question_answer 1) If we move from equator to pole value of g :
A)
first increases then decreases
done
clear
B)
remains same
done
clear
C)
increases
done
clear
D)
decreases
done
clear
View Answer play_arrow
question_answer 2) Wb/m2 is equal to :
A)
dyne
done
clear
B)
tesla
done
clear
C)
watt
done
clear
D)
henry
done
clear
View Answer play_arrow
question_answer 3) A tuning fork vibrates with two beats in 0.4 s, the frequency is :
A)
6 Hz
done
clear
B)
8 Hz
done
clear
C)
5 Hz
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 4) A bus starts from rest with an acceleration of \[1m/{{s}^{2}}.\]A man who is 48 m behind the bus starts with a uniform velocity of 10 m/s. Then the minimum time after which the man will catch the bus?
A)
4s
done
clear
B)
10 s
done
clear
C)
12 s
done
clear
D)
8 s
done
clear
View Answer play_arrow
question_answer 5) If a cyclist moving with a speed of 4,9 m/s on a level road can take a sharp circular turn of radius 4m. Then coefficient of friction between the cycle tyre and the road is:
A)
0.71
done
clear
B)
0.61
done
clear
C)
0.51
done
clear
D)
0.81
done
clear
View Answer play_arrow
question_answer 6) A drum of radius R full of liquid of density d is rotated at (d rad/s. The increase in pressure at die centre of the drum will be :
A)
\[\frac{{{\omega }^{2}}{{R}^{2}}{{d}^{2}}}{2}\]
done
clear
B)
\[\frac{\omega R{{d}^{2}}}{2}\]
done
clear
C)
\[\frac{{{\omega }^{2}}Rd}{2}\]
done
clear
D)
\[\frac{{{\omega }^{2}}{{R}^{2}}d}{2}\]
done
clear
View Answer play_arrow
question_answer 7) A body of mass M at rest explodes into three masses two of which of mass\[\frac{M}{4}\]each are thrown off in perpendicular directions with velocity of 3m/sand 4 m/s respectively. The third piece will be thrown off with a velocity of :
A)
3 m/s
done
clear
B)
2.5 m/s
done
clear
C)
2.0 m/s
done
clear
D)
1.5 m/s
done
clear
View Answer play_arrow
question_answer 8) The distance of a geostationary satellite from the centre of earth (radius R = 6400 km) is nearest 10:
A)
18 R
done
clear
B)
10 R
done
clear
C)
7R
done
clear
D)
5R
done
clear
View Answer play_arrow
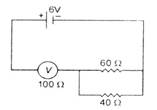
question_answer 9)
The measurement of voltmeter in the following circuit is :
A)
6.0V
done
clear
B)
40V
done
clear
C)
3.4V
done
clear
D)
25V
done
clear
View Answer play_arrow
question_answer 10) Rutherford's M-particle experiment shows that the atoms have :
A)
electrons
done
clear
B)
neutrons
done
clear
C)
nucleus
done
clear
D)
protons
done
clear
View Answer play_arrow
question_answer 11) A simple pendulum is executing simple harmonic motion with a lime period 7', II the length of the pendulum is increased by 21% the increase in the time period of the pendulum of increased length is :
A)
50%
done
clear
B)
30%
done
clear
C)
21 %
done
clear
D)
10 %
done
clear
View Answer play_arrow
question_answer 12) A panicle is thrown vertically upwards. Its velocity at half of the height is 10 nVs. Then the maximum height aimed by it is : (g=10m/s2)
A)
16 m
done
clear
B)
10 m
done
clear
C)
8m
done
clear
D)
18m
done
clear
View Answer play_arrow
question_answer 13) A satellite of mass m is circulating around the earth with constant angular velocity. If radius of the orbit is R0 and mass of earth is M, the angular momentum of the satellite is :
A)
\[M\sqrt{\frac{Gm}{{{R}_{0}}}}\]
done
clear
B)
\[m\sqrt{\frac{GM}{{{R}_{0}}}}\]
done
clear
C)
\[M\sqrt{Gm{{R}_{0}}}\]
done
clear
D)
\[m\sqrt{GM{{R}_{0}}}\]
done
clear
View Answer play_arrow
question_answer 14) The potentiometer consists of a wire of length 4m and resistance \[10{{C}_{2}}.\]It is connected to a cell of emf 2V the potential difference per unit length of the wire will be :
A)
10V/m
done
clear
B)
5 V/m
done
clear
C)
2 V/m
done
clear
D)
0.5 V/m
done
clear
View Answer play_arrow
question_answer 15) The energy released by fission of one atom of \[_{92}{{U}^{235}}\] is 200 MeV the number of fissions required per second to produce a power of 1 kW is :
A)
\[3.125\times {{10}^{9}}\]
done
clear
B)
\[3.125\times {{10}^{12}}\]
done
clear
C)
\[3.125\times {{10}^{13}}\]
done
clear
D)
\[3.125\times {{10}^{11}}\]
done
clear
View Answer play_arrow
question_answer 16) A radioactive element \[_{90}{{X}^{238}}\] decays into \[_{83}{{Y}^{222}}\]. then the number of \[\beta \]-panicles emitted are :
A)
1
done
clear
B)
2
done
clear
C)
4
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 17) The concentric spheres of radii R and r have similar charges with equal surface densities (\[\sigma \]) what is the electric potential at their common centre?
A)
\[\frac{\sigma }{{{\varepsilon }_{0}}}(R-r)\]
done
clear
B)
\[\frac{\sigma }{{{\varepsilon }_{0}}}(R+r)\]
done
clear
C)
\[\frac{R\sigma }{{{\varepsilon }_{0}}}\]
done
clear
D)
\[\frac{\sigma }{{{\varepsilon }_{0}}}\]
done
clear
View Answer play_arrow
question_answer 18) What is the de-Broglie wavelength of 1 kg mass moving with a velocity of 10 ms-1?
A)
\[6.626\times {{10}^{-35}}m\]
done
clear
B)
\[6.626\times {{10}^{-33}}m\]
done
clear
C)
\[6.626\times {{10}^{-34}}m\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 19) Two waves, whose intensities are 9 : 16 are made to interfere, the ratio of maximum and minimum intensities in the interference pattern is :
A)
49 : 1
done
clear
B)
25 : 7
done
clear
C)
10 : 9
done
clear
D)
4 : 3
done
clear
View Answer play_arrow
question_answer 20) A charge Q\[\mu \]C is placed at the centre of cube, the flux coming out from any surface will be :
A)
\[\frac{Q}{24{{\varepsilon }_{0}}}\]
done
clear
B)
\[\frac{Q}{8{{\varepsilon }_{0}}}\]
done
clear
C)
\[\frac{Q}{6{{\varepsilon }_{0}}}\times {{10}^{-6}}\]
done
clear
D)
\[\frac{Q}{6{{\varepsilon }_{0}}}\times {{10}^{-3}}\]
done
clear
View Answer play_arrow
question_answer 21) The reactance of a coil when used in the AC power supply (220 V, 50 cycle/s) is 50 ii. The inductance of the coil is nearly :
A)
0.16 H
done
clear
B)
0.22 H
done
clear
C)
2.2 H
done
clear
D)
1.6 H
done
clear
View Answer play_arrow
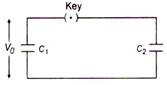
question_answer 22)
A capacitor C1 is charged by a potential difference v0 as shown in the figure. This charging battery is then removed and the capacitor is connected as into an uncharged capacitor C2. What is the final potential difference V across the combination?
