\[6.0\times {{10}^{-44}}kg\,m/s\]
done
clear
C)
\[1.5\times {{10}^{-23}}kg\,m/s\]
done
clear
D)
\[5.9\times {{10}^{-24}}kg\,m/s\]
done
clear
View Answer play_arrow
question_answer 40) Work function of a metal is 2.51 cV. Its threshold frequency is :
A)
\[6.08\times {{10}^{14}}\,cycke/s\]
done
clear
B)
\[4\times {{10}^{11}}\,cycke/s\]
done
clear
C)
\[6.5\times {{10}^{4}}\,cycke/s\]
done
clear
D)
\[6.6\times {{10}^{24}}\,cycke/s\]
done
clear
View Answer play_arrow
question_answer 41) The half-life of a radio-active substance is 10 hours. Its mean life is :
A)
6.93 h
done
clear
B)
20 h
done
clear
C)
7.2 h
done
clear
D)
14.4 h
done
clear
View Answer play_arrow
question_answer 42) The displacement of progressive wave along y-axis is described by the equation \[y-5sin(60\text{ }t+2x).\] Here x,y are in metres and t is in seconds. The wave represented by this equation has :
A)
frequency equal to \[\frac{15}{\pi }Hz\]
done
clear
B)
velocity equals to 30 m/s along -ve direction
done
clear
C)
time period of oscillation equal to \[\frac{\pi }{15}s\]
done
clear
D)
wavelength equal to \[2\pi \]metres
done
clear
View Answer play_arrow
question_answer 43) The slope of isothermal and adiabatic curves are related as :
A)
slope of adiabatic curve = slope of isothermal curve
done
clear
B)
slope of adiabatic curve = y x slope of isothermal curve
done
clear
C)
slope of isothermal curve = y x slope of adiabatic Curve
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 44) A molecule of triatomic gas has :
A)
4 degrees of freedom
done
clear
B)
6 degrees of freedom
done
clear
C)
5 degrees of freedom
done
clear
D)
3 degrees of freedom
done
clear
View Answer play_arrow
question_answer 45) The temperature of body is doubled then thermal conductivity will :
A)
increase or decrease
done
clear
B)
remain same
done
clear
C)
be doubled
done
clear
D)
be halved
done
clear
View Answer play_arrow
question_answer 46) The phase difference between two points separated by 6 m in a wave of frequency 100 Hz is 540° . The wave velocity is:
A)
400 m/s
done
clear
B)
200 m/s
done
clear
C)
100 m/s
done
clear
D)
50 m/s
done
clear
View Answer play_arrow
question_answer 47) A star emitting alight of wavelength 5000\[\overset{0}{\mathop{A}}\,\]moves away from the earth with a speed of 6 km/s. The shift in wavelength due to Dopplers effect will be:
A)
10 \[\overset{0}{\mathop{A}}\,\]
done
clear
B)
1\[\overset{0}{\mathop{A}}\,\]
done
clear
C)
0.1\[\overset{0}{\mathop{A}}\,\]
done
clear
D)
0.01\[\overset{0}{\mathop{A}}\,\]
done
clear
View Answer play_arrow
question_answer 48) The ratio of frequencies of the shortest wavelengths of Balmer and Lyman series of hydrogen is:
A)
4:1
done
clear
B)
1: 4
done
clear
C)
27 : 5
done
clear
D)
5: 27
done
clear
View Answer play_arrow
question_answer 49) If h is the Plancks constant, the momentum of a photon of wavelength 0.01 \[\overset{0}{\mathop{A}}\,\] will be :
A)
\[{{10}^{-3}}h\]
done
clear
B)
\[{{10}^{11}}h\]
done
clear
C)
\[{{10}^{13}}h\]
done
clear
D)
\[{{10}^{12}}h\]
done
clear
View Answer play_arrow
question_answer 50) In npn transistor circuit the collector current is 10 mA. If 90% of the electrons reach the collector, then emitter current will be:
A)
0.1 mA
done
clear
B)
1 mA
done
clear
C)
9 mA
done
clear
D)
near by 11 mA
done
clear
View Answer play_arrow
question_answer 51) Which of the following is most stable among \[C{{u}^{+}},\,F{{e}^{+}},F{{e}^{2+}}\]and \[F{{e}^{3+}}\]?
