 What is one thing that is common
in everyday newspaper headlines? It is the term 'government', written as Govt
in short form. You have heard this word many times from the mouth of elders,
over the radio and on television. You have also read it in the newspapers and
magazines. What is a government? What does it do? Why do we need a government?
Why does our country have a government? Who makes up the government? We will more...
What is one thing that is common
in everyday newspaper headlines? It is the term 'government', written as Govt
in short form. You have heard this word many times from the mouth of elders,
over the radio and on television. You have also read it in the newspapers and
magazines. What is a government? What does it do? Why do we need a government?
Why does our country have a government? Who makes up the government? We will more...  Discuss
? Why do you think the child is
not allowed to play with the rest?
? What do you think about this
kind of behaviour?
? Can you recollect any such
incident where someone was treated this way?
WHAT IS DISCRIMINATION?
Diversity exists naturally while
discrimination is man-made. Discrimination refers to treating a group of people
less favourably than others. This may be because of their skin colour, gender,
nationality, religion, culture, status, disability, age and many more aspects.
In most cases, people who are discriminated against are judged as 'inferior'.
Some are called hurtful names, some are not allowed to participate in events,
some are excluded from jobs and clubs, some are attacked and beaten; some are
not given promotions or unfairly paid less for doing the same work; and some
homes or places of worship are more...
Discuss
? Why do you think the child is
not allowed to play with the rest?
? What do you think about this
kind of behaviour?
? Can you recollect any such
incident where someone was treated this way?
WHAT IS DISCRIMINATION?
Diversity exists naturally while
discrimination is man-made. Discrimination refers to treating a group of people
less favourably than others. This may be because of their skin colour, gender,
nationality, religion, culture, status, disability, age and many more aspects.
In most cases, people who are discriminated against are judged as 'inferior'.
Some are called hurtful names, some are not allowed to participate in events,
some are excluded from jobs and clubs, some are attacked and beaten; some are
not given promotions or unfairly paid less for doing the same work; and some
homes or places of worship are more... 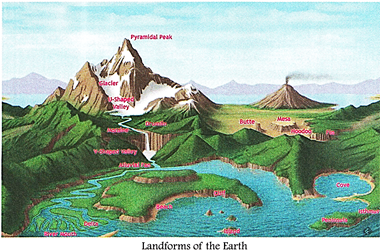 Large and small parts of our Earth
are continuously changing and different landforms are being formed. There are
two types of forces acting on the earth that bring about these changes?internal
forces and external forces.
The ground that you are standing
on is slowly moving. Within the earth a continuous movement is taking place.
The Earth's crust is made up of huge slabs called tectonic plates, which fit
together like a jigsaw puzzle. Internal movements below the earth's surface
cause collision of these plates against each other. Landforms like mountains,
faults, valleys and volcanoes are formed this way. Earthquakes also occur
'due to these movements. You will read about them later in the chapter.
External forces include running H water, moving air, waves and tides at sea,
and movement of H|B frozen ice more...
Large and small parts of our Earth
are continuously changing and different landforms are being formed. There are
two types of forces acting on the earth that bring about these changes?internal
forces and external forces.
The ground that you are standing
on is slowly moving. Within the earth a continuous movement is taking place.
The Earth's crust is made up of huge slabs called tectonic plates, which fit
together like a jigsaw puzzle. Internal movements below the earth's surface
cause collision of these plates against each other. Landforms like mountains,
faults, valleys and volcanoes are formed this way. Earthquakes also occur
'due to these movements. You will read about them later in the chapter.
External forces include running H water, moving air, waves and tides at sea,
and movement of H|B frozen ice more... You need to login to perform this action.
You will be redirected in
3 sec