A)
\[{{V}_{0}}\frac{({{C}_{1}}-{{C}_{2}})}{{{C}_{1}}}\]
done
clear
B)
\[{{V}_{0}}\frac{{{C}_{1}}}{({{C}_{1}}-{{C}_{2}})}\]
done
clear
C)
\[{{V}_{0}}\frac{{{C}_{1}}}{({{C}_{1}}+{{C}_{2}})}\]
done
clear
D)
\[{{V}_{0}}\frac{({{C}_{1}}+{{C}_{2}})}{{{C}_{1}}}\]
done
clear
View Answer play_arrow
question_answer 23) Two rods having thermal conductivity in the ratio of 5 : 3 and having equal length and equal cross-sectional area. are joined face to face. If the temperature of free end of first rod is \[100{}^\circ C\] and temperature of free end of second rod is \[20{}^\circ C\], temperature of junction will be:
A)
\[90{}^\circ C\]
done
clear
B)
\[85{}^\circ C\]
done
clear
C)
\[70{}^\circ C\]
done
clear
D)
\[50{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 24) Two organ pipes sounded together give 5 beats per second. If their lengths are in the ratio of 50 to 51, then the frequency of shorter and longer pipes in Hz are respectively :
A)
250,245
done
clear
B)
245,250
done
clear
C)
250,255
done
clear
D)
255,250
done
clear
View Answer play_arrow
question_answer 25) An organ pipe P1 is closed at one end and vibrating in its first overtone and another pipe P2 opened at both ends vibrating in its third overtone are in resonance with a given tuning fork. Then the ratio of P1 and P2 is :
A)
\[\frac{1}{3}\]
done
clear
B)
\[\frac{2}{3}\]
done
clear
C)
\[\frac{8}{3}\]
done
clear
D)
\[\frac{3}{8}\]
done
clear
View Answer play_arrow
question_answer 26)
Two light springs of force constants \[{{k}_{1}}\] and \[{{k}_{2}}\] a block of mass m are in one line. AB on a smooth horizontal table such that one end of each spring is fixed 10 a required support and other end is attached to block of mass m kg as shown in figure. that frequency of vibration is :
A)
\[n=\frac{1}{2\pi }\sqrt{\frac{{{k}_{1}}-{{k}_{2}}}{m}}\]
done
clear
B)
\[n=\frac{1}{2\pi }\sqrt{\frac{{{k}_{1}}+{{k}_{2}}}{m}}\]
done
clear
C)
\[n=\frac{1}{2\pi }\sqrt{\frac{{{k}_{1}}{{k}_{2}}}{n}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 27) Two panicles are excuting simple harmonic motion. At an instant of lime t their displacements are \[{{Y}_{1}}=\alpha \cos (\omega t)\] and \[{{Y}_{2}}=\alpha \sin (\omega t)\] Then the phase difference between.\[{{Y}_{1}}\] and \[{{Y}_{2}}\] is:
A)
\[120{}^\circ\]
done
clear
B)
\[90{}^\circ\]
done
clear
C)
\[180{}^\circ\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 28) Acapillary tube of radius r is dipped vertically in a liquid of density d, surface tension T and angle of contact 0 then the pressure difference just below the two surfaces, one in the beaker and the other in the capillary tube , is :
A)
\[\frac{2T}{r}\]
done
clear
B)
\[\frac{T}{r\cos \theta }\]
done
clear
C)
\[\frac{2T\cos \theta }{r}\]
done
clear
D)
\[\frac{T\cos \theta }{r}\]
done
clear
View Answer play_arrow
question_answer 29) 1 mole of monoatomic gas \[\left( \gamma =\frac{5}{3} \right)\]value of with 1 mole of diatomic gas \[\left( \gamma =\frac{7}{5} \right)\]what will be the value of y for the mixture?
A)
1, 45
done
clear
B)
1, 4
done
clear
C)
1, 54
done
clear
D)
1, 5
done
clear
View Answer play_arrow
question_answer 30) A pendulum bob has a speed of 3 m/s at its lowest position. The pendulum is 0.5 m long. The speed of the bob, when length makes an angle of 60° to the vertical, is :
A)
2 m/s
done
clear
B)
-m/s
done
clear
C)
\[\frac{1}{3}\] m/s
done
clear
D)
2.5 m/s
done
clear
View Answer play_arrow
question_answer 31) An object of mass 40 kg and having the velocity 4 m/s collides with another object of mass 60 kg having velocity 2 m/s. The collision is perfectly inelastic. The loss in energy is :
A)
110J
done
clear
B)
48J
done
clear
C)
392J
done
clear
D)
440J
done
clear
View Answer play_arrow
question_answer 32) A major-cyclist moving with a velocity of 72 km/h on .1 flat road lakes a turn on the road at a point where the radius of curvature of the road is 20 m. The acceleration due to gravity is 10 m/s2. In order 10 avoid sliding. he must not bend with respect to the vertical plane by an angle greater than :
A)
\[\theta ={{\tan }^{-1}}(4)\]
done
clear
B)
\[\theta ={{\tan }^{-1}}(25,92)\]
done
clear
C)
\[\theta ={{\tan }^{-1}}(2)\]
done
clear
D)
\[\theta ={{\tan }^{-1}}(6)\]
done
clear
View Answer play_arrow
question_answer 33)
Three equal resistors are connected as shown in the figure. The maximum power consumed by each resistor is 18 W. Then maximum power consumed by the combination is :
A)
36 W
done
clear
B)
18 W
done
clear
C)
27 W
done
clear
D)
54 W
done
clear
View Answer play_arrow
question_answer 34) A long vertical current carrying wire produces a neutral point with the earth. Magnetic field at a distance of 5cm from the wire. If the horizontal component of the earth's magnetic induction is 0.18 gauss, then the current in the wire is :
A)
0.036 A
done
clear
B)
0.9 A
done
clear
C)
0.45 A
done
clear
D)
4.5 A
done
clear
View Answer play_arrow
question_answer 35) A point object is 24 cm above the surface of water (\[\mu \] = 4/ 3) in lake. A fish inside the water will observe the image to be at a point:
A)
6 cm above the surface of water
done
clear
B)
6 cm below the surface of water
done
clear
C)
18 cm above the .surface of water
done
clear
D)
32 cm above the surface of water
done
clear
View Answer play_arrow
question_answer 36) The optical length of an astronomical telescope with magnifying power of 10. For normal vision is 44 cm, what is focal length of the objective?