A)
\[C{{u}^{+}}\]
done
clear
B)
\[F{{e}^{+}}\]
done
clear
C)
\[F{{e}^{2+}}\]
done
clear
D)
\[F{{e}^{3+}}\]
done
clear
View Answer play_arrow
question_answer 52) In the ground state of phosphorus total number of unpaired electrons are:
A)
3
done
clear
B)
5
done
clear
C)
8
done
clear
D)
9
done
clear
View Answer play_arrow
question_answer 53) \[H-O-H\]bond angle in \[{{H}_{2}}O\] is \[{{104.5}^{o}}\] and not \[{{109}^{o}}28\] because of:
A)
lone pair-lone pair repulsion
done
clear
B)
lone pair-bond pair repulsion
done
clear
C)
bond pair-bond pair repulsion
done
clear
D)
high electronegativity of oxygen
done
clear
View Answer play_arrow
question_answer 54) Oxidation state of \[I\] in \[{{H}_{4}}IO_{6}^{-}\] is :
A)
\[-1\]
done
clear
B)
\[+5\]
done
clear
C)
\[+6\]
done
clear
D)
\[+7\]
done
clear
View Answer play_arrow
question_answer 55) Hard water is not fit for washing clothes because:
A)
it contains impurities
done
clear
B)
it is acidic in nature
done
clear
C)
it gives precipitate with soap
done
clear
D)
it contains \[N{{a}_{2}}S{{O}_{4}}\] and \[KCl\]
done
clear
View Answer play_arrow
question_answer 56) The half life of a radioactive isotope is 2.5 hour. If the initial mass of the isotope was 16 g, the mass of it remain undecayed after 10 hours will be:
A)
32 g
done
clear
B)
4 g
done
clear
C)
16 g
done
clear
D)
1 g
done
clear
View Answer play_arrow
question_answer 57) Total number of unpaired electrons in \[{{O}_{2}}\] molecule is:
A)
\[0\]
done
clear
B)
\[1\]
done
clear
C)
\[2\]
done
clear
D)
\[3\]
done
clear
View Answer play_arrow
question_answer 58) Conjugate acid of \[{{S}_{2}}{{O}_{8}}^{2-}\] is:
A)
\[{{H}_{2}}{{S}_{2}}{{O}_{8}}\]
done
clear
B)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
\[H{{S}_{2}}O_{8}^{-}\]
done
clear
D)
\[HSO_{4}^{-}\]
done
clear
View Answer play_arrow
question_answer 59) Which of the following element exhibit maximum valency?
A)
\[Cu\]
done
clear
B)
\[Os\]
done
clear
C)
\[Pt\]
done
clear
D)
\[Cs\]
done
clear
View Answer play_arrow
question_answer 60) Which of the following contains maximum ionic radius?
A)
\[{{C}^{4-}}\]
done
clear
B)
\[{{N}^{3-}}\]
done
clear
C)
\[{{O}^{2-}}\]
done
clear
D)
\[M{{g}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 61) Which of the following represents isoelectronic sequence?
A)
\[N,\,O,\,F,\,Ne\]
done
clear
B)
\[N{{a}^{+}},M{{n}^{2+}},A{{l}^{3+}},S{{i}^{4+}}\]
done
clear
C)
\[C{{l}^{-}},Ar,C{{a}^{2+}},T{{i}^{4+}}\]
done
clear
D)
\[Be,M{{g}^{2+}},Ca,S{{i}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 62) The indicator used in the titration of oxalic acid and sodium hydroxide is:
A)
phenolphthalein
done
clear
B)
methyl orange
done
clear
C)
methyl red
done
clear
D)
ammonium hydroxide
done
clear
View Answer play_arrow
question_answer 63) Nitrogen molecule is chemically less active because of the presence of....... between two nitrogen atoms :
A)
single bond
done
clear
B)
double bond
done
clear
C)
triple bond
done
clear
D)
coordinate bond
done
clear
View Answer play_arrow
question_answer 64) To dissolve argentite ore which of the following is used:
A)
\[Na\,[Ag{{(CN)}_{2}}]\]
done
clear
B)
\[NaCN\]
done
clear
C)
\[NaCl\]
done
clear
D)
\[HCl\]
done
clear
View Answer play_arrow
question_answer 65) When \[{{H}_{2}}S\] gas is passed through an acidified solution of ...... yellow precipitate is formed?
A)
\[C{{u}^{2+}}\]ions
done
clear
B)
\[C{{d}^{2+}}\] ions
done
clear
C)
\[N{{i}^{2+}}\] ions
done
clear
D)
\[C{{o}^{2+}}\] ions
done
clear
View Answer play_arrow
question_answer 66) Which of the following has same number of electrons as in \[C{{a}^{2+}}\] ion?
A)
\[C{{u}^{2+}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
C)
\[Ne\]
done
clear
D)
\[M{{g}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 67) Which of the following have highest melting point?
A)
\[NaCl\]
done
clear
B)
\[NaI\]
done
clear
C)
\[NaBr\]
done
clear
D)
\[NaF\]
done
clear
View Answer play_arrow
question_answer 68) Which of the following is not an acid anhydride?
A)
\[C{{O}_{2}}\]
done
clear
B)
\[CO\]
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
\[S{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 69) \[{{H}_{2}}S\] is more acidic than \[{{H}_{2}}O\] because :
A)
atomic number of sulphur is greater than that of oxygen
done
clear
B)
oxygen is more electronegative than sulphur
done
clear
C)
\[H-S\] bond is weaker than \[H-O\] bond
done
clear
D)
\[H-O\] bond is weaker than \[H-S\] bond
done
clear
View Answer play_arrow
question_answer 70) Which of the following halide of calcium is insoluble in water?
A)
\[CaC{{l}_{2}}\]
done
clear
B)
\[Ca{{I}_{2}}\]
done
clear
C)
\[Ca{{F}_{2}}\]
done
clear
D)
\[CaB{{r}_{2}}\]
done
clear
View Answer play_arrow
question_answer 71) Which of the following sulphur containing species cannot act as oxidising agent?
