question_answer 1) The dimensions of electromotive force in terms of current A are
A)
\[\sqrt{2Eq/ml}\]
done
clear
B)
\[\sqrt{2Em/ql}\]
done
clear
C)
\[\sqrt{\frac{Eq}{ml}}\]
done
clear
D)
\[{{10}^{12}}\]
done
clear
View Answer play_arrow
question_answer 2) A body of mass 3 kg is under a force which causes a displacement in it, given by \[s={{t}^{2}}/3\] (in m). Find the work done by the force in 2 s.
A)
2J
done
clear
B)
3.8 J
done
clear
C)
5.2 J
done
clear
D)
2.6 J
done
clear
View Answer play_arrow
question_answer 3) Two blocks of masses \[{{m}_{1}}\]and \[{{m}_{2}}\] are connected by a massless spring and placed atsmooth surface. The spring initially stretchedand released. Then
A)
the momentum of each particle remains constant separately
done
clear
B)
the magnitudes of momenta of bothbodies are same to each other
done
clear
C)
the mechanical energy of system remains constant
done
clear
D)
Both (a) and (b) are correct
done
clear
View Answer play_arrow
question_answer 4) One quarter of the disc of mass m isremoved. If r be the radius of the disc, thenew moment of inertia is
A)
\[2.5\times {{10}^{7}}\]
done
clear
B)
\[1.25\times {{10}^{7}}\]
done
clear
C)
\[2.5\times {{10}^{8}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 5)
How many images are formed by the lens shown; if an object is kept on its axis?
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 6) A convex lens of focal length/is placed somewhere in between an object and a screen.The distance between object and screen is x.If numerical value of magnification producedby lens is m, focal length of lens is
A)
\[1.25\times {{10}^{6}}\]
done
clear
B)
\[\frac{G}{19}\]
done
clear
C)
\[\frac{G}{20}\]
done
clear
D)
\[E=16T-0.04{{T}^{2}}\mu V.\]
done
clear
View Answer play_arrow
question_answer 7) Find the thickness of a plate which willproduce a change in optical path equals to half the wavelength\[{{T}_{i}}\]of the light passing through it normally.
A)
\[{{200}^{0}}C\]
done
clear
B)
\[{{300}^{0}}C\]
done
clear
C)
\[0.18\times {{10}^{-3}}{{C}^{-1}}\]
done
clear
D)
\[\left( \frac{0.4}{\pi } \right)\]
done
clear
View Answer play_arrow
question_answer 8) In double slit experiment, the distancebetween two slits is 0.6 mm and these areilluminated with light of wavelength 4800\[11.4\Omega ,15.5A\].The angular width of first dark fringe on thescreen distant 120 cm from slits will be
A)
\[30.7\Omega ,6.5A\]
done
clear
B)
\[40.4\Omega ,5A\]
done
clear
C)
\[50\Omega ,5A\]
done
clear
D)
\[{{R}_{1}}\]
done
clear
View Answer play_arrow
question_answer 9) In a free space electron is placed in the pathof a plane electromagnetic wave, it will start moving along
A)
centre of earth
done
clear
B)
equator of earth
done
clear
C)
magnetic field
done
clear
D)
electric field
done
clear
View Answer play_arrow
question_answer 10) An electron of mass m and charge q isaccelerated from rest in a uniform electricfield of strength E. The velocity acquired byit as it (ravels a distance I is
A)
\[{{R}_{2}}\]
done
clear
B)
\[{{R}_{1}}>{{R}_{2}}\]
done
clear
C)
\[\frac{{{R}_{1}}}{{{R}_{2}}}\]
done
clear
D)
\[\frac{{{R}_{2}}}{{{R}_{1}}}\]
done
clear
View Answer play_arrow
question_answer 11) 20% of radioactive substance decay in10 days. The amount of the original material left after 30 days is
A)
51.2%
done
clear
B)
62.6%
done
clear
C)
15%
done
clear
D)
21.2%
done
clear
View Answer play_arrow
question_answer 12) 1% of \[\frac{R_{1}^{2}}{{{R}_{2}}}\] Hz of a satellite link was usedfrom telephony. Find the number ofchannels (or subscribers) if each channel is8 kHz.
A)
\[\frac{R_{2}^{2}}{{{R}_{2}}}\]
done
clear
B)
\[{{r}^{-5/2}}\]
done
clear
C)
\[{{T}^{2}}\]
done
clear
D)
\[{{r}^{3}}\]
done
clear
View Answer play_arrow
question_answer 13) In a galvanometer, 5% of the total current inthe circuit passes through it. If the resistance ofthe galvanometer is G, the shunt resistance 5connected to the galvanometer is
A)
19 G
done
clear
B)
\[{{r}^{7/2}}\]
done
clear
C)
20 G
done
clear
D)
\[{{r}^{5/2}}\]
done
clear
View Answer play_arrow
question_answer 14) The temperature of the cold junction of athermocouple is \[0{}^\circ C\] and the temperature of hot junction is \[T{}^\circ C\]. The emf is \[{{r}^{3/2}}\]The inversion temperature \[l\] is
A)
\[\frac{l}{L-l}\]
done
clear
B)
\[400{}^\circ C\]
done
clear
C)
\[100{}^\circ C\]
done
clear
D)
\[\frac{L}{l}\]
done
clear
View Answer play_arrow
question_answer 15) At constant temperature, the volume of a gasis to be decreased by 4%. The pressure mustbe increased by
A)
4%
done
clear
B)
4.16%
done
clear
C)
8%
done
clear
D)
3.86%
done
clear
View Answer play_arrow
question_answer 16) The coefficient of real expansion of mercury is\[\frac{l}{L}\]. If the density of mercury at0°C is 13.6 g/cc, its density at 473 K will be
A)
13.12 g/cc
done
clear
B)
13.65 g/cc
done
clear
C)
13.51 g/cc
done
clear
D)
13.22 g/cc
done
clear
View Answer play_arrow
question_answer 17) A tap supplies water at \[22{}^\circ C\], a man takes 1 Lof water per min at \[37{}^\circ C\] from the geyser.The power of geyser is
A)
525 W
done
clear
B)
1050 W
done
clear
C)
1775 W
done
clear
D)
2100 W
done
clear
View Answer play_arrow
question_answer 18) In an L-R circuit, the value of L is \[\frac{lL}{L+l}\]and the value of R is 30H. If in the circuit, analternating emf of 200 V at 50 cycle per/s isconnected, the impedances of the circuit andcurrent will be
A)
\[\frac{m{{v}^{3}}}{4\sqrt{2g}}\]
done
clear
B)
\[\frac{m{{v}^{2}}}{\sqrt{2g}}\]
done
clear
C)
\[m(2g{{h}^{3}})\]
done
clear
D)
\[mg\left[ \frac{b}{2} \right]\]
done
clear
View Answer play_arrow
question_answer 19) Two conducting circular loops of radii \[mg\left[ a+\frac{b}{2} \right]\] and\[mg\left[ \frac{b-a}{2} \right]\] are placed in the same plane with theircentres coinciding. If \[mg\left[ \frac{b+a}{2} \right]\], the mutual inductance M between them will be directly proportional to
A)
\[i={{i}_{1}}\cos \omega t+{{i}_{2}}\sin \omega t\]
done
clear
B)
\[\frac{{{i}_{1}}+{{i}_{2}}}{2}\]
done
clear
C)
\[\frac{{{({{i}_{1}}+{{i}_{2}})}^{2}}}{\sqrt{2}}\]
done
clear
D)
\[\frac{1}{\sqrt{2}}\sqrt{i_{1}^{2}+i_{2}^{2}}\]
done
clear
View Answer play_arrow
question_answer 20) Imagine a light planet revolving around avery massive star in a circular orbit of radiusr with a period of revolution T. If thegravitational force of attraction between the planet and star is proportional to \[\frac{i_{1}^{2}+i_{2}^{2}}{2}\]then\[\Omega \] is proportional to
A)
\[\frac{30.8}{\sqrt{T}}\overset{0}{\mathop{A}}\,\]
done
clear
B)
\[\frac{3.08}{\sqrt{T}}\overset{0}{\mathop{A}}\,\]
done
clear
C)
\[\frac{0.308}{\sqrt{T}}\overset{0}{\mathop{A}}\,\]
done
clear
D)
\[\frac{0.0308}{\sqrt{T}}\overset{0}{\mathop{A}}\,\]
done
clear
View Answer play_arrow
question_answer 21) A partly hanging uniform chain of length L is resting on a rough horizontal table. If \[{{E}_{e}}\] is the maximum possible length that can hang in equilibrium. The coefficient of friction between the chain and table is
A)
\[{{E}_{ph}}\]
done
clear
B)
\[\frac{{{E}_{e}}}{{{E}_{ph}}}\]
done
clear
C)
\[\frac{v}{c}\]
done
clear
D)
\[\frac{v}{2c}\]
done
clear
View Answer play_arrow
question_answer 22) A particle of mass m is projected with avelocity v making an angle of 45° with thehorizontal. The magnitude of angularmomentum of the projectile about an axis ofprojection when the particle is of maximumheight h is
A)
zero
done
clear
B)
\[\frac{v}{3c}\]
done
clear
C)
\[\frac{v}{4c}\]
done
clear
D)
\[x=7t+4{{t}^{2}}\]
done
clear
View Answer play_arrow
question_answer 23) A plate of mass m, length b and breadth a isinitially lying on a horizontal floor withlength parallel to the floor and breadthperpendicular to the floor. The work done toerect it on its breadth is
A)
\[y=5t,\]
done
clear
B)
\[{{s}^{2}}\]
done
clear
C)
\[{{s}^{2}}\]
done
clear
D)
\[{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 24) An AC is given by equation\[{{s}^{2}}\]. The rms value of thecurrent is given by
A)
\[10\sqrt{8}m/s\]
done
clear
B)
\[\frac{40}{3}m/s\]
done
clear
C)
\[\pi \]
done
clear
D)
\[{{\tan }^{-1}}(\sin )\]
done
clear
View Answer play_arrow
question_answer 25) When ultraviolet light of wavelength 100 nmis incident upon silver plate, a potential of 7.7 V is required to stop the photoelectronsfrom reaching the collector plate. How muchpotential will be required to stop thephotoelectrons when light of wavelength 200 nm is incident upon silver?