A)
4 cm
done
clear
B)
40 cm
done
clear
C)
44 cm
done
clear
D)
440 cm
done
clear
View Answer play_arrow
question_answer 37) The energy of ground electronic state of hydrogen atom is -13.6 eV. The energy of the first excited state will be :
A)
-52,4eV
done
clear
B)
-27.2eV
done
clear
C)
-68eV
done
clear
D)
-3.4 eV
done
clear
View Answer play_arrow
question_answer 38) A block of mass m1 rests on a horizontal table. A string tied to this block is passed over a frictional pulley fixed at one end of the table and another block of mass m1 is hung to the other end of the string. The acceleration (a) of the system is :
A)
\[\frac{{{m}_{1}}{{m}_{2}}g}{{{m}_{1}}-{{m}_{2}}}\]
done
clear
B)
\[\frac{{{m}_{2}}g}{{{m}_{1}}+{{m}_{2}}}\]
done
clear
C)
\[\frac{{{m}_{1}}g}{{{m}_{1}}+{{m}_{2}}}\]
done
clear
D)
\[g\]
done
clear
View Answer play_arrow
question_answer 39) In a p-n-p transistor working as a common base amplifier current gain is 0.96 and emitter current is 7.2 mA. The base current is :
A)
0.2mA
done
clear
B)
0.29mA
done
clear
C)
0.35 mA
done
clear
D)
0.4 mA
done
clear
View Answer play_arrow
question_answer 40) impedance of circuit when a resistance R and a inductor of inductance L are connected in series in an AC circuit of frequency v, is :
A)
\[\sqrt{{{R}^{2}}+2{{\pi }^{2}}{{v}^{2}}{{L}^{2}}}\]
done
clear
B)
\[\sqrt{{{R}^{2}}+4{{\pi }^{2}}{{v}^{2}}{{L}^{2}}}\]
done
clear
C)
\[\sqrt{R+4{{\pi }^{2}}{{v}^{2}}{{L}^{2}}}\]
done
clear
D)
\[\sqrt{R+2{{\pi }^{2}}{{v}^{2}}{{L}^{2}}}\]
done
clear
View Answer play_arrow
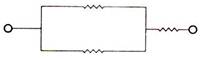
question_answer 41)
In the given figure equivalent resistance between P and Q will be :
A)
\[\frac{14}{9}\Omega \]
done
clear
B)
\[\frac{9}{14}\Omega \]
done
clear
C)
\[\frac{14}{3}\Omega \]
done
clear
D)
\[\frac{3}{14}\Omega \]
done
clear
View Answer play_arrow
question_answer 42) The electric field required to keep a water drop of mass m just to remain suspended, when charged with one electron is :
A)
\[\frac{em}{g}\]
done
clear
B)
\[\frac{mg}{e}\]
done
clear
C)
emg
done
clear
D)
mg
done
clear
View Answer play_arrow
question_answer 43) Which of the following phenomenon shows the transverse nature of light?
A)
Photoelectric effect
done
clear
B)
Interference
done
clear
C)
Polarization
done
clear
D)
Diffraction
done
clear
View Answer play_arrow
question_answer 44) A pure semiconductor behaves, slightly as a conductor at:
A)
high temperature
done
clear
B)
room temperature
done
clear
C)
low temperature
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 45) The angle for which maximum height and horizontal range are same for a projectile is:
A)
\[32{}^\circ\]
done
clear
B)
\[48{}^\circ\]
done
clear
C)
\[76{}^\circ\]
done
clear
D)
\[84{}^\circ\]
done
clear
View Answer play_arrow
question_answer 46) Two particles of equal mass revolve in circular paths of radii r1 and r2 respectively with: he same angular velocity. The ratio of their centripetal forces will be :
A)
\[\frac{{{r}_{1}}}{{{r}_{2}}}\]
done
clear
B)
\[\frac{{{r}_{2}}}{{{r}_{3}}}\]
done
clear
C)
\[\sqrt{\frac{{{r}_{2}}}{{{r}_{1}}}}\]
done
clear
D)
\[{{\left( \frac{{{r}_{2}}}{{{r}_{1}}} \right)}^{2}}\]
done
clear
View Answer play_arrow
question_answer 47) An aeroplane moving horizontally with a speed of 720 km/h drops a food packet, while flying at a height of" 396.9 m. The time taken by a food packet to reach the ground and in horizontal range is : (g = 9.8 m/s2)
A)
9 s and 1800 m
done
clear
B)
8 s and 1500 m
done
clear
C)
3 s and 2000 m
done
clear
D)
5 s and 500 m
done
clear
View Answer play_arrow
question_answer 48) If work done in increasing the size of a soap film from \[10\text{ }cm\times 6\text{ }cm\text{ }to10\text{ }cm\times 11cma2\times {{10}^{-4}}J,\]then the surface tension is :
A)
\[2\times {{10}^{-8}}N{{m}^{-1}}\]
done
clear
B)
\[2\times {{10}^{-6}}N{{m}^{-1}}\]
done
clear
C)
\[2\times {{10}^{-4}}N{{m}^{-1}}\]
done
clear
D)
\[2\times {{10}^{-2}}N{{m}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 49) Three bulbs of 40 W, 60 W and 100 W an arranged in series with 220 V, which bulb ha minimum resistance?
A)
100 W
done
clear
B)
40 W
done
clear
C)
60 W
done
clear
D)
Equal in all bulbs
done
clear
View Answer play_arrow
question_answer 50) A transistor has an \[\alpha \] = 0.95, then \[\beta \] is equal to:
A)
\[\frac{1}{19}\]
done
clear
B)
19
done
clear
C)
1.5
done
clear
D)
0.95
done
clear
View Answer play_arrow
question_answer 51) Isotopes have :
A)
same number of neutrons
done
clear
B)
same number of positron
done
clear
C)
same number of protons
done
clear
D)
same number of nucleus
done
clear
View Answer play_arrow
question_answer 52) 4.6 kJ heat is liberated on burning 0.5 g of sulphur. The enthalpy of formation of \[S{{O}_{2}}\] is: [molecular weight of S = 32, O = 16]
A)
+ 294.4 kJ
done
clear
B)
- 294.4 kJ
done
clear
C)
+ 462.4 kJ
done
clear
D)
- 462.4 kJ
done
clear
View Answer play_arrow
question_answer 53) At \[25{{\,}^{o}}C\]the pH value of a solution is 6, the solution is:
A)
neutral
done
clear
B)
acidic
done
clear
C)
alkaline
done
clear
D)
basic
done
clear
View Answer play_arrow
question_answer 54) The incorrect statement regarding vitamins is:
A)
They help in digestion
done
clear
B)
Their deficiency causes disease
done
clear
C)
They are vital for life
done
clear
D)
They were named by "Funk"
done
clear
View Answer play_arrow
question_answer 55) Nitrobenzene on nitration produce :
A)
o-dinitrobenzene
done
clear
B)
m-dinitrobenzene
done
clear
C)
p-dinitrobenzene
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 56) The boiling point of alcohol is higher than ether due to:
A)
hydrogen bonding
done
clear
B)
large size of alcohol
done
clear
C)
presence of ? OH group
done
clear
D)
high molecular weight
done
clear
View Answer play_arrow
question_answer 57) The volume of \[1.0g\]of hydrogen in litres at NTP is :
A)
6.8 L
done
clear
B)
10.44 L
done
clear
C)
11.2 L
done
clear
D)
14.56 L
done
clear
View Answer play_arrow
question_answer 58) The specific rate constant of a first order reaction depends on :
A)