A)
\[{{H}_{2}}S\]
done
clear
B)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
\[{{H}_{2}}S{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 72) Compared to the mass of lightest nucleus mass an electron is only:
A)
\[1/80\]
done
clear
B)
\[1/1800\]
done
clear
C)
\[1/800\]
done
clear
D)
\[1/1000\]
done
clear
View Answer play_arrow
question_answer 73) Decreasing order of s-character is \[sp,s{{p}^{2}},s{{p}^{3}}\]-hybrid orbitals:
A)
\[s{{p}^{3}}>s{{p}^{2}}>sp\]
done
clear
B)
\[sp>s{{p}^{2}}>s{{p}^{3}}\]
done
clear
C)
\[s{{p}^{3}}>sp>s{{p}^{2}}\]
done
clear
D)
\[s{{p}^{2}}>sp>s{{p}^{3}}\]
done
clear
View Answer play_arrow
question_answer 74) Silver mirror test is given by:
A)
propanone
done
clear
B)
propane
done
clear
C)
propanal
done
clear
D)
propanol
done
clear
View Answer play_arrow
question_answer 75) Total number of isomers for \[{{C}_{8}}{{H}_{8}}{{O}_{2}}\] will be:
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 76) Aniline on heating with solid \[KOH\] and \[C{{S}_{2}}\] produced:
A)
thiourea
done
clear
B)
phenol
done
clear
C)
S-diphenyl thiourea
done
clear
D)
Schiffs base
done
clear
View Answer play_arrow
question_answer 77) Common name of phosgene is:
A)
carbon tetrachloride
done
clear
B)
phosphoryl chloride
done
clear
C)
carbonyl chloride
done
clear
D)
phosphorus trichloride
done
clear
View Answer play_arrow
question_answer 78) Which of the following is the strongest acid:
A)
formic acid
done
clear
B)
acetic acid
done
clear
C)
trichloroacetic acid
done
clear
D)
trifluoroacetic acid
done
clear
View Answer play_arrow
question_answer 79) Identify (Y) in the series: \[CH\equiv CH\xrightarrow{{{O}_{3}}/NaOH}(X)\xrightarrow{Zn/C{{H}_{3}}COOH}(Y)\]
A)
\[C{{H}_{3}}OH\]
done
clear
B)
\[C{{H}_{2}}COOH-C{{H}_{2}}COOH\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}COOH\]
done
clear
D)
\[C{{H}_{2}}OH-C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 80) Which of the following can be sulphonated easily?
A)
Benzene
done
clear
B)
Toluene
done
clear
C)
Nitrobenzene
done
clear
D)
Chlorobenzene
done
clear
View Answer play_arrow
question_answer 81) Benzene diazonium chloride on treatment with hypophosphorus acid and water in presence of \[C{{u}^{+}}\] catalyst produces:
A)
benzene
done
clear
B)
toluene
done
clear
C)
aniline
done
clear
D)
chlorobenzene
done
clear
View Answer play_arrow
question_answer 82) Butene-1 is isomeric with 2-methyl propene. They can be distinguished by:
A)
\[B{{r}_{2}}\]solution
done
clear
B)
Tollens reagent
done
clear
C)
Baeyers reagent
done
clear
D)
\[{{O}_{3}},\,\,{{H}_{2}}O/Zn\]
done
clear
View Answer play_arrow
question_answer 83) Ethyl alcohol exhibits acidic nature on reaction with:
A)
acidic \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]
done
clear
B)
sodium metal
done
clear
C)
\[HCl\]
done
clear
D)
\[C{{H}_{3}}COOH\]
done
clear
View Answer play_arrow
question_answer 84) Which of the following cannot be nitrated directly?
A)
aniline
done
clear
B)
phenol
done
clear
C)
toluene
done
clear
D)
benzoic acid
done
clear
View Answer play_arrow
question_answer 85) Adipic acid on heating form:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 86) The ratio of carbon, hydrogen and oxygen in 2-methyl benzoic acid is:
A)
\[4:2:2\]
done
clear
B)
\[2:4:1\]
done
clear
C)
\[4:4:1\]
done
clear
D)
\[4:4:2\]
done
clear
View Answer play_arrow
question_answer 87) Beckmann thermometer is used for measuring:
A)
boiling point
done
clear
B)
freezing point
done
clear
C)
both a and b
done
clear
D)
elevation in BP or depression in FP
done
clear
View Answer play_arrow
question_answer 88) An equilibrium mixture of the reaction \[2NO(g)+{{O}_{2}}(g)2N{{O}_{2}}(g)\] contains 0.12 moles of \[N{{O}_{2}}\], 0.08 moles of NO and 0.64 moles of 02 in a 4 litre flask at constant temperature. The value of \[{{K}_{C}}\] this temperature is:
A)
7
done
clear
B)
14
done
clear
C)
28
done
clear
D)
24
done
clear
View Answer play_arrow
question_answer 89) A solution of \[NaCl\] in contact with atmosphere has pH of about:
A)
\[3.2\]
done
clear
B)
\[5.4\]
done
clear
C)
\[7\]
done
clear
D)
\[14\]
done
clear
View Answer play_arrow
question_answer 90) A 0.01 M ammonia solution is 5% ionised. Its pH will be:
A)
\[11.80\]
done
clear
B)
\[10.69\]
done
clear
C)
\[7.22\]
done
clear
D)
\[12.24\]
done
clear
View Answer play_arrow
question_answer 91) Equal volumes of \[0.02M\] \[CaC{{l}_{2}}\] and \[0.0004M\] \[N{{a}_{2}}S{{O}_{4}}\] are mixed. If \[{{K}_{sp}}\] for \[CaS{{O}_{4}}=2.4\times {{10}^{-5}},\], whether there would be any precipitation ?