A)
1.5V
done
clear
B)
3.85V
done
clear
C)
2.35V
done
clear
D)
15.4V
done
clear
View Answer play_arrow
question_answer 26) A coil has an inductance of 0.7 H and isjoined in series with a resistance of 220\[{{\sin }^{-1}}(\sin \,r)\].When the AC emf of 220 V, 50 Hz is appliedto it, find the wattless component of currentin the circuit.
A)
0.2 A
done
clear
B)
0.4 A
done
clear
C)
0.5 A
done
clear
D)
0.7 A
done
clear
View Answer play_arrow
question_answer 27) Radiations of two photon energies twice andfive times the work function of metal areincident successively on the metal surface.The ratio of the maximum velocity of thephotoelectrons emitted in the two caseswill be
A)
1 : 1
done
clear
B)
1 : 2
done
clear
C)
1 : 3
done
clear
D)
1 : 4
done
clear
View Answer play_arrow
question_answer 28) The de-Broglie wavelength of neutrons in thermal equilibrium is
A)
\[{{\sin }^{-1}}(tan\,i)\]
done
clear
B)
\[{{\sin }^{-1}}(tan\,r)\]
done
clear
C)
\[\Omega \]
done
clear
D)
\[\Omega \]
done
clear
View Answer play_arrow
question_answer 29) An electron and a photon possess the same de-Broglie wavelength. If \[\Omega \] and\[\frac{{{T}^{2}}}{2x}\]respectively are the energies of electron andphoton and v and c are their respective velocities, then \[\frac{{{T}^{2}}}{2k}\] is equal to
A)
\[\frac{2k}{{{T}^{2}}}\]
done
clear
B)
\[\frac{2{{T}^{2}}}{k}\]
done
clear
C)
\[{{s}^{2}}\]
done
clear
D)
\[{{m}^{2}}/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 30) The x and y coordinates of a particle at anytime t are given by \[{{m}^{2}}/{{s}^{2}}\]and\[{{m}^{2}}/{{s}^{2}}\],where x and y are in metre and t is insecond. The acceleration of the particle att = 5 s is
A)
zero
done
clear
B)
8 m/\[{{m}^{2}}/{{s}^{2}}\]
done
clear
C)
20 m/\[\frac{mgR\sin \theta }{{{B}^{2}}{{l}^{2}}}\]
done
clear
D)
40 m/\[\frac{mgR\sin \theta }{B{{l}^{3}}}\]
done
clear
View Answer play_arrow
question_answer 31) A cart is moving horizontally along a straightline with constant speed 30 m/s. A projectileis to be fired from the moving cart in such away that it will return to the cart after thecart has moved 80 m. At what speed(relative to the cart) must the projectile be fired?\[(Take\text{ }g=\text{ }10\text{ }m/{{s}^{2}})\]
A)
10 m/s
done
clear
B)
\[\frac{mgR\sin \theta }{B{{l}^{4}}}\]
done
clear
C)
\[\Omega \]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 32) A particle has displacement of 12 m towardseast and 5 m towards, north then 6 mvertically upward. The sum of the sed is placements is
A)
12m
done
clear
B)
10.04m
done
clear
C)
14.31m
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 33) A ray of light strikes a plane water-airinterface from within the water at an angleof incidence i. Which one of the following statements is correct?
A)
If i is 90°, the angle of refraction will bethe critical angle
done
clear
B)
If i is equal to the critical angle, thereflected and refracted rays will be atright angles to each other
done
clear
C)
If i is less than the critical angle, all thelight is refracted and the angle of efraction is greater than i
done
clear
D)
If i is less than the critical angle, somelight is refracted and the angle ofreflection is less than the critical angle
done
clear
View Answer play_arrow
question_answer 34) A ray of light from a denser medium strikes ararer medium at an angle of incidence i. Thereflected and refracted rays make an angle of\[t=\infty \]/2 with each other. If the angles ofreflection .end refraction are r and r, thenthe critical angle will be
A)
\[\frac{{{e}^{1/2}}}{{{e}^{1/2}}-1}\]
done
clear
B)
\[\frac{{{e}^{2}}}{{{e}^{2}}-1}\]
done
clear
C)
\[1-{{e}^{-1}}\]
done
clear
D)
\[{{e}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 35) Two tuning forks A and B having a frequencyof 500 Hz each are placed with B to the rightof A. An observer is between the forks and ismoving towards B with a speed of 25 m/s.The speed of sound is 345 m/s and the windspeed is 5 m/s from A to B. Calculate thedifference in the two frequencies heard bythe observer.