time
done
clear
B)
temperature
done
clear
C)
concentration of product
done
clear
D)
concentration of the reactants
done
clear
View Answer play_arrow
question_answer 59) \[CO+{{H}_{2}}\xrightarrow[Co\,or\,Ni]{43\,K,\,1.1\,atmosphere}\]mixture of hydrocarbons \[+{{H}_{2}}O\] The above reaction is :
A)
Bergius process
done
clear
B)
Fischer-Tropsch process
done
clear
C)
Kolbe's process
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 60) The law which explains the law of conservation of mass is:
A)
Avogadro's law
done
clear
B)
Berzelius hypothesis
done
clear
C)
Hund's rule
done
clear
D)
Dalton's theory
done
clear
View Answer play_arrow
question_answer 61) The internal energy of a substance :
A)
increases with increase in temperature
done
clear
B)
decreases with increase in temperature
done
clear
C)
remains constant
done
clear
D)
calculated by \[E=m{{c}^{2}}\]
done
clear
View Answer play_arrow
question_answer 62) Anhydrous \[N{{a}_{2}}C{{O}_{3}}\]is also known as :
A)
fusion mixture
done
clear
B)
soda ash
done
clear
C)
washing soda
done
clear
D)
salt soda
done
clear
View Answer play_arrow
question_answer 63) The type of linkage present in protein molecule is :
A)
ether linkage
done
clear
B)
peptide linkage
done
clear
C)
glycosidic linkage
done
clear
D)
ester linkage
done
clear
View Answer play_arrow
question_answer 64) Two molecules of an ideal gas expand spontaneously into a vacuum. The work done is :
A)
2J
done
clear
B)
4J
done
clear
C)
8J
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 65) In an aqueous solution hydrogen will not reduce :
A)
\[A{{g}^{+}}\]
done
clear
B)
\[C{{u}^{2+}}\]
done
clear
C)
\[Z{{n}^{2+}}\]
done
clear
D)
\[F{{e}^{3+}}\]
done
clear
View Answer play_arrow
question_answer 66) The volume occupied by 9.0 g of nitrogen gas at 300 K and 750 mm Hg pressure is:
A)
5.854
done
clear
B)
6.7432
done
clear
C)
8.8462
done
clear
D)
8.0125
done
clear
View Answer play_arrow
question_answer 67) Toluene and chromyl chloride reacts to produce :
A)
benzoicacid
done
clear
B)
benzaldehyde
done
clear
C)
chlorotoluene
done
clear
D)
benzyl chloride
done
clear
View Answer play_arrow
question_answer 68) The atomic radius decreases in a period due to:
A)
increase in nuclear attraction
done
clear
B)
decrease in nuclear attraction
done
clear
C)
increase in number of electrons
done
clear
D)
decrease in number of electrons
done
clear
View Answer play_arrow
question_answer 69) The change in entropy for the fusion of 1 mole of ice is [melting point of ice = 273 K, molar enthalpy of fusion for ice\[=6.0\,kJ\,mo{{l}^{-1}}\]]:
A)
\[11.73\,J{{K}^{-1}}\,mo{{l}^{-1}}\]
done
clear
B)
\[18.84\,J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
C)
\[21.97J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
D)
\[24.47\,J{{K}^{-1}}\,mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 70) The solutions which have same osmotic concentration are known as :
A)
normal
done
clear
B)
isotonic
done
clear
C)
hypotonic
done
clear
D)
hypertonic
done
clear
View Answer play_arrow
question_answer 71) On reduction secondary amine is given by:
A)
methyl isocyanide
done
clear
B)
methyl cyanide
done
clear
C)
nitrobenzene
done
clear
D)
nitrotoluene
done
clear
View Answer play_arrow
question_answer 72) Schiffs reagent is:
A)
red litmus
done
clear
B)
rosaniline hydrochloride
done
clear
C)
methyl orange
done
clear
D)
2, 4 dinitrophenyl hydrazine
done
clear
View Answer play_arrow
question_answer 73) In nuclear reactors the moderator is :
A)
\[{{D}_{2}}O\]
done
clear
B)
\[{{H}_{2}}O\]
done
clear
C)
\[{{H}_{2}}{{O}_{2}}\]
done
clear
D)
\[{{H}_{2}}S{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 74) 0.2595 g of a substance containing organic compound in a quantitative analysis gives 0.35 g of barium sulphate. The percentage of sulphur is :
A)
10.84%
done
clear
B)
14.64%
done
clear
C)
16.66%
done
clear
D)
18.52%
done
clear
View Answer play_arrow
question_answer 75) Perlon is:
A)
terylene
done
clear
B)
rubber
done
clear
C)
nylon-6
done
clear
D)
polyester
done
clear
View Answer play_arrow
question_answer 76) Phenol and \[N{{H}_{3}}\]reacts in presence of \[ZnC{{l}_{2}}\]at \[300{{\,}^{o}}C\]to produce :
A)
tertiary amine
done
clear
B)
secondary amine
done
clear
C)
primary amine
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 77) Molality does not change with :
A)
temperature
done
clear
B)
concentration
done
clear
C)
pressure
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 78) The pH of \[N/10HCl\]is :
A)
3
done
clear
B)
4
done
clear
C)
2
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 79) The solubility of \[AgCl\]in moles per litre when its solubility product is \[1.56\times {{10}^{-10}}\]at \[25{{\,}^{o}}C\]is :
A)
\[0.576\times {{10}^{-8}}\,mol/L\]
done
clear
B)
\[1.056\times {{10}^{-4}}mol/L\]
done
clear
C)
\[1.249\times {{10}^{-5}}L\]
done
clear
D)
\[1.478\times {{10}^{-6}}mol/L\]
done
clear
View Answer play_arrow
question_answer 80) The amphoteric oxide is :
A)
\[Cr{{O}_{3}}\]
done
clear
B)
\[Cr{{O}_{5}}\]
done
clear
C)
\[C{{r}_{2}}{{O}_{3}}\]
done
clear
D)
\[CrO\]
done
clear
View Answer play_arrow
question_answer 81) Tartarametic is:
A)
sodium potassium tartrate
done
clear
B)
tartaricacid
done
clear
C)
potassium antimony tartrate
done
clear
D)
potassium acid tartrate
done
clear
View Answer play_arrow
question_answer 82) The noble gas which is not found in atmosphere is :
A)
Ar
done
clear
B)
Kr
done
clear
C)
Ne
done
clear
D)
Rn
done
clear
View Answer play_arrow
question_answer 83) Slow oxidation of \[CHC{{l}_{3}}\]in air leads to the formation of:
A)
formic acid
done
clear
B)
carbonyl chloride
done
clear
C)
formyi chloride
done
clear
D)
trichloroacetic acid
done
clear
View Answer play_arrow
question_answer 84) For the manufacture of NI-^ by the following reaction\[{{N}_{2}}+3{{H}_{2}}2N{{H}_{3}}+21.9\,kcal\] the favourable conditions are :
A)
low temperature, high pressure and catalyst
done
clear
B)
high temperature, high pressure and catalyst
done
clear
C)
high temperature, high pressure and catalyst
done
clear
D)