A)
Yes
done
clear
B)
No
done
clear
C)
Unpredictable
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 92) If the vapour pressure of pure solvent is \[{{P}_{0}}\] and the vapour pressure of dilute solution of non-volatile solute is P, then the lowering in vapour pressure is :
A)
\[+ve\]
done
clear
B)
\[-ve\]
done
clear
C)
\[P/{{P}_{0}}\]
done
clear
D)
\[{{P}_{0}}/P\]
done
clear
View Answer play_arrow
question_answer 93) If \[\Delta H\] is the change in enthalpy and \[\Delta E\] is the change in internal energy, then:
A)
\[\Delta H<\Delta E,\], only if the number of moles of gaseous products is greater than the number of moles of gaseous reactants
done
clear
B)
\[\Delta H<\Delta E,\], only if the number of moles of gaseous products is loss than the number of moles of gaseous reactants
done
clear
C)
\[\Delta H\] is always gutter than \[\Delta E\]
done
clear
D)
\[\Delta H\] is always less than \[\Delta E\]
done
clear
View Answer play_arrow
question_answer 94) If the concentration of \[M{{g}^{2+}}\] is 0.01 M, the electrode potential of \[M{{g}^{2+}}/Mg\] electrode will be (Given that \[{{E}^{o}}\,M{{g}^{2+}}\]\[Mg=-2.36V\]):
A)
\[+2.80\]
done
clear
B)
\[-2.80\]
done
clear
C)
\[+2.41\]
done
clear
D)
\[-2.41\]
done
clear
View Answer play_arrow
question_answer 95) The rate of a chemical reaction doubles for every \[{{10}^{o}}C\] increase in temperature, if the temperature of reaction is increased from \[{{30}^{o}}C\] to \[{{80}^{o}}C\]. The rate of reaction increases:
A)
16
done
clear
B)
32
done
clear
C)
64
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 96) A chemical system is in equilibrium. The addition of a catalyst would result is :
A)
increase the amount of heat evolved in the reaction
done
clear
B)
a new reaction pathway to reaction
done
clear
C)
increase in the rate of reverse reaction
done
clear
D)
increase in the rate of forward reaction
done
clear
View Answer play_arrow
question_answer 97) Number of moles occupied by 22.4 litres of Fie gas at \[{{30}^{o}}C\] and one atmospheric pressure are:
A)
\[0.20\]
done
clear
B)
\[1.46\]
done
clear
C)
\[0.90\]
done
clear
D)
\[2.62\]
done
clear
View Answer play_arrow
question_answer 98) What maximum amount of \[BaS{{O}_{4}}\] get precipitated if 0.5 mole of \[BaC{{l}_{2}}\] is mixed with 1 mole of \[{{H}_{2}}S{{O}_{4}}\]?
A)
\[0.5\text{ }mol\]
done
clear
B)
\[1.0\text{ }mol\]
done
clear
C)
\[1.5\text{ }mol~\]
done
clear
D)
\[2.0\text{ }mol\]
done
clear
View Answer play_arrow
question_answer 99) Sodium metal crystallises at room temperature in a body centered cubic lattice with a cell edge \[a=4.29\overset{\text{o}}{\mathop{\text{A}}}\,\]. The radius of sodium atom is:
A)
\[1.40\]
done
clear
B)
\[2.65\]
done
clear
C)
\[1.85\]
done
clear
D)
\[2.15\]
done
clear
View Answer play_arrow
question_answer 100) The half life of a radioactive element is 3.8 days. The time at the end of which 1/20th of the element will remain undecayed will be:
A)
36 day
done
clear
B)
12.86 day
done
clear
C)
24.20 day
done
clear
D)
16.40 day
done
clear
View Answer play_arrow
question_answer 101) Which of the following is an exception of cell theory?
A)
Fungi
done
clear
B)
Lichen
done
clear
C)
Bacteria
done
clear
D)
Virus
done
clear
View Answer play_arrow
question_answer 102) Alga rich in protein :
A)
Ulothrix
done
clear
B)
Spirogyra
done
clear
C)
Oscillatoria
done
clear
D)
Chlorella
done
clear
View Answer play_arrow
question_answer 103) In biotic community, primary consumers are:
A)
herbivores
done
clear
B)
carnivores
done
clear
C)
omnivores
done
clear
D)
decomposers
done
clear
View Answer play_arrow
question_answer 104) Sugarcane Breeding Research Institute (SBKI) is situated at:
A)
Lucknow
done
clear
B)
Coimbatore
done
clear
C)
New Delhi
done
clear
D)
Shimala
done
clear
View Answer play_arrow
question_answer 105) As tree grow older, which of the following will increase more rapidly in thickness?