A)
72.5 Hz
done
clear
B)
55.6 Hz
done
clear
C)
76.2 Hz
done
clear
D)
80.9 Hz
done
clear
View Answer play_arrow
question_answer 36) Wires A and B are made from the samematerial. A has twice the diameter and threetimes the length of B. If the elastic limits arenot reached, when each is stretched by thesame tension, the ratio of energy stored in Ato that in B is
A)
2 : 3
done
clear
B)
3 : 4
done
clear
C)
3 : 2
done
clear
D)
6 :1
done
clear
View Answer play_arrow
question_answer 37) The wave impedance of free space is
A)
zero
done
clear
B)
376.6\[{{y}_{1}}=a\sin \frac{2\pi }{\lambda }(vt-x)\]
done
clear
C)
1883\[{{y}_{2}}=b\sin \frac{2\pi }{\lambda }[(vt-x)+{{x}_{0}}]\]
done
clear
D)
3776\[{{x}_{0}}=(\lambda /2)\]
done
clear
View Answer play_arrow
question_answer 38) If a spring extends by x on loading, thenenergy stored by the spring is (if T is thetension in the spring and k is the springconstant)
A)
\[\left| a-b \right|\]
done
clear
B)
\[a+b\]
done
clear
C)
\[\sqrt{{{a}^{2}}+{{b}^{2}}}\]
done
clear
D)
\[\sqrt{{{a}^{2}}+{{b}^{2}}+2ab\,\cos \,x}\]
done
clear
View Answer play_arrow
question_answer 39) Two cars are moving on two perpendicularroads towards a crossing with uniformspeeds of 72 km/h and 36 km/h. If first carblows horn of frequency 280 Hz, then thefrequency of horn heard by the driver ofsecond car when line joining the car makesangle of 45° with the roads, will be
A)
321 Hz
done
clear
B)
298 Hz
done
clear
C)
289 Hz
done
clear
D)
280 Hz
done
clear
View Answer play_arrow
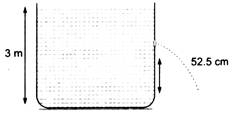
question_answer 40)
Water is filled in a cylindrical container to aheight of3m.A hole is opened at a height of52.5 cm from the bottom. The ratio of thecross-sectional areas of the orifice and thebeaker is 0.1. The square of the speed of theliquid coming out from the orifice is te=10m/\[\frac{{{\varepsilon }_{0}}A}{d}\left[ \frac{{{k}_{1}}}{{{k}_{2}}}+\frac{{{k}_{2}}{{k}_{3}}}{{{k}_{2}}+{{k}_{3}}} \right]\])
A)
50\[\frac{{{\varepsilon }_{0}}A}{d}\left[ \frac{{{k}_{1}}}{{{k}_{2}}}+\frac{({{k}_{2}}+{{k}_{3}})}{{{k}_{2}}{{k}_{3}}} \right]\]
done
clear
B)
51 \[\frac{{{\varepsilon }_{0}}A}{d}\left[ \frac{2}{{{k}_{1}}}+\frac{{{k}_{2}}{{k}_{3}}}{{{k}_{2}}+{{k}_{3}}} \right]\]
done
clear
C)
40.2 \[\frac{{{\varepsilon }_{0}}A}{d}\left[ \frac{2}{{{k}_{1}}}+\frac{{{k}_{2}}+{{k}_{3}}}{{{k}_{2}}{{k}_{3}}} \right]\]
done
clear
D)
60m\[1.81\times {{10}^{-5}}wb\]
done
clear
View Answer play_arrow
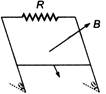
question_answer 41)
A conducting wire of mass m slides downtwo smooth conducting bars, set at an angleto the horizontal as shown in the figure. Theseparation between the bars is equal to I.The system is located in the magnetic field B,perpendicular to the plane of the sliding wireand bars. The velocity of the wire is
A)
\[0.904\times {{10}^{-5}}wb\]
done
clear
B)
\[5.43\times {{10}^{-5}}wb\]
done
clear
C)
\[2\times {{10}^{-2}}{{m}^{2}}\]
done
clear
D)
\[{{10}^{6}}\]
done
clear
View Answer play_arrow
question_answer 42) An emf of 15 V is applied in a circuit containing 5 H inductance and 10\[2.0\times {{10}^{10}}N\]resistance. The ratio of the currents at time\[2.0\times {{10}^{4}}N\] and c = 1 s is
A)
\[2.0\times {{10}^{8}}N\]
done
clear
B)
\[2.0\times {{10}^{6}}N\]
done
clear
C)
\[v=\frac{W}{q}=\frac{[M{{L}^{2}}{{T}^{-2}}]}{[AT]}\]
done
clear
D)
\[v=\frac{W}{q}=\frac{[M{{L}^{2}}{{T}^{-2}}]}{[AT]}\]
done
clear
View Answer play_arrow
question_answer 43) Two waves are passing through a region inthe same direction at the same time. If theequations of these waves are \[\left[ M{{L}^{-2}}{{A}^{-2}} \right]\]And \[\left[ M{{L}^{2}}{{T}^{-2}}{{A}^{-2}} \right]\] then, the amplitude of the resulting wave for \[\left[ M{{L}^{2}}{{T}^{-2}}{{A}^{-2}} \right]\]is
A)
\[\left[ M{{L}^{2}}{{T}^{-3}}{{A}^{-2}} \right]\]
done
clear
B)
\[\frac{3}{2}m{{r}^{2}}\]
done
clear
C)
\[\frac{m{{r}^{2}}}{2}\]
done
clear
D)
\[\frac{3}{8}m{{r}^{2}}\]
done
clear
View Answer play_arrow
question_answer 44) A factory is served by a 220 V supply line. Ina circuit protected by a fuse marked 10 A,the maximum number of 100 W lamps inparallel that can be turned on is
A)
11
done
clear
B)
22
done
clear
C)
33
done
clear
D)
66
done
clear
View Answer play_arrow
question_answer 45) It takes 16 min to boil some water in anelectric kettle. Due to some defect it becomesnecessary to remove 10% turns of heatingcoil of the kettle. After repairs, how muchtime will it take to boil the same mass ofwater?
A)
17.7 min
done
clear
B)
14.4 min
done
clear
C)
20.9mm
done
clear
D)
13.9 min
done
clear
View Answer play_arrow
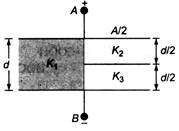
question_answer 46)
Parallel plate capacitor is constructed usingthree different dielectric materials as shownin the figure. The parallel plates, acrosswhich a potential difference is applied are of area A m2 and separated by a distanced metre. The capacitance across AB is
A)
\[\frac{mx}{{{(m+1)}^{2}}}\]
done
clear
B)
\[\frac{mx}{{{(m-1)}^{2}}}\]
done
clear
C)
\[\frac{{{(m+1)}^{2}}}{m}x\]
done
clear
D)
\[\frac{{{(m-1)}^{2}}}{m}x\]
done
clear
View Answer play_arrow
question_answer 47) At what temperature is the rms velocity of ahydrogen molecule equal to that of anoxygen molecule at \[47{}^\circ C\]?
A)
80 K
done
clear
B)
-73 K
done
clear
C)
3K
done
clear
D)
20 K
done
clear
View Answer play_arrow
question_answer 48) An iron rod of \[0.2\text{ }c{{m}^{2}}\] cross-sectional area is subjected to a magnetizing field of \[1200\text{ }A\text{ }{{m}^{-1}}.\] The susceptibility of iron is 599.The magnetic flux produced is
A)
0.904 Wb
done
clear
B)
\[\lambda \]
done
clear
C)
\[\frac{\lambda }{4(\mu -1)}\]
done
clear
D)
\[\frac{2\lambda }{4(\mu -1)}\]
done
clear
View Answer play_arrow
question_answer 49) A coil of 100 turns and area \[\frac{\lambda }{(\mu -1)}\]is pivoted about a vertical diameter in auniform magnetic field and carries a currentof 5A. When the coil is held with its plane innorth-south direction, it experiences acouple of 0.33 Nm. When the plane iseast-west, the corresponding couple is0.4 Nm, the value of magnetic induction is [Neglect earths magnetic field]
A)
0.2 T
done
clear
B)
0.3 T
done
clear
C)
0.4 T
done
clear
D)
0.05 T
done
clear
View Answer play_arrow
question_answer 50) Two copper balls, each weighing 10 g, arekept in air 10 cm apart. If one electron from every \[\frac{\lambda }{2(\mu -1)}\] atoms is transferred from one ballto the other, the coulomb force betweenthem is (atomic weight of copper is 63.5)
A)
\[\overset{0}{\mathop{A}}\,\]
done
clear
B)
\[8\times {{10}^{-4}}red\]
done
clear
C)
\[6\times {{10}^{-4}}red\]
done
clear
D)
\[4\times {{10}^{-4}}red\]
done
clear
View Answer play_arrow
question_answer 51) Which of the following are possible values of n, I and m for an atom having maximum value of \[m=+2\]?
A)
\[n=4,1=3,\text{ }m=+2\]
done
clear
B)
\[n=3,l=2,\text{ }m=-2\]
done
clear
C)
\[n=3,l=3,\text{ }m=+2\]
done
clear
D)
\[n=4,l=3,m=-2\]
done
clear
View Answer play_arrow
question_answer 52) A 1 L vessel initially contains 2.0, 0.5 and 0.0 moles of \[{{N}_{3}}\], \[{{H}_{2}}\] and \[N{{H}_{3}}\] respectively. The system after attaining equilibrium has 0.2 mole of \[N{{H}_{3}}\]. The number of moles of \[{{H}_{2}}\] in the vessel at equilibrium is
A)
\[0.3\]
done
clear
B)
\[0.4\]
done
clear
C)
\[0.2\]
done
clear
D)
\[1.8\]
done
clear
View Answer play_arrow
question_answer 53) For adsorption of a gas on a solid, the plot of log x/m vs log p is linear with slope equal to (n being whole number)
A)
k
done
clear
B)
log k
done
clear
C)
n
done
clear
D)
1/n
done
clear
View Answer play_arrow
question_answer 54) Species acting both as Bronsted acid and base is
A)
\[HSO_{4}^{-}\]
done
clear
B)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
C)
\[N{{H}_{3}}\]
done
clear
D)
\[O{{H}^{-}}\]
done
clear
View Answer play_arrow
question_answer 55) The rate constant of a reaction is \[10.8\times {{10}^{-5}}\] \[mol\,\,d{{m}^{-3}}\,{{s}^{-1}}\]. The order of the reaction is
A)
zero
done
clear
B)
1
done
clear
C)
2
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 56) The half-life period of a radioactive element is 140 days. After 560 days, 1 g of the element will be reduced to
A)
\[\frac{1}{2}g\]
done
clear
B)
\[\frac{1}{4}g\]
done
clear
C)
\[\frac{1}{8}g\]
done
clear
D)
\[\frac{1}{16}g\]
done
clear
View Answer play_arrow
question_answer 57) Mark the wrong statement.