low temperature, low pressure and catalyst
done
clear
View Answer play_arrow
question_answer 85) 4 g caustic soda is dissolved in 100 cc of solution. The normality of solution is:
A)
0
done
clear
B)
0.5
done
clear
C)
1
done
clear
D)
1.5
done
clear
View Answer play_arrow
question_answer 86) Both \[B{{F}_{3}}\]and \[N{{F}_{3}}\]are covalent but \[B{{F}_{3}}\]molecule is non-polar while \[N{{F}_{3}}\] is polar because:
A)
atomic size of boron is smaller than nitrogen
done
clear
B)
\[B{{F}_{3}}\]is planar but \[N{{F}_{3}}\]is pyramidal
done
clear
C)
boron is a metal while nitrogen is a gas
done
clear
D)
BF bond have no dipole moment while NF bond have dipole moment
done
clear
View Answer play_arrow
question_answer 87) Carbon monoxide is a poisonous .gas, the antidote used for this poisoning is :
A)
pure oxygen
done
clear
B)
carbonic acid
done
clear
C)
carborundum
done
clear
D)
carbogen
done
clear
View Answer play_arrow
question_answer 88) \[AlC{{l}_{3}}\]fumes in moist air because it is :
A)
covalent
done
clear
B)
volatile
done
clear
C)
hygroscopic
done
clear
D)
forms \[HCl\]in moist air
done
clear
View Answer play_arrow
question_answer 89) Phenol is more readily soluble in:
A)
NaOH solution
done
clear
B)
dil. HCl
done
clear
C)
both NaOH and HCl
done
clear
D)
\[NaHC{{O}_{3}}\]solution
done
clear
View Answer play_arrow
question_answer 90) Law of electrolysis was given by :
A)
Faraday
done
clear
B)
Ostwald
done
clear
C)
Arrhenius
done
clear
D)
Lamarck
done
clear
View Answer play_arrow
question_answer 91) 10 g of glucose is dissolved in 150 g of water. The mass percentage of glucose is:
A)
2.50%
done
clear
B)
6.25%
done
clear
C)
8.75%
done
clear
D)
10%
done
clear
View Answer play_arrow
question_answer 92) Enzymes are:
A)
moulds
done
clear
B)
inorganic compound
done
clear
C)
proteins
done
clear
D)
micro-organism
done
clear
View Answer play_arrow
question_answer 93) The molecularity of a reaction is :
A)
always two
done
clear
B)
same as its order
done
clear
C)
different than the order
done
clear
D)
may be same or different as the order
done
clear
View Answer play_arrow
question_answer 94) Benzene does not undergo addition reaction easily because :
A)
it has six hydrogen atoms
done
clear
B)
it has a cyclic structure
done
clear
C)
double bonds present in benzene are strong
done
clear
D)
resonance stabilized system is to be preserved
done
clear
View Answer play_arrow
question_answer 95) Metal used for drying organic solvent is :
A)
Na
done
clear
B)
Mg
done
clear
C)
Fe
done
clear
D)
Pt
done
clear
View Answer play_arrow
question_answer 96) Water glass is:
A)
glass made up of water
done
clear
B)
ammonium benzoate
done
clear
C)
sodium silicate
done
clear
D)
calcium silicate
done
clear
View Answer play_arrow
question_answer 97) Soaps are classified as :
A)
carbohydrates
done
clear
B)
salts of fatty acids
done
clear
C)
ether
done
clear
D)
alcohols
done
clear
View Answer play_arrow
question_answer 98) \[{{C}_{3}}{{H}_{9}}N\]cannot represent the following :
A)
primary amine
done
clear
B)
secondary amine
done
clear
C)
tertiary amine
done
clear
D)
quaternary ammonium salt
done
clear
View Answer play_arrow
question_answer 99) Slaked lime and chlorine reacts to produce :
A)
quick lime
done
clear
B)
\[CaC{{l}_{2}}\]
done
clear
C)
\[CaOC{{l}_{2}}\]
done
clear
D)
mixture of \[CaC{{l}_{2}}\]and \[Ca{{(OCl)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 100) The essential constituent of diet is :
A)
amino acid
done
clear
B)
starch
done
clear
C)
glucose
done
clear
D)
carbohydrates
done
clear
View Answer play_arrow
question_answer 101) Spirogyra filaments increase in length due to division in:
A)
basal cell
done
clear
B)
middle cells
done
clear
C)
rhizoidal
done
clear
D)
all green cells
done
clear
View Answer play_arrow
question_answer 102) Zygotic meiosis is found in :
A)
Fern
done
clear
B)
Fucus
done
clear
C)
Funaria
done
clear
D)
Chlamydomonas
done
clear
View Answer play_arrow
question_answer 103) The functional xylem of dicot tree is :
A)
sap wood
done
clear
B)
hard wood
done
clear
C)
heart wood
done
clear
D)
autumn wood
done
clear
View Answer play_arrow
question_answer 104) Which of the following theories was not given by Darwin?
A)
Struggle for existence
done
clear
B)
Natural selection
done
clear
C)
Survival of the fittest
done
clear
D)
Genetic drift
done
clear
View Answer play_arrow
question_answer 105) Fungi are always :
A)
autotrophic
done
clear
B)
heterotrophic
done
clear
C)
parasitic
done
clear
D)
saprophytic
done
clear
View Answer play_arrow
question_answer 106) Bacteria in cold climate can live for :
A)
few years
done
clear
B)
1,000 years
done
clear
C)
2,000 years
done
clear
D)
many thousand years
done
clear
View Answer play_arrow
question_answer 107) The term ?nucleolus? was coined by :
A)
R. Brown
done
clear
B)
R. Hooke
done
clear
C)
Bowman
done
clear
D)
Hanstein
done
clear
View Answer play_arrow
question_answer 108) The organ which gets only oxygenated blood is:
A)
lungs
done
clear
B)
gills
done
clear
C)
spleen
done
clear
D)
liver
done
clear
View Answer play_arrow
question_answer 109) In a pond ecosytem, ?benthos? means :
A)
epineuston
done
clear
B)
periphyton
done
clear
C)
zooplankton on the water surface
done
clear
D)
primary consumers in the depth of a pond
done
clear
View Answer play_arrow
question_answer 110) In malaria, the lever reoccurs due to :
A)
release of merozoites in RBC
done
clear
B)
bursting of RBC
done
clear
C)
formation of sporozoites
done
clear
D)
discharge of excretory products and waste from pathogens
done
clear
View Answer play_arrow
question_answer 111) The condition in which narrowing of arteries take place is :
A)
dotting
done
clear
B)
haemophilia
done
clear
C)
arteriosis
done
clear
D)
arteriosclerosis
done
clear
View Answer play_arrow
question_answer 112) DNA nucleotides are attached by :
A)
hydrogen bonds
done
clear
B)
covalent bonds
done
clear
C)
van der Wall's force
done
clear
D)
electrovalent bonds
done
clear
View Answer play_arrow
question_answer 113) In which of the following will you look for E. coli?