A)
Heart wood
done
clear
B)
Sap wood
done
clear
C)
Xylem
done
clear
D)
Phloem
done
clear
View Answer play_arrow
question_answer 106) Haploid plants can be obtained by culturing :
A)
root tip
done
clear
B)
shoot tip
done
clear
C)
pollen grain
done
clear
D)
endosperm
done
clear
View Answer play_arrow
question_answer 107) Citric acid is produced by :
A)
Aspergilhts niger
done
clear
B)
Candida
done
clear
C)
PeniciHiiim nolatiim
done
clear
D)
Acetobacter xylamcum
done
clear
View Answer play_arrow
question_answer 108) Agar-agar is obtained from :
A)
green algae
done
clear
B)
blue-green algae
done
clear
C)
red algae
done
clear
D)
brown algae
done
clear
View Answer play_arrow
question_answer 109) In angiosperms, how many microspore mother cells are required to produced 100 pollen grains?
A)
100
done
clear
B)
50
done
clear
C)
25
done
clear
D)
10
done
clear
View Answer play_arrow
question_answer 110) Sexual reproduction is absent in:
A)
Volvox
done
clear
B)
Spirogyra
done
clear
C)
Nosloc
done
clear
D)
Mango
done
clear
View Answer play_arrow
question_answer 111) Which of the following is absent in root hairs?
A)
Cell wall
done
clear
B)
Vacuole
done
clear
C)
Nucleus
done
clear
D)
Chloroplast
done
clear
View Answer play_arrow
question_answer 112) Which compound links glycolysis to Krebs cycle?
A)
NADH
done
clear
B)
ATP
done
clear
C)
Pyruvic acid
done
clear
D)
Acetyl CoA
done
clear
View Answer play_arrow
question_answer 113) Uhthrix produces:
A)
isogametes
done
clear
B)
an isogametes
done
clear
C)
ascospores
done
clear
D)
heterogametes
done
clear
View Answer play_arrow
question_answer 114) The floating roots are found in :
A)
Viscum
done
clear
B)
Vanda
done
clear
C)
Tinospora
done
clear
D)
fiissiaea
done
clear
View Answer play_arrow
question_answer 115) Which of the following is absent in phloem of Pinus?
A)
Phloem parenchyma
done
clear
B)
Sieve cells
done
clear
C)
Companion cells
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 116) If a homozygous red flowered plant is crossed with homozygous white flowered plants, the offsprings will be :
A)
all red flowered
done
clear
B)
all white flowered
done
clear
C)
half red and half white flowered
done
clear
D)
all the above
done
clear
View Answer play_arrow
question_answer 117) Cell wall is absent in :
A)
Mycoplasma
done
clear
B)
Bacteria
done
clear
C)
Yeast
done
clear
D)
Rhizopus
done
clear
View Answer play_arrow
question_answer 118) Isobilateral leaf is found in :
A)
dicots
done
clear
B)
monocots
done
clear
C)
hydrophytes
done
clear
D)
xerophytes
done
clear
View Answer play_arrow
question_answer 119) Krebs cycle takes place in :
A)
chloroplast
done
clear
B)
mitochondria
done
clear
C)
ribosome
done
clear
D)
cytoplasm
done
clear
View Answer play_arrow
question_answer 120) Mycorrhiza is a relationship of roots of Pinus and :
A)
fungus
done
clear
B)
alga
done
clear
C)
bacteria
done
clear
D)
ants
done
clear
View Answer play_arrow
question_answer 121) Influenza is caused by?
A)
bacterium
done
clear
B)
virus
done
clear
C)
fungus
done
clear
D)
cyanobacteriurn
done
clear
View Answer play_arrow
question_answer 122) Which is cyanophage?
A)
\[\phi \times -174\]
done
clear
B)
LPP-1
done
clear
C)
S-15
done
clear
D)
SV-30
done
clear
View Answer play_arrow
question_answer 123) Heterothallism was discovered by :
A)
Bessey
done
clear
B)
Alexopolous
done
clear
C)
Blakeslee
done
clear
D)
Mchta
done
clear
View Answer play_arrow
question_answer 124) In \[{{C}_{4}}\] plants, \[C{{O}_{2}}\] combines with :
A)
phosphoglyceric acid
done
clear
B)
ribulose biphosphate
done
clear
C)
phosphoenol pyruvate
done
clear
D)
phosphoglyceraldehyde
done
clear
View Answer play_arrow
question_answer 125) Auxin herbicide is :
A)
NAA
done
clear
B)
IAA
done
clear
C)
2, 4-D
done
clear
D)
IBA
done
clear
View Answer play_arrow
question_answer 126) Soil water available for absorption by roots is:
A)
capillary water
done
clear
B)
chemically combined water
done
clear
C)
gravitational water
done
clear
D)
hygroscopic water
done
clear
View Answer play_arrow
question_answer 127) Mendels dihybrid cross ratio is:
A)
9:3:3:1
done
clear
B)
1:2: 1
done
clear
C)
12:3:1
done
clear
D)
1:1
done
clear
View Answer play_arrow
question_answer 128) Roots which develop from any part of plant except radicle are called?