A)
K boils at the m.p of \[KCl\]
done
clear
B)
K is insoluble in molten \[KCl\]
done
clear
C)
K is extracted by electrolysis of fused \[KOH\]
done
clear
D)
K is extracted by reduction of molten \[KCl\] by metallic Na
done
clear
View Answer play_arrow
question_answer 58) The oxidation number and covalency of sulphur in sulphur molecule \[({{S}_{8}})\] are
A)
0 and 2
done
clear
B)
+ 6 and 8
done
clear
C)
0 and 8
done
clear
D)
+ 6 and 2
done
clear
View Answer play_arrow
question_answer 59) The total number of isomers for \[{{C}_{4}}{{H}_{8}}\] is
A)
5
done
clear
B)
6
done
clear
C)
7
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 60) Benzene does not undergo addition reactions easily because
A)
it has a cyclic structure
done
clear
B)
double bonds in it are very strong
done
clear
C)
resonance stabilised system is to be preserved
done
clear
D)
it has six hydrogen atoms
done
clear
View Answer play_arrow
question_answer 61) Phenol is heated with phthalic anhydride in presence of cone \[{{H}_{2}}S{{O}_{4}}\]. The product gives pink colour with alkali. The product is
A)
phenolphthalein
done
clear
B)
bakelite
done
clear
C)
salicylic acid
done
clear
D)
fluorescein
done
clear
View Answer play_arrow
question_answer 62) When ethyl iodide is heated with dry silver oxide, it forms
A)
ethyl alcohol
done
clear
B)
diethyl ether
done
clear
C)
silver ethoxide
done
clear
D)
ethyl methyl ether
done
clear
View Answer play_arrow
question_answer 63) Oxidation of acetaldehyde with selenium dioxide produces
A)
ethanoic acid
done
clear
B)
methanoic acid
done
clear
C)
glyoxal
done
clear
D)
oxalic acid
done
clear
View Answer play_arrow
question_answer 64) The molal elevation constant of water is \[{{0.52}^{o}}C\]. The boiling point of 1.0 molal aqueous \[KCl\] solution (assuming complete dissociation of \[KCl\]) should be
A)
\[{{100.52}^{o}}C\]
done
clear
B)
\[{{101.04}^{o}}C\]
done
clear
C)
\[{{99.48}^{o}}C\]
done
clear
D)
\[{{98.96}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 65) The relative lowering of the vapour pressure is equal to the ratio between the number
A)
solute molecules to the solvent molecules
done
clear
B)
solute molecules to the total molecules the solution
done
clear
C)
solvent molecules to the total molecules B in the solution
done
clear
D)
solvent molecules to the total number ions of the solute
done
clear
View Answer play_arrow
question_answer 66) When a metal is burnt, its weight increased by 24 per cent. The equivalent weight of the metal will be
A)
2
done
clear
B)
24
done
clear
C)
33.3
done
clear
D)
76
done
clear
View Answer play_arrow
question_answer 67) Arsenic drugs are mainly used in the treatment of
A)
jaundice
done
clear
B)
typhoid
done
clear
C)
syphilis
done
clear
D)
cholera
done
clear
View Answer play_arrow
question_answer 68) Which compound/set of compounds is used in the manufacture of nylon-66?
A)
\[HOOC{{(C{{H}_{2}})}_{4}}COOH+{{H}_{2}}N{{(C{{H}_{2}})}_{6}}N{{H}_{2}}\]
done
clear
B)
\[C{{H}_{3}}=CH-C(C{{H}_{3}})=C{{H}_{2}}\]
done
clear
C)
\[C{{H}_{2}}=C{{H}_{2}}\]
done
clear
D)
done
clear
View Answer play_arrow
question_answer 69) Benzophenone can be converted into benzene using
A)
fused alkali
done
clear
B)
anhydrous \[AlC{{l}_{3}}\]
done
clear
C)
sodium amalgam in water
done
clear
D)
acidified dichromate
done
clear
View Answer play_arrow
question_answer 70) Which of the following carboxylic adds undergoes decarboxylation easily?
A)
\[{{C}_{6}}{{H}_{5}}-CO-C{{H}_{2}}-COOH\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}-CO-COOH\]
done
clear
C)
\[\underset{OH}{\mathop{\underset{|}{\mathop{{{C}_{6}}{{H}_{5}}-CH-COOH}}\,}}\,\]
done
clear
D)
\[\underset{N{{H}_{2}}}{\mathop{\underset{|}{\mathop{{{C}_{6}}{{H}_{5}}-CH-COOH}}\,}}\,\]
done
clear
View Answer play_arrow
question_answer 71) Amines are basic in character because they have
A)
a lone pair of electrons on the nitrogen atom
done
clear
B)
a hydroxyl group in the molecule
done
clear
C)
replaceable hydrogen atom
done
clear
D)
tetrahedral structure
done
clear
View Answer play_arrow
question_answer 72) Across the lanthanide series, the basicity of the lanthanide hydroxides
A)
increases
done
clear
B)
decreases
done
clear
C)
first increases and then decreases
done
clear
D)
first decreases and then increases
done
clear
View Answer play_arrow
question_answer 73) Which one of the following ions is colorless?
A)
\[C{{u}^{+}}\]
done
clear
B)
\[C{{o}^{2+}}\]
done
clear
C)
\[N{{i}^{2+}}\]
done
clear
D)
\[F{{e}^{3+}}\]
done
clear
View Answer play_arrow
question_answer 74) The correct IUPAC name of \[C{{H}_{3}}C{{H}_{2}}CH(C{{H}_{3}})CH{{({{C}_{2}}{{H}_{5}})}_{2}}\] is
A)
4-ethyl-3-methylhexane
done
clear
B)
3-ethyl-4-methylhexane
done
clear
C)
4-methyl-3-ethylhexane
done
clear
D)
2,4-diethylpentane
done
clear
View Answer play_arrow
question_answer 75) 60 g of a compound on analysis produced 24 g carbon, 4 g hydrogen and 32 g oxygen. The empirical formula of the compound is
A)
\[C{{H}_{2}}{{O}_{2}}\]
done
clear
B)
\[C{{H}_{2}}O\]
done
clear
C)
\[C{{H}_{4}}O\]
done
clear
D)
\[{{C}_{2}}{{H}_{4}}{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 76) Consider the isoelectronic series: \[{{K}^{+}}\], \[{{S}^{2-}}\], \[C{{l}^{-}}\] and \[C{{a}^{2+}}\] ,the radii of the ions decrease as
A)
\[C{{a}^{2+}}>{{K}^{+}}>C{{l}^{-}}>{{S}^{2-}}\]
done
clear
B)
\[C{{l}^{-}}>{{S}^{2-}}>{{K}^{+}}>C{{a}^{2+}}\]
done
clear
C)
\[{{S}^{2-}}>C{{l}^{-}}>{{K}^{+}}>C{{a}^{2+}}\]
done
clear
D)
\[{{K}^{+}}>C{{a}^{2+}}>{{S}^{2-}}>C{{l}^{-}}\]
done
clear
View Answer play_arrow
question_answer 77) In electrolysis of dil \[{{H}_{2}}S{{O}_{4}}\] using platinum electrodes
A)
\[{{H}_{2}}\] is evolved at cathode
done
clear
B)
\[S{{O}_{2}}\] is produced at anode
done
clear
C)
\[{{O}_{2}}\] is obtained at cathode
done
clear
D)
\[S{{O}_{2}}\] is produced at cathode
done
clear
View Answer play_arrow
question_answer 78) The heats of neutralisation of four acids A, B, C and D when neutralised against a common base are 13.7, 9.4, 11.2 and 12.4 kcal respectively. The weakest among these acids is
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
question_answer 79) In which of the following reactions, the underlined substance has been oxidised?