A)
Milk
done
clear
B)
Water
done
clear
C)
Human intestine
done
clear
D)
Soil
done
clear
View Answer play_arrow
question_answer 114) The production and storage of energy in mitochondria increases during :
A)
seed germination
done
clear
B)
dormant seed
done
clear
C)
ripening seed
done
clear
D)
seed maturation
done
clear
View Answer play_arrow
question_answer 115) Venom of cobra attacks :
A)
digestive system
done
clear
B)
respiratory system
done
clear
C)
nervous system
done
clear
D)
circulatory system
done
clear
View Answer play_arrow
question_answer 116) Filariform is the larva of:
A)
Arthropod
done
clear
B)
Platyhelminthes
done
clear
C)
Aschelminthes
done
clear
D)
Protozoan
done
clear
View Answer play_arrow
question_answer 117) Type of relationship in which one organism is benefitted while there is not effect on other is
A)
symbiosis
done
clear
B)
commensalism
done
clear
C)
mutualism
done
clear
D)
parasitism
done
clear
View Answer play_arrow
question_answer 118) Silver fish, crab, cockroach, honey bees have which of the following feature common?
A)
Compound eyes
done
clear
B)
Jointed legs
done
clear
C)
Poison glands
done
clear
D)
Metamorphosis
done
clear
View Answer play_arrow
question_answer 119) Amphiblastula is the larva of :
A)
Hydra
done
clear
B)
Sycon
done
clear
C)
Planaria
done
clear
D)
Leucosolenia
done
clear
View Answer play_arrow
question_answer 120) Fertilization in flowering plant was observed by :
A)
Nawaschin
done
clear
B)
Strasburger
done
clear
C)
Hofmeister
done
clear
D)
Maheshwari
done
clear
View Answer play_arrow
question_answer 121) Which of these is absent in anaerobic bacteria?
A)
Cell wall
done
clear
B)
Cell membrane
done
clear
C)
Mesosomes
done
clear
D)
Ribosomes
done
clear
View Answer play_arrow
question_answer 122) Which of the following is present in man but absent in frogs?
A)
Salivary gland
done
clear
B)
Adrenal gland
done
clear
C)
Thyroid gland
done
clear
D)
Pancreas
done
clear
View Answer play_arrow
question_answer 123) Which of the following is not a function of vitamins?
A)
Digestion
done
clear
B)
Metabolism
done
clear
C)
Growth
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 124) Jecobson?s organ is related with :
A)
touch
done
clear
B)
smell
done
clear
C)
heat
done
clear
D)
brain
done
clear
View Answer play_arrow
question_answer 125) Pigmentation of skin is due to :
A)
leucocytes
done
clear
B)
monocytes
done
clear
C)
lymphocytes
done
clear
D)
melanocytes
done
clear
View Answer play_arrow
question_answer 126) Which of the following is a bacterial disease?
A)
Measles
done
clear
B)
Leprosy
done
clear
C)
Diabetes
done
clear
D)
Scurvy
done
clear
View Answer play_arrow
question_answer 127) Broad spectrum antibiotics are one which attack :
A)
pathogens as well as host
done
clear
B)
a wide range of pathogens
done
clear
C)
only pathogens
done
clear
D)
only host
done
clear
View Answer play_arrow
question_answer 128) The disease on the verge of eradication in India is :
A)
kala-azar
done
clear
B)
smallpox
done
clear
C)
poliomyelitis
done
clear
D)
AIDS
done
clear
View Answer play_arrow
question_answer 129) Interferon is a glycoprotein which is :
A)
antiviral
done
clear
B)
anti-bacterial
done
clear
C)
anti-pathogenic
done
clear
D)
anti-oxidant
done
clear
View Answer play_arrow
question_answer 130) Hydra will not be regenerated fully, if it contains :
A)
tentacles, epidermis, gastro dermis
done
clear
B)
gastro dermis, epidermis, hypodermis
done
clear
C)
epidermis, gastro dermis
done
clear
D)
tentacles
done
clear
View Answer play_arrow
question_answer 131) Most abundant lipid in the cell membrane is :
A)
cholesterol
done
clear
B)
phospholipids
done
clear
C)
glycolipids
done
clear
D)
cerebrosides
done
clear
View Answer play_arrow
question_answer 132) ?Power house' of the cell is called :
A)
nucleus
done
clear
B)
ribosome
done
clear
C)
mitochondria
done
clear
D)
Golgi complex
done
clear
View Answer play_arrow
question_answer 133) Ozone hole means :
A)
hole in the ozone layer in stratosphere
done
clear
B)
decrease in the thickness of ozone in the stratosphere
done
clear
C)
decrease in concentration of ozone in trophosphere
done
clear
D)
increase in the concentration of ozone in trophosphere
done
clear
View Answer play_arrow
question_answer 134) Ball and socket joint is found between :
A)
ribs and vertebral
done
clear
B)
femur and tibio-fibula
done
clear
C)
humerus and olecranon fossa
done
clear
D)
humerus and pectoral girdle
done
clear
View Answer play_arrow
question_answer 135) Which of the following is present in mollusks but not in echinodermates?
A)
Flame cell
done
clear
B)
Malpighian tubules
done
clear
C)
Kidney
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 136) Testes descend into scrotum in mammals for:
A)
development of visceral organs
done
clear
B)
spermatogenesis
done
clear
C)
development of sex organs
done
clear
D)
fertilization
done
clear
View Answer play_arrow
question_answer 137) The nerve transmitter, produced at the synapse and neuromuscular junction, is :
A)
GTP
done
clear
B)
ATP
done
clear
C)
acetylcholine
done
clear
D)
phosphokinase
done
clear
View Answer play_arrow
question_answer 138) Which of the following are water soluble vitamins ?
A)
B, C
done
clear
B)
A, D, K
done
clear
C)
A, D, E, K
done
clear
D)
K, B, A, E
done
clear
View Answer play_arrow
question_answer 139) Sprinkling of oil on stagnant water helps in the control of malaria because :
A)
mosquito larvae get asphyxiated (cannot breath)
done
clear
B)
water becomes impure for mosquito
done
clear
C)
specific gravity of water increases
done
clear
D)
oils kills malarial parasite in mosquitoes
done
clear
View Answer play_arrow
question_answer 140) Plants wilt due to excess of:
A)
transpiration
done
clear
B)
photosynthesis
done
clear
C)
absorption
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 141) Cornea transplantation is done on large scale now-a-days because :
A)
it can be easily available
done
clear
B)
it can be easily transplanted
done
clear
C)
it does not link up with blood vessels
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 142) 'Comma' shaped bacteria are known as :
A)
coccus
done
clear
B)
spiral
done
clear
C)
lophotrichus
done
clear
D)
vibrio
done
clear
View Answer play_arrow
question_answer 143) Dance pattern in honey bees is for :
A)
courtship
done
clear
B)
communication
done
clear
C)
recreation
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 144) Bones are mainly made up of :
A)
calcium and phosphorus
done
clear
B)
calcium and sulphur
done
clear
C)
calcium and magnesium
done
clear
D)
calcium and iron
done
clear
View Answer play_arrow
question_answer 145) The process of formation of blood is known as
A)
plasmolysis
done
clear
B)
haemopoiesis
done
clear
C)
haemolysis
done
clear
D)
haemoneogenesis
done
clear
View Answer play_arrow
question_answer 146) Which of the following is a sex-linked disease?