A)
Top root
done
clear
B)
Advantitious roots
done
clear
C)
Pneumatophores
done
clear
D)
Coralloid root
done
clear
View Answer play_arrow
question_answer 129) Which tissue is involved in conduction of water?
A)
Xylem
done
clear
B)
Phloem
done
clear
C)
both a and b
done
clear
D)
Parenchyma
done
clear
View Answer play_arrow
question_answer 130) Ecosystem consists of:
A)
Autotrophs
done
clear
B)
herbivores and carnivores
done
clear
C)
decomposers
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 131) The female sex organs of bryophytes are called :
A)
archegonia
done
clear
B)
ascogonia
done
clear
C)
carpogonia
done
clear
D)
antheridia
done
clear
View Answer play_arrow
question_answer 132) Protein synthesis takes place on :
A)
golgi bodies
done
clear
B)
ribosome
done
clear
C)
chloroplasts
done
clear
D)
mitochondria
done
clear
View Answer play_arrow
question_answer 133) Colchicine prevents mitosis of cells at:
A)
anaphase
done
clear
B)
metaphase
done
clear
C)
prophase
done
clear
D)
interphase
done
clear
View Answer play_arrow
question_answer 134) Virus multiplies in :
A)
culture medium
done
clear
B)
living tissue
done
clear
C)
dead tissue
done
clear
D)
all the above
done
clear
View Answer play_arrow
question_answer 135) The five kingdom classification was proposed by:
A)
Whittaker
done
clear
B)
Eichler
done
clear
C)
Bentham and Hooker
done
clear
D)
Bessey
done
clear
View Answer play_arrow
question_answer 136) Heart shaped prothallus of fern bears :
A)
male sex organs
done
clear
B)
female sex organs
done
clear
C)
bisexual
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 137) Normal secondary growth occurs in :
A)
dicot stem
done
clear
B)
monocot stem
done
clear
C)
both a and b
done
clear
D)
root
done
clear
View Answer play_arrow
question_answer 138) Glycolysis occurs :
A)
in cytoplasm
done
clear
B)
in aerobic respiration
done
clear
C)
in anaerobic respiration
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 139) DNA multiplication is called :
A)
translation
done
clear
B)
transduction
done
clear
C)
transcription
done
clear
D)
replication
done
clear
View Answer play_arrow
question_answer 140) Which of the following is a man made allopolyploid cereal ?
A)
Zea mays
done
clear
B)
Triticale
done
clear
C)
Hordieum vnlgare
done
clear
D)
Onyza saliva
done
clear
View Answer play_arrow
question_answer 141) Big bang theory was proposed by :
A)
Kant
done
clear
B)
Leniantre
done
clear
C)
K. Bahadur
done
clear
D)
Weismann
done
clear
View Answer play_arrow
question_answer 142) Mutation caused by a mutagen is :
A)
natural
done
clear
B)
chemical
done
clear
C)
spontaneous
done
clear
D)
induced
done
clear
View Answer play_arrow
question_answer 143) Triticum aestivum is :
A)
diptoid
done
clear
B)
tetraploid
done
clear
C)
hexaploid
done
clear
D)
haploid
done
clear
View Answer play_arrow
question_answer 144) Alkaptoneuria is a disease where the urine is:
A)
yellow
done
clear
B)
white
done
clear
C)
orange
done
clear
D)
black
done
clear
View Answer play_arrow
question_answer 145) Which of the following is a genetic vector?
A)
House fly
done
clear
B)
Plasmid
done
clear
C)
Mosquito
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 146) Insectivorous plants can survive in:
A)
water rich soil
done
clear
B)
water deficient soil
done
clear
C)
nitrogen deficient soil
done
clear
D)
sulphur deficient soil
done
clear
View Answer play_arrow
question_answer 147) The genes confined to Y-chromosomes are:
A)
hotandric genes
done
clear
B)
mutant genes
done
clear
C)
autosomal gene
done
clear
D)
sex linked genes
done
clear
View Answer play_arrow
question_answer 148) Leydig cells secrete :
A)
Oestrogen
done
clear
B)
Testosterone
done
clear
C)
Progesterone
done
clear
D)
corticosterone
done
clear
View Answer play_arrow
question_answer 149) The larva of Housefly lacks :
A)
eyes
done
clear
B)
wings
done
clear
C)
spiracles
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 150) Excretion in Cockroach occurs through :
A)
Malpighian tubules
done
clear
B)
coxal glands
done
clear
C)
parotid gland
done
clear
D)
nephridium
done
clear
View Answer play_arrow
question_answer 151) Bats belong to which of the following class of Chordates :
A)
Chiroptera
done
clear
B)
Cleroptera
done
clear
C)
Insectivora
done
clear
D)
Edentats
done
clear
View Answer play_arrow
question_answer 152) Reproduction in Paramecium is controlled by:
A)
flagella
done
clear
B)
micronucleus
done
clear
C)
macronucleus
done
clear
D)
cell wall
done
clear
View Answer play_arrow
question_answer 153) Organ of corti is found in :
A)
heart
done
clear
B)
inner ear
done
clear
C)
kidney
done
clear
D)
nasal chamber
done
clear
View Answer play_arrow
question_answer 154) Melanin is a :
A)
pigment
done
clear
B)
enzyme
done
clear
C)
hormone
done
clear
D)
excretory product
done
clear
View Answer play_arrow
question_answer 155) Actin and myosin proteins are related with:
A)
\[N{{a}^{+}}\] and \[{{K}^{+}}\] pump
done
clear
B)
muscle contraction
done
clear
C)
nervous system -
done
clear
D)
excretion of water products
done
clear
View Answer play_arrow
question_answer 156) Aspergillosis is a disease of:
A)
kidney
done
clear
B)
heart
done
clear
C)
lungs
done
clear
D)
eyes
done
clear
View Answer play_arrow
question_answer 157) Spleen is :
A)
lymphoid
done
clear
B)
haemopoitic
done
clear
C)
celluloid
done
clear
D)
reproductive
done
clear
View Answer play_arrow
question_answer 158) A condition of failure of kidney to form urine is called :
A)
hematuria
done
clear
B)
creatinine
done
clear
C)
aneuria
done
clear
D)
ketonuria
done
clear
View Answer play_arrow
question_answer 159) Pouched mammals are called as :
A)
euthfrians
done
clear
B)
metatherians
done
clear
C)
prototherians
done
clear
D)
atherians
done
clear
View Answer play_arrow
question_answer 160) Which of the following has radial symmetry in adult and bilateral symmetry in larva?