A)
\[\underline{B{{r}_{2}}}+{{H}_{2}}S\xrightarrow{{}}\underline{2HBr}+S\]
done
clear
B)
\[\underline{2HgC{{l}_{2}}}+SnC{{l}_{2}}\xrightarrow{{}}\underline{H{{g}_{2}}C{{l}_{2}}}+SnC{{l}_{4}}\]
done
clear
C)
\[C{{l}_{2}}+\underline{2KI}\xrightarrow{{}}2KCl+\underline{{{I}_{2}}}\]
done
clear
D)
\[\underline{2C{{u}^{2+}}}+4{{I}^{-}}\xrightarrow{{}}\underline{C{{u}_{2}}{{I}_{2}}}+{{I}_{2}}\]
done
clear
View Answer play_arrow
question_answer 80) A real gas most closely approaches the behaviour of an ideal gas at
A)
15 atm and 200 K
done
clear
B)
1 atm and 273 K
done
clear
C)
0.5 atm and 500 K
done
clear
D)
15 atm and 500 K
done
clear
View Answer play_arrow
question_answer 81) Average KE of \[C{{O}_{2}}\] at \[{{27}^{o}}C\] is E. The average kinetic energy of \[{{N}_{2}}\] at the same temperature will be
A)
\[E\]
done
clear
B)
\[22E\]
done
clear
C)
\[E/22\]
done
clear
D)
\[E/\sqrt{2}\]
done
clear
View Answer play_arrow
question_answer 82) The bond energies of \[C-C\], \[C=C\], \[H-H\] and \[C-H\] linkages are 350, 600, 400 and 410 kJ/mol respectively. The heat of hydrogenation of ethylene is
A)
\[-170\,kJ\,mo{{l}^{-1}}\]
done
clear
B)
\[-260\,kJ\,mo{{l}^{-1}}\]
done
clear
C)
\[-400\,kJ\,mo{{l}^{-1}}\]
done
clear
D)
\[-450\,kJ\,mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 83) The pH of \[{{10}^{-3}}M\,NaOH\] is
A)
\[3\]
done
clear
B)
\[11\]
done
clear
C)
\[4\]
done
clear
D)
\[-11\]
done
clear
View Answer play_arrow
question_answer 84) The digestion of fats in the intestine is aided by
A)
diffusion
done
clear
B)
protection
done
clear
C)
peptisation
done
clear
D)
emulsification
done
clear
View Answer play_arrow
question_answer 85) When \[Zn\] is treated with excess of \[NaOH\], the product obtained is
A)
\[Zn{{(OH)}_{2}}\]
done
clear
B)
\[ZnOH\]
done
clear
C)
\[N{{a}_{2}}Zn{{O}_{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 86) Which of the following alkyl halides is hydrolysed by \[{{S}_{N}}2\] mechanism?
A)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}Br\]
done
clear
B)
\[C{{H}_{3}}Br\]
done
clear
C)
\[C{{H}_{2}}=CHC{{H}_{2}}Br\]
done
clear
D)
\[{{(C{{H}_{3}})}_{3}}CBr\]
done
clear
View Answer play_arrow
question_answer 87) Ethylene combines with sulphur monochloride to form
A)
phosgene
done
clear
B)
mustard gas
done
clear
C)
methyl isocyanate (MIC)
done
clear
D)
lewisite
done
clear
View Answer play_arrow
question_answer 88) Which of the following has the highest solubility product?
A)
\[KOH\]
done
clear
B)
\[CsOH\]
done
clear
C)
\[LiOH\]
done
clear
D)
\[RbOH\]
done
clear
View Answer play_arrow
question_answer 89) Fluorine is not prepared by general methods because
A)
HF can be easily oxidized
done
clear
B)
HF cannot be easily oxidized
done
clear
C)
HF is highly poisonous
done
clear
D)
HF is a good conductor of electricity
done
clear
View Answer play_arrow
question_answer 90) A major constituent of Portland cement (except lime) is
A)
silica
done
clear
B)
alumina
done
clear
C)
iron oxide
done
clear
D)
magnesia
done
clear
View Answer play_arrow
question_answer 91) Which statement is not correct?
A)
A sigma bond is weaker than a \[\pi \] -bond
done
clear
B)
A sigma bond is stronger than a \[\pi \] -bond
done
clear
C)
A double bond is stronger than a sigma bond
done
clear
D)
A double bond is shorter than a single bond
done
clear
View Answer play_arrow
question_answer 92) Which pair of the elements will have the same chemical properties?
A)
\[13,22\]
done
clear
B)
\[3,11\]
done
clear
C)
\[4,24\]
done
clear
D)
\[2,1\]
done
clear
View Answer play_arrow
question_answer 93) The rate constant for the reaction,\[2{{N}_{2}}{{O}_{5}}\xrightarrow{{}}4N{{O}_{2}}+{{O}_{2}}\] is \[3.0\times {{10}^{-5}}{{s}^{-1}}\]. If the rate is \[2.40\times {{10}^{-5}}\,mol\,{{L}^{-1}}\,{{s}^{-1}}\] then the concentration of \[{{N}_{2}}{{O}_{5}}\] (in \[mol\,{{L}^{-1}}\]) is
A)
\[1.4\]
done
clear
B)
\[1.2\]
done
clear
C)
\[0.04\]
done
clear
D)
\[0.8\]
done
clear
View Answer play_arrow
question_answer 94) Which one of the following compounds has\[s{{p}^{2}}\]- hybridisation?
A)
\[C{{O}_{2}}\]
done
clear
B)
\[S{{O}_{2}}\]
done
clear
C)
\[{{N}_{2}}O\]
done
clear
D)
\[CO\]
done
clear
View Answer play_arrow
question_answer 95) Which of the following is used in dating archaeological findings?
A)
\[_{92}^{235}U\]
done
clear
B)
\[_{6}^{14}C\]
done
clear
C)
\[_{1}^{3}H\]
done
clear
D)
\[_{8}^{18}O\]
done
clear
View Answer play_arrow
question_answer 96) Standard reduction potentials for,\[L{{i}^{+}}/Li;\] \[Z{{n}^{2+}}/Zn;\]\[{{H}^{+}}/{{H}_{2}}\]and \[A{{g}^{+}}/Ag\] are \[-3.05,\] \[-0.762,\] \[0.00\] and \[+0.80\]V respectively. Which of the following has highest reducing capacity?