A)
Syphillis
done
clear
B)
Hepatitis
done
clear
C)
Phenyketonuria
done
clear
D)
Colour blindness
done
clear
View Answer play_arrow
question_answer 147) The egg apparatus usually consists of :
A)
egg and antipodal cells
done
clear
B)
egg and central cell
done
clear
C)
egg and two synergids
done
clear
D)
egg and one synergid
done
clear
View Answer play_arrow
question_answer 148) Biologically marriage should be avoided between :
A)
Rh' male and Rh' female?
done
clear
B)
Rh male and Rh' female
done
clear
C)
Rh' male and Rh female
done
clear
D)
Rh' female and Rh" male
done
clear
View Answer play_arrow
question_answer 149) The functional unit of kidney is called :
A)
neuron
done
clear
B)
nephron
done
clear
C)
axon
done
clear
D)
nephridia
done
clear
View Answer play_arrow
question_answer 150) The correct sequence of taxonomic categories is :
A)
class-phylum -order-family-genus - species
done
clear
B)
phylum -order-class-genus-family- species
done
clear
C)
division-class-family-genus-species- order
done
clear
D)
phylum-class-order -family-genus- species
done
clear
View Answer play_arrow
question_answer 151) Who is the constitutional head of our century?
A)
The president
done
clear
B)
The Lt. General
done
clear
C)
The Chief Minister
done
clear
D)
The Chief Justice
done
clear
View Answer play_arrow
question_answer 152) The period known as the ?Goldern Age" of India :
A)
Gupta's regime
done
clear
B)
Mughal's regime
done
clear
C)
Maurya?s regime
done
clear
D)
English regime
done
clear
View Answer play_arrow
question_answer 153) If time is to clock then pressure is to :
A)
Potometer
done
clear
B)
Hydrometer
done
clear
C)
Barometer
done
clear
D)
Lactometer
done
clear
View Answer play_arrow
question_answer 154) Who decides whether a member of parliament is subject to any disqualification?
A)
Speaker
done
clear
B)
President
done
clear
C)
Prime Minister
done
clear
D)
Chief Justice
done
clear
View Answer play_arrow
question_answer 155) First general elections were held in India in :
A)
1966
done
clear
B)
1951
done
clear
C)
1956
done
clear
D)
1949
done
clear
View Answer play_arrow
question_answer 156) Who gave the slogan ?InquilabZindabad??
A)
Bhagat Singh
done
clear
B)
S.C. Bose
done
clear
C)
Sardar Patel
done
clear
D)
Mahatma Gandhi
done
clear
View Answer play_arrow
question_answer 157) ?National Song? is written by :
A)
Iquabal
done
clear
B)
BankimChander Chatter Ji
done
clear
C)
Tagore
done
clear
D)
M.F. Hussain
done
clear
View Answer play_arrow
question_answer 158) The ?God of Small Things? is written by :
A)
Indira Gandhi
done
clear
B)
Benazir Bhutto
done
clear
C)
Tasleema Nasreen
done
clear
D)
(d ) Arundhati Roy
done
clear
View Answer play_arrow
question_answer 159) Two arms of a watch show time of 3.30 in a mirror. What is the actual time in watch?
A)
12 : 30
done
clear
B)
8 : 30
done
clear
C)
4 : 30
done
clear
D)
9 : 30
done
clear
View Answer play_arrow
question_answer 160) If in a certain code MAGAZINE is coded as AMAGIZEN, then ENVELOPE will be coded as :
A)
ENEVOLEP
done
clear
B)
NEVEOLEP
done
clear
C)
NEEVOLEP
done
clear
D)
NEEVOLPE
done
clear
View Answer play_arrow
question_answer 161) If in a certain code STUDENT is coded as TSDUNET then TEACHER will be coded as :
A)
ETCAEHR
done
clear
B)
REHACTA
done
clear
C)
ETCHAER
done
clear
D)
ATECHER
done
clear
View Answer play_arrow
question_answer 162) Hoof: Horse : : ? : Man
A)
Leg
done
clear
B)
Arm
done
clear
C)
Hand
done
clear
D)
Foot
done
clear
View Answer play_arrow
question_answer 163) A woman introduces a man as the son of the brother of her mother. How is the man related to the woman?
A)
Nephew
done
clear
B)
Son
done
clear
C)
Cousin
done
clear
D)
Uncle
done
clear
View Answer play_arrow
question_answer 164) If B says that his mother is the only daughter of A?s mother. How is A related to B?
A)
Uncle
done
clear
B)
Father
done
clear
C)
Brother
done
clear
D)
Grand-father
done
clear
View Answer play_arrow
question_answer 165) Find out the wrong number in each series. 100, 97, 90, 86, 76, 71, 62, 55:
A)
55
done
clear
B)
62
done
clear
C)
76
done
clear
D)
86
done
clear
View Answer play_arrow
question_answer 166) Find out the wrong number in each series. 1, 2, 5, 14, 41, 124:
A)
2
done
clear
B)
5
done
clear
C)
124
done
clear
D)
41
done
clear
View Answer play_arrow
question_answer 167) If a match starts between India and England at 10.00 a.m. in London, at what time it will be telecaste in India?
A)
2.30 a.m.
done
clear
B)
3.30 p.m.
done
clear
C)
6.00 p.m.
done
clear
D)
10.00 a.m.
done
clear
View Answer play_arrow
question_answer 168) If 35846 is coded as 63458, then how 24321 will be coded as :
A)
12234
done
clear
B)
14223
done
clear
C)
12243
done
clear
D)
12423
done
clear
View Answer play_arrow
question_answer 169) If 35698 is coded as 53766, then 67284 will be coded as :
A)
84332
done
clear
B)
86263
done
clear
C)
84352
done
clear
D)
85352
done
clear
View Answer play_arrow
question_answer 170) What is the number which multiplied with itself gives a number which is 5 greater than 59?
A)
5
done
clear
B)
8
done
clear
C)
9
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 171) 94, 88, 82, 96, 70, 104.......:
A)
58
done
clear
B)
60
done
clear
C)
64
done
clear
D)
78
done
clear
View Answer play_arrow
question_answer 172) Find the number which will come next in the series: 3, 14 , 47.......:
A)
89
done
clear
B)
119
done
clear
C)
137
done
clear
D)
146
done
clear
View Answer play_arrow
question_answer 173) Find the number which will come next in the series: 2, 5, 1 1, 23..........:
A)
42
done
clear
B)
47
done
clear
C)
36
done
clear
D)
34
done
clear
View Answer play_arrow
question_answer 174) Find the number which will come next in the series: 2, 9, 28, 65,.........:
A)
121
done
clear
B)
195
done
clear
C)
126
done
clear
D)
103
done
clear
View Answer play_arrow
question_answer 175) Find the number which will come next in the series: 26, 23, 20, 17, 14, 11...........:
A)
6
done
clear
B)
5
done
clear
C)
7
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 176) Which of the following represents the central idea of the passage?
A)
Life is full of pain and suffering
done
clear
B)
Struggle or without struggle life is an enjoyable game
done
clear
C)
Life is an unending struggle
done
clear
D)
Sufffering is not really necessary for achievement in life
done
clear
View Answer play_arrow
question_answer 177) Which of the following statements is not true?