A)
Annelida
done
clear
B)
Coelenterata
done
clear
C)
Echinoderms
done
clear
D)
Platyhelminthes
done
clear
View Answer play_arrow
question_answer 161) Similarity between fish and tadpole is:
A)
legs
done
clear
B)
fins
done
clear
C)
lateral line
done
clear
D)
scales
done
clear
View Answer play_arrow
question_answer 162) Cleidoic eggs are found in :
A)
birds
done
clear
B)
fish
done
clear
C)
frogs
done
clear
D)
reptiles
done
clear
View Answer play_arrow
question_answer 163) Limulus belongs to :
A)
Chilopoda
done
clear
B)
Crustacea
done
clear
C)
Arachnida
done
clear
D)
Merostomata
done
clear
View Answer play_arrow
question_answer 164) Which of the following is a transparent tissue?
A)
Tendon
done
clear
B)
Hyaline cartilage
done
clear
C)
Fibrous cartilage
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 165) Omithine cycle takes place in :
A)
liver
done
clear
B)
heart
done
clear
C)
kidney
done
clear
D)
muscles
done
clear
View Answer play_arrow
question_answer 166) The vagus nerve is the cranial nerve numbering :
A)
5th
done
clear
B)
7th
done
clear
C)
3rd
done
clear
D)
10th
done
clear
View Answer play_arrow
question_answer 167) The deficiency of which vitamin can cause scurvy?
A)
B
done
clear
B)
C
done
clear
C)
D
done
clear
D)
K
done
clear
View Answer play_arrow
question_answer 168) Which of the following amino acid is metabolised by brain ?
A)
Alanine
done
clear
B)
Clutamic acid
done
clear
C)
Histidine
done
clear
D)
Glycine
done
clear
View Answer play_arrow
question_answer 169) In micturition:
A)
urethera relaxes
done
clear
B)
ureter contracts
done
clear
C)
ureter relaxes
done
clear
D)
urethra contracts
done
clear
View Answer play_arrow
question_answer 170) The egg of a frog is :
A)
centrolecithal
done
clear
B)
telolecithal
done
clear
C)
macrolecithal
done
clear
D)
microlecithal
done
clear
View Answer play_arrow
question_answer 171) The main function of Henles loop is:
A)
passage of urine
done
clear
B)
filtration of blood
done
clear
C)
formation of urine
done
clear
D)
conservation of water
done
clear
View Answer play_arrow
question_answer 172) Which of the following is a sesamoid bone?
A)
Patella
done
clear
B)
Palatine
done
clear
C)
Pterygoid
done
clear
D)
Presphenoid
done
clear
View Answer play_arrow
question_answer 173) Terminating codons are :
A)
UAG
done
clear
B)
UGA
done
clear
C)
UAA
done
clear
D)
All the above
done
clear
View Answer play_arrow
question_answer 174) Choanocytes perform:
A)
Nutritions
done
clear
B)
Excretion
done
clear
C)
Reproduction
done
clear
D)
secretion of spicules
done
clear
View Answer play_arrow
question_answer 175) Mouth parts of housefly are called as :
A)
biting and sucking type
done
clear
B)
sponging and sucking type
done
clear
C)
biting and chewing type
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 176) Which parasite shows alternation of generation and alternations of hosts in its life cycle ?