A)
\[Ag\]
done
clear
B)
\[{{H}_{2}}\]
done
clear
C)
\[Zn\]
done
clear
D)
\[Li\]
done
clear
View Answer play_arrow
question_answer 97) The ratio between the neutrons in C and Si with respect to atomic masses 12 and 28 is
A)
\[2:3\]
done
clear
B)
\[3:2\]
done
clear
C)
\[3:7\]
done
clear
D)
\[7:3\]
done
clear
View Answer play_arrow
question_answer 98) Fluorine reacts with water to give
A)
HF and \[{{O}_{2}}\]
done
clear
B)
HF and \[O{{F}_{2}}\]
done
clear
C)
HF and \[{{O}_{3}}\]
done
clear
D)
HF, \[{{O}_{2}}\] and \[{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 99) Identify Z in the following series.\[{{C}_{2}}{{H}_{5}}I\xrightarrow{Alc.\,KOH}X\xrightarrow{B{{r}_{2}}}Y\xrightarrow{KCN}Z\]
A)
\[C{{H}_{3}}C{{H}_{2}}CN\]
done
clear
B)
\[NCC{{H}_{2}}-C{{H}_{2}}CN\]
done
clear
C)
\[BrC{{H}_{2}}-C{{H}_{2}}CN\]
done
clear
D)
\[BrCH=CHCN\]
done
clear
View Answer play_arrow
question_answer 100) 1-butyne on oxidation with hot alkaline \[KMn{{O}_{4}}\] would yield
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}COOH\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}COOH\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}COOH+C{{O}_{2}}+{{H}_{2}}O\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}COOH+HCOOH\]
done
clear
View Answer play_arrow
question_answer 101) The IV, V and IX cranial nerves are
A)
olfactory, spinal accessory and vagus
done
clear
B)
trigeminal, vagus and glossopharyngeal
done
clear
C)
oculomotor, trigeminal and hypoglossal
done
clear
D)
pathetic (trochlear), trigeminal and glossopharyngeal
done
clear
View Answer play_arrow
question_answer 102) The cranial capacity of Australopithecus was about 500 cc. It existed in
A)
Miocene
done
clear
B)
Pliocene
done
clear
C)
Pleistocene
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 103) Hormones differ from enzymes in that they are
A)
found only in animals
done
clear
B)
found only in plants
done
clear
C)
not used up at all in metabolism
done
clear
D)
used up in metabolism
done
clear
View Answer play_arrow
question_answer 104) Hyaluronic acid is a hetero polysaccharide and has acetyl glucosamine and glucoronic acid. It is a cementing material and found in
A)
ovum and synovial fluid
done
clear
B)
vitreous humour
done
clear
C)
cerebrospinal fluid
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 105) Mammals having traits of both reptiles and mammals are
A)
monotremes
done
clear
B)
marsupials
done
clear
C)
whales
done
clear
D)
bats
done
clear
View Answer play_arrow
question_answer 106) In man, the sperms are stored and nourished in
A)
testes
done
clear
B)
vasa efferentia
done
clear
C)
epididymis
done
clear
D)
spermatic cord
done
clear
View Answer play_arrow
question_answer 107) The major excretory product of arthropods is
A)
ammonia
done
clear
B)
urea
done
clear
C)
uric acid
done
clear
D)
trimethylamine oxide
done
clear
View Answer play_arrow
question_answer 108) The famous book origin of species was written by Charles Darwin in
A)
1809
done
clear
B)
1859
done
clear
C)
1885
done
clear
D)
1871
done
clear
View Answer play_arrow
question_answer 109) Test tube baby implies
A)
fertilisation and development of ovum in test tube
done
clear
B)
fertilisation in test tube and development in uterus
done
clear
C)
fertilisation in uterus and development in test tube
done
clear
D)
fertilisation and development in uterus
done
clear
View Answer play_arrow
question_answer 110) The enzymes responsible for the digestion of starch in food of man is present in
A)
salivary and gastric secretions
done
clear
B)
salivary and pancreatic secretions
done
clear
C)
gastric and pancreatic secretions
done
clear
D)
gastric and duodenal secretions
done
clear
View Answer play_arrow
question_answer 111) What is the main difference between RBCs of human and frog?
A)
Human RBCs are non-nucleated
done
clear
B)
Haemoglobin is found only in human RBCs
done
clear
C)
Human RBCs have nucleus
done
clear
D)
Human RBCs are multi-nucleated
done
clear
View Answer play_arrow
question_answer 112) Wauchereria bancrofti is transmitted by
A)
sandfly
done
clear
B)
tse-tse fly
done
clear
C)
Anopheles mosquito
done
clear
D)
Culex mosquito
done
clear
View Answer play_arrow
question_answer 113) It is not possible to create life because
A)
we can not match creativity and intelligence of God
done
clear
B)
nature of molecular organisation is complex and not fully known
done
clear
C)
all mode of life are not fully known
done
clear
D)
most of modes are known except a few
done
clear
View Answer play_arrow
question_answer 114) Ageing is characterised by
A)
decline in metabolic activity
done
clear
B)
increased metabolic activity
done
clear
C)
increased anabolism
done
clear
D)
increased catabolism
done
clear
View Answer play_arrow
question_answer 115) Which organ of the body acts as filter of blood?
A)
Bone marrow
done
clear
B)
Liver
done
clear
C)
Lymph glands
done
clear
D)
Spleen
done
clear
View Answer play_arrow
question_answer 116) Honey has three sugars, they are
A)
glucose, fructose and lactose
done
clear
B)
glucose, galactose and inulin
done
clear
C)
dextrose, laevulose and maltose
done
clear
D)
dextrose, lactose and ribose
done
clear
View Answer play_arrow
question_answer 117) The clitellum of earthworm is for
A)
reproduction
done
clear
B)
helping in respiration
done
clear
C)
helping in locomotion
done
clear
D)
forming a wall of cocoon
done
clear
View Answer play_arrow
question_answer 118) Trophocytes, mycetocytes, oenocytes and urate cells are found in the fat body of cockroach. Which statement is correct?
A)
Trophocytes contain reserve food
done
clear
B)
Mycetocytes contain symbiotic bacteria
done
clear
C)
Oenocytes sex wax and urate cells contain uric acid
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 119) Auxetic growth is increase in
A)
cell volume only
done
clear
B)
cell number only
done
clear
C)
fatty tissue
done
clear
D)
intercellular material
done
clear
View Answer play_arrow
question_answer 120) The epithelial cells lining the stomach of vertebrates is protected from damage by HCl because
A)
HCl is too dilute
done
clear
B)
the epithelial cells are resistant to the action of HCl
done
clear
C)
HCl is neutralised by alkaline gastric juice
done
clear
D)
the epithelial cells are covered with a mucous secretion
done
clear
View Answer play_arrow
question_answer 121) Reason, will, memory, intelligence and emotions are governed by
A)
cerebellum
done
clear
B)
mid brain
done
clear
C)
medulla oblongata
done
clear
D)
cerebral hemispheres
done
clear
View Answer play_arrow
question_answer 122) The drugs which do not develop physiological dependence are
A)
opiates
done
clear
B)
sedatives
done
clear
C)
stimulants
done
clear
D)
hallucinogens
done
clear
View Answer play_arrow
question_answer 123) In Ascaris, eggs are fertilised in the
A)
vagina
done
clear
B)
oviducts
done
clear
C)
lower pans of uteri
done
clear
D)
seminal vesicles
done
clear
View Answer play_arrow
question_answer 124) Iron free compound of haemoglobin is
A)
globin
done
clear
B)
haematin
done
clear
C)
bilirubin
done
clear
D)
haemotoxin
done
clear
View Answer play_arrow
question_answer 125) The expulsion of completely developed foetus from the uterus is known as
A)
ovulation
done
clear
B)
oviposition
done
clear
C)
gestation
done
clear
D)
parturition
done
clear
View Answer play_arrow
question_answer 126) The banding pattern of chromosome three and six of human beings and chimpanzee show that they had
A)
different origin
done
clear
B)
common origin
done
clear
C)
same number of chromosomes
done
clear
D)
similar blood groups
done
clear
View Answer play_arrow
question_answer 127) Blood of Pheretima is
A)
blue with haemocyanin in corpuscles
done
clear
B)
blue with haemoglobin in corpuscles
done
clear
C)
red with haemoglobin in corpuscles
done
clear
D)
red with haemoglobin in plasma
done
clear
View Answer play_arrow
question_answer 128) If parathyroid gland degenerates which activity is disturbed?
A)
Growth
done
clear
B)
Calcium concentration
done
clear
C)
Potassium concentration
done
clear
D)
Sodium concentration
done
clear
View Answer play_arrow
question_answer 129) Some times immune system of body start behaving against the own body or self. This lead to
A)
autoimmune disease
done
clear
B)
bacterial disease
done
clear
C)
viral disease
done
clear
D)
hereditary disease
done
clear
View Answer play_arrow
question_answer 130) Genetic drift is the elimination of
A)
individuals
done
clear
B)
chromosomes
done
clear
C)
species
done
clear
D)
gene of some original characteristic of a species due to epidemics
done
clear
View Answer play_arrow
question_answer 131) Nucleosome gives beaded appearance to chromosomes. They help in packing of DNA in the chromosomes. A nucleosome has
A)
about two turns of DNA
done
clear
B)
8 histone molecules (2 molecuies each of \[{{H}_{2}}A,\text{ }{{H}_{2}}B,\text{ }{{H}_{3}}\]and \[{{H}_{4}}\])
done
clear
C)
166 nitrogen base pairs
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 132) Pesticides ingredient which killed 2,500 people in Bhopal gas tragedy in 1984 is
A)
mustard gas
done
clear
B)
DDT
done
clear
C)
methyl isocyanate
done
clear
D)
carbon tetrachloride
done
clear
View Answer play_arrow
question_answer 133) According to Haeckels biogenetic law
A)
development of individual metazoan shows embryonic characters of ancestors
done
clear
B)
ontogeny repeats phylogeny
done
clear
C)
germplasm is immortal
done
clear
D)
every organism is produced by its parents
done
clear
View Answer play_arrow
question_answer 134) Which of the following pair is characterized by swollen lip, thick pigmented skin of hands and legs and irritability?