A)
Hardships add to the interest of life
done
clear
B)
A game becomes really interesting if one knows beforehand how it is going to end up
done
clear
C)
Games become dull if they are not fully contested
done
clear
D)
The spectators as well as the teams enjoy a game only if there is struggle for the result
done
clear
View Answer play_arrow
question_answer 178) Which of the following nearly sums up the meaning of the first sentence?
A)
Hard labour on man?s part usually ends in suffering
done
clear
B)
Even after undergoing suffering, man can hardly achieve anything
done
clear
C)
Man cannot achieve anything unless he suffers for it
done
clear
D)
Achievement in life is disproportionate of* the suffering one undergoes for it
done
clear
View Answer play_arrow
question_answer 179) The statement the foe is worthy of the steel, in the last but one sentence means that :
A)
the other fellow is capable of defending himself
done
clear
B)
the opposite team is equipped with deadly arms
done
clear
C)
the enemy shows a lot of fact and flexibility
done
clear
D)
the enemy is capable of showing determined hardline and resistance
done
clear
View Answer play_arrow
question_answer 180) Which of the following is opposite in meaning to the word dull in the passage?
A)
Attractive
done
clear
B)
Beautiful
done
clear
C)
Interesting
done
clear
D)
Famous
done
clear
View Answer play_arrow
question_answer 181) Choose the synonyms of given words from set of the words. SHORTFALL:
A)
Shortage
done
clear
B)
Infringement
done
clear
C)
Over stepping
done
clear
D)
Violation
done
clear
View Answer play_arrow
question_answer 182) Choose the synonyms of given words from set of the words. RAISING :
A)
Uplift
done
clear
B)
Humiliation
done
clear
C)
Levelling
done
clear
D)
Depression
done
clear
View Answer play_arrow
question_answer 183) Choose the synonyms of given words from set of the words. INDICT :
A)
Guilty
done
clear
B)
Blame
done
clear
C)
Attack
done
clear
D)
Accuse
done
clear
View Answer play_arrow
question_answer 184) Choose the synonyms of given words from set of the words. EXTRAVAGANT :
A)
Pompous
done
clear
B)
Reckless
done
clear
C)
Unreasonable
done
clear
D)
Wasteful
done
clear
View Answer play_arrow
question_answer 185) Choose the synonyms of given words from set of the words. TRANSPARENT:
A)
Verbose
done
clear
B)
Lucid
done
clear
C)
Involved
done
clear
D)
Witty
done
clear
View Answer play_arrow
question_answer 186) Choose the antonyms of given words from set of the words. HOMAGE:
A)
Good
done
clear
B)
Creative
done
clear
C)
Insult
done
clear
D)
Dull
done
clear
View Answer play_arrow
question_answer 187) Choose the antonyms of given words from set of the words. DENSITY:
A)
Compactness
done
clear
B)
Bulk
done
clear
C)
Solidity
done
clear
D)
Thinness
done
clear
View Answer play_arrow
question_answer 188) Choose the antonyms of given words from set of the words. EARTHY:
A)
Surly
done
clear
B)
Taciturn
done
clear
C)
Refined
done
clear
D)
Valuable
done
clear
View Answer play_arrow
question_answer 189) Choose the antonyms of given words from set of the words. AUSTERE :
A)
Competitive
done
clear
B)
Aggressive
done
clear
C)
Blase
done
clear
D)
Soft
done
clear
View Answer play_arrow
question_answer 190) 1 : Chankya, by his foresight and strength of character, built up the Maury an Empire. 6 : It is not true as some people said that Sardar Patel was not particular about the means as long as he achieved his end. P : They were men of iron. Q : Therefore, they were impervious to criticism and had implicit confidence in themselves. R : Both of them were guided by stern realism and would tolerate no deviations from the objectives which they pursued relentlessly. 5: Similarly, Patel refashioned a compact India out of the fragments left by the British.
A)
SQRP
done
clear
B)
SRQP
done
clear
C)
SRPQ
done
clear
D)
PQSR
done
clear
View Answer play_arrow
question_answer 191) 1 : Much of our adultbehaviour and our attitudes are determined by our upbringing. 6: Physiologists have studied these forces in depth. P : But the process does not stop here. Q : In particular by the effects of that small part of society which is our family. R : As we grow, we are constantly increasingly affected by new forces such as the social pressure of our friends and the larger world of society. 5: The family and our early life have profound effect on our later life.
A)
SRPQ
done
clear
B)
PRSQ
done
clear
C)
QPSR
done
clear
D)
QSPR
done
clear
View Answer play_arrow
question_answer 192) 1 : There is nothing strange in the fact that so many foreign students should wish to learn English. 6: This key will open to him whatever is valuable in the literature of the world. P : If any valuable book is written in another language an English translation of its is sure to be speedily published. Q : Anyone who masters the English tongue acquires a key. R : Most books found to be generally useful are written in English. 5 : English speaking want no monopoly of knowledge.
A)
PSQR
done
clear
B)
SQRP
done
clear
C)
RPSQ
done
clear
D)
PQSR
done
clear
View Answer play_arrow
question_answer 193) 1 : There was a boy named Jack. 6: At last she turned him out of the house. P : So the mother asked him to find work. Q : They were very poor. R : He lived with his mother. S : But Jack refused to work.
A)
RQPS
done
clear
B)
PQRS
done
clear
C)
QPRS
done
clear
D)
RPSQ
done
clear
View Answer play_arrow
question_answer 194) Choose the correct form of the adjective from the given choices. She is the ...........of the two sisters.
A)
tall
done
clear
B)
taller
done
clear
C)
tallest
done
clear
D)
tallish
done
clear
View Answer play_arrow
question_answer 195) Choose the correct form of the adjective from the given choices. ...........staff members attended the meeting.
A)
A few
done
clear
B)
Few
done
clear
C)
Any
done
clear
D)
The few
done
clear
View Answer play_arrow
question_answer 196) In the following sentences, choose the most appropriate preposition. People were alarmed.......the tiger.
A)
at
done
clear
B)
in
done
clear
C)
on
done
clear
D)
for
done
clear
View Answer play_arrow
question_answer 197) In the following sentences, choose the most appropriate preposition. He lives......... Pune.
A)
towards
done
clear
B)
from
done
clear
C)
in
done
clear
D)
to
done
clear
View Answer play_arrow
question_answer 198) In the following sentences, choose the most appropriate preposition. She is very affectionate........her children.
A)
towards
done
clear
B)
over
done
clear
C)
with
done
clear
D)
upon
done
clear
View Answer play_arrow
question_answer 199) In the following sentences, choose the most appropriate preposition. He is not in the office......the moment.
A)
ill
done
clear
B)
for
done
clear
C)
with
done
clear
D)
from
done
clear
View Answer play_arrow
question_answer 200) In the following sentences, choose the most appropriate preposition. Kiran was ashamed..........her rude behaviour.
A)
at
done
clear
B)
of Ashamed is usually followed by ?of.
done
clear
C)
over
done
clear
D)
onto
done
clear
View Answer play_arrow