A)
Fasciola
done
clear
B)
Ascaris
done
clear
C)
Taenia
done
clear
D)
Wuchereria
done
clear
View Answer play_arrow
question_answer 177) The special sound producing organ in birds is:
A)
syrinx
done
clear
B)
glottis
done
clear
C)
larynx
done
clear
D)
oesophagus
done
clear
View Answer play_arrow
question_answer 178) The function of sweat gland is :
A)
excretion of salts
done
clear
B)
water regulation
done
clear
C)
then no regulation
done
clear
D)
excretion
done
clear
View Answer play_arrow
question_answer 179) Insulin is secreted by :
A)
\[\beta \]cells of islets of Langerhans in pancreas
done
clear
B)
spleen
done
clear
C)
\[\alpha \]cells of Langerhans of pancreas
done
clear
D)
mucosa of oesophagus
done
clear
View Answer play_arrow
question_answer 180) The jumping gene in Maize were discovered by:
A)
Khorana
done
clear
B)
Morgan
done
clear
C)
Barbara McCIintock
done
clear
D)
Watson and Crirk
done
clear
View Answer play_arrow
question_answer 181) Nissls granules are found in :
A)
liver cells
done
clear
B)
nerve cells
done
clear
C)
intestinal cells
done
clear
D)
kidney
done
clear
View Answer play_arrow
question_answer 182) Bone is connected to muscle with the help of :
A)
ligament
done
clear
B)
tendon
done
clear
C)
cartilage
done
clear
D)
fibre
done
clear
View Answer play_arrow
question_answer 183) Melanocyte stimulating hormone is secreted by:
A)
intermediate lobe of pituitary
done
clear
B)
posterior lobe of pituitary
done
clear
C)
anterior lobe of pituitary
done
clear
D)
all the above
done
clear
View Answer play_arrow
question_answer 184) Prostate gland secretion help in formation of:
A)
larvae
done
clear
B)
seman
done
clear
C)
cocoon
done
clear
D)
cartilage
done
clear
View Answer play_arrow
question_answer 185) Reabsorption of water by kidney is controlled by:
A)
GH
done
clear
B)
ADH
done
clear
C)
oxytocin
done
clear
D)
steroids
done
clear
View Answer play_arrow
question_answer 186) 5th cranial nerve of frog is called :
A)
optic
done
clear
B)
vagus
done
clear
C)
trigeminal
done
clear
D)
opthalamus
done
clear
View Answer play_arrow
question_answer 187) The contraction of gall bladder is due to :
A)
cholecystokinen
done
clear
B)
gastrin
done
clear
C)
secretin
done
clear
D)
enterokinase
done
clear
View Answer play_arrow
question_answer 188) Theory of pangenesis was given by :
A)
Darwin
done
clear
B)
Lamarck
done
clear
C)
Oparin
done
clear
D)
de Vries
done
clear
View Answer play_arrow
question_answer 189) Morphine is an alkaloid, this is obtained from:
A)
Cinchona
done
clear
B)
Opium
done
clear
C)
Rauwolfia
done
clear
D)
Cannnbis
done
clear
View Answer play_arrow
question_answer 190) The lower jaw of a mammal is made up of:
A)
dentary
done
clear
B)
maxilla
done
clear
C)
premaxilla
done
clear
D)
post maxilla
done
clear
View Answer play_arrow
question_answer 191) Who called birds as glorified reptiles ?
A)
Huxley
done
clear
B)
Romer
done
clear
C)
Mendel
done
clear
D)
Robert Hooke
done
clear
View Answer play_arrow
question_answer 192) Biological catalyst is :
A)
an enzyme
done
clear
B)
an amino acid
done
clear
C)
ail amino sugar
done
clear
D)
an antacid
done
clear
View Answer play_arrow
question_answer 193) A nucleotide consists of:
A)
Sugar
done
clear
B)
nitrogen base
done
clear
C)
phosphoric acid
done
clear
D)
all the above
done
clear
View Answer play_arrow
question_answer 194) Soma clonal variations are :
A)
produced in tissue culture
done
clear
B)
caused by gamma rays
done
clear
C)
produced in sexual reproduction
done
clear
D)
caused by mutagcns
done
clear
View Answer play_arrow
question_answer 195) Response to contact is called as :
A)
Thermotaxis
done
clear
B)
Thigmotaxis
done
clear
C)
Chemotaxis
done
clear
D)
geotropism
done
clear
View Answer play_arrow
question_answer 196) Indicator of pollution in water is :
A)
E. coli
done
clear
B)
Lichen
done
clear
C)
Amoeba
done
clear
D)
Nostoc
done
clear
View Answer play_arrow
question_answer 197) The function of contractile vacuole in Protozoa is:
A)
Locomotion
done
clear
B)
Digestion
done
clear
C)
Osmorcgulation
done
clear
D)
Reproduction
done
clear
View Answer play_arrow
question_answer 198) Hydra recognises its prey by:
A)
nematocyst
done
clear
B)
chemical stimulus
done
clear
C)
smell
done
clear
D)
sensitivity
done
clear
View Answer play_arrow
question_answer 199) Aristotle lantern is found in :
A)
phiuroidia
done
clear
B)
Asteriodea
done
clear
C)
Echinoidea
done
clear
D)
Holothuroidea
done
clear
View Answer play_arrow
question_answer 200) Salmon fish is :
A)
anadromous
done
clear
B)
catndromous
done
clear
C)
both a and b
done
clear
D)
Amphibious
done
clear
View Answer play_arrow