A)
Thiamine - Beri beri
done
clear
B)
Protein - Kwashiorkor
done
clear
C)
Nicotinamide - Pellagra
done
clear
D)
Iodine - Goitre
done
clear
View Answer play_arrow
question_answer 135) Ascaris protects itself from effect of digestive enzymes of the host by means of
A)
antienzymes
done
clear
B)
cuticle
done
clear
C)
Both (a) and (b)
done
clear
D)
mucus
done
clear
View Answer play_arrow
question_answer 136) Which set includes all homologous organs?
A)
Hind legs of dogs, duck and kangaroo
done
clear
B)
Wings of bat, butterflies and birds
done
clear
C)
String of honey bee, scorpion and mosquito
done
clear
D)
Tail of rat, peacock and earthworm
done
clear
View Answer play_arrow
question_answer 137) Which stages of Plasmodium are likely to be found in the stomach of a female Anopheles. Which has just taken a blood meal from a patient of malaria?
A)
Merozoites, trophozoites and gametocytes
done
clear
B)
Sporozoites, trophozoites and gametes
done
clear
C)
Merozoites, gametocytes and zygotes
done
clear
D)
Gametocytes, ookinetes and gametes
done
clear
View Answer play_arrow
question_answer 138) Which of the following cells have lost their capacity to divided in order to get specialised?
A)
Muscle cells and nerve cells
done
clear
B)
Bone marrow cells and liver cells
done
clear
C)
Kidney and intestinal cells
done
clear
D)
Retinal cells
done
clear
View Answer play_arrow
question_answer 139) The blood flow through human heart visualised by
A)
magnetic resonance imaging
done
clear
B)
DSA
done
clear
C)
sonography
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 140) Chromosome are best observed ai metaphase. For studying the shape, best stage is
A)
meca phase
done
clear
B)
anaphase
done
clear
C)
telophase
done
clear
D)
late prophase
done
clear
View Answer play_arrow
question_answer 141) Selection will not eliminate lethal recessive gene from a large population because
A)
there are always some heterozygotes carriers of the alleles
done
clear
B)
gene fixation occurs in populations
done
clear
C)
heterozygotes are at a selective advantage
done
clear
D)
rate of mutation to the lethal allele is high
done
clear
View Answer play_arrow
question_answer 142) Population 1 and II are growing in the same habitat and are morphologically similar but these are inter sterile. In term of biological concept of species, they may be considered as
A)
one species
done
clear
B)
two distinct species
done
clear
C)
two sibling species
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 143) Diagrammatic representation of all chromosomes at metaphase is called
A)
ideogram
done
clear
B)
cardiogram
done
clear
C)
karyotype
done
clear
D)
leotype
done
clear
View Answer play_arrow
question_answer 144) Which one is the most important characteristic feature of birds?
A)
Forelimbs modified into wings
done
clear
B)
Bipedal vertebrate
done
clear
C)
Homoiothermal
done
clear
D)
Exoskeleton of feathers
done
clear
View Answer play_arrow
question_answer 145) Inter-phalangial joints are also called as
A)
fixed joints
done
clear
B)
hinge joints
done
clear
C)
movable joints
done
clear
D)
straight joints
done
clear
View Answer play_arrow
question_answer 146) The protoplasmic segment of a striated muscle fibre is termed as
A)
sarcoplasm
done
clear
B)
sarcomere
done
clear
C)
neuromere
done
clear
D)
metamere
done
clear
View Answer play_arrow
question_answer 147) Steroid hormones are similar in structure to
A)
tryosine
done
clear
B)
cholesterol
done
clear
C)
coenzyme-A
done
clear
D)
glycerol
done
clear
View Answer play_arrow
question_answer 148) Meroblastic cleavage refers to which type of division of egg?
A)
Complete
done
clear
B)
Spiral
done
clear
C)
Incomplete
done
clear
D)
Horizontal
done
clear
View Answer play_arrow
question_answer 149) Outer covering of cartilage is called
A)
perichondrium
done
clear
B)
periosteum
done
clear
C)
endostemum
done
clear
D)
perilonertum
done
clear
View Answer play_arrow
question_answer 150) Toxic substances are detoxified in human body in
A)
kidney
done
clear
B)
lungs
done
clear
C)
liver
done
clear
D)
stomach
done
clear
View Answer play_arrow
question_answer 151) Pyrenoids are centre of
A)
fat storage
done
clear
B)
starch storage
done
clear
C)
protein formation
done
clear
D)
enzyme formation
done
clear
View Answer play_arrow
question_answer 152) Damping off of seedling is due to
A)
nematode
done
clear
B)
Albugo Candida
done
clear
C)
Fusarium oxysporium
done
clear
D)
Pythium debaryanum
done
clear
View Answer play_arrow
question_answer 153) Sulphur containing amino acids are
A)
valine, lysine and cysteine
done
clear
B)
tryptophan, glutamic acid and aspartic acid
done
clear
C)
citrulline, methionine and giumeric acid
done
clear
D)
cysreine, cystine and methionine
done
clear
View Answer play_arrow
question_answer 154) Which one of the following not used in production of yoghurt?
A)
Streptococcus lact is
done
clear
B)
Streptococcus the rmophilus
done
clear
C)
Lactobacillus vulgaricus
done
clear
D)
Acetobacter aceti
done
clear
View Answer play_arrow
question_answer 155) Which of the following statements is correct?
A)
Xylem is made of all living cells
done
clear
B)
Xylem is made of living and non-living cells
done
clear
C)
Xylem is made of non-living cells
done
clear
D)
Xylem does not contain cells
done
clear
View Answer play_arrow
question_answer 156) Age of the plant can be calculated by
A)
measuring its height
done
clear
B)
counting leaf bases
done
clear
C)
counting annual rings
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 157) The cell cultured in vitro gives rise to complete plant. This ability of plant cell is known as
A)
growth
done
clear
B)
development
done
clear
C)
regeneration
done
clear
D)
totipotency
done
clear
View Answer play_arrow
question_answer 158) Cohesion force theory was proposed by
A)
JC Bose (1850)
done
clear
B)
Dixon and Jolly (1950)
done
clear
C)
Benson and Calvin (1855)
done
clear
D)
Dixon and Jolly (1894)
done
clear
View Answer play_arrow
question_answer 159) Living fossils means
A)
primitive organised organisms
done
clear
B)
extinct organisms
done
clear
C)
organisms with ancestral characters and time has not changed them
done
clear
D)
connecting link between two groups
done
clear
View Answer play_arrow
question_answer 160) The process of photosynthesis is
A)
reductive, exergonic and catabolic
done
clear
B)
reductive, endergonic and catabolic
done
clear
C)
reductive, exergonic and anabolic
done
clear
D)
reductive, endergonic and anabolic
done
clear
View Answer play_arrow
question_answer 161) Unit of distance between genes in a chromosome is known as
A)
DNA
done
clear
B)
Morgan
done
clear
C)
Centimorgan
done
clear
D)
Spacer
done
clear
View Answer play_arrow
question_answer 162) Which is the correct sequence of code transfer involved in the formation of polypeptide?
A)
DNA-rRNA-rRNA-mRNA
done
clear
B)
tRNA-DNA-mRNA-rRNA
done
clear
C)
mRNA-fRNA-DNA-Amino acid
done
clear
D)
DNA-mRNA-fRNA-Amino acid
done
clear
View Answer play_arrow
question_answer 163) Study of pollen grains is called
A)
Spermology
done
clear
B)
Anatomy
done
clear
C)
Palynology
done
clear
D)
Aerobiology
done
clear
View Answer play_arrow
question_answer 164) Golgi complex is specialised for
A)
energy transduction
done
clear
B)
glycosidation of lipids
done
clear
C)
digestion
done
clear
D)
conversion of energy
done
clear
View Answer play_arrow
question_answer 165) Which is not a intracellular compartment in the cell?
A)
Nucleus
done
clear
B)
Mitochondria
done
clear
C)
Chloroplast
done
clear
D)
Centriole
done
clear
View Answer play_arrow
question_answer 166) The conservation of local (endemic) varieties of crop is must because
A)
these are source of gene for genetic diversity
done
clear
B)
these are high yielding and nutritious
done
clear
C)
we want to conserve all plants and animals of the past for our knowledge
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 167) The term taxonomy was coined by
A)
de Candolle
done
clear
B)
Linnaeus
done
clear
C)
John Ray
done
clear
D)
Bauchin
done
clear
View Answer play_arrow
question_answer 168) A lichen responsible for forest fire is
A)
Lecanora
done
clear
B)
Usnea
done
clear
C)
Cladonia
done
clear
D)
Ramalina
done
clear
View Answer play_arrow
question_answer 169) The carbon dioxide acceptor in \[{{\text{C}}_{\text{3}}}\text{-}\]plants is
A)
phosphoenol pyruvate (PEP)
done
clear
B)
ribulose 1, 5-diphosphate (RuDP)
done
clear
C)
phosphoglyceric acid
done
clear
D)
ribulose mono phosphate CRuMP)
done
clear
View Answer play_arrow
question_answer 170) Fusion of two vegetative nuclei represents the phenomenon of
A)
parasexual reproduction
done
clear
B)
partheno apogamy
done
clear
C)
apogamy
done
clear
D)
apospory
done
clear
View Answer play_arrow
question_answer 171) In a given RNA segment AUG. ACC, UGG, ACC, CCA, UCA, if the first base gets mutated the effect of this on coding by this RNA segment will result in
A)
a change of first amino acid only
done
clear
B)
a complete change in the types as well as the sequence of almost all amino acids
done
clear
C)
no change in the sequence of amino acids
done
clear
D)
one amino acid less in protein chain
done
clear
View Answer play_arrow
question_answer 172) When certain solute panicles are added to the pure solvent, the diffusion pressure of the resulting solution lowered by an amount repressed in term of
A)
Turgor Pressure (TP)
done
clear
B)
Diffusion Pressure (DP)
done
clear
C)
Osmotic Pressure (OP)
done
clear
D)
Diffusion Pressure Deficit (DPD)
done
clear
View Answer play_arrow
question_answer 173) The callus culture and suspension culture produce plantlets. They are distinguished by
A)
callus culture is in vitro while suspension is in vivo
done
clear
B)
in callus culture shoots are organized whereas in suspension culture somatic embryo is organized
done
clear
C)
in callus culture hormone is present but in suspension culture hormone is absent
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 174) Angiosperms differ from gymnosperms in having
A)
broad leaves
done
clear
B)
tracheids
done
clear
C)
fruit(ovary)
done
clear
D)
cotyledons
done
clear
View Answer play_arrow
question_answer 175) Undecomposed organic matter in soil is called
A)
litter
done
clear
B)
humus
done
clear
C)
mull
done
clear
D)
duft
done
clear
View Answer play_arrow
question_answer 176) Meristematic cells are
A)
thin-walled, less protoplasmic, isodiametric and nucleated
done
clear
B)
thin-walled, densely protoplasmic, isodiametric and nucleated
done
clear
C)
thick-walled, less protoplasmic, isodiametric and nucleated
done
clear
D)
thick-walled, densely protoplasmic, isodiametric and non-nucleated
done
clear
View Answer play_arrow
question_answer 177) Tubulin protein occur in
A)
enzymes of Krebs cycle
done
clear
B)
rough endoplasmic reticulum
done
clear
C)
microtubules
done
clear
D)
microfilaments
done
clear
View Answer play_arrow
question_answer 178) Vaccines prepared by genetic engineering are safe to man because they are
A)
least active form of virus
done
clear
B)
active form of virus
done
clear
C)
coat protein formed as antibody
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 179) The principle agent of alcoholic fermentation and bread making is
A)
Schizosaccharomyces octosporus
done
clear
B)
Saccharomyces cerevisiae
done
clear
C)
Sacc/iaromyces ellipsiodeus
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 180) Which one of the following is a pseudo cereals?
A)
Wheat
done
clear
B)
Rice
done
clear
C)
Buck wheat
done
clear
D)
Pisum sativum
done
clear
View Answer play_arrow
question_answer 181) Carrying capacity of a population is determined by its
A)
population growth rate
done
clear
B)
birth rate
done
clear
C)
death rate
done
clear
D)
limiting resource
done
clear
View Answer play_arrow
question_answer 182) Succession is
A)
gradual, convergent, directional and continuous process
done
clear
B)
series of biotic communities that appear gradually in a barren area
done
clear
C)
orderly process of community change till stability
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 183) Ovule is inverted with body fused to funicle, micropyle lying close to hilum and facing the placenta, it is
A)
hemitropous
done
clear
B)
orthon-opous
done
clear
C)
anatropous
done
clear
D)
campylotropous
done
clear
View Answer play_arrow
question_answer 184) During elongation of polypeptide chain, the sigma factor
A)
is released to take part again
done
clear
B)
is retained and performs special function
done
clear
C)
its function is not known
done
clear
D)
is used during the closing of chain
done
clear
View Answer play_arrow
question_answer 185) Which one of the following belong to the same category?
A)
Cashewnut, coconut and chestnut
done
clear
B)
Coconut, orange and tomato
done
clear
C)
Betelnut, chestnut and coconut
done
clear
D)
Mango, almond and coconut
done
clear
View Answer play_arrow
question_answer 186) The basic unit of classification is
A)
species
done
clear
B)
taxon
done
clear
C)
category
done
clear
D)
sub-species
done
clear
View Answer play_arrow
question_answer 187) As the rice deficient in vitamin-A, the rice which have the vitamin-A transformed by Agro bacrerium. Its common name is
A)
basmati rice
done
clear
B)
white rice
done
clear
C)
golden rice
done
clear
D)
yellow rice
done
clear
View Answer play_arrow
question_answer 188) Operon unit consists of
A)
regulator, operator and repressive gene
done
clear
B)
regulator, structural and operator gene
done
clear
C)
regulator, structural, operator and promoter gene
done
clear
D)
regulator, structural and promoter gene
done
clear
View Answer play_arrow
question_answer 189) Artificial application of auxins like IAA, IBA and NAA to unpollinated pistils can form
A)
fruits with much flesh
done
clear
B)
larger fruits
done
clear
C)
sweet fruits
done
clear
D)
seedless fruits
done
clear
View Answer play_arrow
question_answer 190) Site of formation of ribosomal precursor or ribosomal subunits in cell is
A)
nucleus
done
clear
B)
nucleolus
done
clear
C)
nucleus body
done
clear
D)
stroma
done
clear
View Answer play_arrow
question_answer 191) Which cycle is directly driven by solar radiations?
A)
Phosphorus
done
clear
B)
Carbon
done
clear
C)
Water
done
clear
D)
Nitrogen
done
clear
View Answer play_arrow
question_answer 192) Zygospore of Spirogyra, at the time of meiosis, is divided into four nuclei. How many nuclei degenerate out of these four?
A)
One
done
clear
B)
Two
done
clear
C)
Three
done
clear
D)
Four
done
clear
View Answer play_arrow
question_answer 193) Sometimes, the fern plant arises from fern prothallus without fertilisation. This is an example of
A)
apospory
done
clear
B)
apogamy
done
clear
C)
parthenocarpy
done
clear
D)
gametogenesis
done
clear
View Answer play_arrow
question_answer 194) Clove is
A)
flower bud
done
clear
B)
axillary bud
done
clear
C)
thalamus
done
clear
D)
ovule
done
clear
View Answer play_arrow
question_answer 195) Cycas is
A)
monoecious
done
clear
B)
bisexual
done
clear
C)
dioecious
done
clear
D)
hermaphrodite
done
clear
View Answer play_arrow
question_answer 196) The drug belladonna is obtained from
A)
Atropa
done
clear
B)
Opium
done
clear
C)
Rauwolffia
done
clear
D)
Solanum
done
clear
View Answer play_arrow
question_answer 197) Which part of embryo comes out first during seed germination?
A)
Radicle
done
clear
B)
Plumule
done
clear
C)
Hypocotyl
done
clear
D)
Epicotyl
done
clear
View Answer play_arrow
question_answer 198) Porous wood contains
A)
vessels
done
clear
B)
tracheids
done
clear
C)
fibres
done
clear
D)
parenchyma
done
clear
View Answer play_arrow
question_answer 199) In rainy season, door get swelled due to
A)
imbibitions
done
clear
B)
diffusion
done
clear
C)
transpiration
done
clear
D)
respiration
done
clear
View Answer play_arrow
question_answer 200) Monocarpic plants flower
A)
once
done
clear
B)
twice
done
clear
C)
many times
done
clear
D)
never
done
clear
View Answer play_arrow